Vitamin D in Wild and Farmed Atlantic Salmon (Salmo Salar)—What Do We Know?
Abstract
1. Introduction
2. Materials and Method
2.1. Wild Salmon
Sampling
2.2. Farmed Salmon
2.2.1. Experimental Procedure
2.2.2. Sampling
2.3. Review for Data on Vitamin D in Salmonids
2.4. Laboratory Analyses
2.4.1. Vitamin D in Salmon Fillets and Fishfeed
2.4.2. Fat Content in Wild Salmon
2.5. Statistical Analysis
3. Results
3.1. Wild Salmon
3.2. Farmed Salmon
3.2.1. Performance of Fish
3.2.2. Vitamin D Content of Farmed Salmon
3.3. Published Data on Vitamin D in Wild and Farmed Salmonids
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Spiro, A.; Buttriss, J.L. Vitamin D: An overview of vitamin D status and intake in Europe. Nutr. Bull. 2014, 39, 322–350. [Google Scholar] [CrossRef]
- Holick, M.F. Vitamin D and health: Evolution, biologic functions, and recommended dietary intakes for vitamin D. Clin. Rev. Bone Miner. MeTable 2009, 7, 2–19. [Google Scholar] [CrossRef]
- O’Neill, C.M.; Kazantzidis, A.; Ryan, M.J.; Barber, N.; Sempos, C.T.; Durazo-Arvizu, R.A.; Jorde, R.; Grimnes, G.; Eiriksdottir, G.; Gudnason, V.; et al. Seasonal changes in vitamin D-effective UVB availability in Europe and associations with population serum 25-hydroxyvitamin D. Nutrients 2016, 8, 533. [Google Scholar] [CrossRef] [PubMed]
- Cashman, K.D.; Dowling, K.G.; Skrabakova, Z.; Gonzalez-Gross, M.; Valtuena, J.; De Henauw, S.; Moreno, L.; Damsgaard, C.T.; Michaelsen, K.F.; Mølgaard, C.; et al. Vitamin D deficiency in Europe: Pandemic? Am. J. Clin. Nutr. 2016, 103, 1033–1044. [Google Scholar] [CrossRef] [PubMed]
- Nordic Council of Ministers. Nordic Nutrition Recommendations 2012, 5th ed.; Norden: Copenhagen, Denmark, 2012. [Google Scholar]
- Scientific Advisory Committee on Nutrition (SACN). Report on Vitamin D and Health. 2016. Available online: https://www.gov.uk/government/groups/scientific-advisory-committee-on-nutrition (accessed on 11 March 2019).
- EFSA Panel on Dietetic Products, Nutrition and Allergies (NDA). Dietary reference values for vitamin D. EFSA J. 2016, 14, 4547. [Google Scholar]
- Institute of Medicine (IOM). Dietary Reference Intakes for Calcium and Vitamin D; The National Academics Press: Washington, DC, USA, 2011. [Google Scholar]
- Kiely, M.; Black, L.J. Scandinavian journal of clinical and laboratory investigation dietary strategies to maintain adequacy of circulating 25-Hydroxyvitamin D concentrations background and scope. Scand. J. Clin. Lab. Investig. 2012, 72, 14–23. [Google Scholar]
- Cashman, K.D.; Kiely, M. Tackling inadequate vitamin D intakes within the population: Fortification of dairy products with vitamin D may not be enough. Endocrine 2016, 51, 38–46. [Google Scholar] [CrossRef] [PubMed]
- Purser, J.; Forsyth, N. Aquaculture: Farming Aquatic Animals and Plants, 2nd ed.; Lucas, J.S., Southgate, P.C., Eds.; Wiley Blackwell: Chichester, UK, 2012; pp. 313–337. [Google Scholar]
- EUMOFA. The EU Fish Market, 2018. Available online: https://www.eumofa.eu/ (accessed on 25 February 2019).
- FAO. The State of World Fisheries and Aquaculture 2018. Available online: https://www.fao.org/publications (accessed on 5 February 2019).
