Vitamin D and Anti-Müllerian Hormone Levels in Infertility Treatment: The Change-Point Problem
Abstract
1. Introduction
2. Material and Methods
2.1. Study Design and Participants
2.2. Ethical Consideration
2.3. Data Collection
2.4. Statistical Analysis
3. Results
3.1. Sample Size Calculation
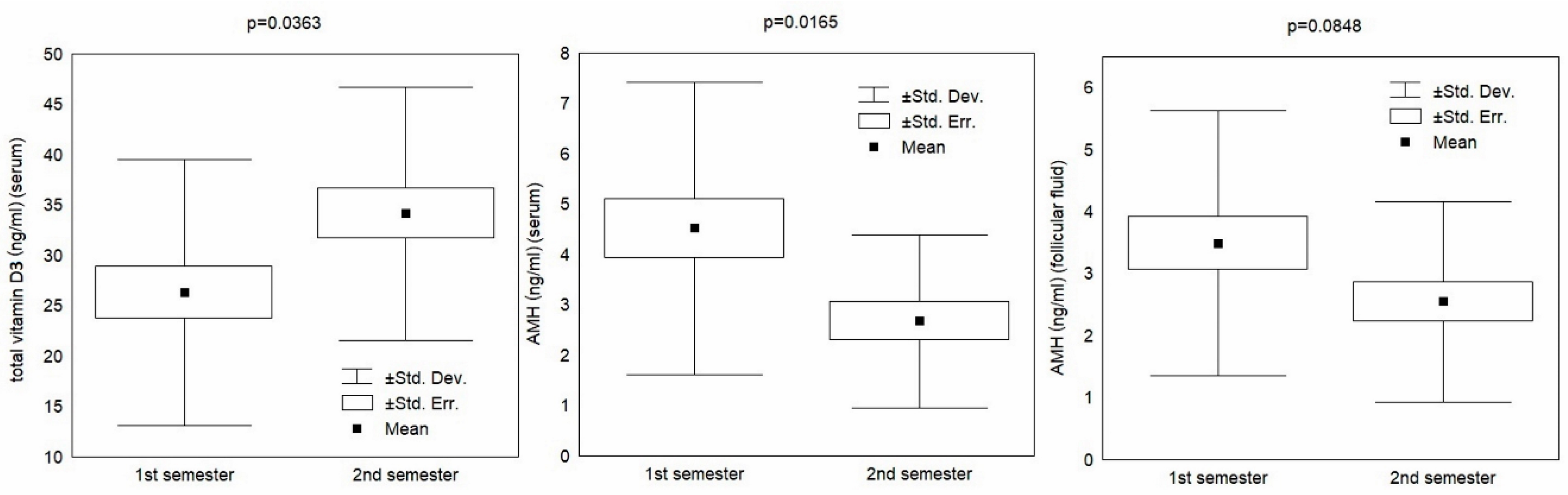
3.2. Student’s t-Test
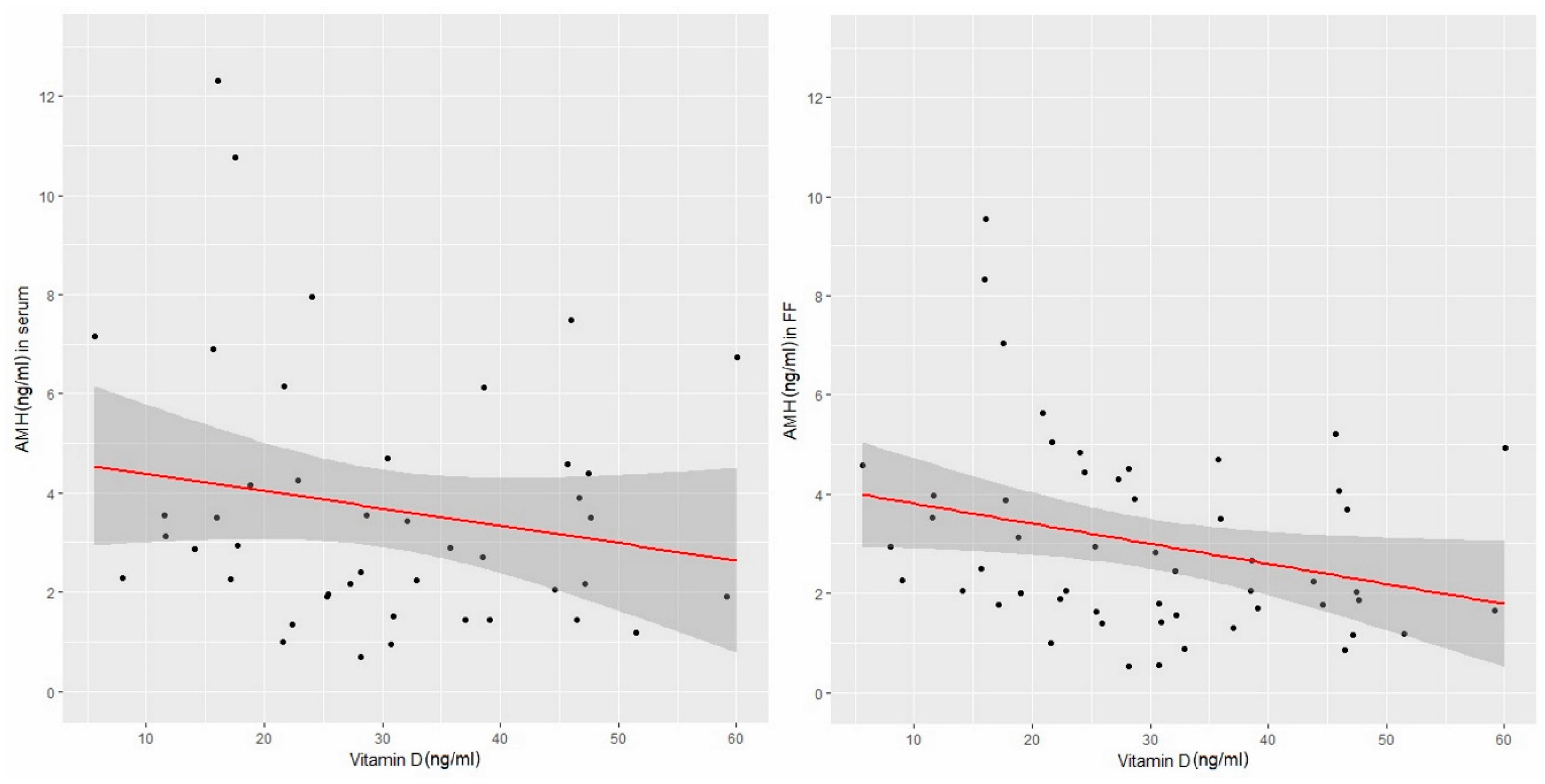
3.3. Pearson’s Linear Correlations
3.4. Change-Point Problem
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- La Marca, A.; Giulini, S.; Orvieto, R.; De Leo, V.; Volpe, A. Anti-Müllerian hormone concentrations in maternal serum during pregnancy. Hum. Reprod. 2005, 20, 1569–1572. [Google Scholar] [CrossRef] [PubMed]
- Dennis, N.A.; Houghton, L.A.; Jones, G.T.; van Rij, A.M.; Morgan, K.; McLennan, I.S. The level of serum anti-Müllerian hormone correlates with vitamin D status in men and women but not in boys. J. Clin. Endocrinol. Metab. 2012, 97, 2450–2455. [Google Scholar] [CrossRef] [PubMed]
- Dumont, A.; Robin, G.; Catteau-Jonard, S.; Dewailly, D. Role of anti-Müllerian hormone in pathophysiology, diagnosis and treatment of polycystic ovary syndrome: A review. Reprod. Biol. Endocrinol. 2015, 13, 137. [Google Scholar] [CrossRef]
- Broer, S.L.; Eijkemans, M.J.; Scheffer, G.J.; van Rooij, I.A.; de Vet, A.; Themmen, A.P.; Laven, J.S.; de Jong, F.H.; Te Velde, E.R.; Fauser, B.C.; et al. Anti-mullerian hormone predicts menopause: A long-term follow-up study in normoovulatory women. J. Clin. Endocrinol. Metab. 2011, 96, 2532–2539. [Google Scholar] [CrossRef] [PubMed]
- Barbakadze, L.; Kristesashvili, J.; Khonelidze, N.; Tsagareishvili, G. The correlations of anti-mullerian hormone, follicle-stimulating hormone and antral follicle count in different age groups of infertile women. Int. J. Fertil. Steril. 2015, 8, 393–398. [Google Scholar] [PubMed]
- von Websky, K.; Hasan, A.A.; Reichetzeder, C.; Tsuprykov, O.; Hocher, B. Impact of vitamin D on pregnancy-related disorders and on offspring outcome. J. Steroid Biochem. Mol. Biol. 2018, 180, 51–64. [Google Scholar] [CrossRef]
- Tsuprykov, O.; Buse, C.; Skoblo, R.; Hocher, B. Comparison of free and total 25-hydroxyvitamin D in normal human pregnancy. J. Steroid Biochem. Mol. Biol. 2019. [Google Scholar] [CrossRef]
