Nutrition and Supplement Update for the Endurance Athlete: Review and Recommendations
Abstract
1. Introduction
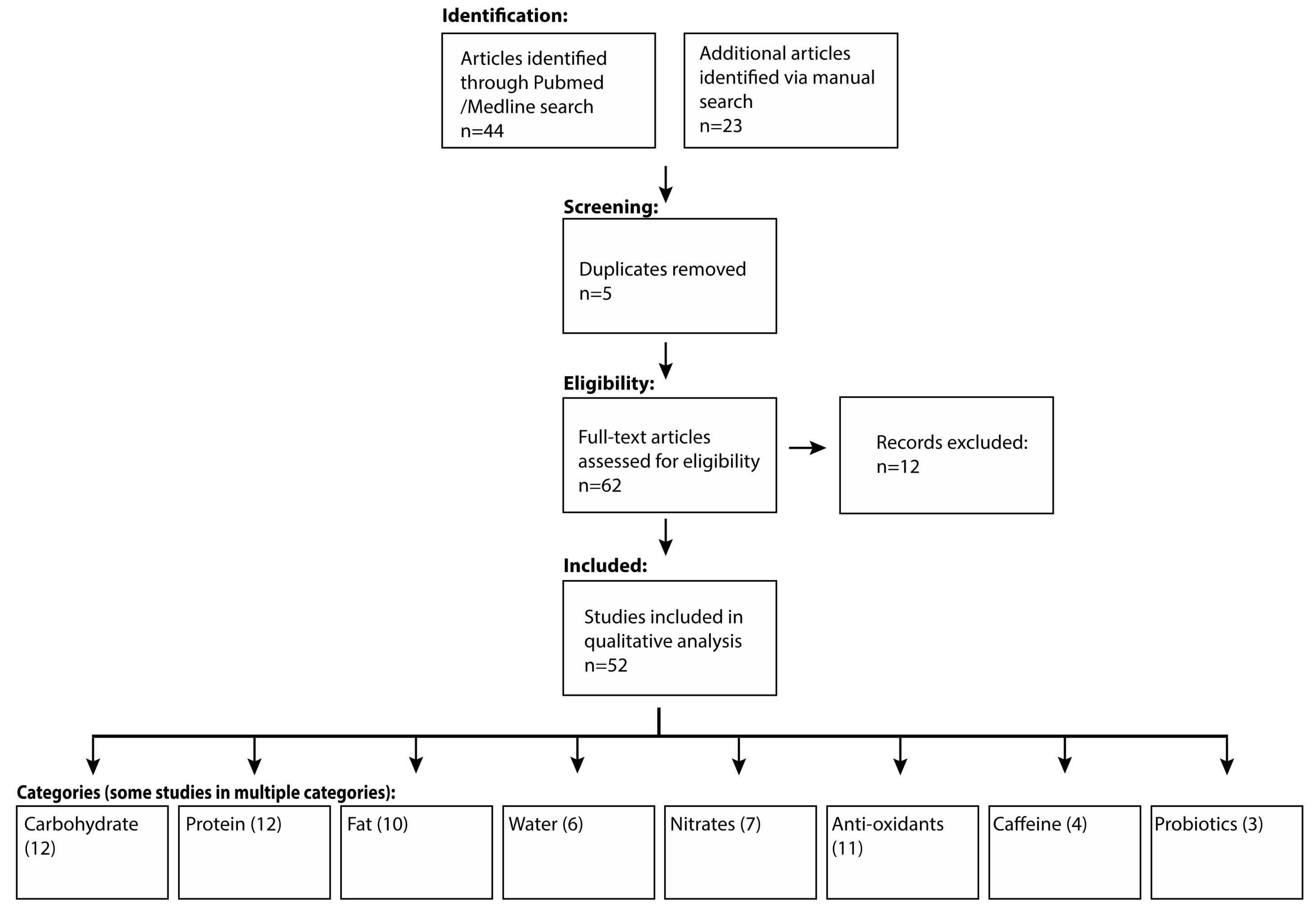
2. Materials and Methods
3. Results
3.1. Carbohydrate
3.1.1. Pre-Competition, “Loading”
3.1.2. During Competition, “Fueling”
3.2. Protein
3.2.1. Daily Protein Requirements
3.2.2. Pre-, During, and Post-Exercise Protein Requirements
3.3. Fat
3.4. Hydration
3.5. Supplements and “Hot Topics”
3.5.1. Nitrates
3.5.2. Antioxidants
3.5.3. Caffeine
3.5.4. Probiotics
3.5.5. Recovery Nutrition
3.5.6. Concern with High Carbohydrate Diets?
4. Discussion
Author Contributions
Funding
Conflicts of Interest
References
- Behind USAT and Ironman’s Efforts to Grow the Sport. March 2017. Available online: https://www.triathlete.com/2017/03/lifestyle/behind-usat-ironmans-efforts-grow-sport_299133 (accessed on 25 May 2019).
