Effects of Intermittent Energy Restriction Combined with a Mediterranean Diet on Reducing Visceral Adiposity: A Randomized Active Comparator Pilot Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design and Participants
2.2. Randomization and Masking
2.3. Diet and Physical Activity Prescriptions
2.4. Study Measurements
2.5. 6-month Post-Intervention Telephone Interview
2.6. Statistical Analysis
3. Results
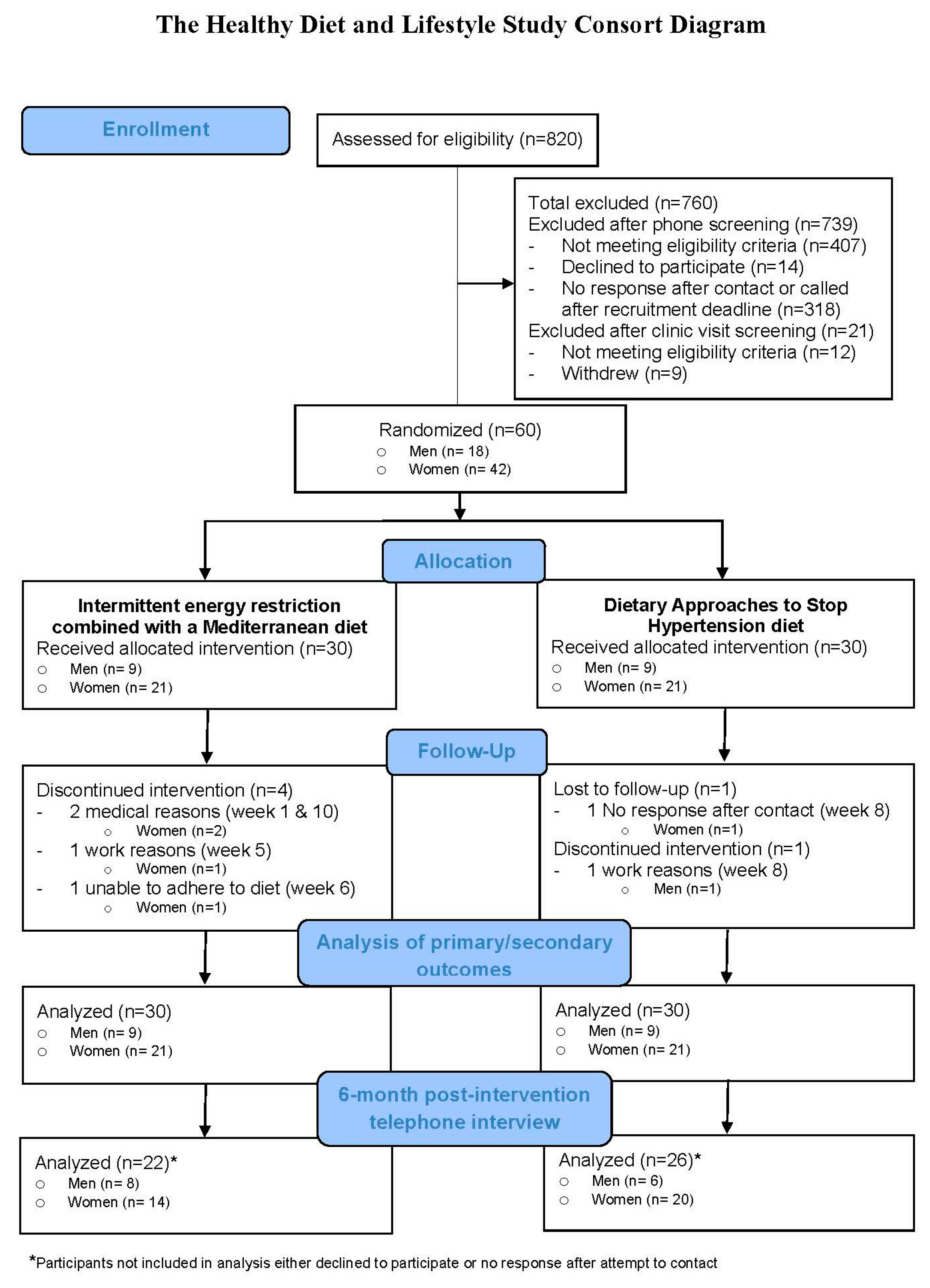
3.1. Study Population
3.2. Intervention Adherence
3.3. Changes in Anthropometric, DXA, and Biomarker Measurements at Week 12
3.4. 6-month Post-Intervention Telephone Interview
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Gallagher, E.J.; LeRoith, D. Obesity and diabetes: The increased risk of cancer and cancer-related mortality. Physiol. Rev. 2015, 95, 727–748. [Google Scholar] [CrossRef] [PubMed]
- The GBD 2015 Obesity Collaborators. Health effects of overweight and obesity in 195 countries over 25 years. New Engl. J. Med. 2017, 377, 13–27. [Google Scholar] [CrossRef] [PubMed]
- Bergman, R.N.; Kim, S.P.; Catalano, K.J.; Hsu, I.R.; Chiu, J.D.; Kabir, M.; Hucking, K.; Ader, M. Why visceral fat is bad: Mechanisms of the metabolic syndrome. Obesity 2006, 14, 16S–19S. [Google Scholar] [CrossRef] [PubMed]
- Mirza, M.S. Obesity, visceral fat, and NAFLD: Querying the role of adipokines in the progression of nonalcoholic fatty liver disease. ISRN Gastroenterol. 2011, 2011, 592404. [Google Scholar] [CrossRef] [PubMed]
- Lehrke, M.; Reilly, M.P.; Millington, S.C.; Iqbal, N.; Rader, D.J.; Lazar, M.A. An inflammatory cascade leading to hyperresistinemia in humans. PLoS Med. 2004, 1, 161–168. [Google Scholar] [CrossRef] [PubMed]
- Arner, P. Differences in lipolysis between human subcutaneous and omental adipose tissues. Ann. Med. 1995, 27, 435–438. [Google Scholar] [CrossRef] [PubMed]
- Lyon, C.J.; Law, R.E.; Hsueh, W.A. Minireview: Adiposity, inflammation, and atherogenesis. Endocrinology 2003, 144, 2195–2200. [Google Scholar] [CrossRef] [PubMed]
- Ziccardi, P.; Nappo, F.; Giugliano, G.; Esposito, K.; Marfella, R.; Cioffi, M.; D’Andrea, F.; Molinari, A.M.; Giugliano, D. Reduction of inflammatory cytokine concentrations and improvement of endothelial functions in obese women after weight loss over one year. Circulation 2002, 105, 804–809. [Google Scholar] [CrossRef]
- Sanna, C.; Rosso, C.; Marietti, M.; Bugianesi, E. Non-alcoholic fatty liver diease and extra-hepatic cancers. Int. J. Mol. Sci. 2016, 17, 717. [Google Scholar] [CrossRef]
- Morimoto, Y.; Schembre, S.M.; Steinbrecher, A.; Erber, E.; Pagano, I.; Grandinetti, A.; Kolonel, L.N.; Maskarinec, G. Ethnic differences in weight gain and diabetes risk: The Multiethnic Cohort Study. Eur. PMC 2010, 37, 230–236. [Google Scholar] [CrossRef]
- Lim, U.; Monroe, K.R.; Buchthal, S.; Fan, B.; Cheng, I.; Kristal, B.S.; Lampe, J.W.; Hullar, M.A.; Franke, A.A.; Stram, D.O.; et al. Propensity for intra-abdominal and hepatic adiposity varies among ethnic groups. Gastroenterology 2019, 156, 966–975. [Google Scholar] [CrossRef] [PubMed]
- Setiawan, V.W.; Lim, U.; Lipworth, L.; Lu, S.C.; Shepherd, J.; Ernst, T.; Wilkens, L.R.; Henderson, B.E.; Le Marchand, L. Sex and ethnic differences in the association of obesity with risk of hepatocellular carcinoma. Clin. Gastroenterol. Hepatol. 2016, 14, 309–316. [Google Scholar] [CrossRef] [PubMed]
