Effect of Nut Consumption on Erectile and Sexual Function in Healthy Males: A Secondary Outcome Analysis of the FERTINUTS Randomized Controlled Trial
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Anthropometric, Dietary, Blood Parameters, and Seminogram Measurements
2.3. Erectile Function Questionnaire
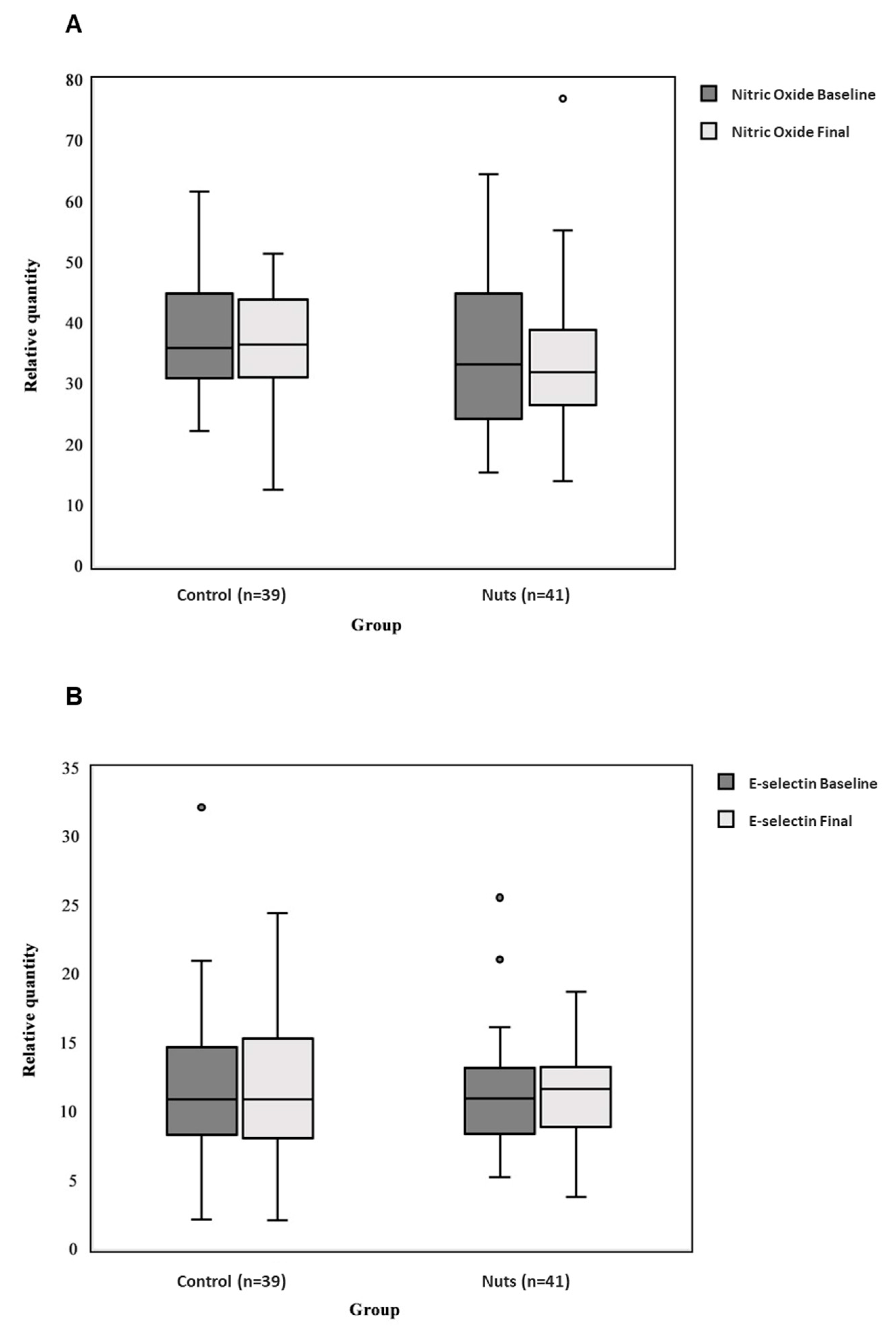
2.4. Surrogated Measures of Erectile Endothelial Function
2.5. Statistical Analyses
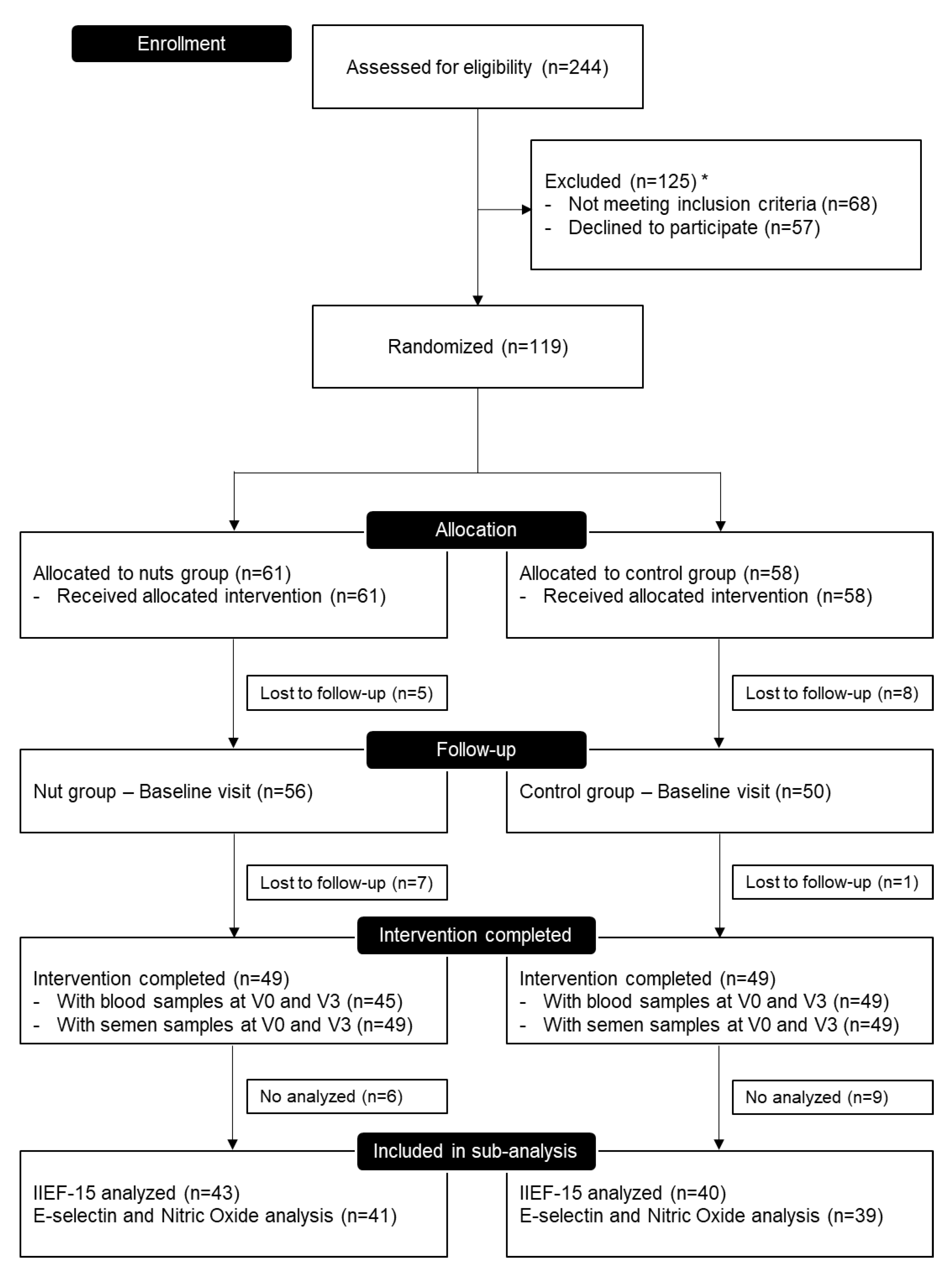
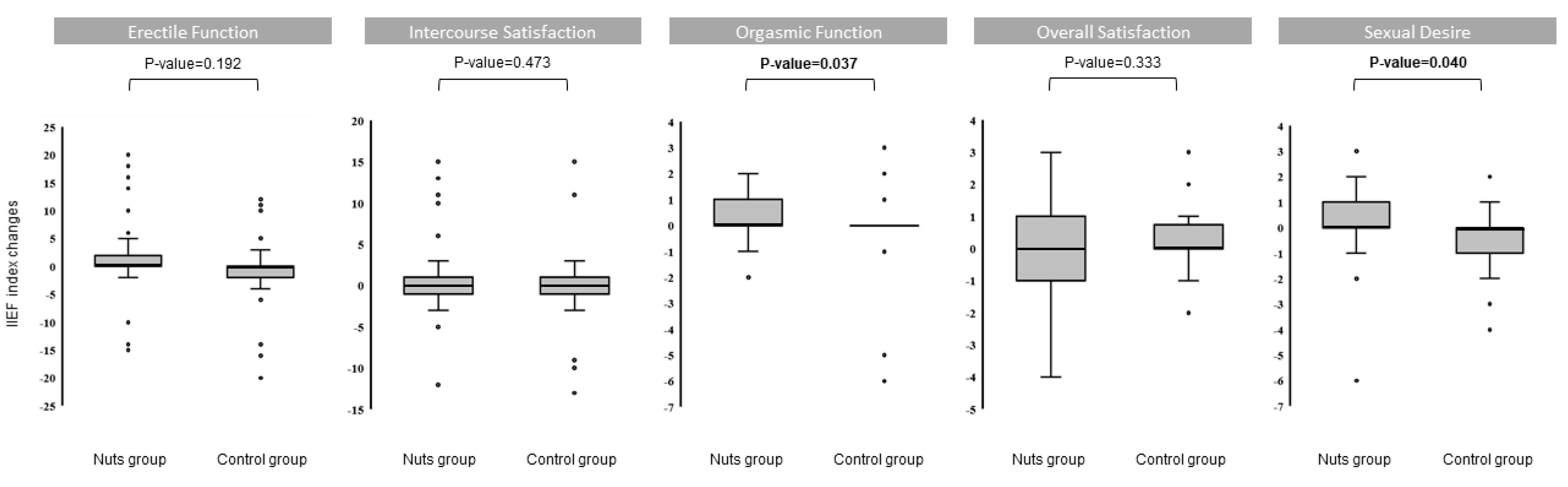
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- NIH Consensus NIH Consensus Conference: NIH Consensus Development Panel on Impotence. JAMA 1993, 270, 83–90. [CrossRef]
- Prins, J.; Blanker, M.H.; Bohnen, A.M.; Thomas, S.; Bosch, J. Prevalence of erectile dysfunction: A systematic review of population-based studies. Int. J. Impot. Res. 2002, 14, 422–432. [Google Scholar] [CrossRef]
- Aytac, I.; McCkinlay, J.; Krane, R. The likely worldwide increase in erectile dysfunction between 1995 and 2025 and some possible policy consequences. BJU Int. 1999, 84, 50–56. [Google Scholar] [CrossRef]
- Maiorino, M.; Bellastella, G.; Esposito, K. Lifestyle modifications and erectile dysfunction: What can be expected? Asian J. Androl. 2015, 17, 5–10. [Google Scholar]
- Kirby, M. The circle of lifestyle and erectile dysfunction. Sex. Med. Rev. 2015, 3, 169–182. [Google Scholar] [CrossRef] [PubMed]
- Hehemann, M.C.; Kashania, J.A. Can lifestyle modification affect men’s erectile function? Transl. Androl. Urol. 2016, 5, 187–194. [Google Scholar] [CrossRef]
