Increment in Dietary Potassium Predicts Weight Loss in the Treatment of the Metabolic Syndrome
Abstract
1. Introduction
2. Methods
2.1. Study Design and Population
2.2. Study Outcomes
2.3. Study and Nutritional Intervention
2.4. Nutritional Assessment
2.5. Quality Control for Nutritional Assessment
2.6. Statistical Analysis
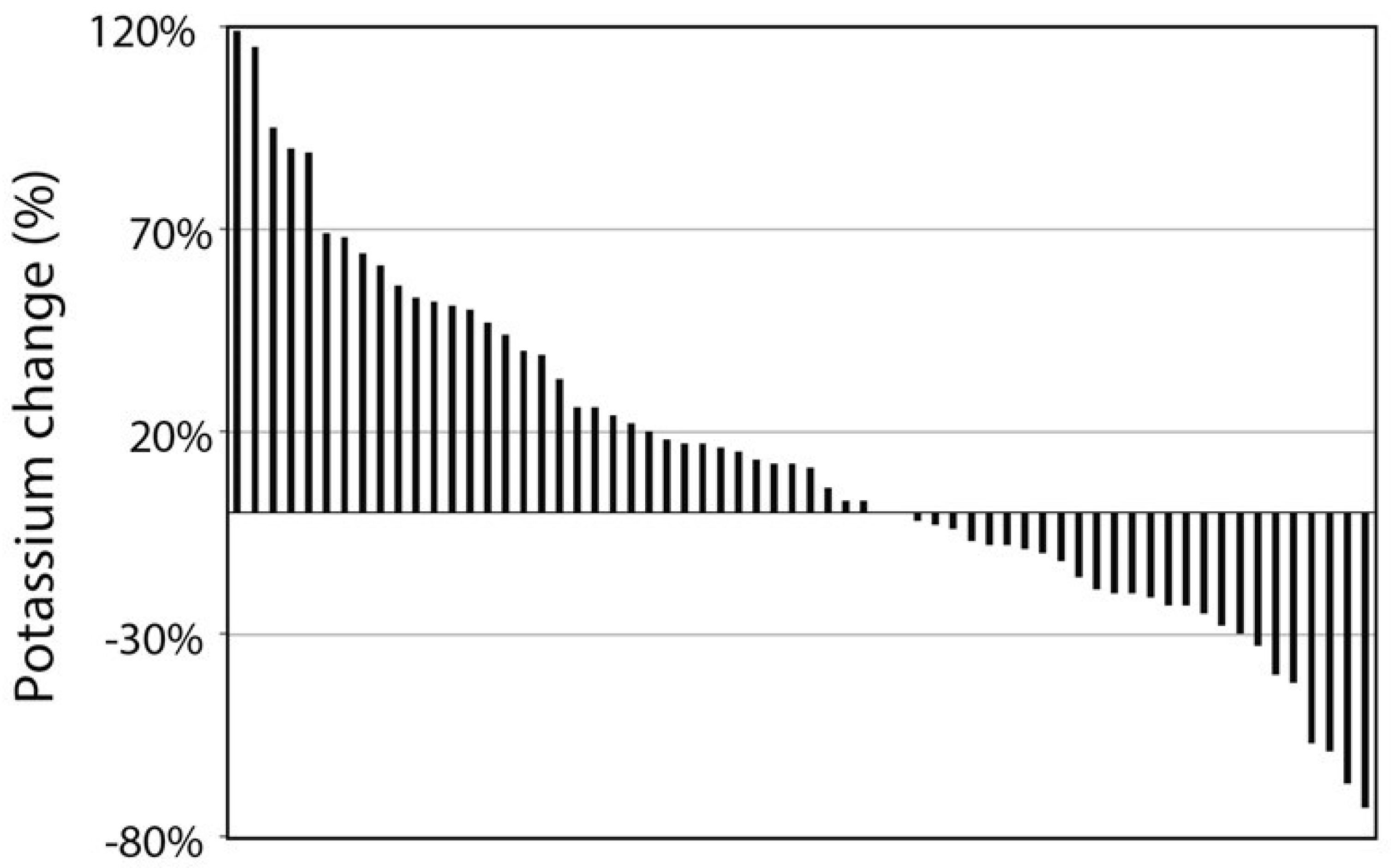
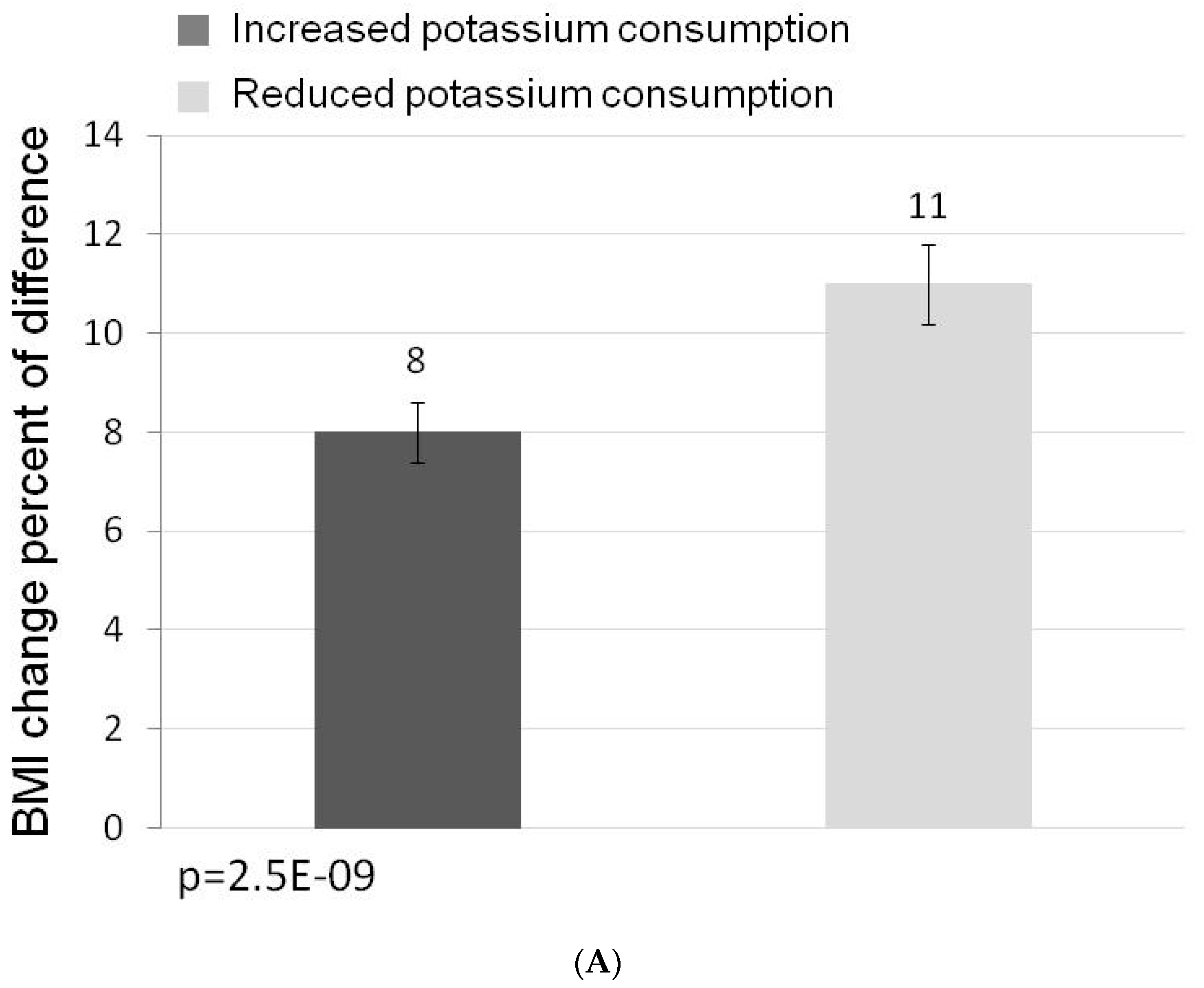
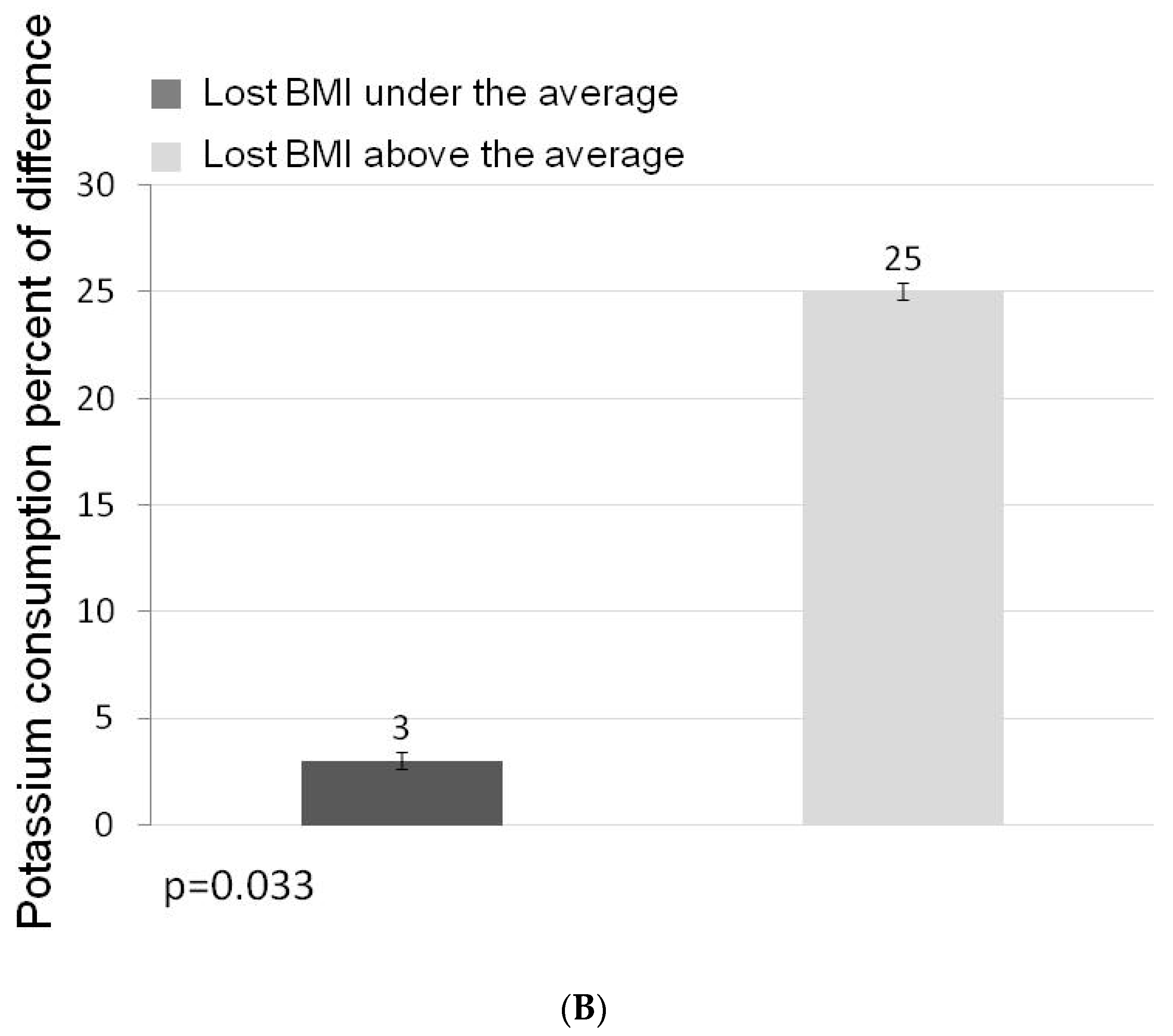
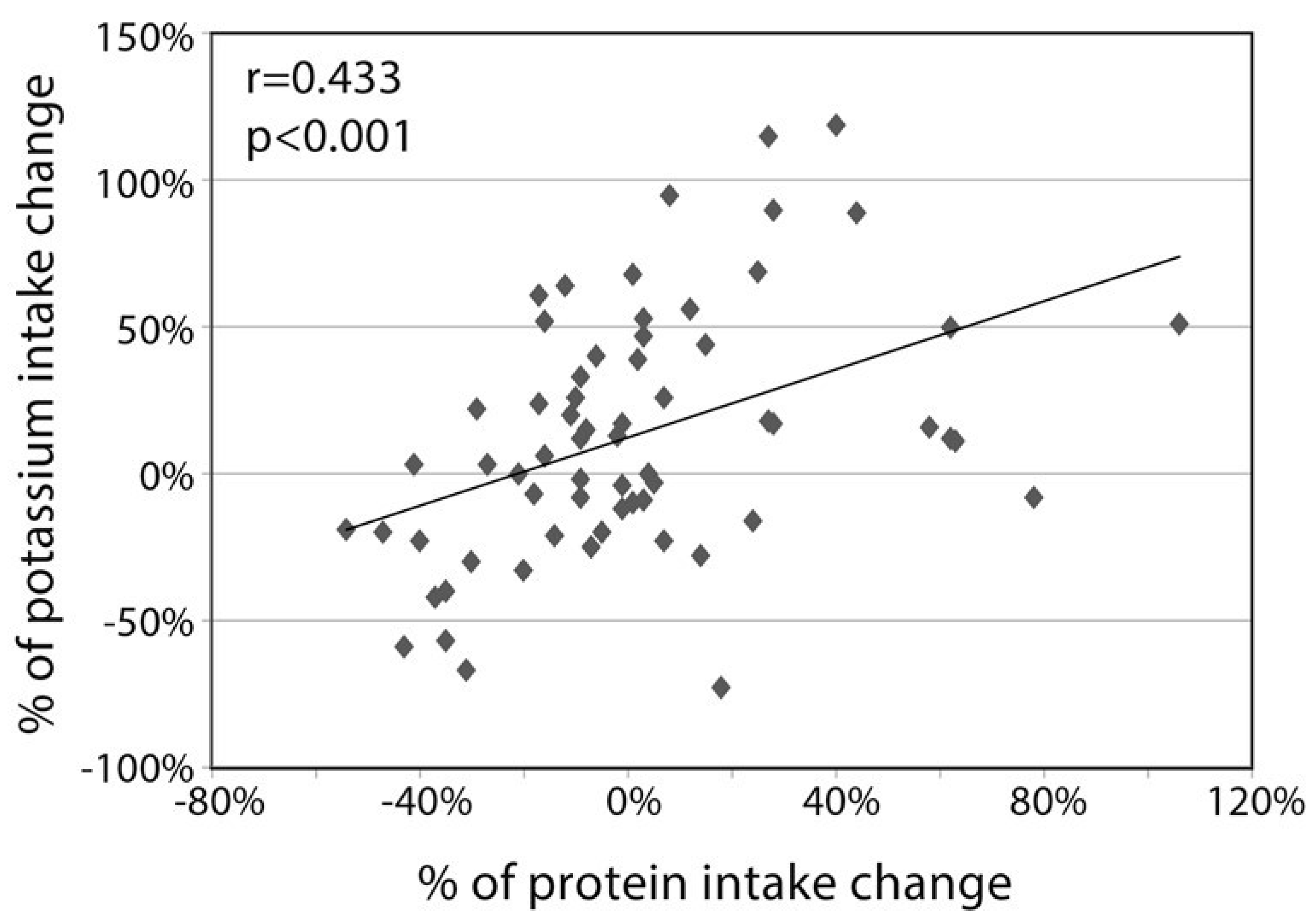