- Nazare, J.; Smith, J.D.; Borel, A.; Haffner, S.M.; Balkau, B.; Ross, R.; Massien, C.; Almeras, N.; Despres, J. Ethnic influences on the relations between abdominal subcutaneous and visceral adiposity, liver fat, and cardiometabolic risk profile: The International Study of Prediction of Intra-Abdominal Adiposity and Its Relationship With Cardiometabolic Risk/Intra-Abdominal Adiposity. Am. J. Clin. Nutr. 2012, 96, 714–726. [Google Scholar] [PubMed]
- Kadowaki, T.; Sekikawa, A.; Murata, K.; Maegawa, H.; Takamiya, T.; Okamura, T.; El-Saed, A.; Miyamatsu, N.; Edmundowicz, D.; Kita, Y.; et al. Japanese men have larger areas of visceral adipose tissue than Caucasian men in the same levels of waist circumference in a population-based study. Int. J. Obes. 2006, 30, 1163–1165. [Google Scholar] [CrossRef] [PubMed]
- Chaston, T.B.; Dixon, J.B. Factors associated with percent change in visceral versus subcutaneous abdominal fat during weight loss: Findings from a systematic review. Int. J. Obes. 2008, 32, 619–628. [Google Scholar] [CrossRef] [PubMed]
- Middleton, K.R.; Anton, S.D.; Perri, M.G. Long-term adherence to health behavior change. Am. J. Lifestyle Med. 2013, 7, 395–404. [Google Scholar] [CrossRef] [PubMed]
- Harvie, M.; Howell, A. Potential benefits and harms of intermittent energy restriction and intermittent fasting amongst obese, overweight and normal weight subjects -- A narrative review of human and animal evidence. Behav. Sci. 2017, 7, 4. [Google Scholar] [CrossRef]
- Harvey, J.; Howell, A.; Morris, J.; Harvie, M. Intermittent energy restriction for weight loss: Spontaneous reduction of energy intake on unrestricted days. Food. Sci. Nutr. 2018, 6, 674–680. [Google Scholar] [CrossRef]
- Harris, L.; Hamilton, S.; Azevedo, L.B.; Olajide, J.; De Brun, C.; Waller, G.; Whittaker, V.; Sharp, T.; Lean, M.; Hankey, C.; et al. Intermittent fasting interventions for the treatment of overweight and obesity in adults: A systematic review and meta-analysis. JBI Database Syst. Rev. Implement. Rep. 2018, 16, 507–547. [Google Scholar] [CrossRef]
- Ells, L.J.; Atkinson, G.; McGowan, V.J.; Hamilton, S.; Waller, G.; Harrison, S. Intermittent fasting interventions for the treatment of overweight and obesity in adults aged 18 years and over: A systematic review protocol. JBI Database Syst. Rev. Implement. Rep. 2015, 13, 60–68. [Google Scholar]
- Harris, L.; McGarty, A.; Hutchinson, L.; Ells, L.; Hankey, C. Short-term intermittent energy restriction interventions for weight management: A systematic review and metaanalysis. Obes. Rev. 2018, 19, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Harvie, M.; Wright, C.; Pegington, M.; McMullan, D.; Mitchell, E.; Martin, B.; Cutler, R.G.; Evans, G.; Whiteside, S.; Maudsley, S.; et al. The effect of intermittent energy and carbohydrate restriction v. daily energy restriction on weight loss and metabolic disease risk markers in overweight women. Br. J. Nutr. 2013, 110, 1534–1547. [Google Scholar] [CrossRef] [PubMed]
- Dernini, S.; Berry, E.M. Mediterranean diet: From a healthy diet to a sustainable dietary pattern. Front. Nutr. 2015, 2, 15. [Google Scholar] [CrossRef] [PubMed]
- Trichopoulou, A.; Lagiou, P. Healthy traditional Mediterranean diet: An expression of culture, history, and lifestyle. Nutr. Rev. 1997, 55, 383–389. [Google Scholar] [CrossRef] [PubMed]
- Willett, W.C.; Sacks, F.; Trichopoulou, A.; Drescher, G.; Ferro-Luzzi, A.; Helsing, E.; Trichopoulos, D. Mediterranean diet pyramid: A cultural model for healthy eating. Am. J. Clin. Nutr. 1995, 61, 1402S–1406S. [Google Scholar] [CrossRef] [PubMed]
