Comparison of the Effectiveness of Lifestyle Modification with Other Treatments on the Incidence of Type 2 Diabetes in People at High Risk: A Network Meta-Analysis
Abstract
1. Introduction
2. Materials and Methods
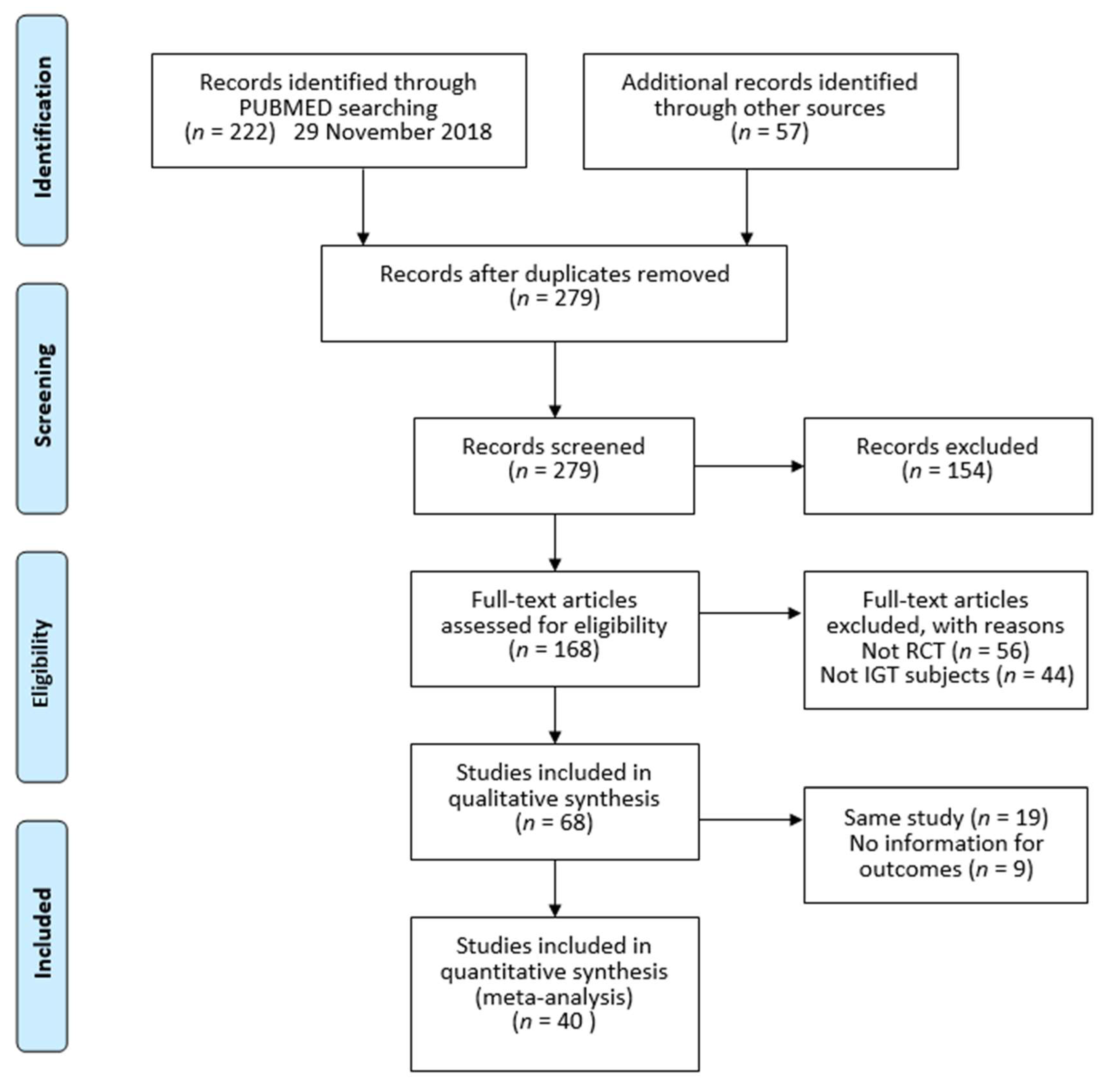
2.1. Study Design, Search Strategy, and Information Sources
2.2. Study Selection
2.3. Data Extraction and Risk of Bias within Individual Studies
2.4. Statistical Analysis
3. Results
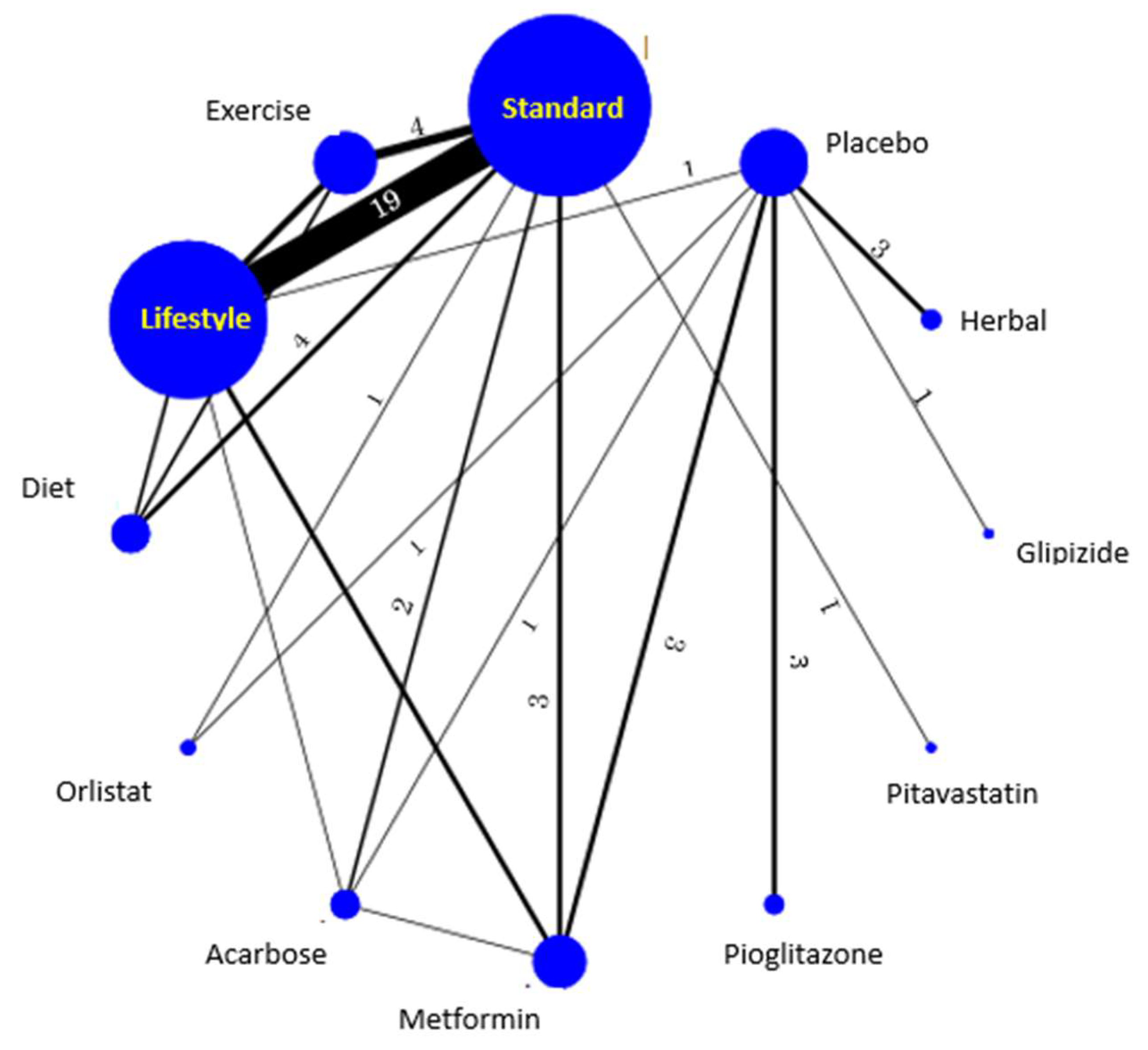
3.1. Type of Intervention
3.2. Risk of Bias
3.3. Odds Ratio with Onset of Type 2 Diabetes
4. Discussion
4.1. Findings in the Context of the Literature
4.2. Strengths and Limitations
4.3. Implications for Practice and Research
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- American Diabetes Association. Economic costs of diabetes in the U.S. in 2017. Diabetes Care 2018, 41, 917–928. [Google Scholar] [CrossRef] [PubMed]
- Kolb, H.; Martin, S. Environmental/lifestyle factors in the pathogenesis and prevention of type 2 diabetes. BMC Med. 2017, 15, 131. [Google Scholar] [CrossRef] [PubMed]
- Gillies, C.L.; Abrams, K.R.; Lambert, P.C.; Cooper, N.J.; Sutton, A.J.; Hsu, R.T.; Khunti, K. Parmaological and liestyle interventions to prevent or delay type 2 diabetes in people with impaired glucose tolerance: Systematic review and meta-analysis. BMJ 2007, 334, 299. [Google Scholar] [CrossRef] [PubMed]
- Haw, J.S.; Galaviz, K.I.; Straus, A.N.; Kowalski, A.J.; Magee, M.J.; Weber, M.B.; Wei, J.; Narayan, K.M.V.; Ali, M.D. Long-term Sustainability of Diabetes Prevention Approaches: A Systematic Review and Meta-analysis of Randomized Clinical Trials. JAMA Intern. Med. 2017, 177, 1808–1817. [Google Scholar] [CrossRef] [PubMed]
- Yeung, S.; Soliternik, J.; Mazzola, N. Nutritional supplements for the prevention of diabetes mellitus and its complications. J. Nutr. Intermed. Metab. 2018, 14, 16–21. [Google Scholar] [CrossRef]
- Aune, D.; Norat, T.; Leitzmann, M.; Tonstad, S.; Vatten, L.J. Physical activity and the risk of type 2 diabetes: A systematic review and dose-response meta-analysis. Eur. J. Epidemiol. 2015, 30, 529–542. [Google Scholar] [CrossRef] [PubMed]
- Pang, B.; Zhang, Y.; Liu, J.; He, L.; Zheng, Y.; Lian, F.; Tong, X. Prevention of Type 2 Diabetes with the Chinese Herbal Medicine Tianqi Capsule: A Systematic Review and Meta-Analysis. Diabetes Ther. 2017, 8, 1227–1242. [Google Scholar] [CrossRef]
- Chaudhury, A.; Duvoor, C.; Reddy, D.V.S.; Kraleti, S.; Chada, A.; Ravilla, R.; Marco, A.; Shekhawat, N.S.; Montales, M.T.; Kuriakose, K.; et al. Clinical Review of Antidiabetic Drugs: Implications for Type 2 Diabetes Mellitus Management. Clinical Review of Antidiabetic Drugs: Implications for Type 2 Diabetes Mellitus Management. Front. Endocrinol. 2017, 8, 6. [Google Scholar] [CrossRef]
- Yamaoka, K.; Tango, T. Efficacy of Lifestyle Education to Prevent Type 2 Diabetes: A meta-analysis of randomized controlled trials. Diabetes Care 2005, 28, 2780–2786. [Google Scholar] [CrossRef]