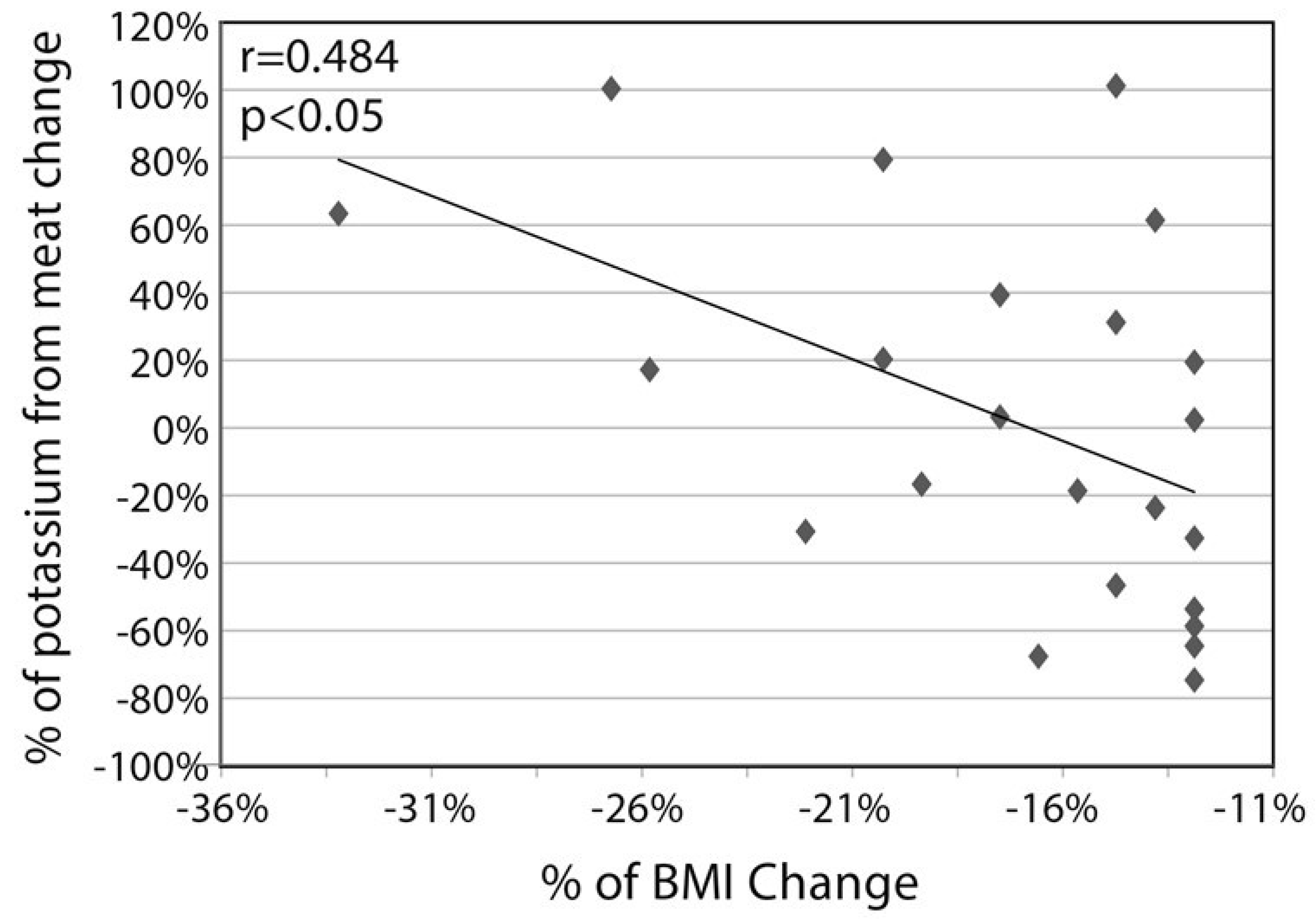
3. Results
3.1. Participants
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Cogswell, M.E.; Zhang, Z.; Carriquiry, A.L.; Gunn, J.P.; Kuklina, E.V.; Saydah, S.H.; Yang, Q.; Moshfegh, A.J. Sodium and potassium intakes among US adults: NHANES 2003–2008. Am. J. Clin. Nutr. 2012, 96, 647–657. [Google Scholar] [CrossRef] [PubMed]
- Institute of Medicine. Dietary Reference Intakes: Water, Potassium, Sodium Chloride, and Sulfate, 1st ed.; National Academy Press: Washington, DC, USA, 2004. [Google Scholar]
- US Department of Health and Human Services; USDA. Dietary Guidelines for Americans, 2010, 7th ed.; USDA: Washington, DC, USA, 2011.