- Allen, M.S.; Walter, E.E. Erectile Dysfunction: An Umbrella Review of Meta-Analyses of Risk-Factors, Treatment, and Prevalence Outcomes. J. Sex. Med. 2019, 16, 531–541. [Google Scholar] [CrossRef]
- Vlachopoulos, C.; Rokkas, K.; Ioakeimidis, N. Inflammation, metabolic syndrome, erectile dysfunction, and coronary artery disease: Common links. Eur. Urol. 2007, 52, 1590–1600. [Google Scholar] [CrossRef] [PubMed]
- Esposito, K.; Ciotola, M.; Giugliano, F.; De Sio, M.; Giugliano, G.; D’Armiento, M.; Giugliano, D. Mediterranean diet improves erectile function in subjects with the metabolic syndrome. Int. J. Impot. Res. 2006, 18, 405–410. [Google Scholar] [CrossRef]
- Schwingshackl, L.; Hoffmann, G. Mediterranean dietary pattern, inflammation and endothelial function: A systematic review and meta-analysis of intervention trials. Nutr. Metab. Cardiovasc. Dis. 2014, 24, 929–939. [Google Scholar] [CrossRef]
- Neale, E.P.; Tapsell, L.C.; Guan, V.; Batterham, M.J. The effect of nut consumption on markers of inflammation and endothelial function: A systematic review and meta-analysis of randomised controlled trials. BMJ Open 2017, 7, e016863. [Google Scholar] [CrossRef] [PubMed]
- Yu, Z.; Malik, V.S.; Keum, N.; Hu, F.B.; Giovannucci, E.L.; Stampfer, M.J.; Willett, W.C.; Fuchs, C.S.; Bao, Y. Associations between nut consumption and inflammatory biomarkers. Am. J. Clin. Nutr. 2016, 104, 722–728. [Google Scholar] [CrossRef] [PubMed]
- Aldemir, M.; Okulu, E.; Neşelioğlu, S.; Erel, O.; Kayıgil, Ö. Pistachio diet improves erectile function parameters and serum lipid profiles in patients with erectile dysfunction. Int. J. Impot. Res. 2011, 23, 32–38. [Google Scholar] [CrossRef] [PubMed]