- Yamaoka, K.; Tango, T. Effects of Lifestyle Modification on Metabolic Syndrome: A Systematic Review and Meta-analysis. BMC Med. 2012, 10, 138. [Google Scholar] [CrossRef]
- White, I.R.; Barrett, J.K.; Jackson, D.; Higgins, J.P. Consistency and inconsistency in network meta-analysis: Model estimation using multivariate meta-regression. Res. Synth. Methods 2012, 3, 111–125. [Google Scholar] [CrossRef] [PubMed]
- Li, T.; Puhan, M.A.; Vedula, S.S.; Singh, S.; Dickersin, K. The Ad Hoc Network Meta-analysis Methods Meeting Working Group. Network meta-analysis-highly attractive but more methodological research is needed. BMC Med. 2011, 9, 79. [Google Scholar] [CrossRef] [PubMed]
- Riley, R.D.; Jackson, D.; Salanti, G.; Burke, D.L.; Price, M.; Kirkham, J.; White, I.R. Multivariate and network meta-analysis of multiple outcomes and multiple treatments: Rationale, concepts, and examples. BMJ 2017, 358, j3932. [Google Scholar] [CrossRef] [PubMed]
- Higgins, J.P.T.; Green, S. Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0 [updated March 2011]; Higgins, J.P.T., Green, S., Eds.; The Cochrane Collaboration: London, UK, 2011; Available online: http://handbook.cochrane.org (accessed on 27 March 2019).
- White, I.R. Multivariate random-effects meta-regression: Updates to mvmeta. Stata J. 2011, 11, 255–270. [Google Scholar] [CrossRef]
- Higgins, J.P.T.; Jackson, D.; Barrett, J.K.; Lu, G.; Ades, A.E.; White, I.R. Consistency and inconsistency in network meta-analysis: Concepts and models for multi-arm studies. Res. Synth. Methods 2012, 3, 98–110. [Google Scholar] [CrossRef] [PubMed]
- Duval, S.J.; Tweedie, R.L. Trim and Fill: A Simple Funnel-Plot-Based Method of Testing and Adjusting for Publication Bias in Meta-Analysis. Biometrics 2000, 56, 455–463. [Google Scholar] [CrossRef] [PubMed]
- Duval, S.J.; Tweedie, R.L. A nonparametric “trim and fill” method of assessing publication bias in meta-analysis. J. Am. Stat. Assoc. 2000, 95, 89–98. [Google Scholar]
- Salanti, G.; Ades, A.E.; Ioannidis, J.P. Graphical methods and numerical summaries for presenting results from multiple-treatment meta-analysis: An overview and tutorial. J. Clin. Epidemiol. 2011, 64, 163–171. [Google Scholar] [CrossRef] [PubMed]
- Hutton, B.; Salanti, G.; Caldwell, D.M.; Chaimani, A.; Schmid, C.H.; Cameron, C.; Ioannidis, J.P.; Straus, S.; Thorlund, K.; Jansen, J.P.; et al. The PRISMA Extension Statement for Reporting of Systematic Reviews Incorporating Network Meta-analyses of Health Care Interventions: Checklist and Explanations. Ann. Intern. Med. 2015, 162, 777–784. [Google Scholar] [CrossRef]
- Li, C.L.; Pan, C.Y.; Lu, J.M.; Zhu, Y.; Wang, J.H.; Deng, X.X.; Xia, F.C.; Wang, H.Z.; Wang, H.Y. Effect of metformin on patients with impaired glucose tolerance. Diabet. Med. 1999, 16, 477–481. [Google Scholar] [CrossRef]