- USDA; Agricultural Research Service. Nutrient Intakes from Food. Mean Amounts Consumed Per Individual, by Gender and Age. What We Eat in America; NHANES 2007–2008 (Updated 14 April 2011). 2010. Available online: www.ars.usda.gov/ba/bhnrc/fsrg (accessed on 10 August 2011).
- Rumawas, M.E.; Dwyer, J.T.; McKeown, N.M.; Meigs, J.B.; Rogers, G.; Jacques, P.F. The development of the Mediterranean-style dietary pattern score and its application to the American diet in the Framingham Offspring Cohort. J. Nutr. 2009, 139, 1150–1156. [Google Scholar] [CrossRef] [PubMed]
- Nowson, C.A.; Worsley, A.; Margerison, C.; Jorna, M.K.; Godfrey, S.J.; Booth, A. Blood pressure change with weight loss is affected by diet type in men. Am. J. Clin. Nutr. 2005, 81, 983–989. [Google Scholar] [CrossRef] [PubMed]
- Blumenthal, J.A.; Babyak, M.A.; Hinderliter, A.; Watkins, L.L.; Craighead, L.; Lin, P.H.; Caccia, C.; Johnson, J.; Waugh, R.; Sherwood, A. Effects of the DASH diet alone and in combination with exercise and weight loss on blood pressure and cardiovascular biomarkers in men and women with high blood pressure: The ENCORE study. Arch. Intern. Med. 2010, 170, 126–135. [Google Scholar] [CrossRef] [PubMed]
- Whelton, P.K.; He, J.; Cutler, J.A.; Brancati, F.L.; Appel, L.J.; Follmann, D.; Klag, M.J. Effects of oral potassium on blood pressure. Meta-analysis of randomized controlled clinical trials. JAMA 1997, 277, 1624–1632. [Google Scholar] [CrossRef] [PubMed]
- He, J.; Gu, D.; Kelly, T.N.; Hixson, J.E.; Rao, D.C.; Jaquish, C.E.; Chen, J.; Zhao, Q.; Gu, C.; Huang, J.; et al. GenSalt Collaborative Research Group Genetic variants in the renin-angiotensin-aldosterone system and blood pressure responses to potassium intake. J. Hypertens. 2011, 29, 1719–1730. [Google Scholar] [CrossRef] [PubMed]
- Marcus, Y.; Segev, E.; Shefer, G.; Sack, J.; Tal, B.; Yaron, M.; Carmeli, E.; Shefer, L.; Margaliot, M.; Limor, R.; et al. Multidisciplinary Treatment of the MSLowers Blood Pressure Variability Independent of Blood Pressure Control. J. Clin. Hypertens. 2016, 18, 19–24. [Google Scholar] [CrossRef] [PubMed]
- National Cholesterol Education Program (NCEP). Third Report of the National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (Adult Treatment Panel III) final report. Circulation 2002, 106, 3143–3421. [Google Scholar] [CrossRef]
- Ganz, T.; Wainstein, J.; Gilad, S.; Limor, R.; Boaz, M.; Stern, N. Serum asymmetric dimethylarginine and arginine levels predict microvascular and macrovascular complications in type 2 diabetes mellitus. Diabetes Metab. Res. Rev. 2017, 33, e2836. [Google Scholar] [CrossRef] [PubMed]
- Cai, X.; Li, X.; Fan, W.; Yu, W.; Wang, S.; Li, Z.; Scott, E.M.; Li, X. Potassium and Obesity/Metabolic Syndrome: A Systematic Review and Meta-Analysis of the Epidemiological Evidence. Nutrients 2016, 183, 8. [Google Scholar] [CrossRef] [PubMed]