- Søndergaard, H.; Leerbech, E. The Content of Vitamin D in Danish Foods; Statens Levnedsmiddelinstitute: Søborg, Denmark, 1982. [Google Scholar]
- Ostermeyer, U.; Schmidt, T. Vitamin D and provitamin D in fish. Eur. Food Res. Technol. 2006, 222, 403–413. [Google Scholar] [CrossRef]
- Małesa-Ciećwierz, M.; Usydus, Z. Vitamin D: Can fish food-based solutions be used for reduction of vitamin D deficiency in Poland? Nutrition 2015, 31, 187–192. [Google Scholar] [CrossRef] [PubMed]
- Myhre, J.B.; Borgejordet, Å.; Nordbotten, A.; Løken, E.B.; Fagerli, R.A. Nutritional Composition of Selected Wild and Farmed Raw Fish. Matttilsynet Norwegian Safety Authority: Norway, 2012. Available online: http://www.matportalen.no/verktoy/matvaretabellen/ (accessed on 5 February 2019).
- Jakobsen, J.; Bysted, A.; Langwagen, M.; Nielsen, C.W.; Ygil, K.H.; Trolle, E. Nutrient Content in Fish and Fish Products—With an Extra Focus on Farmed Salmon; National Food Institute, Technical University of Denmark: Lyngby, Denmark, 2019. [Google Scholar]
- Padula, D.; Greenfield, H.; Cunningham, J.; Kiermeier, A.; McLeod, C. Australian seafood compositional profiles: A pilot study. Vitamin D and mercury content. Food Chem. 2016, 193, 106–111. [Google Scholar] [CrossRef] [PubMed]
- USDA Food Composition Databases. Available online: https://ndb.nal.usda.gov/ndb/ (accessed on 4 February 2019).
- Takeuchi, A.; Okano, T.; Tanda, M.; Kobayashi, T. Possible origin of extremely high contents of vitamin D3 in some kinds of fish liver. Comp. Biochem. Physiol. Part. A Physiol. 1991, 100, 483–487. [Google Scholar]
- Lock, E.J.; Waagbø, R.; Wendelaar Bonga, S.; Flik, G. The significance of vitamin D for fish: A review. Aquac. Nutr. 2010, 16, 100–116. [Google Scholar] [CrossRef]
- Jacobsen, J.A.; Hansen, L.P. Feeding habits of wild and escaped farmed Atlantic salmon, Salmo salar L., in the Northeast Atlantic. ICES J. Mar. Sci. 2001, 58, 916–933. [Google Scholar] [CrossRef]
- EFSA Panel on Additives and Products or Substances used in Animal Feed. Scientific opinion on the safety and efficacy of vitamin D3 (cholecalciferol) as a feed additive for chickens for fattening turkeys other poultry, pigs, piglets (suckling), calves. EFSA J. 2012, 10, 2968–2994. [Google Scholar]
- EFSA Panel on Additives and Products or Substances used in Animal Feed. Scientific opinion on the safety and efficacy of vitamin D3 (cholecalciferol) as a feed additive for pigs, piglets, bovines, ovines, calves, equines, chickens for fattening, turkeys. EFSA J. 2013, 11, 3289–3315. [Google Scholar]
- EFSA Panel on Additives and Products or Substances used in Animal Feed. Scientific opinion on the safety and efficacy of vitamin D3(cholecalciferol) as a feed additive for all animal species. EFSA J. 2014, 12, 3568–3592. [Google Scholar] [CrossRef]
- EFSA Panel on Additives and Products or Substances used in Animal Feed. Scientific opinion on the safety of vitamin D3 addition to feedingstuffs for fish. EFSA J. 2017, 15, 4713–4732. [Google Scholar]
- Horvli, O.; Lie, Ø.; Aksnes, L. Tissue distribution of vitamin D3 in Atlantic salmon Salmo salar: Effect of dietary level. Aquac. Nutr. 1998, 4, 127–131. [Google Scholar] [CrossRef]
- Graff, I.E.; Høie, S.; Totland, G.K.; Lie, Ø. Three different levels of dietary vitamin D3 fed to first-feeding fry of Atlantic salmon (Salmo salar L.): Effect on growth, mortality, calcium content and bone formation. Aquac. Nutr. 2002, 8, 103–111. [Google Scholar] [CrossRef]
- Mattila, P.; Piironen, V.; Hakkarainen, T.; Hirvi, T.; UusiRauva, E.; Eskelinen, P. Possibilities to raise vitamin D content of rainbow trout (Oncorhynchus mykiss) by elevated feed cholecalciferol contents. J. Sci. Food Agric. 1999, 79, 195–198. [Google Scholar] [CrossRef]
- DTU Library DTU FindIT. Available online: http://libguides.dtu.dk/ (accessed on 18 February 2019).