- Shilton, A. Let’s Try a Triathlon—The New York Times. Available online: https://www.nytimes.com/guides/well/triathlon-training (accessed on 2 April 2019).
- Miller, J.A. The Running Bubble Has Popped. (You Couldn’t Hear It in New York.)—The New York Times. Available online: https://www.nytimes.com/2017/11/05/sports/ny-marathon-running.html (accessed on 2 April 2019).
- Costa, R.J.S.; Hoffman, M.D.; Stellingwerff, T. Considerations for Ultra-Endurance Activities: Part 1—Nutrition. Res. Sports Med. 2019, 27, 166–181. [Google Scholar] [CrossRef] [PubMed]
- Nikolaidis, P.T.; Veniamakis, E.; Rosemann, T.; Knechtle, B. Nutrition in Ultra-Endurance: State of the Art. Nutrients 2018, 10, 1995. [Google Scholar] [CrossRef] [PubMed]
- Ebell, M.H.; Siwek, J.; Weiss, B.D.; Woolf, S.H.; Susman, J.; Ewigman, B.; Bowman, M. Strength of Recommendation Taxonomy (SORT): A Patient-Centered Approach to Grading Evidence in the Medical Literature. AFP 2004, 69, 548. [Google Scholar] [CrossRef]
- U.S. Department of Health & Human Services, Agency for Healthcare Research and Quality. Clinical Guidelines and Recommendations. Available online: https://www.ahrq.gov/professionals/clinicians-providers/guidelines-recommendations/index.html (accessed on 25 May 2019).
- Jäger, R.; Kerksick, C.M.; Campbell, B.I.; Cribb, P.J.; Wells, S.D.; Skwiat, T.M.; Purpura, M.; Ziegenfuss, T.N.; Ferrando, A.A.; Arent, S.M.; et al. International Society of Sports Nutrition Position Stand: Protein and Exercise. J. Int. Soc. Sports Nutr. 2017, 14, 20. [Google Scholar] [CrossRef] [PubMed]
- Thomas, D.T.; Erdman, K.A.; Burke, L.M. Position of the Academy of Nutrition and Dietetics, Dietitians of Canada, and the American College of Sports Medicine: Nutrition and Athletic Performance. J. Acad. Nutr. Diet. 2016, 116, 501–528. [Google Scholar] [CrossRef]
- Spriet, L.L. New Insights into the Interaction of Carbohydrate and Fat Metabolism during Exercise. Sports Med. 2014, 44 (Suppl. 1), S87–S96. [Google Scholar] [CrossRef]
- Jeukendrup, A.E.; Jentjens, R.L.P.G.; Moseley, L. Nutritional Considerations in Triathlon. Sports Med. 2005, 35, 163–181. [Google Scholar] [CrossRef] [PubMed]
- Getzin, A.R.; Milner, C.; Harkins, M. Fueling the Triathlete: Evidence-Based Practical Advice for Athletes of All Levels. Curr. Sports Med. Rep. 2017, 16, 240–246. [Google Scholar] [CrossRef]
- Noakes, T.D. Physiological Models to Understand Exercise Fatigue and the Adaptations That Predict or Enhance Athletic Performance. Scand. J. Med. Sci. Sports 2000, 10, 123–145. [Google Scholar] [CrossRef]
- Burke, L.M.; Hawley, J.A.; Wong, S.H.S.; Jeukendrup, A.E. Carbohydrates for Training and Competition. J. Sports Sci. 2011, 29 (Suppl. 1), S17–S27. [Google Scholar] [CrossRef]
- Bergström, J.; Hermansen, L.; Hultman, E.; Saltin, B. Diet, Muscle Glycogen and Physical Performance. Acta Physiol. Scand. 1967, 71, 140–150. [Google Scholar] [CrossRef] [PubMed]