- Shin, D.; Joh, H.K.; Kim, K.H.; Park, S.M. Benefits of potassium intake on metabolic syndrome: The fourth Korean national health and nutrition examination survey (KNHANES IV). Atherosclerosis 2013, 230, 80–85. [Google Scholar] [CrossRef] [PubMed]
- Murakami, K.; Livingstone, M.B.; Sasaki, S.; Uenishi, K. Ability of self-reported estimates of dietary sodium, potassium and protein to detect an association with general and abdominal obesity: Comparison with the estimates derived from 24 h urinary excretion. Br. J. Nutr. 2015, 113, 1308–1318. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Hwang, S.S.; Kim, S.; Chin, H.J.; Han, J.S.; Heo, N.J. Potassium intake and the prevalence of metabolic syndrome: The Korean national health and nutrition examination survey 2008–2010. PLoS ONE 2013, 8, e55106. [Google Scholar] [CrossRef] [PubMed]
- Jain, N.; Minhajuddin, A.T.; Neeland, I.J.; Elsayed, E.F.; Vega, G.L.; Hedayati, S.S. Association of urinary sodium-to-potassium ratio with obesity in a multiethnic cohort. Am. J. Clin. Nutr. 2014, 99, 992–998. [Google Scholar] [CrossRef] [PubMed]
- Elfassy, T.; Mossavar-Rahmani, Y.; Van Horn, L.; Gellman, M.; Sotres-Alvarez, D.; Schneiderman, N.; Daviglus, M.; Beasley, J.M.; Llabre, M.M.; Shaw, P.A.; et al. Associations of Sodium and Potassium with Obesity Measures Among Diverse US Hispanic/Latino Adults: Results from the Hispanic Community Health Study/Study of Latinos. Obesity 2018, 26, 442–450. [Google Scholar] [CrossRef] [PubMed]
- Wen, W.; Wan, Z.; Zhou, D.; Zhou, J.; Yuan, Z. The Amelioration of Insulin Resistance in Salt Loading Subjects by Potassium Supplementation is Associated with a Reduction in Plasma IL-17A Levels. Exp. Clin. Endocrinol. Diabetes 2017, 125, 571–576. [Google Scholar] [CrossRef] [PubMed]
- Chatterjee, R.; Colangelo, L.A.; Yeh, H.C.; Anderson, C.A.; Daviglus, M.L.; Liu, K.; Brancati, F.L. Potassium intake and risk of incident type 2 diabetes mellitus: The Coronary Artery Risk Development in Young Adults (CARDIA) Study. Diabetologia 2012, 55, 1295–1303. [Google Scholar] [CrossRef] [PubMed]
- United States Department of Agriculture; Agricultural Research Service. USDA Food Composition Databases. Available online: https://ndb.nal.usda.gov/ndb/nutrients/index (accessed on 1 January 2016).
- Youn, J.H. Gut sensing of potassium intake and its role in potassium homeostasis. Semin. Nephrol. 2013, 33, 248–256. [Google Scholar] [CrossRef] [PubMed]
| Feature (n) | Baseline | 1 Year | p Value a |
|---|---|---|---|
| Age, years (68) | 52 ± 12 | ||
| Weight, kg (68) | 99 ± 17 | 90 ± 17 | <0.000 |
| BMI, kg/m² (68) | 35 ± 4 | 31 ± 4 | <0.000 |
| FBM, Kg (62) | 40 ± 93 | 33 ± 8 | <0.000 |
| FBM, % (62) | 41 ± 1 | 38.4 ± 1 | <0.000 |
| LBM, Kg (62) | 56 ± 12 | 54 ± 12 | <0.000 |
| LBM, % (62) | 59 ± 11 | 62 ± 1 | <0.000 |
| RMR (43) | 1831 ± 404 | 1759 ± 404 | 0.128 |
| ADMA, ug/mL (49) | 0.57 ± 0.17 | 0.45 ± 0.10 | <0.000 |
| Arginine, ug/mL (49) | 42.19 ± 14.17 | 50.30 ± 19.11 | 0.025 |