- Burild, A.; Frandsen, H.L.; Poulsen, M.; Jakobsen, J. Quantification of physiological levels of vitamin D3 and 25-hydroxyvitamin D3 in porcine fat and liver in subgram sample sizes. J. Sep. Sci. 2014, 37, 2659–2663. [Google Scholar] [CrossRef]
- Barnkob, L.L.; Petersen, P.M.; Nielsen, J.P.; Jakobsen, J. Vitamin D enhanced pork from pigs exposed to artificial UVB light in indoor facilities. Eur. Food Res. Technol. 2019, 245, 411–418. [Google Scholar] [CrossRef]
- Nordisk Metodikkomite for Næringsmidler. Fat. Determination According to SBR (Schmid-Bondyzki-Ratslaff) in Meat and Meat Products; NMKL: Lyngby, Denmark, 1989. [Google Scholar]
- Chen, T.C.; Chimeh, F.; Lu, Z.; Mathieu, J.; Person, K.S.; Zhang, A.; Kohn, N.; Martinello, S.; Berkowitz, R.; Holick, M.F. Factors that influence the cutaneous synthesis and dietary sources of vitamin D. Arch. Biochem. Biophys. 2007, 460, 213–217. [Google Scholar] [CrossRef] [PubMed]
- Jacobsen, J.S.; Leth, T. Overvaagningsprogram for Næringsstoffer, Fisk 2. Runde. ILE 1994.2; National Food Administration: Søborg, Denmark, 1994.
- Mattila, P.; Puronen, V.; Uusi-Rauva, E.; Koivistoinen, P. Cholecalciferol and 25-Hydroxycholecalciferol contents in fish and fish products. J. Food Compos. Anal. 1995, 43, 232–243. [Google Scholar] [CrossRef]
- Jacobsen, J.S.; Knuthsen, P. Food Monitoring System for Nutrients. Fish. 3rd Cycle; National Food Administration: Søborg, Denmark, 1999.
- Roe, M.; Church, S.; Pinchen, H.; Finglas, P. Nutrient Analysis of Fish and Fish Products; Analytical report; Department of Health: London, UK, 2013. Available online: https://www.gov.uk/government/publications (accessed on 5 February 2019).
- Jakobsen, J. Vitamin D in wild salmon—distribution within a salmon and dependence on weight and size. Unpublished (in prep)
- Graff, I.E.; Øyen, J.; Kjellevold, M.; Frøyland, L.; Gjesdal, C.G.; Almås, B.; Rosenlund, G.; Lie, Ø. Reduced bone resorption by intake of dietary vitamin D and K from tailor-made Atlantic salmon: A randomized intervention trial. Oncotarget 2016, 7, 68200–69215. [Google Scholar] [CrossRef]
- Burild, A.; Lauridsen, C.; Faqir, N.; Sommer, H.M.; Jakobsen, J. Vitamin D3 and 25-hydroxyVitamin D3 in pork and their relationship to Vitamin D status in pigs. J. Nutr. Sci. 2016, 5. [Google Scholar] [CrossRef]
- Mattila, P.; Lehikoinen, K.; Kiiskinen, T.; Piironen, V. Cholecalciferol and 25-hydroxycholecalciferol content of chicken egg yolk as affected by the cholecalciferol content of feed. J. Agric. Food Chem. 1999, 47, 4089–4092. [Google Scholar] [CrossRef]
- Duffy, S.K.; O’Doherty, J.V.; Rajauria, G.; Clarke, L.C.; Hayes, A.; Dowling, K.G.; O’Grady, M.N.; Kerry, J.P.; Jakobsen, J.; Cashman, K.D.; et al. Vitamin D-biofortified beef: A comparison of cholecalciferol with synthetic versus UVB-mushroom-derived ergosterol as feed source. Food Chem. 2018, 256, 18–24. [Google Scholar] [CrossRef]
- Wolters, W.R.; Barrows, F.T.; Burr, G.S.; Hardy, R.W. Growth parameters of wild and selected strains of Atlantic salmon, Salmo salar, on two experimental diets. Aquaculture 2009, 297, 136–140. [Google Scholar] [CrossRef]
- Thorarinsdottir, R.I.; Jokumsen, A.; Bjornsson, B.T.; Torrissen, O. Local Raw Materials for Productin of Fish Feed for Aquaculture. Nord. Innov. Proj. no 10102. Nordic Innovation Centre, Norden. 2011. Available online: http://nordicinnovation.org/Global/_Publications/Reports/2011/2011_lokal_raw_material_fish_feed_rep.pdf (accessed on 11 March 2019).