- Bussau, V.A.; Fairchild, T.J.; Rao, A.; Steele, P.; Fournier, P.A. Carbohydrate Loading in Human Muscle: An Improved 1 Day Protocol. Eur. J. Appl. Physiol. 2002, 87, 290–295. [Google Scholar] [CrossRef] [PubMed]
- Jeukendrup, A.E.; Moseley, L.; Mainwaring, G.I.; Samuels, S.; Perry, S.; Mann, C.H. Exogenous Carbohydrate Oxidation during Ultraendurance Exercise. J. Appl. Physiol. 2006, 100, 1134–1141. [Google Scholar] [CrossRef] [PubMed]
- Jeukendrup, A. A Step Towards Personalized Sports Nutrition: Carbohydrate Intake During Exercise. Sports Med. 2014, 44 (Suppl. 1), 25–33. [Google Scholar] [CrossRef] [PubMed]
- Getzin, A.R.; Milner, C.; LaFace, K.M. Nutrition Update for the Ultraendurance Athlete. Curr. Sports Med. Rep. 2011, 10, 330–339. [Google Scholar] [CrossRef] [PubMed]
- Hansen, A.K.; Fischer, C.P.; Plomgaard, P.; Andersen, J.L.; Saltin, B.; Pedersen, B.K. Skeletal Muscle Adaptation: Training Twice Every Second Day vs. Training Once Daily. J. Appl. Physiol. 2005, 98, 93–99. [Google Scholar] [CrossRef] [PubMed]
- Silva, T.d.A.e.; de Souza, M.E.D.C.A.; de Amorim, J.F.; Stathis, C.G.; Leandro, C.G.; Lima-Silva, A.E. Can Carbohydrate Mouth Rinse Improve Performance during Exercise? A Systematic Review. Nutrients 2013, 6, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Jeukendrup, A.E.; Chambers, E.S. Oral Carbohydrate Sensing and Exercise Performance. Curr. Opin. Clin. Nutr. Metab. Care 2010, 13, 447–451. [Google Scholar] [CrossRef]
- Cox, G.R.; Clark, S.A.; Cox, A.J.; Halson, S.L.; Hargreaves, M.; Hawley, J.A.; Jeacocke, N.; Snow, R.J.; Yeo, W.K.; Burke, L.M. Daily Training with High Carbohydrate Availability Increases Exogenous Carbohydrate Oxidation during Endurance Cycling. J. Appl. Physiol. 2010, 109, 126–134. [Google Scholar] [CrossRef]
- Jeukendrup, A.; Brouns, F.; Wagenmakers, A.J.; Saris, W.H. Carbohydrate-Electrolyte Feedings Improve 1 h Time Trial Cycling Performance. Int. J. Sports Med. 1997, 18, 125–129. [Google Scholar] [CrossRef]
- Carter, J.M.; Jeukendrup, A.E.; Mann, C.H.; Jones, D.A. The Effect of Glucose Infusion on Glucose Kinetics during a 1-h Time Trial. Med. Sci. Sports Exerc. 2004, 36, 1543–1550. [Google Scholar] [CrossRef]
- Carter, J.M.; Jeukendrup, A.E.; Jones, D.A. The Effect of Carbohydrate Mouth Rinse on 1-h Cycle Time Trial Performance. Med. Sci. Sports Exerc. 2004, 36, 2107–2111. [Google Scholar] [CrossRef]
- Phillips, S.M.; Van Loon, L.J.C. Dietary Protein for Athletes: From Requirements to Optimum Adaptation. J. Sports Sci. 2011, 29 (Suppl. 1), S29–S38. [Google Scholar] [CrossRef]
- Phillips, S.M. Dietary Protein Requirements and Adaptive Advantages in Athletes. Br. J. Nutr. 2012, 108 (Suppl. 2), S158–S167. [Google Scholar] [CrossRef]
- Burd, N.A.; West, D.W.D.; Moore, D.R.; Atherton, P.J.; Staples, A.W.; Prior, T.; Tang, J.E.; Rennie, M.J.; Baker, S.K.; Phillips, S.M. Enhanced Amino Acid Sensitivity of Myofibrillar Protein Synthesis Persists for up to 24 h after Resistance Exercise in Young Men. J. Nutr. 2011, 141, 568–573. [Google Scholar] [CrossRef]
