Naked Oat (Avena nuda L.) Oligopeptides: Immunomodulatory Effects on Innate and Adaptive Immunity in Mice via Cytokine Secretion, Antibody Production, and Th Cells Stimulation
Abstract
:1. Introduction
2. Materials and Methods
2.1. OOPs Preparation
2.2. Animals and Treatment
2.3. Sample Collection and Preparation
2.4. Assessment Protocols of Immunomodulatory Activity
2.4.1. Phagocytic Capacity Assessment of Peritoneal Macrophages on Chicken Red Blood Cell (CRBCs) Clearance
2.4.2. The Carbon Clearance Test
2.4.3. Spleen Natural Killer (NK) Cell Activity Analysis
2.4.4. Delayed-Type Hypersensitivity (DTH) Reaction, Immunoglobulin (Ig) M-Plaque-Forming Cell (IgM-PFC) Test, and Serum Hemolysin Level Measurement
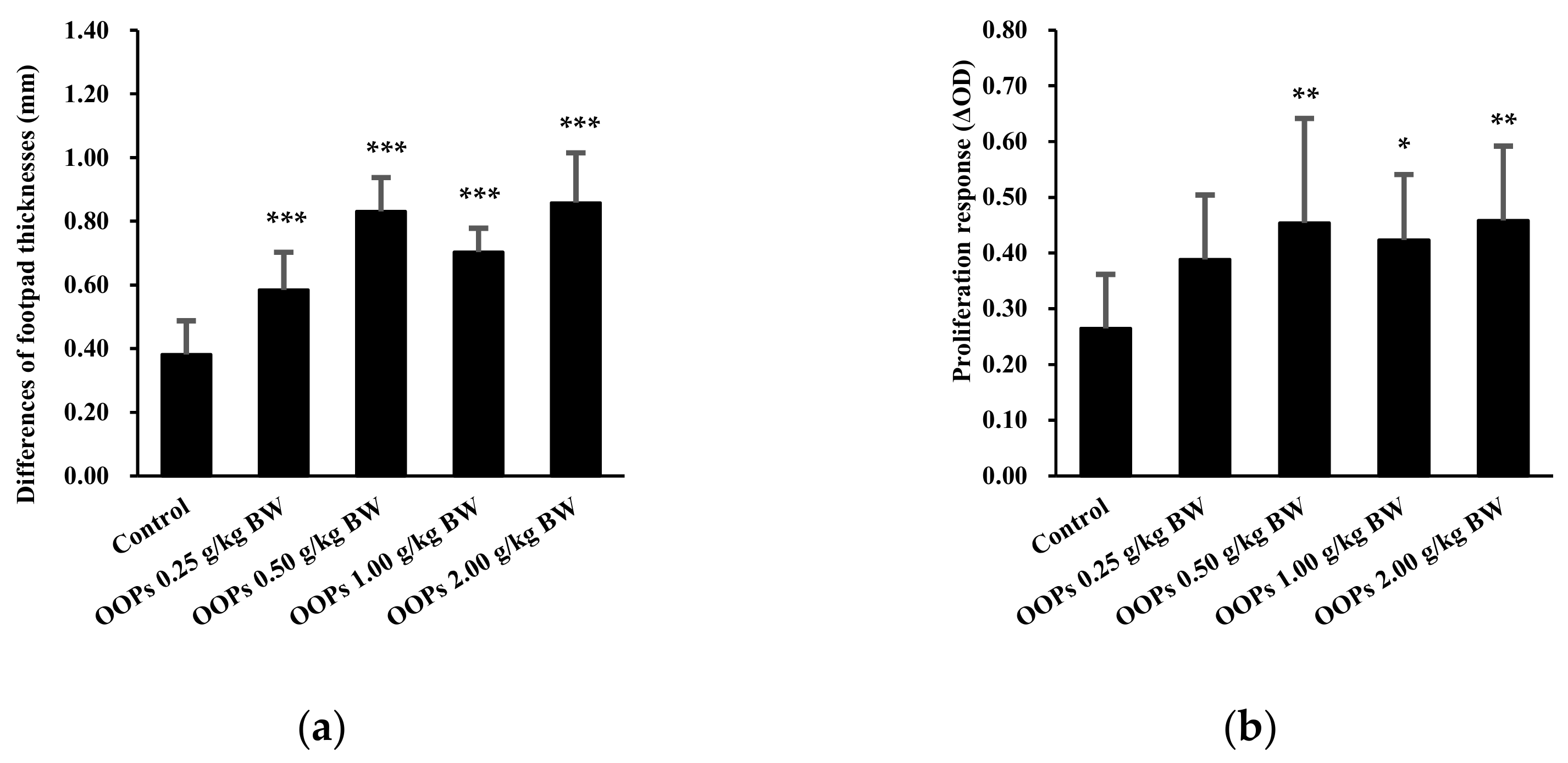
2.4.5. Splenocyte proliferation assay
2.4.6. Splenic T Lymphocyte Subpopulation Assay
2.4.7. Multiplex Sandwich Immunoassay of Cytokine and Immunoglobulin Levels
2.5. Statistical Analysis
3. Results
3.1. Effects of OOPs on Body Weight and Immune Organs