- Grzechocinska, B.; Dabrowski, F.A.; Cyganek, A.; Wielgos, M. The role of vitamin D in impaired fertility treatment. Neuroendocrinol. Endocrinol. Lett. 2013, 34, 756–762. [Google Scholar]
- Fabris, A.M.; Cruz, M.; Iglesias, C.; Pacheco, A.; Patel, A.; Patel, J.; Fatemi, H.; García-Velasco, J.A. Impact of vitamin D levels on ovarian reserve and ovarian response to ovarian stimulation in oocyte donors. Reprod. Biomed. Online 2017, 35, 139–144. [Google Scholar] [CrossRef]
- Pearce, K.; Gleeson, K.; Tremellen, K. Serum anti-Mullerian hormone production is not correlated with seasonal fluctuations of vitamin D status in ovulatory or PCOS women. Hum. Reprod. 2015, 30, 2171–2177. [Google Scholar] [CrossRef]
- Irani, M.; Merhi, Z. Role of vitamin D in ovarian physiology and its implication in reproduction: A systematic review. Fertil. Steril. 2014, 102, 460–468. [Google Scholar] [CrossRef] [PubMed]
- Revelli, A. Follicular fluid content and oocyte quality: From single biochemical markers tometabolomics. Reprod. Biol. Endocrinol. 2009, 7, 40. [Google Scholar] [CrossRef] [PubMed]
- Khodadadi, A.; Asghariany, M. Change-point Problem and Regression: An Annotated Bibliography. Collection of Biostatistics Research Archiv; The Berkeley Electronic Press: Berkeley, CA, USA, 2008; p. 44. [Google Scholar]
- Hulley, S.B.; Cummings, S.R.; Browner, W.S.; Grady, D.G.; Newman, T.B. Designing Clinical Research: An Epidemiologic Approach, 4th ed.; Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2013; Appendix 6C:79. [Google Scholar]
- Holick, M.F.; Binkley, N.C.; Bischoff-Ferrari, H.A.; Gordon, C.M.; Hanley, D.A.; Heaney, R.P.; Murad, M.H.; Weaver, C.M. Evaluation, treatment, and prevention of vitamin D deficiency: An Endocrine Society clinical practice guideline. J. Clin. Endocrinol. Metab. 2011, 96, 1911–1930. [Google Scholar] [CrossRef]
- Muggeo, V.M.R. Segmented: An R package to fit regression models with broken-line relationships. R News 2008, 8, 20–25. [Google Scholar]
- Ozkan, S.; Jindal, S.; Greenseid, K.; Shu, J.; Zeitlian, G.; Hickmon, C.; Pal, L. Replete vitamin D stores predict reproductive success following in vitro fertilization. Fertil. Steril. 2010, 94, 1314–1319. [Google Scholar] [CrossRef] [PubMed]