- Kerksick, C.M.; Arent, S.; Schoenfeld, B.J.; Stout, J.R.; Campbell, B.; Wilborn, C.D.; Taylor, L.; Kalman, D.; Smith-Ryan, A.E.; Kreider, R.B.; et al. International Society of Sports Nutrition Position Stand: Nutrient Timing. J. Int. Soc. Sports Nutr. 2017, 14, 33. [Google Scholar] [CrossRef]
- Wilmore, J.H.; Costill, D.L.; Kenney, W.L. Fuel for Exercising Muscle: Metabolism and Hormonal Control. In Physiology of Sport and Exercise, 4th ed.; Wilmore, J.H., Costill, D.L., Kenney, W.L., Eds.; Human Kinetics: Champaign, IL, USA, 2008; ISBN-13: 978-0-7360-5583-3. [Google Scholar]
- Volek, J.S.; Noakes, T.; Phinney, S.D. Rethinking Fat as a Fuel for Endurance Exercise. Eur. J. Sport Sci. 2015, 15, 13–20. [Google Scholar] [CrossRef]
- Institute of Medicine, Food and Nutrition Board. Total fat and fatty acids. In Dietary Reference Intakes for Energy, Carbohydrate, Fiber, Fat, Fatty Acids, Cholesterol, Protein, and Amino Acids; Institute of Medicine (U.S.), Ed.; National Academies Press: Washington, DC, USA, 2005. [Google Scholar]
- Terasawa, N.; Okamoto, K.; Nakada, K.; Masuda, K. Effect of Conjugated Linoleic Acid Intake on Endurance Exercise Performance and Anti-Fatigue in Student Athletes. J. Oleo Sci. 2017, 66, 723–733. [Google Scholar] [CrossRef]
- Tajmanesh, M.; Aryaeian, N.; Hosseini, M.; Mazaheri, R.; Kordi, R. Conjugated Linoleic Acid Supplementation Has No Impact on Aerobic Capacity of Healthy Young Men. Lipids 2015, 50, 805–809. [Google Scholar] [CrossRef]
- Kerksick, C.M.; Wilborn, C.D.; Roberts, M.D.; Smith-Ryan, A.; Kleiner, S.M.; Jäger, R.; Collins, R.; Cooke, M.; Davis, J.N.; Galvan, E.; et al. ISSN Exercise & Sports Nutrition Review Update: Research & Recommendations. J. Int. Soc. Sports Nutr. 2018, 15, 38. [Google Scholar] [CrossRef]
- Macaluso, F.; Barone, R.; Catanese, P.; Carini, F.; Rizzuto, L.; Farina, F.; Di Felice, V. Do Fat Supplements Increase Physical Performance? Nutrients 2013, 5, 509–524. [Google Scholar] [CrossRef]
- Convertino, V.A.; Armstrong, L.E.; Coyle, E.F.; Mack, G.W.; Sawka, M.N.; Senay, L.C.; Sherman, W.M. American College of Sports Medicine Position Stand. Exercise and Fluid Replacement. Med. Sci. Sports Exerc. 1996, 28, i–vii. [Google Scholar] [CrossRef]
- Noakes, T.D.; Sharwood, K.; Speedy, D.; Hew, T.; Reid, S.; Dugas, J.; Almond, C.; Wharam, P.; Weschler, L. Three Independent Biological Mechanisms Cause Exercise-Associated Hyponatremia: Evidence from 2,135 Weighed Competitive Athletic Performances. Proc. Natl. Acad. Sci. USA 2005, 102, 18550–18555. [Google Scholar] [CrossRef]
- American College of Sports Medicine Position Stand. Exercise and Fluid Replacement. -PubMed-NCBI. Available online: https://www.ncbi.nlm.nih.gov/pubmed/17277604 (accessed on 3 April 2019).