- European Association for the Study of the Liver (EASL); European Association for the Study of Diabetes (EASD); European Association for the Study of Obesity (EASO). EASL-EASD-EASO Clinical practice guidelines for the management of non-alcoholic fatty liver disease. Obes. Facts 2016, 9, 65–90. [Google Scholar] [CrossRef] [PubMed]
- Maskarinec, G.; Lim, U.; Jacobs, S.; Monroe, K.R.; Ernst, T.; Buchthal, S.D.; Shepherd, J.A.; Wilkens, L.R.; LeMarchand, L.; Boushey, C.J. Diet quality in midadulthood predicts visceral adiposity and liver fatness in older ages: The Multiethnic Cohort Study. Obesity 2017, 25, 1442–1450. [Google Scholar] [CrossRef] [PubMed]
- Six, B.L.; Schap, T.E.; Zhu, F.M.; Mariappan, A.; Bosch, M.; Delp, E.J.; Ebert, D.S.; Kerr, D.A.; Boushey, C.J. Evidence-based development of a mobile telephone food record. J. Am. Diet. Assoc. 2010, 110, 74–79. [Google Scholar] [CrossRef]
- Kerr, D.A.; Pollard, C.M.; Howat, P.; Delp, E.J.; Pickering, M.; Kerr, K.R.; Dhaliwal, S.S.; Pratt, I.S.; Wright, J.; Boushey, C.J. Connecting Health and Technology (CHAT): Protocol of a randomized controlled trial to improve nutrition behaviours using mobile devices and tailored text messaging in young adults. BMC Public Health 2012, 12, 477. [Google Scholar] [CrossRef]
- Daugherty, B.L.; Schap, T.E.; Ettienne-Gittens, R.; Zhu, F.M.; Bosch, M.; Delp, E.J.; Ebert, D.S.; Kerr, D.A.; Boushey, C.J. Novel technologies for assessing dietary intake: Evaluating the usability of a mobile telephone food record among adults and adolescents. J. Med. Internet Res. 2012, 14, e58. [Google Scholar] [CrossRef]
- Boushey, C.J.; Spoden, M.; Delp, E.J.; Zhu, F.; Bosch, M.; Ahmad, Z.; Shvetsov, Y.B.; DeLany, J.P.; Kerr, D.A. Reported energy intake accuracy compared to doubly labeled water and usability of the mobile food record among community dwelling adults. Nutrients 2017, 9, 312. [Google Scholar] [CrossRef] [PubMed]
- Albright, C.L.; Steffen, A.; Wilkens, L.R.; White, K.K.; Novotny, R.; Nigg, C.; Saiki, K.; Brown, W.J. The randomization process. In Fundamentals of Clinical Trials, 3rd ed.; Friedman, L.S., Furberg, C.D., Demets, D.L., Eds.; Springer Science + Business Media, LLC: New York, NY, USA, 1998; ISBN 0-387-98586-7. [Google Scholar]
- Cha, S.; Lim, S.; Kim, K.; Lee, E.; Kang, B.; Choi, Y.; Yoon, K.; Ahn, Y.; Lee, J.; Ko, S. Community-based randomized controlled trial of diabetes prevention study for high-risk individuals of type 2 diabetes: Lifestyle intervention using web-based system. BMC Public Health 2017, 17, 387. [Google Scholar] [CrossRef] [PubMed]
- Lund, J.L.; Richardson, D.B.; Sturmer, T. The active comparator, new user study design in pharmacoepidemiology: Historical foundations and contemporary application. Curr. Epidemiol. Rep. 2015, 2, 221–228. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, K.; Solomon, D.H.; Kim, S.C. Active-comparator design and new-user design in observational studies. Nat. Rev. Rheumatol. 2015, 11, 437–441. [Google Scholar] [CrossRef] [PubMed]
- Sacks, F.M.; Svetkey, L.P.; Vollmer, W.M.; Appel, L.J.; Bray, G.A.; Harsha, D.; Obarzanek, E.; Conlin, P.R.; Miller, E.R.; Simons-Morton, D.G.; et al. DASH-Sodium Collaborative Research Group. Effects on blood pressure of reduced dietary sodium and the Dietary Approaches to Stop Hypertension (DASH) diet. New Eng. J. Med. 2001, 344, 3–10. [Google Scholar] [CrossRef] [PubMed]