| Systolic Blood Pressure, mmHg (59) | 125 ± 11 | 122 ± 9 | 0.024 |
| Diastolic Blood Pressure, mmHg (59) | 76 ± 9 | 73 ± 7 | 0.011 |
| Fasting plasma glucose (mg/dL) (57) | 101 ± 15 | 86 ± 14 | <0.000 |
| Total cholesterol (mg/dl) (54) | 191 ± 44 | 172 ± 34 | <0.000 |
| Triglycerides (mg/dl) (55) | 195 ± 74 | 134 ± 66 | <0.000 |
| HDL (mg/dl) (55) | 43 ± 10 | 47 ± 13 | 0.001 |
| LDL (mg/dl) (50) | 108 ± 33 | 98 ± 29 | 0.006 |
| HbA1C (%) (40) | 5.9 ± 0.5 | 5.7 ± 0.4 | 0.004 |
| Food Components (n = 63) | before Treatment | after one Year | p Value a |
|---|---|---|---|
| Food Energy (Kcal/day) | 2999 ± 1071 | 1970 ± 641 | <0.000 |
| % of carbohydrates (of total calories) | 40 ± 11 | 29 ± 8 | <0.000 |
| % of protein (of total calories) | 19 ± 5 | 27 ± 5 | <0.000 |
| % of fat (of total calories) | 39 ± 7 | 41 ± 7 | <0.030 |
| Potassium (mg/day) | 3973 ± 2287 | 3911 ± 1455 | 0.752 |
| Potassium density (mg/Kcal/day) | ± 1.30.5 | 0.5 ± 2 | <0.000 |
| Sodium (mg/day) | 5257 ± 2703 | 4111 ± 793 | <0.000 |
| Step | Predicting Variable | t | β | F change | R2 Change | F | R2 | p |
|---|---|---|---|---|---|---|---|---|
| 1 | Caproic acid, % change | −4.010 | −0.423 *** | 7.412 ** | 0.119 | 7.412 ** | 0.119 | <0.000 |
| 2 | Calcium, % change | 2.87 | 0.335 ** | 6.005 * | 0.082 | 6.658 ** | 0.274 | 0.006 |
| 3 | Food energy, % change | 2.228 | 0.238 * | 4.086 * | 0.053 | 6.305 ** | 0.327 | 0.03 |
| 4 | Potassium, % change | −5.739 | −0.865 *** | 4.659 * | 0.056 | 6.331 *** | 0.383 | <0.000 |
| 5 | Vitamin B6, % of change | 3.87 | 0.542 *** | 9.856 ** | 0.102 | 7.835 *** | 0.485 | <0.000 |
| 6 | Total sugars, % change | 2.374 | 0.239 * | 5.634 * | 0.055 | 8.822 *** | 0.514 | 0.022 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tal, B.; Sack, J.; Yaron, M.; Shefer, G.; Buch, A.; Ben Haim, L.; Marcus, Y.; Shenkerman, G.; Sofer, Y.; Shefer, L.; et al. Increment in Dietary Potassium Predicts Weight Loss in the Treatment of the Metabolic Syndrome. Nutrients 2019, 11, 1256. https://doi.org/10.3390/nu11061256
Tal B, Sack J, Yaron M, Shefer G, Buch A, Ben Haim L, Marcus Y, Shenkerman G, Sofer Y, Shefer L, et al. Increment in Dietary Potassium Predicts Weight Loss in the Treatment of the Metabolic Syndrome. Nutrients. 2019; 11(6):1256. https://doi.org/10.3390/nu11061256
Chicago/Turabian StyleTal, Brurya, Jessica Sack, Marianna Yaron, Gabi Shefer, Assaf Buch, Limor Ben Haim, Yonit Marcus, Galina Shenkerman, Yael Sofer, Lili Shefer, and et al. 2019. "Increment in Dietary Potassium Predicts Weight Loss in the Treatment of the Metabolic Syndrome" Nutrients 11, no. 6: 1256. https://doi.org/10.3390/nu11061256
APA StyleTal, B., Sack, J., Yaron, M., Shefer, G., Buch, A., Ben Haim, L., Marcus, Y., Shenkerman, G., Sofer, Y., Shefer, L., Margaliot, M., & Stern, N. (2019). Increment in Dietary Potassium Predicts Weight Loss in the Treatment of the Metabolic Syndrome. Nutrients, 11(6), 1256. https://doi.org/10.3390/nu11061256