- Paffoni, A.; Ferrari, S.; Viganò, P.; Pagliardini, L.; Papaleo, E.; Candiani, M.; Tirelli, A.; Fedele, L.; Somigliana, E. Vitamin D deficiency and infertility: Insights from in vitro fertilization cycles. J. Clin. Endocrinol. Metab. 2014, 99, E2372–E2376. [Google Scholar] [CrossRef]
- Dixon, K.M.; Mason, R.S. Vitamin D. Int. J. Biochem. Cell Biol. 2009, 5, 982–985. [Google Scholar] [CrossRef]
- Rojansky, N.; Brzezinski, A.; Schenker, J.G. Seasonality in human reproduction: An update. Hum. Reprod. 1992, 7, 735–745. [Google Scholar] [CrossRef]
- Dennis, N.A.; Houghton, L.A.; Pankhurst, M.W.; Harper, M.J.; McLennan, I.S. Acute supplementation with high dose vitamin D3 increases serum anti-Müllerian hormone in young women. Nutrients 2017, 9, 719. [Google Scholar] [CrossRef]
- Merhi, Z.; Doswell, A.; Krebs, K.; Cipolla, M. Vitamin D alters genes involved in follicular development and steroidogenesis in human cumulus granulosa cells. J. Clin. Endocrinol. Metab. 2014, 99, E1137–E1145. [Google Scholar] [CrossRef]
- Malloy, P.J.; Peng, L.; Wang, J.; Feldman, D. Interaction of the vitamin D receptor with a vitamin D response element in the Mullerian-inhibiting substance (MIS) promoter: Regulation of MIS expression by calcitriol in prostate cancer cells. Endocrinology 2008, 150, 1580–1587. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.M.; Donahoe, P.K.; Hasegawa, T.; Silverman, B.; Crist, G.B.; Best, S.; Hasegawa, Y.; Noto, R.A.; Schoenfeld, D.; MacLaughlin, D.T. Mullerian inhibiting substance in humans: Normal levels from infancy to adulthood. J. Clin. Endocrinol. Metab. 1996, 81, 571–576. [Google Scholar] [PubMed]
- Xu, J.; Hennebold, J.D.; Seifer, D.B. Direct vitamin D3 actions on rhesus macaque follicles in three-dimensional culture: Assessment of follicle survival, growth, steroid, and antimüllerian hormone production. Fertil. Steril. 2016, 106, 1815–1820. [Google Scholar] [CrossRef] [PubMed]
- Laganà, A.S.; Vitale, S.G.; Ban Frangež, H.; Vrtačnik-Bokal, E.; D’Anna, R. Vitamin D in human reproduction: The more, the better? An evidence-based critical appraisal. Eur. Rev. Med. Pharmacol. Sci. 2017, 21, 4243–4251. [Google Scholar]