- Levine, H.; Jørgensen, N.; Martino-Andrade, A.; Mendiola, J.; Weksler-Derri, D.; Mindlis, I.; Pinotti, R.; Swan, S.H. Temporal trends in sperm count: A systematic review and meta-regression analysis. Hum. Reprod. Update 2017, 23, 646–659. [Google Scholar] [CrossRef] [PubMed]
- Ilacqua, A.; Izzo, G.; Emerenziani, G.P.; Baldari, C.; Aversa, A. Lifestyle and fertility: The influence of stress and quality of life on male fertility. Reprod. Biol. Endocrinol. 2018, 16, 115. [Google Scholar] [CrossRef] [PubMed]
- Salas-Huetos, A.; Moraleda, R.; Giardina, S.; Anton, E.; Blanco, J.; Salas-Salvadó, J.; Bulló, M. Effect of nut consumption on semen quality and functionality in healthy men consuming a Western-style diet: A randomized controlled trial. Am. J. Clin. Nutr. 2018, 108, 953–962. [Google Scholar] [CrossRef] [PubMed]
- Estruch, R.; Ros, E.; Salas-Salvadó, J.; Covas, M.-I.; Corella, D.; Arós, F.; Gómez-Gracia, E.; Ruiz-Gutiérrez, V.; Fiol, M.; Lapetra, J.; et al. Primary Prevention of Cardiovascular Disease with a Mediterranean Diet Supplemented with Extra-Virgin Olive Oil or Nuts. N. Engl. J. Med. 2018, 378, e34. [Google Scholar] [CrossRef] [PubMed]
- Salas-Salvadó, J.; Díaz-López, A.; Ruiz-Canela, M.; Basora, J.; Fitó, M.; Corella, D.; Serra-Majem, L.; Warnberg, J.; Romaguera, D.; Estruch, R.; et al. Effect of a lifestyle intervention program with energy-restricted Mediterranean diet and exercise on weight loss and cardiovascular risk factors: One-year results of the PREDIMED-Plus trial. Diabetes Care 2018, 42, 777–788. [Google Scholar] [CrossRef] [PubMed]