- Watanabe, M.; Yamaoka, K.; Yokotsuka, M.; Tango, T. Randomized controlled trial of a new dietary education program to prevent type 2 diabetes in a high-risk group of Japanese male workers. Diabetes Care 2003, 26, 3209–3214. [Google Scholar] [CrossRef] [PubMed]
- Fan, G.J.; Luo, G.B.; Qin, M.L. Effect of jiangtang bushen recipe in intervention treatment of patients with impaired glucose tolerance. Zhongguo Zhong Xi Yi Jie He Za Zhi 2004, 24, 317–320. (In Chinese) [Google Scholar] [PubMed]
- Yates, T.; Davies, M.; Gorely, T.; Bull, F.; Khunti, K. Effectiveness of a pragmatic education program designed to promote walking activity in individuals with impaired glucose tolerance: A randomized controlled trial. Diabetes Care 2009, 32, 1404–1410. [Google Scholar] [CrossRef] [PubMed]
- Gagnon, C.; Brown, C.; Couture, C.; Kamga-Ngande, C.N.; Hivert, M.F.; Baillargeon, J.P.; Carpentier, M.F.; Langlois, M.F. A cost-effective moderate-intensity interdisciplinary weight-management programme for individuals with prediabetes. Diabetes Metab. 2011, 37, 410–418. [Google Scholar] [CrossRef]
- Zhou, J. Life style interventions study on the effects of impaired glucose regulations in Shanghai urban communities. Wei Sheng Yan Jiu 2011, 40, 331–333. (In Chinese) [Google Scholar]
- Xu, D.F.; Sun, J.Q.; Chen, M.; Chen, Y.Q.; Xie, H.; Sun, W.J.; Lin, Y.F.; Jiang, J.J.; Sun, W.; Chen, A.F.; et al. Effects of lifestyle intervention and meal replacement on glycaemic and body-weight control in Chinese subjects with impaired glucose regulation: A 1-year randomised controlled trial. Br. J. Nutr. 2013, 109, 487–494. [Google Scholar] [CrossRef]
- Fang, Z.; Zhao, J.; Shi, G.; Shu, Y.; Ni, Y.; Wang, H.; Ding, L.; Lu, R.; Li, J.; Zhu, X.; et al. Shenzhu Tiaopi granule combined with lifestyle intervention therapy for impaired glucose tolerance: A randomized controlled trial. Complement. Ther. Med. 2014, 22, 842–850. [Google Scholar] [CrossRef]
- Lian, F.; Li, G.; Chen, X.; Wang, X.; Piao, C.; Wang, J.; Hong, Y.; Ba, A.; Wu, S.; Zhou, X.; et al. Chinese herbal medicine Tianqi reduces progression from impaired glucose tolerance to diabetes: A double-blind, randomized, placebo-controlled, multicenter trial. J. Clin. Endocrinol. Metab. 2014, 99, 648–655. [Google Scholar] [CrossRef]
- Pan, X.R.; Li, G.W.; Hu, Y.H.; Wang, J.X.; Yang, W.Y.; An, Z.X.; Hu, Z.X.; Lin, J.; Xiao, J.Z.; Cao, H.B.; et al. Effects of diet and exercise in preventing NIDDM in people with impaired glucose tolerance, The Da Qing IGT and Diabetes Study. Diabetes Care 1997, 20, 537–544. [Google Scholar] [CrossRef]
- Wein, P.; Beischer, N.; Harris, C.; Permezel, M. Trial of Simple versus Intensified Dietary Modification for Prevention of Progression to Diabetes Mellitus in Women with Impaired Glucose Tolerance. Aust. N. Z. J. Obstet. Gynaecol. 1999, 39, 162–166. [Google Scholar] [CrossRef]