- Ytrestøyl, T.; Aas, T.S.; Åsgård, T. Utilisation of feed resources in production of Atlantic salmon (Salmo salar) in Norway. Aquaculture 2015, 448, 365–374. [Google Scholar] [CrossRef]
- Sprague, M.; Dick, J.R.; Tocher, D.R. Impact of sustainable feeds on omega-3 long chain fatty acid levels in farmed Atlantic salmon, 2006–2015. Sci. Rep. 2016, 6, 21892. [Google Scholar] [CrossRef] [PubMed]
- Sissener, N.H.; Julshamn, K.; Espe, M.; Lunestad, B.T.; Hemre, G.-I.; Waagbø, R.; Måge, A. Surveillance of selected nutrients, additives and undesirables in commercial Norwegian fish feeds in the years 2000-2010. Aqua. Nutr. 2013, 19, 555–572. [Google Scholar] [CrossRef]
- Sele, V.; Sanden, M.; Berntssen, M.H.G.; Lunestad, B.T.; Espe, M.; Lie, K.K.; Amlund, H.; Lundebye, A.-K.; Hemre, G.-I.; Waagbø, R.; et al. Monitoring Program for Fish Feed. Rapport fra Havforskningen, 25-2018. Årsrapport for Prøver Innsamlet i 2017; Havforskningsinstituttet: Bergen, Norway, 2017. [Google Scholar]