- Martínez-González, M.A.; García-Arellano, A.; Toledo, E.; Salas-Salvadó, J.; Buil-Cosiales, P.; Corella, D.; Covas, M.I.; Schröder, H.; Arós, F.; Gómez-Gracia, E.; et al. A 14-item mediterranean diet assessment tool and obesity indexes among high-risk subjects: The PREDIMED trial. PLoS ONE 2012, 7, e43134. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Ballart, J.D.; Piñol, J.L.; Zazpe, I.; Corella, D.; Carrasco, P.; Toledo, E.; Perez-Bauer, M.; Martínez-González, M.A.; Salas-Salvadó, J.; Martín-Moreno, J.M. Relative validity of a semi-quantitative food-frequency questionnaire in an elderly Mediterranean population of Spain. Br. J. Nutr. 2010, 103, 1808–1816. [Google Scholar] [CrossRef] [PubMed]
- Moreiras, O.; Carvajal, A. Tablas de Composición de Alimentos (Food Composition Tables), 9th ed.; Ediciones Pirámide: Madrid, Spain, 2005. [Google Scholar]
- Mataix Verdú, J. Tabla de Composición de Alimentos Españoles (Food Composition Tables), 4th ed.; Universidad de Granada: Granada, Spain, 2003. [Google Scholar]
- World Health Organization. WHO Laboratory Manual for the Examination and Processing of Human Semen, 5th ed.; World Health Organization: Geneva, Switzerland, 2010. [Google Scholar]
- Björndahl, L.; Barratt, C.L.R.; Mortimer, D.; Jouannet, P. “How to count sperm properly”: Checklist for acceptability of studies based on human semen analysis. Hum. Reprod. 2016, 31, 227–232. [Google Scholar] [CrossRef] [PubMed]
- Rosen, R.C.; Riley, A.; Wagner, G.; Osterloh, I.H.; Kirkpatrick, J.; Mishra, A. The international index of erectile function (IIEF): A multidimensional scale for assessment of erectile dysfunction. Urology 1997, 49, 822–830. [Google Scholar] [CrossRef]