- Heymsfield, S.B.; Segal, K.R.; Hauptman, J.; Lucas, C.P.; Boldrin, M.N.; Rissanen, A.; Wilding, J.P.; Sjöström, L. Effects of weight loss with orlistat on glucose tolerance and progression to type 2 diabetes in obese adults. Arch. Intern. Med. 2000, 160, 1321–1326. [Google Scholar] [CrossRef] [PubMed]
- Lindström, J.; Louheranta, A.; Mannelin, M.; Rastas, M.; Salminen, V.; Eriksson, J.; Uusitupa, M.; Tuomilehto, J.; the Finnish Diabetes Prevention Study Group. The Finnish Diabetes Prevention Study (DPS): Lifestyle intervention and 3-year results on diet and physical activity. Diabetes Care 2003, 26, 3230–3236. [Google Scholar]
- Chiasson, J.L.; Josse, R.G.; Gomis, R.; Gomis, R.; Hanefeld, M.; Karasik, A.; Laakso, M.; The STOP-NIDDM Trial Research Group. Acarbose for prevention of type 2 diabetes mellitus: The STOP-NIDDM randomised trial. Lancet 2002, 359, 2072–2077. [Google Scholar] [CrossRef]
- Knowler, W.C.; Barrett-Connor, E.; Fowler, S.E.; Hamman, R.F.; Lachin, J.M.; Walker, E.A.; Nathan, D.M. Reduction in the incidence of type 2 diabetes with lifestyle intervention or metformin. N. Engl. J. Med. 2002, 346, 393–403. [Google Scholar] [PubMed]
- Liao, D.; Asberry, P.J.; Shofer, J.B.; Callahan, H.; Matthys, C.; Boyko, E.J.; Leonetti, D.; Kahn, S.E.; Austin, M.; Newell, L.; et al. Improvement of BMI, body composition, and body fat distribution with lifestyle modification in Japanese Americans with impaired glucose tolerance. Diabetes Care 2002, 25, 1504–1510. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Tao, L.L.; Deng, Y.B.; Fan, X.B.; Bao, Q.D. Effect of exercise training in patients with impaired glucose tolerance. Chin. J. Clin. Rehabil. 2004, 8, 2912–2913. (In Chinese) [Google Scholar]
- Torgerson, J.S.; Hauptman, J.; Boldrin, M.N.; Sjöström, L. XENical in the prevention of diabetes in obese subjects (XENDOS) study: A randomized study of orlistat as an adjunct to lifestyle changes for the prevention of type 2 diabetes in obese patients. Diabetes Care 2004, 27, 155–161. [Google Scholar] [CrossRef]
- Kosaka, K.; Noda, M.; Kuzuya, T. Prevention of type 2 diabetes by lifestyle intervention: A Japanese trial in IGT males. Diabetes Res. Clin. Pract. 2005, 67, 152–162. [Google Scholar] [CrossRef]
- Oldroyd, J.C.; Unwin, N.C.; White, M.; Imrie, K.; Mathers, J.C.; Alberti, K.G.M.M. Randomised controlled trial evaluating the effectiveness of behavioural interventions to modify cardiovascular risk factors in men and women with impaired glucose tolerance: Outcomes at 6 months. Diabetes Res. Clin. Pract. 2001, 52, 29–43. [Google Scholar] [CrossRef]
- DREAM Trial Investigators. Effect of ramipril on the incidence of diabetes. N. Engl. J. Med. 2006, 355, 1551–1562. [Google Scholar] [CrossRef]
- Eriksson, J.G.; Lehtovirta, M.; Ehrnström, B.; Salmela, S.; Groop, L. Long-term beneficial effects of glipizide treatment on glucose tolerance in subjects with impaired glucose tolerance. J. Intern. Med. 2006, 259, 553–560. [Google Scholar] [CrossRef] [PubMed]