- Hong, S.H.; Lee, J.E.; Kim, H.S.; Jung, Y.J.; Hwang, D.; Lee, J.H.; Yang, S.Y.; Kim, S.C.; Cho, S.K.; An, B.S. Effect of vitamin D3 on production of progesterone in porcine granulosa cells by regulation of steroidogenic enzymes. J. Biomed. Res. 2016, 30, 203–208. [Google Scholar]
- Parikh, G.; Varadinova, M.; Suwandhi, P.; Araki, T.; Rosenwaks, Z.; Poretsky, L.; Seto-Young, D. Vitamin D regulates steroidogenesis and insulin-like growth factor binding protein-1 (IGFBP-1) production in human ovarian cells. Horm. Metab. Res. 2010, 42, 754–757. [Google Scholar] [CrossRef]
- Aleyasin, A.; Hosseini, M.A.; Mahdavi, A.; Safdarian, L.; Fallahi, P.; Mohajeri, M.R.; Abbasi, M.; Esfahani, F. Predictive value of the level of vitamin D in follicular fluid on the outcome of assisted reproductive technology. Eur. J. Obstet. Gynecol. Reprod. Biol. 2011, 159, 132–137. [Google Scholar] [CrossRef] [PubMed]
- Firouzabadi, R.D.; Rahmani, E.; Rahsepar, M.; Firouzabadi, M.M. Value of follicular fluid vitamin D in predicting the pregnancy rate in an IVF program. Arch. Gynecol. Obstet. 2014, 289, 201–206. [Google Scholar] [CrossRef] [PubMed]
- Estes, S.J.; Ye, B.; Qiu, W.; Cramer, D.; Hornstein, M.D.; Missmer, S.A. A proteomic analysis of IVF follicular fluid in women ≤32 years old. Fertil Steril. 2009, 92, 1569–1578. [Google Scholar]
- Ciepiela, P.; Dulęba, A.J.; Kowaleczko, E.; Chełstowski, K.; Kurzawa, R. Vitamin D as a follicular marker of human oocyte quality and a serum marker of in vitro fertilization outcome. J. Assist. Reprod. Genet. 2018, 35, 1265–1276. [Google Scholar] [CrossRef]
- Skrobot, A.; Demkow, U.; Wachowska, M. Immunomodulatory Role of Vitamin D: A Review. Adv. Exp. Med. Biol. 2018, 1108, 13–23. [Google Scholar] [PubMed]


| Overall | 1st Semester | 2nd Semester | t-Test | ||
|---|---|---|---|---|---|
| Origin | Biomarker | Mean ± SD | Mean ± SD | Mean ± SD | p-value |
| Serum | vitamin D total (ng/mL) | 29.7 ± 13.3 | 26.3 ± 13.2 | 34.2 ± 12.6 | 0.0363 |
| AMH (ng/mL) | 3.68 ± 2.58 | 4.52 ± 2.90 | 2.67 ± 1.72 | 0.0165 | |
| FSH (mIU/mL) | 6.87 ± 1.74 | 6.61 ± 1.79 | 7.25 ± 1.66 | 0.2476 | |
| LH (mIU/mL) | 6.12 ± 2.16 | 6.25 ± 1.74 | 5.94 ± 2.66 | 0.6611 | |
| estradiol (pg/mL) | 1269 ± 754 | 1447 ± 662 | 1076 ± 814 | 0.0957 | |