- Rosen, R.C.; Cappelleri, J.C.; Iii, N.G. The International Index of Erectile Function (IIEF): A state-of-the-science review. Int. J. Impot. Res. 2002, 14, 226–244. [Google Scholar] [CrossRef] [PubMed]
- Robbins, W.A.; Xun, L.; FitzGerald, L.Z.; Esguerra, S.; Henning, S.M.; Carpenter, C.L. Walnuts improve semen quality in men consuming a Western-style diet: Randomized control dietary intervention trial. Biol. Reprod. 2012, 87, 101–111. [Google Scholar] [CrossRef] [PubMed]
- R Development Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2013; ISBN 3-900051-07-0. Available online: http://www.R-project.org/ (accessed on 11 March 2019).
- Fellows, I. Deducer: A data analysis GUI for R. J. Stat. Softw. 2012, 49, 1–15. [Google Scholar] [CrossRef]
- Ros, E. Nuts and novel biomarkers of cardiovascular disease. Am. J. Clin. Nutr. 2009, 89, 1649S–1656S. [Google Scholar] [CrossRef] [PubMed]
- Del Gobbo, L.C.; Falk, M.C.; Feldman, R.; Lewis, K.; Mozaffarian, D. Effects of tree nuts on blood lipids, apolipoproteins, and blood pressure: Systematic review, meta-analysis, and dose-response of 61 controlled intervention trials. Am. J. Clin. Nutr. 2015, 102, 1347–1356. [Google Scholar] [CrossRef] [PubMed]
- Sabaté, J.; Oda, K.; Ros, E. Nut Consumption and Blood Lipid Levels: A Pooled Analysis of 25 Intervention Trials. Arch. Intern. Med. 2010, 170, 821–827. [Google Scholar] [CrossRef] [PubMed]
- US Department of Agriculture, Agricultural Research Service, Nutrient Data Laboratory. USDA National Nutrient Database for Standard Reference. Current Version: April 2018. Available online: https://ndb.nal.usda.gov/ndb/search/list (accessed on 25 April 2019).