- Ramachandran, A.; Snehalatha, C.; Mary, S.; Mukesh, B.; Bhaskar, A.D.; Vijay, V.; Indian Diabetes Prevention Programme (IDPP). The Indian Diabetes Prevention Programme shows that lifestyle modification and metformin prevent type 2 diabetes in Asian Indian subjects with impaired glucose tolerance (IDPP-1). Diabetologia 2006, 49, 289–297. [Google Scholar] [CrossRef] [PubMed]
- Roumen, C.; Corpeleijn, E.; Feskens, E.J.; Mensink, M.; Saris, W.H.M.; Blaak, E. Impact of 3-year lifestyle intervention on postprandial glucose metabolism: The SLIM study. Diabet. Med. 2008, 25, 597–605. [Google Scholar] [CrossRef] [PubMed]
- Kawahara, T.; Takahashi, K.; Inazu, T.; Arao, T.; Kawahara, C.; Tabata, T.; Moriyama, H.; Okada, Y.; Morita, E.; Tanaka, Y. Reduced progression to type 2 diabetes from impaired glucose tolerance after a 2-day in-hospital diabetes educational program: The Joetsu Diabetes Prevention Trial. Diabetes Care 2008, 31, 1949–1954. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Kawamori, R.; Tajima, N.; Iwamoto, Y.; Kashiwagi, A.; Shimamoto, K.; Kaku, K. Voglibose for prevention of type 2 diabetes mellitus: A randomised, double-blind trial in Japanese individuals with impaired glucose tolerance. Lancet 2009, 73, 1607–1614. [Google Scholar] [CrossRef]
- Penn, L.; White, M.; Oldroyd, J.; Walker, K.; Alberti, G.M.M.; Mathers, J.C. Prevention of type 2 diabetes in adults with impaired glucose tolerance: The European Diabetes Prevention RCT in Newcastle upon Tyne, UK. BMC Public Health 2009, 9, 342. [Google Scholar] [CrossRef]
- Ramachandran, A.; Snehalatha, C.; Mary, S.; Selvam, S.; Kumar, C.K.; Seeli, A.C.; Shetty, A.S. Pioglitazone does not enhance the effectiveness of lifestyle modification in preventing conversion of impaired glucose tolerance to diabetes in Asian Indians: Results of the Indian Diabetes Prevention Programme-2 (IDPP-2). Diabetologia 2009, 52, 1019–1026. [Google Scholar] [CrossRef]
- Zinman, B.; Harris, S.B.; Neuman, J.; Gerstein, H.C.; Retnakaran, R.R.; Raboud, J.; Qi, Y.; Hanley, A.J.G. Low-dose combination therapy with rosiglitazone and metformin to prevent type 2 diabetes mellitus (CANOE trial): A double-blind randomised controlled study. Lancet 2010, 376, 103–111. [Google Scholar] [CrossRef]
- DeFronzo, R.A.; Tripathy, D.; Schwenke, D.C.; Banerji, M.A.; Bray, G.A.; Buchanan, T.A.; Clement, S.C.; Henry, R.R.; Hodis, H.N.; Kitabchi, A.E.; et al. Pioglitazone for diabetes prevention in impaired glucose tolerance. N. Engl. J. Med. 2011, 364, 1104–1115. [Google Scholar] [CrossRef]
- Lu, Y.H.; Lu, J.M.; Wang, S.Y.; LiZheng, R.P.; Tian, H.; Wang, X.L. Outcome of intensive integrated intervention in participants with impaired glucose regulation in China. Adv. Ther. 2011, 28, 511–519. [Google Scholar] [CrossRef]