- Wyndham, C.H.; Strydom, N.B. The Danger of an Inadequate Water Intake during Marathon Running. S. Afr. Med. J. 1969, 43, 893–896. [Google Scholar]
- Almond, C.S.D.; Shin, A.Y.; Fortescue, E.B.; Mannix, R.C.; Wypij, D.; Binstadt, B.A.; Duncan, C.N.; Olson, D.P.; Salerno, A.E.; Newburger, J.W.; et al. Hyponatremia among Runners in the Boston Marathon. N. Engl. J. Med. 2005, 352, 1550–1556. [Google Scholar] [CrossRef]
- Chorley, J.; Cianca, J.; Divine, J. Risk Factors for Exercise-Associated Hyponatremia in Non-Elite Marathon Runners. Clin. J. Sport Med. 2007, 17, 471–477. [Google Scholar] [CrossRef]
- Hew-Butler, T.; Ayus, J.C.; Kipps, C.; Maughan, R.J.; Mettler, S.; Meeuwisse, W.H.; Page, A.J.; Reid, S.A.; Rehrer, N.J.; Roberts, W.O.; et al. Statement of the Second International Exercise-Associated Hyponatremia Consensus Development Conference, New Zealand, 2007. Clin. J. Sport Med. 2008, 18, 111–121. [Google Scholar] [CrossRef]
- Krabak, B.J.; Parker, K.M.; DiGirolamo, A. Exercise-Associated Collapse: Is Hyponatremia in Our Head? PM R 2016, 8 (Suppl. 3), S61–S68. [Google Scholar] [CrossRef]
- Noakes, T.D.; Goodwin, N.; Rayner, B.L.; Branken, T.; Taylor, R.K. Water Intoxication: A Possible Complication during Endurance Exercise. Med. Sci. Sports Exerc. 1985, 17, 370–375. [Google Scholar] [CrossRef]
- Noakes, T.; IMMDA. Fluid Replacement during Marathon Running. Clin. J. Sport Med. 2003, 13, 309–318. [Google Scholar] [CrossRef]
- Montain, S.J.; Cheuvront, S.N.; Sawka, M.N. Exercise Associated Hyponatraemia: Quantitative Analysis to Understand the Aetiology. Br. J. Sports Med. 2006, 40, 98–105. [Google Scholar] [CrossRef]
- Shaltout, H.A.; Eggebeen, J.; Marsh, A.P.; Brubaker, P.H.; Laurienti, P.J.; Burdette, J.H.; Basu, S.; Morgan, A.; Dos Santos, P.C.; Norris, J.L.; et al. Effects of Supervised Exercise and Dietary Nitrate in Older Adults with Controlled Hypertension and/or Heart Failure with Preserved Ejection Fraction. Nitric Oxide 2017, 69, 78–90. [Google Scholar] [CrossRef]
- Larsen, F.J.; Weitzberg, E.; Lundberg, J.O.; Ekblom, B. Effects of Dietary Nitrate on Oxygen Cost during Exercise. Acta Physiol. Oxf. 2007, 191, 59–66. [Google Scholar] [CrossRef]
- Domínguez, R.; Cuenca, E.; Maté-Muñoz, J.L.; García-Fernández, P.; Serra-Paya, N.; Estevan, M.C.L.; Herreros, P.V.; Garnacho-Castaño, M.V. Effects of Beetroot Juice Supplementation on Cardiorespiratory Endurance in Athletes. A Systematic Review. Nutrients 2017, 9, 43. [Google Scholar] [CrossRef]
- McMahon, N.F.; Leveritt, M.D.; Pavey, T.G. The Effect of Dietary Nitrate Supplementation on Endurance Exercise Performance in Healthy Adults: A Systematic Review and Meta-Analysis. Sports Med. 2017, 47, 735–756. [Google Scholar] [CrossRef]
- Jonvik, K.L.; Nyakayiru, J.; van Loon, L.J.C.; Verdijk, L.B. Can Elite Athletes Benefit from Dietary Nitrate Supplementation? J. Appl. Physiol. 2015, 119, 759–761. [Google Scholar] [CrossRef]
- Porcelli, S.; Pugliese, L.; Rejc, E.; Pavei, G.; Bonato, M.; Montorsi, M.; La Torre, A.; Rasica, L.; Marzorati, M. Effects of a Short-Term High-Nitrate Diet on Exercise Performance. Nutrients 2016, 8, 534. [Google Scholar] [CrossRef]
- Clifford, T.; Constantinou, C.M.; Keane, K.M.; West, D.J.; Howatson, G.; Stevenson, E.J. The Plasma Bioavailability of Nitrate and Betanin from Beta Vulgaris Rubra in Humans. Eur. J. Nutr. 2017, 56, 1245–1254. [Google Scholar] [CrossRef]