- Quesada-Gomez, J.M.; Bouillon, R. Is calcifediol better than cholecalciferol for vitamin D supplementation? Osteoporos. Int. 2018, 29, 1697–1711. [Google Scholar] [CrossRef] [PubMed]
- Jakobsen, J.; Andersen, E.A.W.; Christensen, T.; Andersen, R.; Bügel, S. Vitamin D vitamers affect vitamin D status differently in young healthy males. Nutrients 2018, 10, 12. [Google Scholar] [CrossRef] [PubMed]
- Pedersen, A.N.; Christensen, T.; Matthiessen, J.; Knudsen, V.K.; Rosenlund-Sorensen, M.; Biltoft-Jensen, A.; Hinsch, H.-J.; Ygil, K.H.; Krørup, K.; Saxholt, E.; et al. Dietary Habits in Denmark 2011–2013. Main Results; DTU Fødevareinstitutet: Søborg, Denmark, 2015. [Google Scholar]
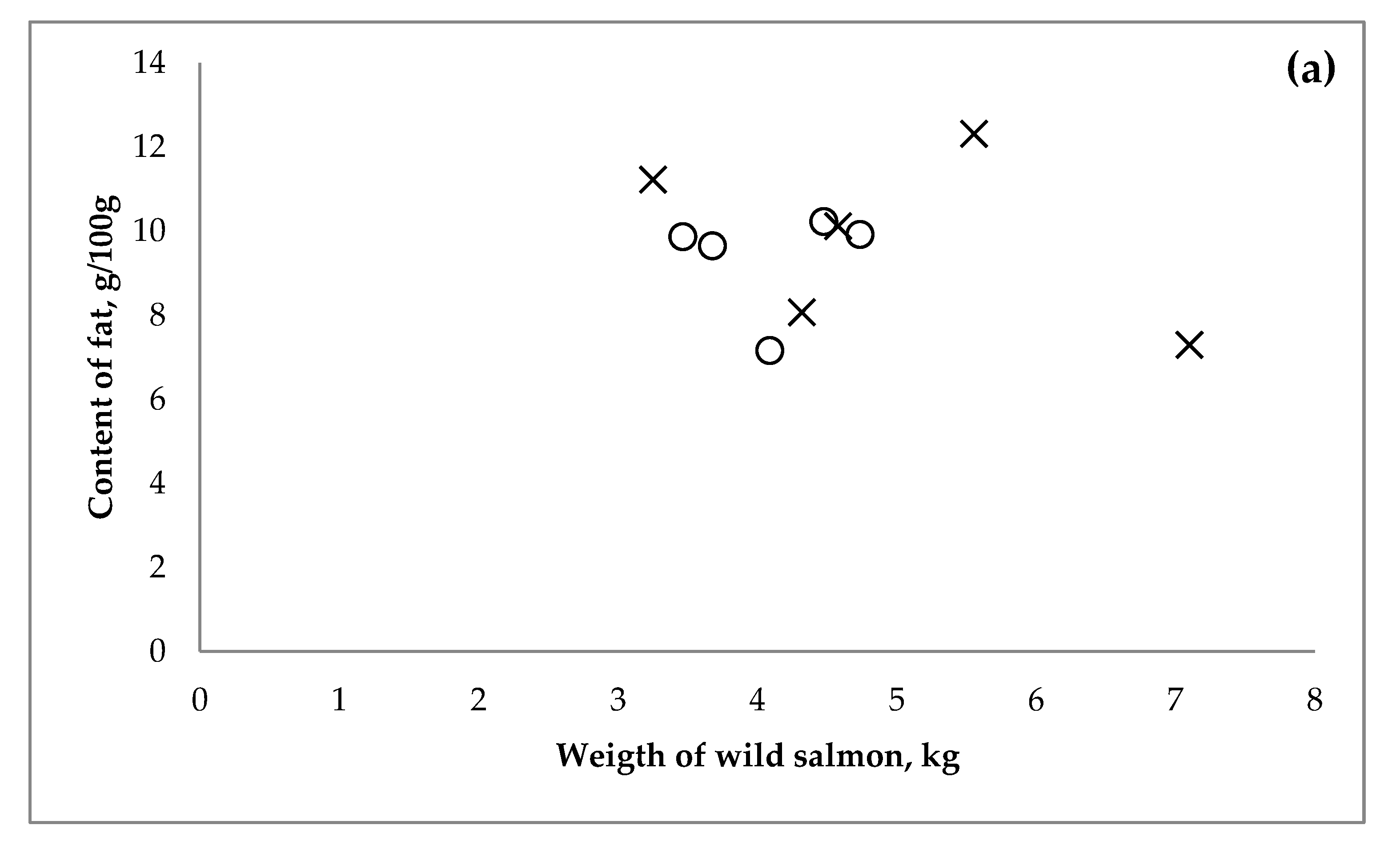
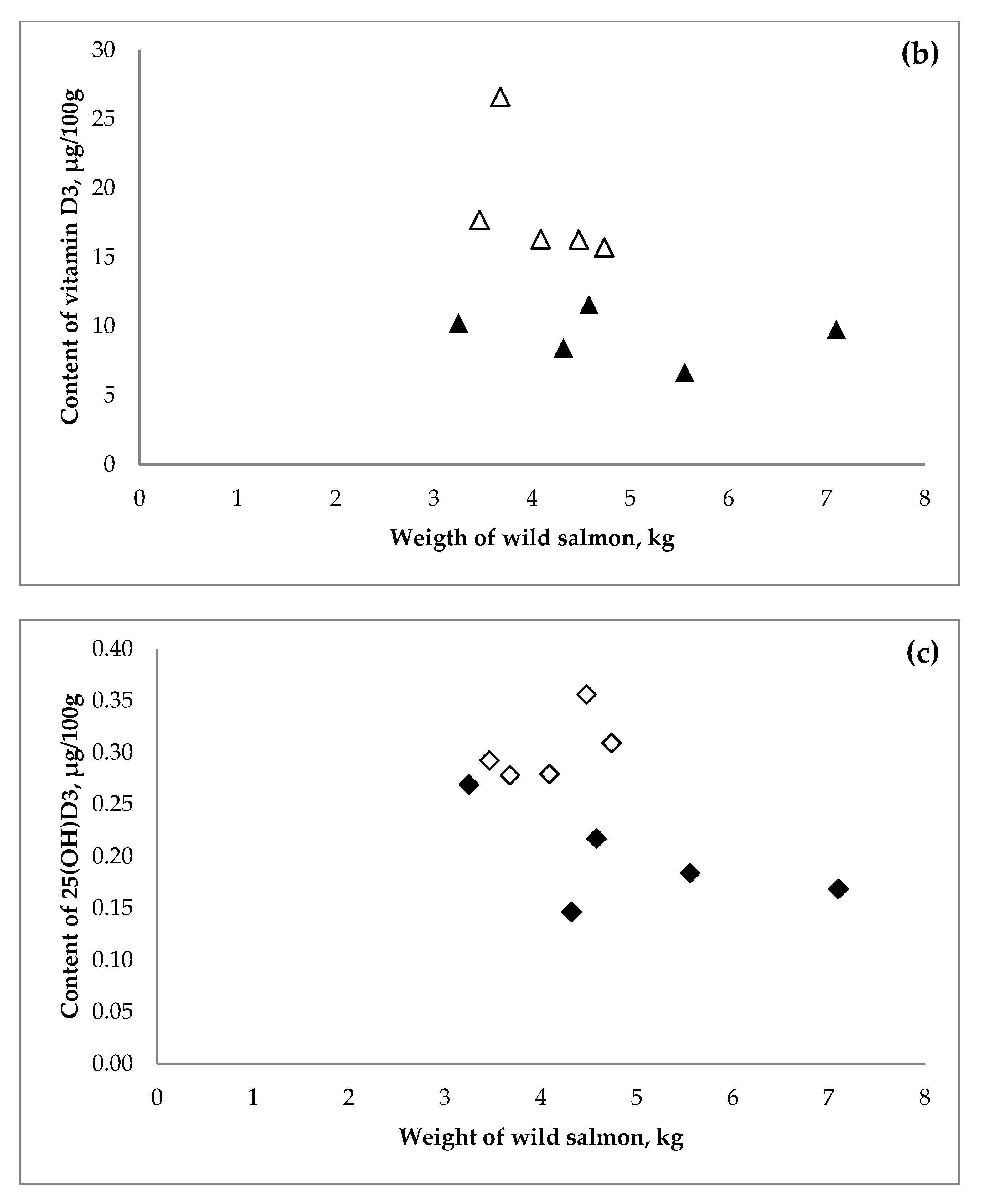
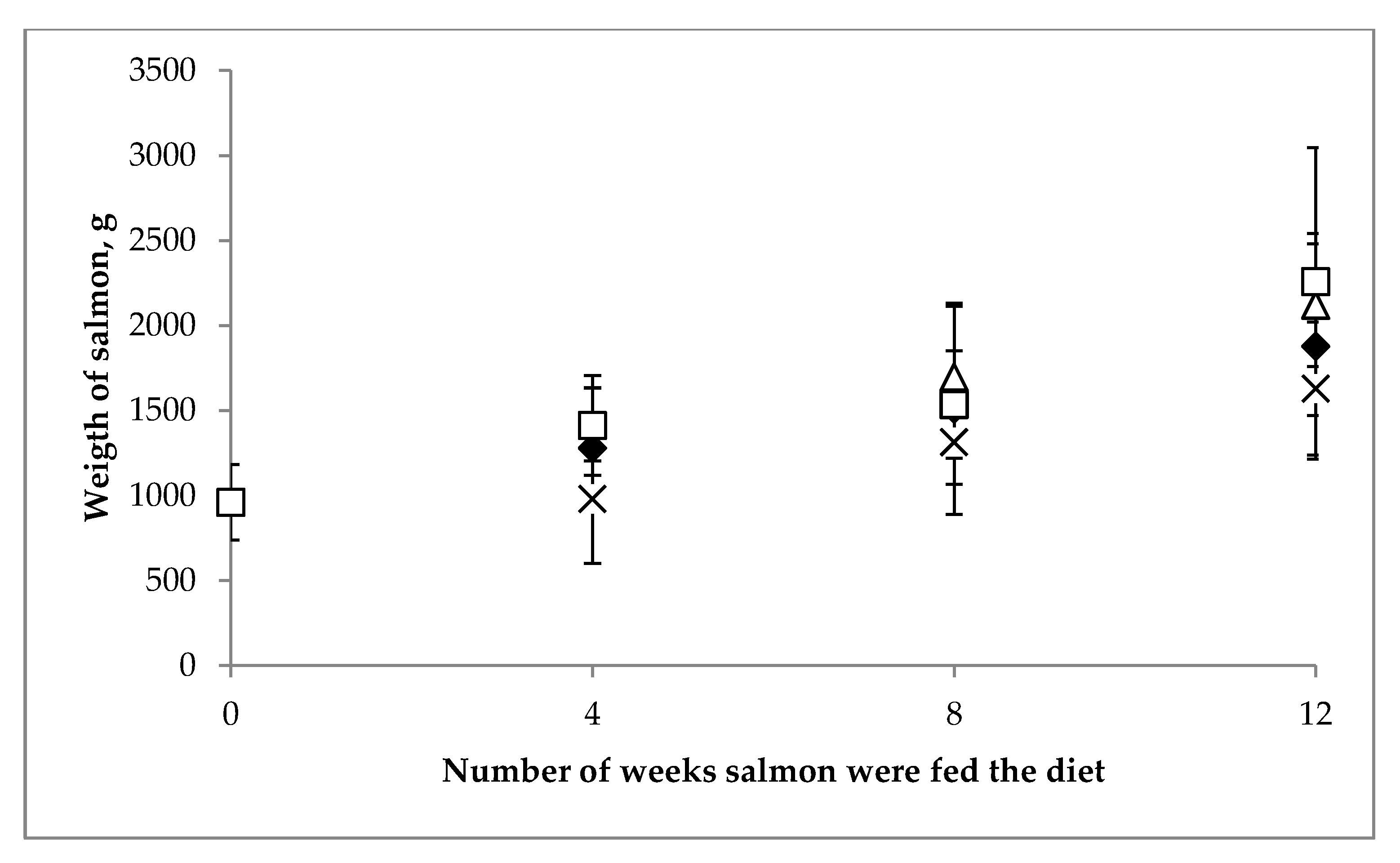