- Salas-Salvadó, J.; Bulló, M.; Babio, N.; Martinez-Gonzalez, M.A.; Ibarrola-Jurado, N.; Basora, J.; Estruch, R.; Govas, M.I.; Corella, D.; Arós, F.; et al. Reduction in the incidence of type 2 diabetes with the Mediterranean diet: Results of the PREDIMED-Reus nutrition intervention randomized trial. Diabetes Care 2011, 34, 14–19. [Google Scholar] [CrossRef] [PubMed]
- Sakane, N.; Sato, J.; Tsushita, K.; Tsujii, S.; Kotani, K.; Tsuzaki, K.; Tominaga, M.; Kawazu, S.; Sato, Y.; Usui, T.; et al. Prevention of type 2 diabetes in a primary healthcare setting: Three-year results of lifestyle intervention in Japanese subjects with impaired glucose tolerance. BMC Public Health 2011, 11, 40. [Google Scholar] [CrossRef] [PubMed]
- Saito, T.; Watanabe, M.; Nishida, J.; Izumi, T.; Omura, M.; Takagi, T.; Fukunaga, R.; Bandai, Y.; Tajima, N.; Nakamura, Y.; et al. Lifestyle modification and prevention of type 2 diabetes in overweight Japanese with impaired fasting glucose levels: A randomized controlled trial. Arch. Intern. Med. 2011, 171, 1352–1360. [Google Scholar] [CrossRef] [PubMed]
- Ockene, I.S.; Tellez, T.L.; Rosal, M.C.; Reed, G.W.; Mordes, J.; Olendzki, B.C.; Handelman, G.; Nicolosi, R.; Ma, Y. Outcomes of a Latino community-based intervention for the prevention of diabetes: The Lawrence Latino Diabetes Prevention Project. Am. J. Public Health 2012, 102, 336–342. [Google Scholar] [CrossRef] [PubMed]
- Vermunt, P.W.; Milder, I.E.; Wielaard, F.; de Vries, J.J.M.; Baan, C.A.; van Oers, J.A.M.; Westert, G.P. A lifestyle intervention to reduce Type 2 diabetes risk in Dutch primary care: 2.5-year results of a randomized controlled trial. Diabet. Med. 2012, 29, e223–e231. [Google Scholar] [CrossRef]
- Odawara, M.; Yamazaki, T.; Kishimoto, J.; Noda, M.; Terauchi, Y.; Shiba, T.; Kitazato, J.; Maemura, K.; Tobe, K.; Iwamoto, Y.; et al. Effect of pitavastatin on the incidence of diabetes in Japanese individuals with impaired glucose tolerance. Diabetologia 2013, 56, S59. [Google Scholar]
- Wong, C.K.; Fung, C.S.; Siu, S.C.; Lo, Y.Y.C.; Wong, K.W.; Fong, D.Y.T.; Lam, C.L.K. A short message service (SMS) intervention to prevent diabetes in Chinese professional drivers with pre-diabetes: A pilot single-blinded randomized controlled trial. Diabetes Res. Clin. Pract. 2013, 102, 158–166. [Google Scholar] [CrossRef] [PubMed]
- Weber, M.B.; Ranjani, H.; Staimez, L.R.; Anjana, R.M.; Ali, M.K.; Venkat Narayan, K.M.; Mohan, V. The Stepwise Approach to Diabetes Prevention: Results From the D-CLIP Randomized Controlled Trial. Diabetes Care 2016, 39, 1760–1767. [Google Scholar] [CrossRef] [PubMed]
- Hemmingsen, B.; Gimenez-Perez, G.; Mauricio, D.; Roquéi Figuls, M.; Metzendorf, M.I.; Richter, B. Diet, physical activity or both for prevention or delay of type 2 diabetes mellitus and its associated complications in people at increased risk of developing type 2 diabetes mellitus. Cochrane Database Syst. Rev. 2017, 12. [Google Scholar] [CrossRef]