- McIlvenna, L.C.; Monaghan, C.; Liddle, L.; Fernandez, B.O.; Feelisch, M.; Muggeridge, D.J.; Easton, C. Beetroot Juice versus Chard Gel: A Pharmacokinetic and Pharmacodynamic Comparison of Nitrate Bioavailability. Nitric Oxide 2017, 64, 61–67. [Google Scholar] [CrossRef]
- Larsen, F.J.; Ekblom, B.; Sahlin, K.; Lundberg, J.O.; Weitzberg, E. Effects of Dietary Nitrate on Blood Pressure in Healthy Volunteers. N. Engl. J. Med. 2006, 355, 2792–2793. [Google Scholar] [CrossRef]
- Gomez-Cabrera, M.-C.; Borrás, C.; Pallardó, F.V.; Sastre, J.; Ji, L.L.; Viña, J. Decreasing Xanthine Oxidase-Mediated Oxidative Stress Prevents Useful Cellular Adaptations to Exercise in Rats. J. Physiol. 2005, 567(Pt. 1), 113–120. [Google Scholar] [CrossRef]
- Gomez-Cabrera, M.-C.; Martínez, A.; Santangelo, G.; Pallardó, F.V.; Sastre, J.; Viña, J. Oxidative Stress in Marathon Runners: Interest of Antioxidant Supplementation. Br. J. Nutr. 2006, 96 (Suppl. 1), S31–S33. [Google Scholar] [CrossRef]
- Gomez-Cabrera, M.-C.; Domenech, E.; Romagnoli, M.; Arduini, A.; Borras, C.; Pallardo, F.V.; Sastre, J.; Viña, J. Oral Administration of Vitamin C Decreases Muscle Mitochondrial Biogenesis and Hampers Training-Induced Adaptations in Endurance Performance. Am. J. Clin. Nutr. 2008, 87, 142–149. [Google Scholar] [CrossRef]
- Gomez-Cabrera, M.-C.; Domenech, E.; Viña, J. Moderate Exercise Is an Antioxidant: Upregulation of Antioxidant Genes by Training. Free Radic. Biol. Med. 2008, 44, 126–131. [Google Scholar] [CrossRef]
- Vitale, K.C.; Hueglin, S.; Broad, E. Tart Cherry Juice in Athletes: A Literature Review and Commentary. Curr. Sports Med. Rep. 2017, 16, 230–239. [Google Scholar] [CrossRef]
- Bentley, D.J.; Ackerman, J.; Clifford, T.; Slattery, K.S.; Lamprecht, M. Green Tea Catechins and Sport Performance. In Antioxidants in Sport Nutrition; Lamprecht, M., Ed.; CRC Press/Taylor & Francis: Boca Raton, FL, USA, 2015; ISBN-13 978-1-4665-6757-3. [Google Scholar]
- Rourke, S. Drinking Tea: Are the Health Benefits Real? Available online: http://www.medscape.com/viewarticle/907456 (accessed on 3 April 2019).
- Kim, J.; Park, J.; Lim, K. Nutrition Supplements to Stimulate Lipolysis: A Review in Relation to Endurance Exercise Capacity. J. Nutr. Sci. Vitaminol. 2016, 62, 141–161. [Google Scholar] [CrossRef]
- Hursel, R.; Viechtbauer, W.; Dulloo, A.G.; Tremblay, A.; Tappy, L.; Rumpler, W.; Westerterp-Plantenga, M.S. The Effects of Catechin Rich Teas and Caffeine on Energy Expenditure and Fat Oxidation: A Meta-Analysis. Obes. Rev. 2011, 12, e573–e581. [Google Scholar] [CrossRef]
- Palmatier, M.A.; Kang, A.M.; Kidd, K.K. Global Variation in the Frequencies of Functionally Different Catechol-O-Methyltransferase Alleles. Biol. Psychiatry 1999, 46, 557–567. [Google Scholar] [CrossRef]
- Murase, T.; Haramizu, S.; Shimotoyodome, A.; Nagasawa, A.; Tokimitsu, I. Green Tea Extract Improves Endurance Capacity and Increases Muscle Lipid Oxidation in Mice. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2005, 288, R708–R715. [Google Scholar] [CrossRef]
- Murase, T.; Haramizu, S.; Shimotoyodome, A.; Tokimitsu, I.; Hase, T. Green Tea Extract Improves Running Endurance in Mice by Stimulating Lipid Utilization during Exercise. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2006, 290, R1550–R1556. [Google Scholar] [CrossRef]
- Partnerships Informed Choice. Available online: https://www.informed-choice.org/partnerships (accessed on 3 April 2019).