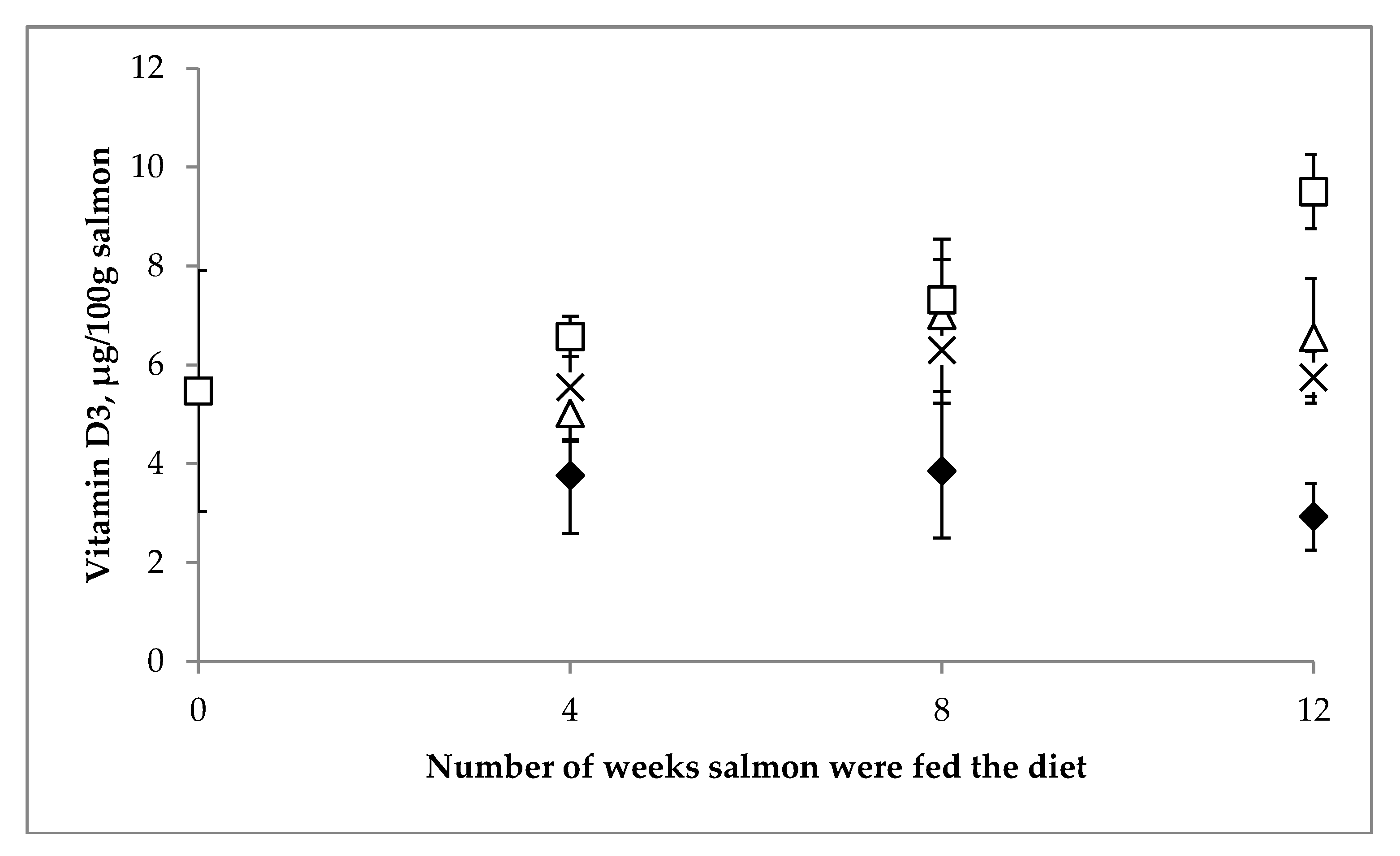
| Year of | Salmon (Salmo Salar) | Salmon (Oncorhynchus sp.) | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Sampling | Wild | Farmed | Wild x | Farmed y | |||||||||
| N | mean ± SD | Range | N | mean ± SD | Range | N | mean ± SD | Range | N | mean ± SD | Range | Ref | |
| 1953–1980 | 3 | 30 ± 24 a,c | 8–55 | [14] | |||||||||
| 1991 | 9 | 13 ± 3 | 8–16 | [36] | |||||||||
| 1993 | * 2 × 10 | 7.6 | 7.3–7.8 | [37] | |||||||||
| 1996 | 11 | 18.7 ± 3.8 | 10–23 | [38] | |||||||||
| 2002–2003 | * 2 × 10 | 9.6 | 8.4–10.7 | * 4 × 10 | 7.6 | 4.2–9.1 | * 4 × 20 | 8.1 | 3.8–10.7 | [15] | |||
| 2007 b | 24 | 6.2 ± 1.0 c | 12 | 9.3 ± 1.6 c | [35] | ||||||||
| 2007–2008 | * 20 | 10 | * 12 | 6.9 | [17] | ||||||||
| 2007–2008 | * 2 × 10 | 6.7 | 6.5–7.0 | * 2 × 10 | 14.2 | 12.7–15.6 | [18] | ||||||
| 2011 | * 9 | 7.0 | [39] | ||||||||||