- Galaviz, K.I.; Weber, M.B.; Straus, A.; Haw, J.S.; Narayan, K.M.V.; Ali, M.K. Global Diabetes Prevention Interventions: A Systematic Review and Network Meta-analysis of the Real-World Impact on Incidence, Weight, and Glucose. Diabetes Care 2018, 41, 1526–1534. [Google Scholar] [CrossRef]
- Chiavaroli, L.; Viguiliouk, E.; Nishi, S.K.; Mejia, S.B.; Rahelic, D.; Kahleova, H.; Salas-Salvado, J.; Kendall, C.W.C.; Sievenpiper, J.L. DASH dietary pattern and cardiometabolic outcomes; an umbrella review of systematic reviews and meta-analyses. Nutrients 2019, 11, 338. [Google Scholar] [CrossRef] [PubMed]
- NICE. Type 2 Diabetes: Prevention in People at High Risk; Public Health Guideline [PH38]; Last updated: September 2017; NICE: London, UK, 2012; Available online: https://www.nice.org.uk/guidance/ph38 (accessed on 27 March 2019).
- CDC. The National Diabetes Prevention Program. National DPP Coverage Toolkit; CDC: Washington, DC, USA, 2019; Available online: https://coveragetoolkit.org/about-national-dpp/ (accessed on 27 March 2019).
- WHO. Diabetes Activities; WHO: Geneva, Switzerland, 2019; Available online: http://www.euro.who.int/en/health-topics/noncommunicable-diseases/diabetes/activities (accessed on 27 March 2019).
| Interventions | Pair-Wise | NMA (Frequentist Approach) (r = 0.5) | NMA (Bayesian Approach) $ | ||
|---|---|---|---|---|---|
| n# | OR (95% CI) | I2 | OR (95% CI) | OR (95% CI) | |
| Comparison for Lifestyle | |||||
| Lifestyle vs. Exercise (indirect) | 1.18 (0.74,1.93) | 1.57 (0.81,3.28) | |||
| Lifestyle vs. Diet (indirect) | 0.93 (0.60,1.43) | 1.01 (0.53,1.94) | |||
| Lifestylevs. Orlistat (indirect) | 1.19 (0.55,2.58) | 1.01 (0.37,2.77) | |||
| Lifestyle vs. Acarbose/Voglibose (indirect) | 0.92 (0.57,1.48) | 0.89 (0.44,1.84) | |||
| Lifestyle vs. Metformin/Flumamine (indirect) | 0.86 (0.60,1.25) | 0.96 (0.58,1.73) | |||
| Lifestyle vs. Pioglitazone, Rosiglitazone (indirect) | 0.57 (0.32,1.00) | 0.57 (0.26,1.36) | |||
| Lifestyle vs. Pitavastatin (indirect) | 0.68 (0.36,1.29) | 0.53 (0.20,1.37) | |||
| Lifestyle vs. Glipizide (indirect) | 2.07 (0.24,17.7) | 3.21 (0.31,99.3) | |||
| Lifestyle vs. Herbal medicine (indirect) | 0.71 (0.38,1.36) | 0.73 (0.29,1.87) | |||
| Lifestyle vs. Standard | 19 | 0.65 (0.56,0.75) | 4.7 | 0.60 (0.48,0.76) | 0.46 (0.33,0.61) |
| Lifestyle vs. Placebo | 1 | 0.44 (0.31,0.61) | 0.41 (0.27,0.63) | 0.38 (0.20,0.71) | |
| Comparison for Lifestyle | |||||
| Diet vs. Standard | 4 | 0.71 (0.55,0.90) | 35.7 | 0.65 (0.43,0.98) | 0.46 (0.24,0.84) |
| Exercise vs. Standard | 4 | 0.45 (0.24,0.84) | 29.9 | 0.50 (0.31,0.81) | 0.29 (0.14,0.55) |
| Orlistat vs. Standard | 1 | 0.49 (0.25,0.95) | 0.51 (0.24,1.07) | 0.45 (0.17,1.19) | |
| Acarbose/Voglibose vs. Standard | 2 | 0.70 (0.31,1.57) | 13.4 | 0.66 (0.42,1.04) | 0.51 (0.25,0.99) |
| Metformin/Flumamine vs. Standard | 3 | 0.40 (0.10,1.68) | 75.4 | 0.70 (0.47,1.03) | 0.48 (0.26,0.79) |
| Pioglitazone, Rosiglitazone vs. Standard (indirect) | 1.06 (0.60,1.90) | 0.80 (0.33,1.76) | |||