- Appel, L.J.; Moore, T.J.; Obarzanek, E.; Vollmer, W.M.; Svetkey, L.P.; Sacks, F.M.; Bray, G.A.; Vogt, T.M.; Cutler, J.A.; Windhauser, M.M.; et al. DASH Collaborative Research Group. A clinical trial of the effects of dietary patterns on blood pressure. New Engl. J. Med. 1997, 336, 1117–1124. [Google Scholar] [CrossRef] [PubMed]
- Albright, C.L.; Steffen, A.D.; Wilkens, L.R.; White, K.K.; Novotny, R.; Nigg, C.R.; Saiki, K.; Brown, W.J. Effectiveness of a 12-month randomized clinical trial to increase physical activity in multiethnic postpartum women: Results from Hawaii’s Na Mikimiki Project. Prev. Med. 2014, 69, 214–223. [Google Scholar] [CrossRef] [PubMed]
- Institute of Medicine. Dietary Reference Intakes for Energy, Carbohydrate, Fiber, Fat, Fatty Acids, Cholesterol, Protein, Amino Acids (Macronutrients), 2nd ed.; National Academies Press: Washington, DC, USA, 2005; ISBN 0-309-08525. [Google Scholar]
- DRI Calculator for Healthcare Professionals. Available online: https://fnic.nal.usda.gov/fnic/dri-calculator/ (accessed on 6 January 2018).
- Harvie, M.N.; Pegington, M.; Mattson, M.P.; Frystyk, J.; Dillon, B.; Evans, G.; Cuzick, J.; Jebb, S.A.; Martin, B.; Cutler, R.G.; et al. The effects of intermittent or continuous energy restriction on weight loss and metabolic disease risk markers: A randomized trial in young overweight women. Int. J. Obes. 2011, 35, 714–727. [Google Scholar] [CrossRef]
- Resnicow, K.; Jackson, A.; Wang, T.; De, A.K.; McCarty, F.; Dudley, W.N.; Baranowski, T. A motivational interviewing intervention to increase fruit and vegetable intake through black churches: Results of the Eat for Life trial. Am. J. Public Health 2001, 91, 1686–1693. [Google Scholar] [CrossRef]
- Resnicow, K.; Campbell, M.K.; Carr, C.; McCarty, F.; Wang, T.; Periasamy, S.; Rahotep, S.; Doyle, C.; Williams, A.; Stables, G. Body and soul A dietary intervention conducted through African-American churches. Am. J. Prev. Med. 2004, 27, 97–105. [Google Scholar] [CrossRef]
- Resnicow, K.; Jackson, A.; Blissett, D.; Wang, T.; McCarty, F.; Rahotep, S.; Periasamy, S. Results of the healthy body healthy spirit trial. Health Psychol. 2005, 24, 339–348. [Google Scholar] [CrossRef] [PubMed]
- Fialkowski, M.K.; McCrory, M.A.; Roberts, S.M.; Tracy, J.K.; Grattan, L.M.; Boushey, C.J. Evaluation of dietary assessment tools used to assess the diet of adults participating in the Communities Advancing the Studies of Tribal Nations Across the Lifespan (CoASTAL) cohort. J. Am. Diet. Assoc. 2010, 110, 65–73. [Google Scholar] [CrossRef] [PubMed]
- Novotny, R.; Nigg, C.; McGlone, K.; Renda, G.; Jung, N.; Matsunaga, M.; Karanja, N. Pacific Tracker 2—Expert System (PacTrac2-ES) behavioural assessment and intervention tool for the Pacific Kids DASH for Health (PacDASH) study. Food Chem. 2013, 140, 471–477. [Google Scholar] [CrossRef] [PubMed]
- Bathgate, K.E.; Sherriff, J.L.; Leonard, H.; Dhaliwal, S.S.; Delp, E.J.; Boushey, C.J.; Kerr, D.A. Feasibility of assessing diet with a mobile food record for adolescents and young adults with Down Syndrome. Nutrients 2017, 9, 273. [Google Scholar] [CrossRef] [PubMed]
- Murphy, S.P. Unique nutrition support for research at the Cancer Research Center of Hawaii. Hawaii Med. J. 2002, 61, 15. [Google Scholar] [PubMed]
- Murphy, S.P.; Martin, C.L.; Davison, N.; Cheung, L.W.; Au, D.L.; Novotny, R. A comparison of two systems for entering and assessing dietary data for a research study. J. Am. Diet. Assoc. 2009, 109, 905–908. [Google Scholar] [CrossRef]