| 2014 | 5 | 18.5 ± 4.6 | 15.7–26.6 | - | |||||||||
| 2015 b | 10 | 26.5 ± 5.6 | 10 | 5.9 ± 3.6 | 10 | 8.0 ± 3.4 | [16] | ||||||
| 2016 b | 3 | 5.8 | 3.6–8.2 | [19] | |||||||||
| 2016 | 5 | 9.4 ± 1.9 | 6.7–11.6 | - | |||||||||
| 2018 | 12 | 6.0 ± 1.4 | 2.3–7.3 | [18] | |||||||||
| Year of | Salmon (Salmo Salar) | Salmon (Oncorhynchus sp.) | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Sampling | Wild | Farmed | Wild x | Farmed y | |||||||||
| N | mean ± SD | Range | N | mean ± SD | Range | N | mean ± SD | Range | N | mean ± SD | Range | Ref | |
| 2007–2008 | * 20 | 0.49 | * 12 | 0.22 | [17] | ||||||||
| 2007–2008 | * 2 × 10 | 0.38 | 0.37 × 0.38 | * 2 × 10 | 0.054 | <0.1–0.11 | [18] | ||||||
| 2011 | * 9 | 0.18 | [39] | ||||||||||
| 2014 | 5 | 0.30 ± 0.03 | 0.28–0.36 | - | |||||||||
| 2016 b | 3 | nd | [19] | ||||||||||
| 2016 | 5 | 0.22 ± 0.05 | 0.15–0.27 | - | |||||||||
| 2018 | 12 | 0.78 ± 0.30 | 0.37–1.5 | [18] | |||||||||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jakobsen, J.; Smith, C.; Bysted, A.; Cashman, K.D. Vitamin D in Wild and Farmed Atlantic Salmon (Salmo Salar)—What Do We Know? Nutrients 2019, 11, 982. https://doi.org/10.3390/nu11050982
Jakobsen J, Smith C, Bysted A, Cashman KD. Vitamin D in Wild and Farmed Atlantic Salmon (Salmo Salar)—What Do We Know? Nutrients. 2019; 11(5):982. https://doi.org/10.3390/nu11050982
Chicago/Turabian StyleJakobsen, Jette, Cat Smith, Anette Bysted, and Kevin D. Cashman. 2019. "Vitamin D in Wild and Farmed Atlantic Salmon (Salmo Salar)—What Do We Know?" Nutrients 11, no. 5: 982. https://doi.org/10.3390/nu11050982
APA StyleJakobsen, J., Smith, C., Bysted, A., & Cashman, K. D. (2019). Vitamin D in Wild and Farmed Atlantic Salmon (Salmo Salar)—What Do We Know? Nutrients, 11(5), 982. https://doi.org/10.3390/nu11050982