- Paluska, S.A. Caffeine and Exercise. Curr. Sports Med. Rep. 2003, 2, 213–219. [Google Scholar] [CrossRef]
- Goldstein, E.R.; Ziegenfuss, T.; Kalman, D.; Kreider, R.; Campbell, B.; Wilborn, C.; Taylor, L.; Willoughby, D.; Stout, J.; Graves, B.S.; et al. International Society of Sports Nutrition Position Stand: Caffeine and Performance. J. Int. Soc. Sports Nutr. 2010, 7, 5. [Google Scholar] [CrossRef]
- Glaister, M.; Gissane, C. Caffeine and Physiological Responses to Submaximal Exercise: A Meta-Analysis. Int. J. Sports Physiol. Perform. 2018, 13, 402–411. [Google Scholar] [CrossRef]
- Ivy, J.L.; Costill, D.L.; Fink, W.J.; Lower, R.W. Influence of Caffeine and Carbohydrate Feedings on Endurance Performance. Med. Sci. Sports 1979, 11, 6–11. [Google Scholar] [CrossRef]
- Graham, T.E.; Hibbert, E.; Sathasivam, P. Metabolic and Exercise Endurance Effects of Coffee and Caffeine Ingestion. J. Appl. Physiol. 1998, 85, 883–889. [Google Scholar] [CrossRef]
- Graham, T.E.; Spriet, L.L. Metabolic, Catecholamine, and Exercise Performance Responses to Various Doses of Caffeine. J. Appl. Physiol. 1995, 78, 867–874. [Google Scholar] [CrossRef]
- Spriet, L.L. Exercise and Sport Performance with Low Doses of Caffeine. Sports Med. 2014, 44 (Suppl. 2), 175–184. [Google Scholar] [CrossRef]
- Armstrong, L.E.; Casa, D.J.; Maresh, C.M.; Ganio, M.S. Caffeine, Fluid-Electrolyte Balance, Temperature Regulation, and Exercise-Heat Tolerance. Exerc. Sport Sci. Rev. 2007, 35, 135–140. [Google Scholar] [CrossRef]
- Lara, B.; Ruiz-Moreno, C.; Salinero, J.J.; Del Coso, J. Time Course of Tolerance to the Performance Benefits of Caffeine. PLoS ONE 2019, 14. [Google Scholar] [CrossRef]
- Beaumont, R.; Cordery, P.; Funnell, M.; Mears, S.; James, L.; Watson, P. Chronic Ingestion of a Low Dose of Caffeine Induces Tolerance to the Performance Benefits of Caffeine. J. Sports Sci. 2017, 35, 1920–1927. [Google Scholar] [CrossRef]
- Gonçalves, L.S.; Painelli, V.S.; Yamaguchi, G.; Oliveira, L.F.; Saunders, B.; da Silva, R.P.; Maciel, E.; Artioli, G.G.; Roschel, H.; Gualano, B. Dispelling the Myth That Habitual Caffeine Consumption Influences the Performance Response to Acute Caffeine Supplementation. J. Appl. Physiol. 2017, 123, 213–220. [Google Scholar] [CrossRef]
- Talanian, J.L.; Spriet, L.L. Low and Moderate Doses of Caffeine Late in Exercise Improve Performance in Trained Cyclists. Appl. Physiol. Nutr. Metab. 2016, 41, 850–855. [Google Scholar] [CrossRef]
- Nichols, A.W. Probiotics and Athletic Performance: A Systematic Review. Curr. Sports Med. Rep. 2007, 6, 269–273. [Google Scholar] [CrossRef]
- Spence, L.; Brown, W.J.; Pyne, D.B.; Nissen, M.D.; Sloots, T.P.; McCormack, J.G.; Locke, A.S.; Fricker, P.A. Incidence, Etiology, and Symptomatology of Upper Respiratory Illness in Elite Athletes. Med. Sci. Sports Exerc. 2007, 39, 577–586. [Google Scholar] [CrossRef]
- Leite, G.S.F.; Resende Master Student, A.S.; West, N.P.; Lancha, A.H. Probiotics and Sports: A New Magic Bullet? Nutrition 2019, 60, 152–160. [Google Scholar] [CrossRef]
- Coqueiro, A.Y.; de Oliveira Garcia, A.B.; Rogero, M.M.; Tirapegui, J. Probiotic Supplementation in Sports and Physical Exercise: Does It Present Any Ergogenic Effect? Nutr. Health 2017, 23, 239–249. [Google Scholar] [CrossRef]
- Pedersen, D.J.; Lessard, S.J.; Coffey, V.G.; Churchley, E.G.; Wootton, A.M.; Ng, T.; Watt, M.J.; Hawley, J.A. High Rates of Muscle Glycogen Resynthesis after Exhaustive Exercise When Carbohydrate Is Coingested with Caffeine. J. Appl. Physiol. 2008, 105, 7–13. [Google Scholar] [CrossRef]
- Davis, W. Wheat Belly: Lose the Wheat, Lose the Weight, and Find. Your Path Back to Health; Rodale Books: Emmaus, PA, USA, 2014; ISBN 13: 978-1-60961-479-9. [Google Scholar]
- Perlmutter, D. Grain Brain: The Surprising Truth about Wheat, Carbs, and Sugar—Your Brain’s Silent Killers; Hachette: London, UK, 2018; ISBN 13: 978-0316234801. [Google Scholar]
- Piercy, K.L.; Troiano, R.P.; Ballard, R.M.; Carlson, S.A.; Fulton, J.E.; Galuska, D.A.; George, S.M.; Olson, R.D. The Physical Activity Guidelines for Americans. JAMA 2018, 320, 2020–2028. [Google Scholar] [CrossRef]
- Duckworth, A. Grit: The Power of Passion and Perseverance (Vol. 124); Scribner: New York, NY, USA, 2016; ISBN 13: 978-1-5011-1110-5. [Google Scholar]
- What You Need to Know about Dietary Supplements. Available online: https://www.fda.gov/food/dietarysupplements/usingdietarysupplements/ucm109760.htm (accessed on 3 April 2019).