| Pitavastatin vs. Standard | 1 | 0.88 (0.65,1.20) | 0.88 (0.49,1.59) | 0.87 (0.34,2.17) | |
| Glipizide vs. Standard (indirect) | 0.29 (0.03,2.51) | 0.14 (0.00,1.45) | |||
| Herbal medicine vs. Standard (indirect) | 0.85 (0.44,1.61) | 0.63 (0.24,1.58) | |||
| Placebo vs. Standard (indirect) | 1.46 (0.95,2.25) | 1.23 (0.63,2.23) | |||
| Comparison with Placebo | |||||
| Diet vs. Placebo (indirect) | 0.45 (0.25,0.79) | 0.37 (0.16,0.89) | |||
| Exercise vs. Placebo (indirect) | 0.35(0.19,0.64) | 0.38 (0.16,0.88) | |||
| Orlistat vs. Placebo | 1 | 0.40 (0.07,2.24) | 0.35 (0.15,0.79) | 0.35 (0.14,0.90) | |
| Acarbose/Voglibose vs. Placebo | 1 | 0.38 (0.25,0.58) | 0.45 (0.28,0.74) | 0.42 (0.20,0.87) | |
| Metformin/Flumamine vs Placebo | 3 | 0.52 (0.30,0.91) | 64.4 | 0.48 (0.32,0.71) | 0.39 (0.21,0.67) |
| Pioglitazone, Rosiglitazone vs. Placebo | 3 | 0.70 (0.44,1.11) | 87.3 | 0.73 (0.50,1.05) | 0.65 (0.37,1.11) |
| Pitavastatin vs. Placebo (indirect) | 0.64 (0.29,1.42) | 0.71 (0.24,2.23) | |||
| Glipizide vs. Placebo | 1 | 0.22 (0.02,1.90) | 0.20 (0.02,1.64) | 0.12 (0.00,1.13) | |
| Herbal medicine vs. Placebo | 3 | 0.60 (0.45,0.82) | 0.0 | 0.60 (0.29,1.26) | 0.51 (0.25,1.02) |
| Log Likelihood Ratio/p-values for inconsistency | χ2 = 7.24, p = 0.78 (df = 11) | Tau = 0.42 (0.23,0.66) | |||
| Study rank | Lifestyle | Exercise | Diet | Orlistat | Acarbose/Voglibose | Metformin/Flumamine | Pioglitazone/Rosiglitazone | Pitavastatin | Glipizide | Herbal medicine | Placebo | Standard |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Worst | 0.0 | 0.0 | 0.1 | 0.4 | 0.0 | 0.0 | 4.1 | 8.0 | 6.7 | 1.3 | 77.0 | 2.5 |
| Mean | 4.4 | 3.1 | 5.4 | 3.6 | 5.4 | 6.0 | 9.6 | 8.2 | 3.0 | 7.7 | 11.7 | 9.5 |
| SUCRA | 0.7 | 0.8 | 0.6 | 0.8 | 0.6 | 0.5 | 0.2 | 0.3 | 0.8 | 0.4 | 0.0 | 0.2 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yamaoka, K.; Nemoto, A.; Tango, T. Comparison of the Effectiveness of Lifestyle Modification with Other Treatments on the Incidence of Type 2 Diabetes in People at High Risk: A Network Meta-Analysis. Nutrients 2019, 11, 1373. https://doi.org/10.3390/nu11061373
Yamaoka K, Nemoto A, Tango T. Comparison of the Effectiveness of Lifestyle Modification with Other Treatments on the Incidence of Type 2 Diabetes in People at High Risk: A Network Meta-Analysis. Nutrients. 2019; 11(6):1373. https://doi.org/10.3390/nu11061373
Chicago/Turabian StyleYamaoka, Kazue, Asuka Nemoto, and Toshiro Tango. 2019. "Comparison of the Effectiveness of Lifestyle Modification with Other Treatments on the Incidence of Type 2 Diabetes in People at High Risk: A Network Meta-Analysis" Nutrients 11, no. 6: 1373. https://doi.org/10.3390/nu11061373
APA StyleYamaoka, K., Nemoto, A., & Tango, T. (2019). Comparison of the Effectiveness of Lifestyle Modification with Other Treatments on the Incidence of Type 2 Diabetes in People at High Risk: A Network Meta-Analysis. Nutrients, 11(6), 1373. https://doi.org/10.3390/nu11061373