- Lue, T. Erectile dysfunction. N. Engl. J. Med. 2000, 342, 1802–1813. [Google Scholar] [CrossRef] [PubMed]
- Patel, D.P.; Craig, R.C., Jr.; Myers, J.B.; Brant, W.O.; Hotaling, J.M. Serum Biomarkers of Erectile Dysfunction in Diabetes Mellitus : A Systematic Review of Current Literature. Sex. Med. Rev. 2017, 5, 339–348. [Google Scholar] [CrossRef] [PubMed]
- Araña Rosaínz, M.; Ojeda, M.O.; Acosta, J.R.; Elías-Calles, L.; González, N.; Herrera, O.T.; Álvarez, C.; Rodríguez, E.; Báez, M.; Seijas, E.; et al. Imbalanced Low-Grade Inflammation and Endothelial Activation in Patients with Type 2 Diabetes Mellitus and Erectile Dysfunction. J. Sex. Med. 2011, 8, 2017–2030. [Google Scholar] [CrossRef] [PubMed]
- Gulati, S.; Misra, A.; Pandey, R.M. Effect of Almond Supplementation on Glycemia and Cardiovascular Risk Factors in Asian Indians in North India with Type 2 Diabetes Mellitus: A 24–Week Study. Metab. Syndr. Relat. Disord. 2017, 15, 98–105. [Google Scholar] [CrossRef] [PubMed]
- Nehra, A.; Jackson, G.; Miner, M.; Billups, K.L.; Burnett, A.L.; Buvat, J.; Carson, C.C.; Cunningham, G.R.; Ganz, P.; Goldstein, I.; et al. The Princeton III Consensus Recommendations for the Management of Erectile Dysfunction and Cardiovascular Disease. Mayo Clin. Proc. 2012, 87, 766–778. [Google Scholar] [CrossRef] [PubMed]
- Jenkins, D.J.A.; Kendall, C.W.C.; Banach, M.S.; Srichaikul, K.; Vidgen, E.; Mitchell, S.; Parker, T.; Nishi, S.; Bashyam, B.; De Souza, R.; et al. Nuts as a replacement for carbohydrates in the diabetic diet: A reanalysis of a randomised controlled trial. Diabetologia 2018, 61, 1734–1747. [Google Scholar] [CrossRef] [PubMed]
- Alderson, P. Absence of evidence is not evidence of absence. BMJ 1995, 328, 476–477. [Google Scholar] [CrossRef] [PubMed]
| Variable | Nuts Group (n = 43) | Control Group (n = 40) | p-Value |
|---|---|---|---|
| General characteristics; mean (SD) | |||
| Age (years) | 24.05 (4.82) | 25.83 (4.58) | 0.066 |
| Weight (kg) | 73.44 (10.16) | 76.82 (12.04) | 0.270 |
| Height (cm) | 176.46 (6.21) | 177.73 (6.66) | 0.409 |
| BMI (kg/m2) | 23.53 (2.59) | 24.33 (3.64) | 0.379 |
| Waist circumference (cm) | 79.98 (7.41) | 83.26 (8.98) | 0.067 |
| Systolic blood pressure (mmHg) | 129.38 (11.37) | 126.50 (11.79) | 0.425 |
| Diastolic blood pressure (mmHg) | 73.34 (7.90) | 70.90 (8.49) | 0.174 |
| Score for adherence to Western-style diet a | 8.11 (2.16) | 8.80 (2.26) | 0.182 |
| Blood parameters; median (IQR) | |||
| Fasting plasma glucose (mg/dl) | 87.0 (82.0, 93.5) | 85.5 (81.5, 91.0) | 0.384 |
| Total cholesterol (mg/dl) | 167.0 (149.5, 188.5) | 173.5 (150.0, 196.0) | 0.374 |
| HDL-c (mg/dl) | 58.0 (49.5, 66.5) | 55.5 (50.0, 67.0) | 0.736 |
| LDL-c (mg/dl) | 87.0 (74.5, 105.5) | 98.0 (77.8, 119.3) | 0.202 |
| VLDL-c (mg/dl) | 13.0 (12.0, 19.0) | 13.0 (9.0, 17.3) | 0.240 |
| Triglycerides (mg/dl) | 66.0 (59.0, 95.5) | 64.0 (47.0, 85.0) | 0.258 |