| Follicular Fluid | AMH (ng/mL) | 3.01 ± 1.90 | 3.49 ± 2.14 | 2.55 ± 1.62 | 0.0848 |
| FSH (mIU/mL) | 4.75 ± 2.04 | 4.34 ± 1.73 | 4.87 ± 2.03 | 0.3255 | |
| LH (mIU/mL) | 0.81 ± 0.82 | 0.74 ± 0.66 | 0.77 ± 0.90 | 0.9174 | |
| estradiol (µg/mL) | 542 ± 419 | 492 ± 346 | 568 ± 496 | 0.5344 |
| Vitamin D Total | ≤30 ng/mL | >30 ng/mL | t-test |
|---|---|---|---|
| Patient Characteristics | Mean ± SD | Mean ± SD | p-value |
| Age | 34.1 ± 4.2 | 35.4 ± 4 | 0.2782 |
| BMI (kg/m2) | 22.5 ± 2.9 | 21.8 ± 2.8 | 0.5208 |
| AMH (ng/mL) (serum) | 4.14 ± 3.05 | 3.18 ± 1.90 | 0.2137 |
| AMH (ng/mL) (follicular fluid) | 3.63 ± 2.14 | 2.32 ± 1.32 | 0.0094 |
| AMH | r (CI 95%) | p-value |
|---|---|---|
| serum | −0.19 (−0.46, 0.12) | 0.2211 |
| follicular fluid | −0.28 (−0.51, 0.02) | 0.0391 |
| AMH Origin | Regression Parameter | Mean (CI 95%) | p-value |
|---|---|---|---|
| Serum | change-point | 31 (15,47) | 0.0001 |
| slope I | −0.14 (0.00, 0.28) | 0.0605 | |
| slope II | 0.05 (−0.11, 0.21) | 0.5017 | |
| Follicular Fluid | change-point | 33 (15, 51) | 0.0003 |
| slope I | −0.09 (−0.17, −0.01) | 0.0340 | |
| slope II | 0.02 (−0.10, 0.14) | 0.7432 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bednarska-Czerwińska, A.; Olszak-Wąsik, K.; Olejek, A.; Czerwiński, M.; Tukiendorf, A. Vitamin D and Anti-Müllerian Hormone Levels in Infertility Treatment: The Change-Point Problem. Nutrients 2019, 11, 1053. https://doi.org/10.3390/nu11051053
Bednarska-Czerwińska A, Olszak-Wąsik K, Olejek A, Czerwiński M, Tukiendorf A. Vitamin D and Anti-Müllerian Hormone Levels in Infertility Treatment: The Change-Point Problem. Nutrients. 2019; 11(5):1053. https://doi.org/10.3390/nu11051053
Chicago/Turabian StyleBednarska-Czerwińska, Anna, Katarzyna Olszak-Wąsik, Anita Olejek, Michał Czerwiński, and Andrzej Tukiendorf. 2019. "Vitamin D and Anti-Müllerian Hormone Levels in Infertility Treatment: The Change-Point Problem" Nutrients 11, no. 5: 1053. https://doi.org/10.3390/nu11051053
APA StyleBednarska-Czerwińska, A., Olszak-Wąsik, K., Olejek, A., Czerwiński, M., & Tukiendorf, A. (2019). Vitamin D and Anti-Müllerian Hormone Levels in Infertility Treatment: The Change-Point Problem. Nutrients, 11(5), 1053. https://doi.org/10.3390/nu11051053