- Howes, E.; Boushey, C.J.; Kerr, D.A.; Tomayko, E.J.; Cluskey, M. Image-based dietary assessment ability of dietetics students and interns. Nutrients 2017, 9, 114. [Google Scholar] [CrossRef] [PubMed]
- Shepherd, J.; Ng, B.; Sommer, M.; Heymsfield, S.B. Body composition by DXA. Bone 2017, 104, 101–105. [Google Scholar] [CrossRef] [PubMed]
- Kelly, T.L.; Wilson, K.E.; Heymsfield, S.B. Dual Energy X-Ray Absorptiometry Body Composition Reference Values from NHANES. PLoS ONE 2009, 4, e7038. [Google Scholar] [CrossRef]
- Iannuzzi-Sucic, M.; Prestwood, K.M.; Kenny, A.M. Prevalence of Sarcopenia and Predictors of Skeletal Muscle Mass in Healthy, Older Men and Women. J. Gerontol. 2002, 57, M772–M777. [Google Scholar] [CrossRef]
- Kolonel, L.N.; Henderson, B.E.; Hankin, J.H.; Nomura, A.M.Y.; Wilkens, L.R.; Pike, M.C.; Stram, D.O.; Monroe, K.R.; Earle, M.E.; Nagamine, F.S. A multiethnic cohort in Hawaii and Los Angeles: Baseline characteristics. Am. J. Epidemiol. 2000, 151, 346–357. [Google Scholar] [CrossRef] [PubMed]
- Schubel, R.; Nattenmuller, J.; Sookthai, D.; Nonnenmacher, T.; Graf, M.E.; Riedl, L.; Schlett, C.L.; von Stackelberg, O.; Johnson, T.; Nabers, D.; et al. Effects of intermittent and continuous calorie restriction on body weight and metabolism over 50 wk: A randomized controlled trial. Am. J. Clin. Nutr. 2018, 108, 933–945. [Google Scholar] [CrossRef] [PubMed]
- Baraldi, A.N.; Enders, C.K. An introduction to modern missing data analyses. J. Sch. Psychol. 2010, 48, 5–37. [Google Scholar] [CrossRef] [PubMed]
- National Institutes of Health: National Heart, Lung, and Blood Institute. Managing Overweight and Obesity in Adults: Systematic Evidence Review from the Obesity Expert Panel. Available online: https://www.nhlbi.nih.gov/health-topics/managing-overweight-obesity-in-adults (accessed on 4 April 2019).
- National Institutes of Health: National Heart, Lung, and Blood Institute. Clinical Guidelines on the Identification, Evaluation, and Treatment of Overweight and Obesity in Adults: The Evidence Report. Available online: https://www.nhlbi.nih.gov/files/docs/guidelines/ob_gdlns.pdf. (accessed on 4 April 2019).
- Liu, Z.; Que, S.; Xu, J.; Peng, T. Alanine aminotransferase-old biomarker and new concept: A review. Int. J. Med. Sci. 2014, 11, 925–935. [Google Scholar] [CrossRef] [PubMed]
- Conley, M.; Le Fevre, L.; Haywood, C.; Proietto, J. Is two days of intermittent energy restriction per week a feasible weight loss approach in obese males? A randomised pilot study. Nutr. Diet. 2017, 75, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Carter, S.; Clifton, P.M.; Keogh, J.B. Effect of intermittent compared with continuous energy restricted diet on glycemic control in patients with type 2 diabetes: A randomized noninferiority trial. JAMA Netw. Open 2018, 1, e180756. [Google Scholar] [CrossRef] [PubMed]
- Hall, K.D.; Ayuketah, A.; Brychta, R.; Walter, P.J.; Yang, S.; Zhou, M. Ultra-processed diets cause excess calorie intake and weight gain: An inpatient randomized controlled trial of ad libitum food intake. Cell Metab. 2019, 30, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Hartmann, C.; Dohle, S.; Siegrist, M. Importance of cooking skills for balanced food choices. Appetite 2013, 65, 125–131. [Google Scholar] [CrossRef] [PubMed]
- Hsu, C.C.; Ness, E.; Kowdley, K.V. Nutritional approaches to achieve weight loss in nonalcoholic fatty liver disease. Adv. Nutr. 2017, 8, 253–265. [Google Scholar] [CrossRef] [PubMed]