| Fasting plasma insulin (mcUl/ml) | 5.40 (2.60, 8.60) | 5.20 (2.75, 6.80) | 0.654 |
| C-Reactive protein (mg/dl) | 0.20 (0.20, 0.20) | 0.20 (0.13, 0.20) | 0.144 |
| Folate (ng/ml) | 6.30 (4.80, 8.70) | 6.30 (4.88, 7.80) | 0.616 |
| Semen characteristics; median (IQR) | |||
| pH | 8.0 (8.0, 8.5) | 8.0 (8.0, 8.5) | 0.940 |
| Volume (mL) | 3.50 (1.95, 4.55) | 3.40 (2.50, 5.13) | 0.179 |
| Total spermatozoa (×106) | 75.20 (28.10, 104.50) | 72.05 (40.90, 125.50) | 0.497 |
| Spermatozoa concentration (×106) | 25.20 (14.50, 41.80) | 19.80 (9.70, 37.95) | 0.402 |
| Viability (%) | 78.68 (70.26, 82.52) | 80.21 (73.66, 85.87) | 0.257 |
| Total motility (progressive and non-progressive motility) (%) | 64.66 (45.91, 71.34) | 70.11 (62.73, 78.56) | 0.097 |
| Progressive motility (%) | 44.67 (28.27, 53.97) | 49.72 (35.39, 61.67) | 0.086 |
| Non-progressive motility (%) | 13.26 (9.79, 16.15) | 11.64 (7.58, 14.31) | 0.103 |
| Immotile spermatozoa (%) | 35.33 (28.58, 52.06) | 29.89 (21.44, 37.27) | 0.094 |
| Normal forms (%) | 6.33 (4.91, 8.17) | 6.27 (5.23, 7.57) | 0.935 |
| Abnormal head (%) | 52.59 (43.06, 66.28) | 55.07 (41.22, 67.81) | 0.771 |
| Abnormal midpiece (%) | 10.71 (8.63, 15.05) | 12.71 (8.46, 14.23) | 0.705 |
| Abnormal principal piece (%) | 12.94 (5.44, 29.66) | 9.46 (4.53, 25.27) | 0.529 |
| Combined abnormality (%) | 8.40 (6.49, 12.88) | 7.85 (6.71, 14.07) | 0.985 |
| Variables | Nut Group (n = 43) | Control Group (n = 40) | Treatment Effect | ||
|---|---|---|---|---|---|
| Baseline | Changes | Baseline | Changes | p-Value | |
| Energy | |||||
| Energy intake (kcal/d) | 2699.13 (994.43) | −145.19 (31.74) | 2359.57 (565.72) | −215.22 (18.18) | 0.029 |
| Macronutrients | |||||
| Proteins (g/d) | 112.20 (39.37) | −7.68 (1.44) | 103.13 (22.51) | −10.45 (0.34) | 0.065 |
| Carbohydrates (g/d) | 305.24 (134.16) | −50.48 (4.83) | 254.34 (63.04) | −11.21 (0.05) | 0.333 |
| Simple carbohydrates (g/d) | 112.70 (79.80) | −13.47 (3.33) | 92.54 (28.75) | −6.31 (0.06) | 0.696 |
| Complex carbohydrates (g/d) | 176.44 (68.26) | −34.36 (0.84) | 145.29 (46.70) | −12.09 (1.30) | 0.604 |
| Total fat (g/d) | 106.89 (40.75) | 12.19 (0.66) | 97.98 (30.47) | −11.95 (0.92) | <0.001 |
| MUFA (g/d) | 42.87 (18.36) | 5.89 (0.32) | 37.97 (15.82) | −6.14 (0.48) | <0.001 |
| SFA (g/d) | 33.33 (17.87) | −3.19 (0.21) | 31.17 (10.64) | −3.67 (0.04) | 0.589 |
| PUFA (g/d) | 13.51 (6.41) | 11.03 (0.27) | 11.37 (4.50) | −2.60 (0.23) | <0.001 |
| Cholesterol (mg/d) | 426.64 (253.96) | −70.48 (8.95) | 386.09 (131.85) | −56.08 (2.37) | 0.745 |
| Fiber (g/d) | 24.69 (11.21) | −0.77 (0.39) | 19.84 (9.45) | −2.39 (0.41) | 0.002 |
| Alcohol (g/d) | 9.43 (14.01) | −2.79 (0.73) | 7.91 (13.14) | −2.25 (0.89) | 0.693 |
| Glycemic load | 172.69 (75.43) | −31.38 (2.55) | 141.20 (37.69) | −5.52 (0.32) | 0.384 |
| Glycemic index | 56.16 (4.50) | −1.48 (0.18) | 55.13 (5.45) | 0.29 (0.08) | 0.346 |