| Nutrient | Daily Requirements | Pre-Exercise | During Exercise | Post-Exercise |
|---|---|---|---|---|
| Carbohydrate | 5–7 g/kg/day (1 h/day) 6–10 g/kg/day (1–3 h/day) 8–12 g/kg/day (4≥ h/day) | 6 g/kg/day (<90 min) 10–12 g/kg/day (> 90 min) + 1–4 g/kg (1–4 h prior to event) | 30–60 g/h (<2.5 h) 60–70 g/h (>2.5 h) 90 g/h (>2.5 h, if tolerable) | 8–10 g/kg/day (first 24 h) 1.0–1.2 g/kg/h (first 3–5 h) or 0.8 g/kg/h + protein (0.3 mg/kg/h) or caffeine (3 mg/kg) |
| Protein | 1.4 g/kg/day 0.3 g/kg every 3–5 h | 0.3 g/kg immediately prior (or post–exercise) | 0.25 g/kg/h (if high intensity/eccentric exercise) | 0.3 g/kg within 0–2 h (or pre-exercise) |
| Fat | Do not restrict to <20% total caloric energy Unclear role of CLA, omega-3, MCT supplements Consider limiting fat intake only during carbohydrate loading, or pre-race if GI comfort concerns | |||
| Water | Try initial hydration plan at ~400–800 mL/h; Adjust according to individual athlete variations (sweat rates, sweat sodium content, exercise intensity, body temperature, ambient temperature, bodyweight, kidney function) Follow thirst mechanism, monitor parameters (bodyweight, urine color) | Replace fluid with 150% of fluid lost | ||
| Sodium | Try initial sodium plan at 300–600 mg/h if high sweat rate (>1.2 L/h), subjective “salty sweater,” or prolonged exercise >2 h Adjust intake according to individual athlete variations (sweat rates, sweat sodium content, exercise intensity, body temperature, ambient temperature, bodyweight, kidney function) | Improved water repletion observed with >60 mmol/L sodium content (~1380 mg/L) | ||
| Nitrates | 300–600 mg of nitrate (up to 10 mg/kg or 0.1 mmol/kg) or 500 mL beetroot juice or 3–6 whole beets within 90 min of exercise onset Consider multi-day dosing e.g., 6 days of a high-nitrate diet prior to event | |||
| Antioxidants | Avoid prior to exercise to maximize training adaptation Take prior to exercise only if recovery needed within 24 h Many options: whole foods, dark berries, dark greens, green tea e.g., 8–12oz tart cherry juice twice a day (1oz if concentrate) 4–5 days prior and 2–3 days after event e.g., green tea extract (270–1200 mg/day) | |||
| Caffeine | 3–6 mg/kg taken 30–90 min prior to exercise Consider “topping-up” every 1–2 h as needed ≥9 mg/kg does not further enhance performance, may have undesirable side effects, + drug test ≤3 mg/kg can also be ergogenic without side effects | 3 mg/kg with carbohydrate enhances glycogen repletion | ||
| Probiotics | Lactobacillus and Bifidobacteria may help with upper respiratory and/or GI symptoms | |||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vitale, K.; Getzin, A. Nutrition and Supplement Update for the Endurance Athlete: Review and Recommendations. Nutrients 2019, 11, 1289. https://doi.org/10.3390/nu11061289
Vitale K, Getzin A. Nutrition and Supplement Update for the Endurance Athlete: Review and Recommendations. Nutrients. 2019; 11(6):1289. https://doi.org/10.3390/nu11061289
Chicago/Turabian StyleVitale, Kenneth, and Andrew Getzin. 2019. "Nutrition and Supplement Update for the Endurance Athlete: Review and Recommendations" Nutrients 11, no. 6: 1289. https://doi.org/10.3390/nu11061289
APA StyleVitale, K., & Getzin, A. (2019). Nutrition and Supplement Update for the Endurance Athlete: Review and Recommendations. Nutrients, 11(6), 1289. https://doi.org/10.3390/nu11061289