- Boushey, C.J.; Spoden, M.; Zhu, F.M.; Delp, E.J.; Kerr, D.A. New mobile methods for dietary assessment: Review of image-assisted and image-based dietary assessment methods. Proc. Nutr. Soc. 2016, 76, 283–294. [Google Scholar] [CrossRef]
- Bartholome, L.T.; Peterson, R.E.; Raatz, S.K.; Raymond, N.C. A comparison of the accuracy of self-reported intake with measured intake of a laboratory overeating episode in overweight and obese women with and without binge eating disorder. Eur. J. Nutr. 2013, 52, 193–202. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Karelis, A.D.; Lavoie, M.; Fontaine, J.; Messier, V.; Strychar, I.; Rabasa-Lhoret, R.; Doucet, E. Anthropometric, metabolic, dietary and psychosocial profiles of underreporters of energy intake: A doubly labeled water study among overweight/obese postmenopausal women–A Montreal Ottawa New Emerging Team study. Eur. J. Clin. Nutr. 2010, 64, 68–74. [Google Scholar] [CrossRef] [PubMed]
- Mohammad, A.; De Lucia Rolfe, E.; Sleigh, A.; Kivisild, T.; Behbehani, K.; Wareham, N.J.; Brage, S.; Mohammad, T. Validity of visceral adiposity estimates from DXA against MRI in Kuwaiti men and women. Nutr. Diabetes 2017, 7, e238. [Google Scholar] [CrossRef] [PubMed]
- Kaul, S.; Rothney, M.P.; Peters, D.M.; Wacker, W.K.; Davis, C.E.; Shapiro, M.D.; Ergun, D.L. Dual-energy X-Ray Absorptiometry for quantification of visceral fat. Obesity 2012, 20, 1313–1318. [Google Scholar] [CrossRef] [PubMed]
- Lin, H.; Yan, H.; Rao, S.; Xia, M.; Zhou, Q.; Xu, H.; Rothney, M.P.; Xia, Y.; Wacker, W.K.; Ergun, D.L.; et al. Quantification of visceral adipose tissue using lunar dual-energy X-ray absorptiometry in Asian Chinese. Obesity 2013, 21, 2112–2117. [Google Scholar] [CrossRef] [PubMed]
| Variable | Randomization Group | |
|---|---|---|
| IER+MED (n = 30) | DASH (n = 30) | |
| Men | 9 | 9 |
| Visceral adipose tissue category (%) | ||
| High (90 < 150 cm2) | 6 (67%) | 6 (67%) |
| Very high (≥ 150 cm2) | 3 (33%) | 3 (33%) |
| Women | 21 | 21 |
| Visceral adipose tissue category (%) | ||
| High (80 < 150 cm2) | 13 (62%) | 14 (67%) |
| Very high (≥ 150 cm2) | 8 (38) | 7 (33%) |
| Characteristics (Men and Women) | ||
| Age (years) | 48.4 ± 4.7 | 46.2 ± 5.4 |
| Height (m) | 1.6 ± 0.1 | 1.6 ± 0.1 |
| Weight (kg) | 79.3 ± 12.5 | 81.0 ± 12.5 |
| Visceral adipose tissue area (cm2) | 134.6 ± 6.4 | 135.3 ± 6.4 |
| Body mass index (kg/m2) | 30.5 ± 3.5 | 30.8 ± 3.3 |
| Moderate or vigorous physical activity (hours/day) | 1.6 ± 0.2 | 1.4 ± 0.3 |
| Ethnicity (%) | ||
| Chinese | 23.3 | 6.7 |
| Japanese | 56.7 | 63.3 |
| Korean | 10.0 | 13.3 |
| Mixed Asian | 10.0 | 16.7 |
| Variable | Baseline | Weeks 5–6 | p2 | Week 11 | p3 |
|---|---|---|---|---|---|
| Energy (kcal) | |||||
| IER+MED 4 | 1590 ± 078 | 1227 ± 085 | <0.0001 | 1155 ± 077 | <0.0001 |
| DASH | 1803 ± 111 | 1414 ± 096 | 0.001 | 1507 ± 100 | 0.001 |
| Protein (g) | |||||
| IER+MED | 73.0 ± 3.6 | 74.9 ± 5.0 | 0.692 | 72.7 ± 4.2 | 0.946 |
| DASH | 76.5 ± 4.6 | 66.1 ± 4.8 | 0.055 | 70.6 ± 4.7 | 0.128 |
| Protein (% energy) | |||||
| IER+MED | 18.7 ± 0.7 | 24.9 ± 1.0 | <0.0001 | 25.7 ± 1.1 | <0.0001 |
| DASH | 17.3 ± 0.6 | 18.7 ± 0.5 | 0.115 | 18.9 ± 0.5 | 0.011 |
| Carbohydrate (g) | |||||
| IER+MED | 180 ± 11 | 115 ± 10 | <0.0001 | 103 ± 09 | <0.0001 |
| DASH | 198 ± 13 | 160 ± 12 | 0.013 | 169 ± 12 | 0.008 |
| Carbohydrate (% energy) | |||||
| IER+MED | 44.8 ± 1.5 | 37.3 ± 1.6 | <0.0001 | 35.1 ± 1.6 | <0.0001 |
| DASH | 44.1 ± 1.2 | 44.8 ± 1.1 | 0.650 | 45.2 ± 1.3 | 0.418 |
| Total fat (g) | |||||