| Micronutrients | |||||
| Sodium (mg/d) | 3452.88 (1455.36) | −407.37 (26.13) | 3178.08 (958.75) | −174.95 (23.37) | 0.743 |
| Potassium (mg/d) | 3639.91 (1624.75) | −217.88 (58.80) | 3126.17 (968.16) | −140.77 (21.62) | 0.289 |
| Linoleic (g/d) | 26.08 (26.64) | 4.79 (1.35) | 31.40 (31.79) | −4.66 (1.24) | 0.311 |
| Magnesium (mg/d) | 407.62 (199.96) | 32.17 (7.96) | 329.53 (108.61) | −16.92 (0.93) | <0.001 |
| Calcium (mg/d) | 987.45 (514.83) | −98.27 (2.79) | 898.70 (324.53) | −51.64 (13.16) | 0.843 |
| Iron (mg/d) | 20.27 (14.55) | −2.04 (0.60) | 18.35 (13.33) | −0.11 (0.47) | 0.864 |
| Selenium (mg/d) | 222.04 (162.30) | −48.94 (13.01) | 209.25 (140.86) | −46.06 (11.51) | 0.563 |
| β-carotene equivalents (µg/d) | 4711.11 (5096.91) | −1008.75 (348.71) | 3913.42 (4116.64) | 22.34 (72.89) | 0.642 |
| Retinol (µg/d) | 592.05 (1409.67) | −230.55 (169.57) | 402.76 (166.28) | 141.07 (141.29) | 0.254 |
| Vitamin D (µg/d) | 7.37 (13.90) | 0.55 (0.31) | 5.05 (5.65) | 4.50 (3.67) | 0.606 |
| Vitamin E (µg/d) | 13.18 (5.12) | 6.10 (1.20) | 15.19 (18.38) | −3.03 (0.93) | 0.014 |
| Vitamin K (µg/d) | 130.94 (163.71) | −10.50 (0.30) | 115.38 (111.22) | −32.51 (6.99) | 0.213 |
| Omega-3 (g/d) | 1.72 (0.89) | 1.53 (0.02) | 1.68 (0.73) | −0.10 (0.02) | <0.001 |
| ALA (g/d) | 1.10 (0.61) | 1.55 (0.02) | 1.00 (0.39) | −0.22 (0.02) | <0.001 |
| EPA (g/d) | 0.51 (2.74) | 0.02 (0.00) | 1.03 (3.96) | 0.65 (0.12) | 0.236 |
| DHA (g/d) | 0.15 (0.19) | 0.02 (0.01) | 0.21 (0.31) | 0.10 (0.03) | 0.161 |
| Omega-6 (g/d) | 18.03 (14.80) | 10.27 (0.08) | 24.64 (23.36) | −3.76 (0.94) | 0.016 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Salas-Huetos, A.; Muralidharan, J.; Galiè, S.; Salas-Salvadó, J.; Bulló, M. Effect of Nut Consumption on Erectile and Sexual Function in Healthy Males: A Secondary Outcome Analysis of the FERTINUTS Randomized Controlled Trial. Nutrients 2019, 11, 1372. https://doi.org/10.3390/nu11061372
Salas-Huetos A, Muralidharan J, Galiè S, Salas-Salvadó J, Bulló M. Effect of Nut Consumption on Erectile and Sexual Function in Healthy Males: A Secondary Outcome Analysis of the FERTINUTS Randomized Controlled Trial. Nutrients. 2019; 11(6):1372. https://doi.org/10.3390/nu11061372
Chicago/Turabian StyleSalas-Huetos, Albert, Jananee Muralidharan, Serena Galiè, Jordi Salas-Salvadó, and Mònica Bulló. 2019. "Effect of Nut Consumption on Erectile and Sexual Function in Healthy Males: A Secondary Outcome Analysis of the FERTINUTS Randomized Controlled Trial" Nutrients 11, no. 6: 1372. https://doi.org/10.3390/nu11061372
APA StyleSalas-Huetos, A., Muralidharan, J., Galiè, S., Salas-Salvadó, J., & Bulló, M. (2019). Effect of Nut Consumption on Erectile and Sexual Function in Healthy Males: A Secondary Outcome Analysis of the FERTINUTS Randomized Controlled Trial. Nutrients, 11(6), 1372. https://doi.org/10.3390/nu11061372