| IER+MED | 65 ± 4 | 53 ± 5 | 0.015 | 52 ± 4 | 0.001 |
| DASH | 78 ± 6 | 57 ± 4 | <0.0001 | 63 ± 5 | 0.001 |
| Total fat (% energy) | |||||
| IER+MED | 36.6 ± 1.1 | 39.1 ± 1.4 | 0.140 | 40.2 ± 1.2 | 0.014 |
| DASH | 38.3 ± 1.1 | 36.7 ± 1.1 | 0.183 | 36.8 ± 1.3 | 0.234 |
| Saturated fatty acids (% energy) | |||||
| IER+MED | 11.5 ± 0.4 | 11.1 ± 0.6 | 0.560 | 11.6 ± 0.6 | 0.854 |
| DASH | 11.7 ± 0.5 | 11.0 ± 0.4 | 0.092 | 11.8 ± 0.5 | 0.830 |
| Monounsaturated fatty acids (% energy) | |||||
| IER+MED | 13.6 ± 0.4 | 15.0 ± 0.6 | 0.055 | 15.9 ± 0.6 | 0.001 |
| DASH | 14.5 ± 0.5 | 13.6 ± 0.5 | 0.111 | 13.9 ± 0.6 | 0.357 |
| Polyunsaturated fatty acids (% energy) | |||||
| IER+MED | 8.4 ± 0.5 | 9.6 ± 0.5 | 0.093 | 8.8 ± 0.5 | 0.426 |
| DASH | 9.0 ± 0.5 | 9.0 ± 0.6 | 0.994 | 7.7 ± 0.4 | 0.093 |
| Dietary fiber (g) | |||||
| IER+MED | 13.0 ± 1.0 | 12.8 ± 0.9 | 0.874 | 11.9 ± 1.0 | 0.297 |
| DASH | 13.4 ± 1.1 | 14.1 ± 1.3 | 0.661 | 13.7 ± 1.0 | 0.814 |
| Variable | Baseline | Week 12 | p2 | Change | p3 |
|---|---|---|---|---|---|
| Weight (kg) | |||||
| IER+MED | 79.3 ± 2.2 | 73.4 ± 2.2 | <0.0001 | −5.9 ± 0.7 | 0.007 |
| DASH | 81.0 ± 2.2 | 77.8 ± 2.2 | <0.0001 | −3.3 ± 0.6 | |
| Body mass index (kg/m2) | |||||
| IER+MED | 30.5 ± 0.6 | 28.3 ± 0.6 | <0.0001 | −2.2 ± 0.2 | 0.002 |
| DASH | 30.8 ± 0.6 | 29.6 ± 0.6 | <0.0001 | −1.2 ± 0.2 | |
| Waist circumference (cm) | |||||
| IER+MED | 100.3 ± 1.6 | 93.3 ± 1.6 | <0.0001 | −6.9 ± 0.8 | 0.026 |
| DASH | 100.7 ± 1.6 | 96.2 ± 1.6 | <0.0001 | −4.5 ± 0.7 | |
| Hip circumference (cm) | |||||
| IER+MED | 107.7 ± 1.3 | 102.5 ± 1.3 | <0.0001 | −5.3 ± 0.5 | 0.021 |
| DASH | 107.3 ± 1.3 | 103.9 ± 1.3 | <0.0001 | −3.4 ± 0.5 | |
| Body fat (%) | |||||
| IER+MED | 33.4 ± 1.2 | 31.3 ± 1.2 | <0.0001 | −2.0 ± 0.4 | 0.021 |
| DASH | 33.0 ± 1.2 | 32.1 ± 1.2 | 0.023 | −0.8 ± 0.4 | |
| Fat mass (kg) | |||||
| IER+MED | 26.4 ± 1.1 | 23.1 ± 1.1 | <0.0001 | −3.3 ± 0.4 | 0.005 |
| DASH | 26.4 ± 1.1 | 24.9 ± 1.1 | <0.0001 | −1.6 ± 0.4 | |
| Muscle mass (kg) | |||||
| IER+MED | 21.9 ± 0.9 | 20.8 ± 0.9 | <0.0001 | −1.1 ± 0.2 | 0.013 |
| DASH | 22.3 ± 0.9 | 21.8 ± 0.9 | 0.005 | −0.5 ± 0.2 | |
| Total lean body mass (kg) | |||||
| IER+MED | 52.6 ± 1.8 | 50.4 ± 1.8 | <0.0001 | −2.3 ± 0.4 | 0.040 |
| DASH | 54.3 ± 1.8 | 53.1 ± 1.8 | 0.002 | −1.2 ± 0.4 | |
| Visceral adipose tissue area (cm2) | |||||
| IER+MED | 134.6 ± 6.4 | 112.0 ± 6.5 | <0.0001 | −22.6 ± 3.6 | 0.022 |
| DASH | 135.3 ± 6.4 | 124.5 ± 6.5 | 0.003 | −10.7 ± 3.5 | |
| Subcutaneous adipose tissue area (cm2) | |||||
| IER+MED | 373.1 ± 16.2 | 324.9 ± 16.4 | <0.0001 | −48.2 ± 6.4 | <0.0001 |
| DASH | 359.0 ± 16.2 | 344.0 ± 16.3 | 0.018 | −15.0 ± 6.1 | |
| VAT/SAT ratio 4 | |||||
| IER+MED | 0.38 ± 0.02 | 0.36 ± 0.02 | 0.157 | −0.01 ± 0.01 | 0.825 |
| DASH | 0.39 ± 0.02 | 0.37 ± 0.02 | 0.076 | −0.01 ± 0.01 |
| Variable | Baseline | Week 12 | p2 | Change | p3 |
|---|---|---|---|---|---|
| Cholesterol (mg/dL) | |||||
| IER+MED | 237.0 ± 10.3 | 219.5 ± 10.3 | 0.009 | −17.4 ± 6.4 | 0.356 |
| DASH | 250.0 ± 10.0 | 240.9 ± 10.0 | 0.149 | −9.1 ± 6.2 | |
| HDL cholesterol (mg/dL) | |||||
| IER+MED | 38.1 ± 2.4 | 39.6 ± 2.4 | 0.396 | 1.5 ± 1.8 | 0.610 |
| DASH | 32.1 ± 2.3 | 34.9 ± 2.3 | 0.110 | 2.8 ± 1.7 | |
| LDL cholesterol (mg/dL) | |||||
| IER+MED | 178.5 ± 9.2 | 164.5 ± 9.2 | 0.019 | −14.0 ± 5.8 | 0.585 |
| DASH | 188.6 ± 9.1 | 179.1 ± 9.1 | 0.104 | −9.5 ± 5.8 | |
| Triglycerides (mg/dL) | |||||
| IER+MED | 101.9 ± 26.2 | 77.1 ± 26.2 | 0.004 | −24.8 ± 8.2 | 0.809 |
| DASH | 165.5 ± 25.2 | 143.5 ± 25.2 | 0.008 | −22.0 ± 7.9 | |
| Alanine transaminase (U/L) | |||||
| IER+MED | 33.8 ± 3.2 | 20.1 ± 3.2 | 0.001 | −13.8 ± 3.7 | 0.038 |
| DASH | 19.5 ± 3.1 | 16.6 ± 3.1 | 0.419 | −2.9 ± 3.6 | |
| Glucose (mg/dL) | |||||
| IER+MED | 104.3 ± 4.2 | 102.2 ± 4.2 | 0.374 | −2.1 ± 2.4 | 0.928 |
| DASH | 104.9 ± 4.1 | 102.5 ± 4.1 | 0.294 | −2.4 ± 2.3 | |
| Aspartame transaminase (U/L) | |||||
| IER+MED | 23.8 ± 1.8 | 18.1 ± 1.8 | 0.012 | −5.7 ± 2.2 | 0.179 |
| DASH | 18.9 ± 1.7 | 17.3 ± 1.7 | 0.462 | −1.6 ± 2.1 | |
| Insulin (mU/L) | |||||
| IER+MED | 13.9 ± 1.7 | 8.8 ± 1.7 | <0.001 | −5.1 ± 1.2 | 0.134 |
| DASH | 14.6 ± 1.6 | 12.0 ± 1.6 | 0.027 | −2.5 ± 1.7 | |
| Systolic blood pressure (mm Hg) | |||||
| IER+MED | 133.2 ± 2.5 | 124.3 ± 2.7 | <0.001 | −9.0 ± 2.5 | 0.345 |
| DASH | 133.4 ± 2.5 | 127.7 ± 2.6 | 0.024 | −5.7 ± 2.4 | |
| Diastolic blood pressure (mm Hg) | |||||
| IER+MED | 84.2 ± 1.7 | 77.5 ± 1.8 | <0.001 | −6.7 ± 1.5 | 0.124 |
| DASH | 86.2 ± 1.7 | 82.8 ± 1.8 | 0.021 | −3.4 ± 1.4 |
| Variable | IER+MED | DASH |
|---|---|---|
| Weight (kg) | 75.5 ± 2.7 | 77.9 ± 3.2 |
| Change in weight between Week 12 1 and 6-month post-intervention (kg) 2 | 1.0 ± 3.8 | 1.1 ± 2.1 * |
| Physical activity (hours/week) | 3.8 ± 0.4 | 3.6 ± 0.6 |
| Able to follow the diet prescription for longer than 3 months (%) | ||
| No | 28.6 | 12.0 |
| Yes | 71.4 | 88.0 |
| If yes, for how many more months? | 5.0 ± 0.6 | 3.6 ± 0.5 |
| Still following the diet prescription (%) | ||
| No | 33.3 | 56.0 |
| Yes | 66.7 | 44.0 |
| If yes, following the diet the same, better, not as well, or other (%) | ||
| Same | 6.7 | 25.0 |
| Better | 0.0 | 0.0 |
| Not as well | 80.0 | 66.7 |
| Same and not as well | 13.3 | 0.0 |
| Other | 0.0 | 8.3 |
| Would nutrition classes and/or food preparation classes been helpful (%) | ||
| No | 13.6 | 24.0 |
| Yes | 86.4 | 76.0 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Panizza, C.E.; Lim, U.; Yonemori, K.M.; Cassel, K.D.; Wilkens, L.R.; Harvie, M.N.; Maskarinec, G.; Delp, E.J.; Lampe, J.W.; Shepherd, J.A.; et al. Effects of Intermittent Energy Restriction Combined with a Mediterranean Diet on Reducing Visceral Adiposity: A Randomized Active Comparator Pilot Study. Nutrients 2019, 11, 1386. https://doi.org/10.3390/nu11061386
Panizza CE, Lim U, Yonemori KM, Cassel KD, Wilkens LR, Harvie MN, Maskarinec G, Delp EJ, Lampe JW, Shepherd JA, et al. Effects of Intermittent Energy Restriction Combined with a Mediterranean Diet on Reducing Visceral Adiposity: A Randomized Active Comparator Pilot Study. Nutrients. 2019; 11(6):1386. https://doi.org/10.3390/nu11061386
Chicago/Turabian StylePanizza, Chloe E., Unhee Lim, Kim M. Yonemori, Kevin D. Cassel, Lynne R. Wilkens, Michelle N. Harvie, Gertraud Maskarinec, Edward J. Delp, Johanna W. Lampe, John A. Shepherd, and et al. 2019. "Effects of Intermittent Energy Restriction Combined with a Mediterranean Diet on Reducing Visceral Adiposity: A Randomized Active Comparator Pilot Study" Nutrients 11, no. 6: 1386. https://doi.org/10.3390/nu11061386
APA StylePanizza, C. E., Lim, U., Yonemori, K. M., Cassel, K. D., Wilkens, L. R., Harvie, M. N., Maskarinec, G., Delp, E. J., Lampe, J. W., Shepherd, J. A., Le Marchand, L., & Boushey, C. J. (2019). Effects of Intermittent Energy Restriction Combined with a Mediterranean Diet on Reducing Visceral Adiposity: A Randomized Active Comparator Pilot Study. Nutrients, 11(6), 1386. https://doi.org/10.3390/nu11061386