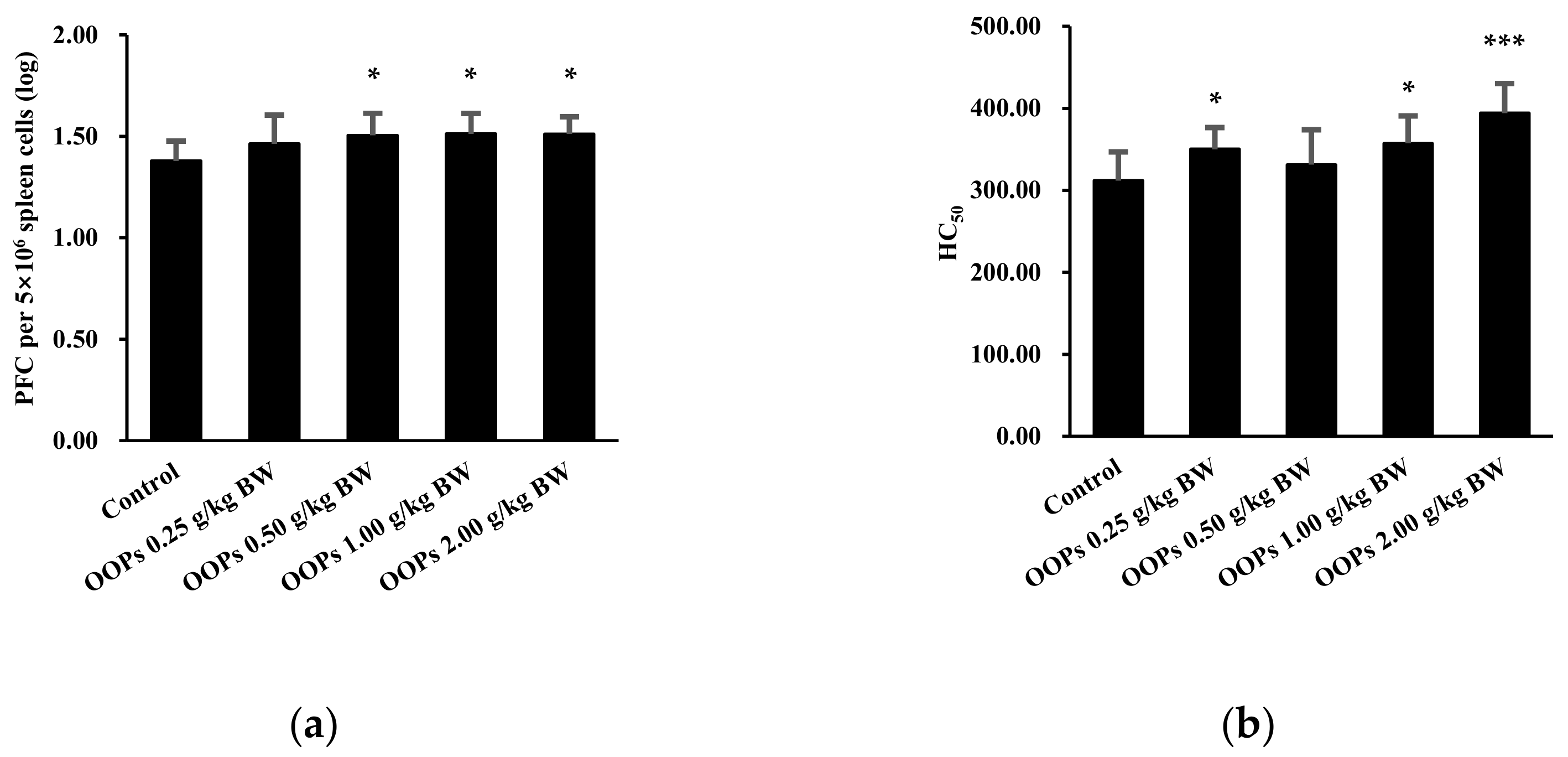
3.2. Effects of OOPs on Humoral Immunity
3.3. Effects of OOPs on Cell-Mediated Immunity
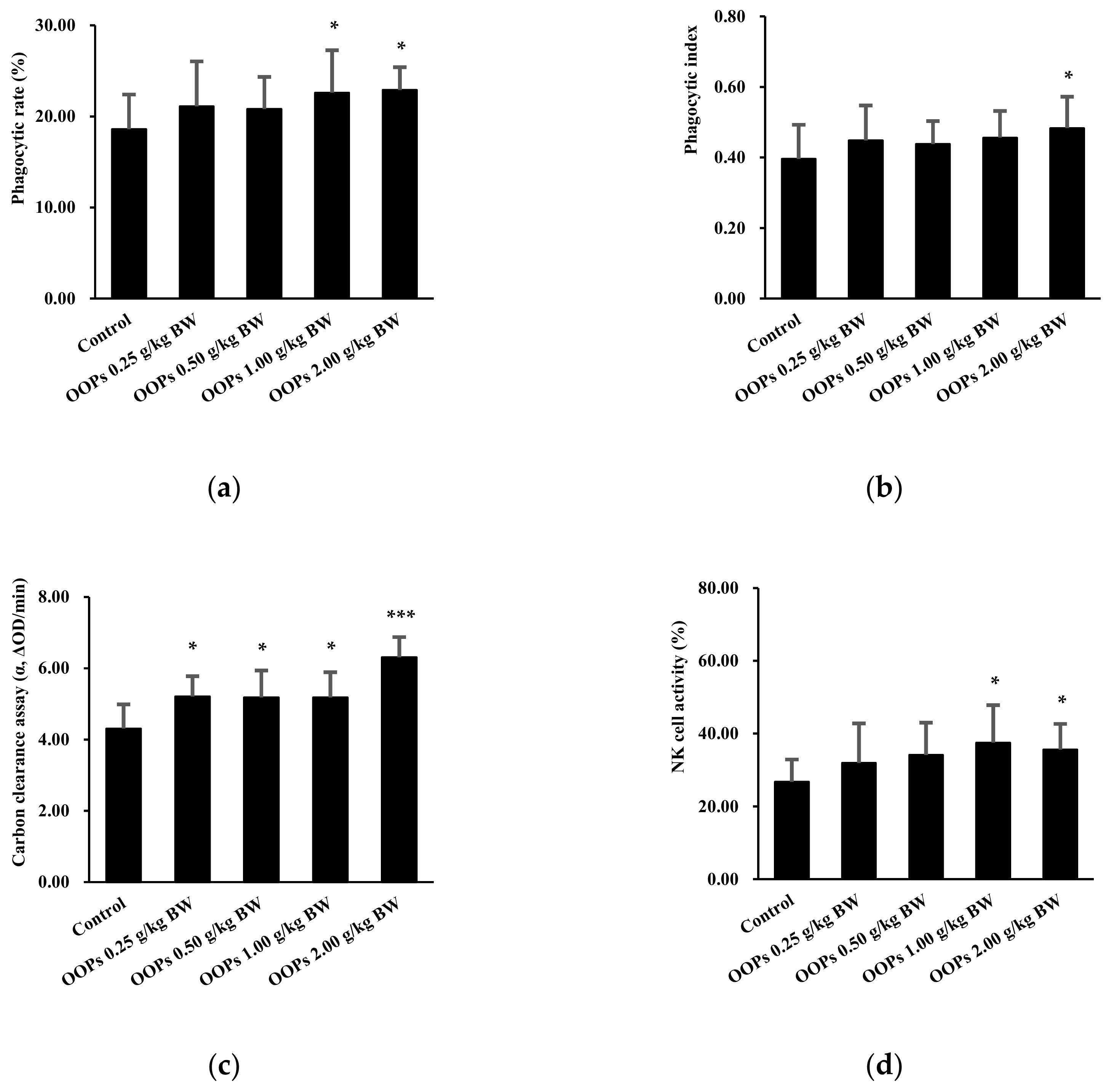
3.4. Effects of OOPs on Macrophage Phagocytosis
3.5. Effects of OOPs on NK Cell Activity
3.6. Effects of OOPs on Splenic T Lymphocyte Subpopulations
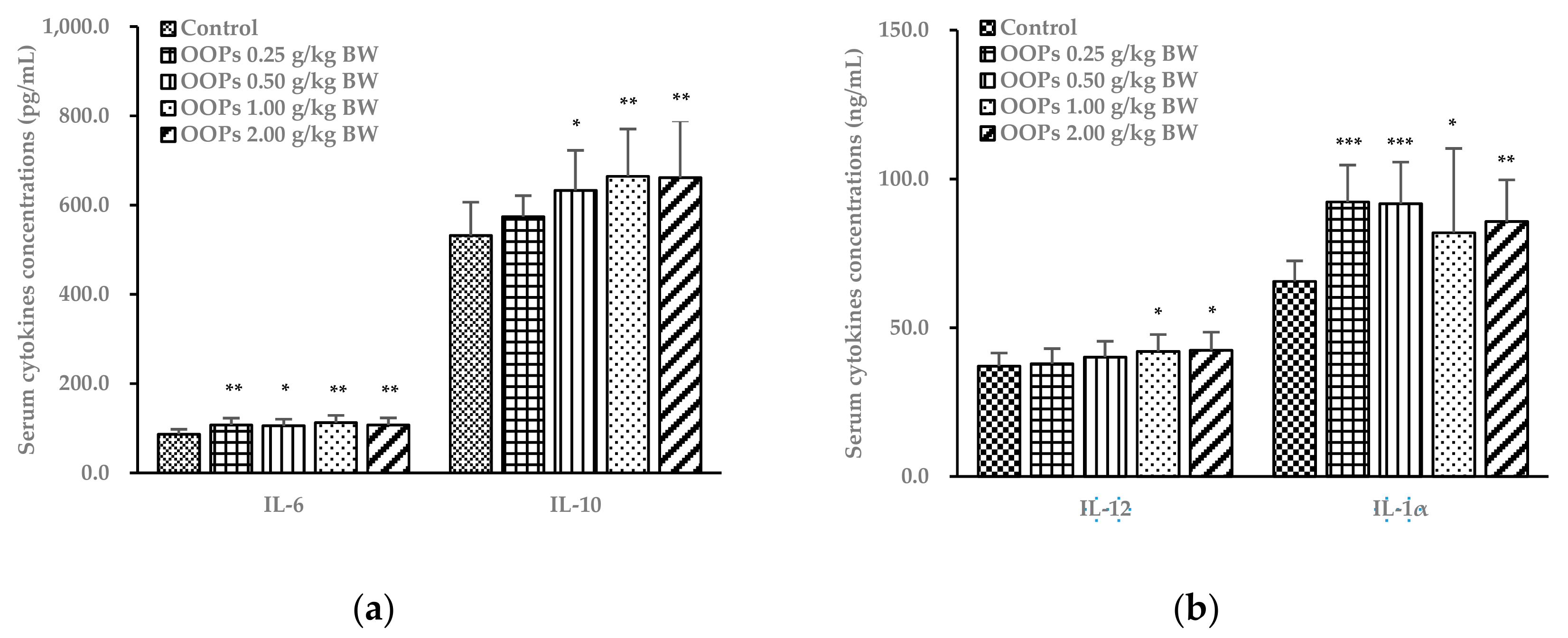
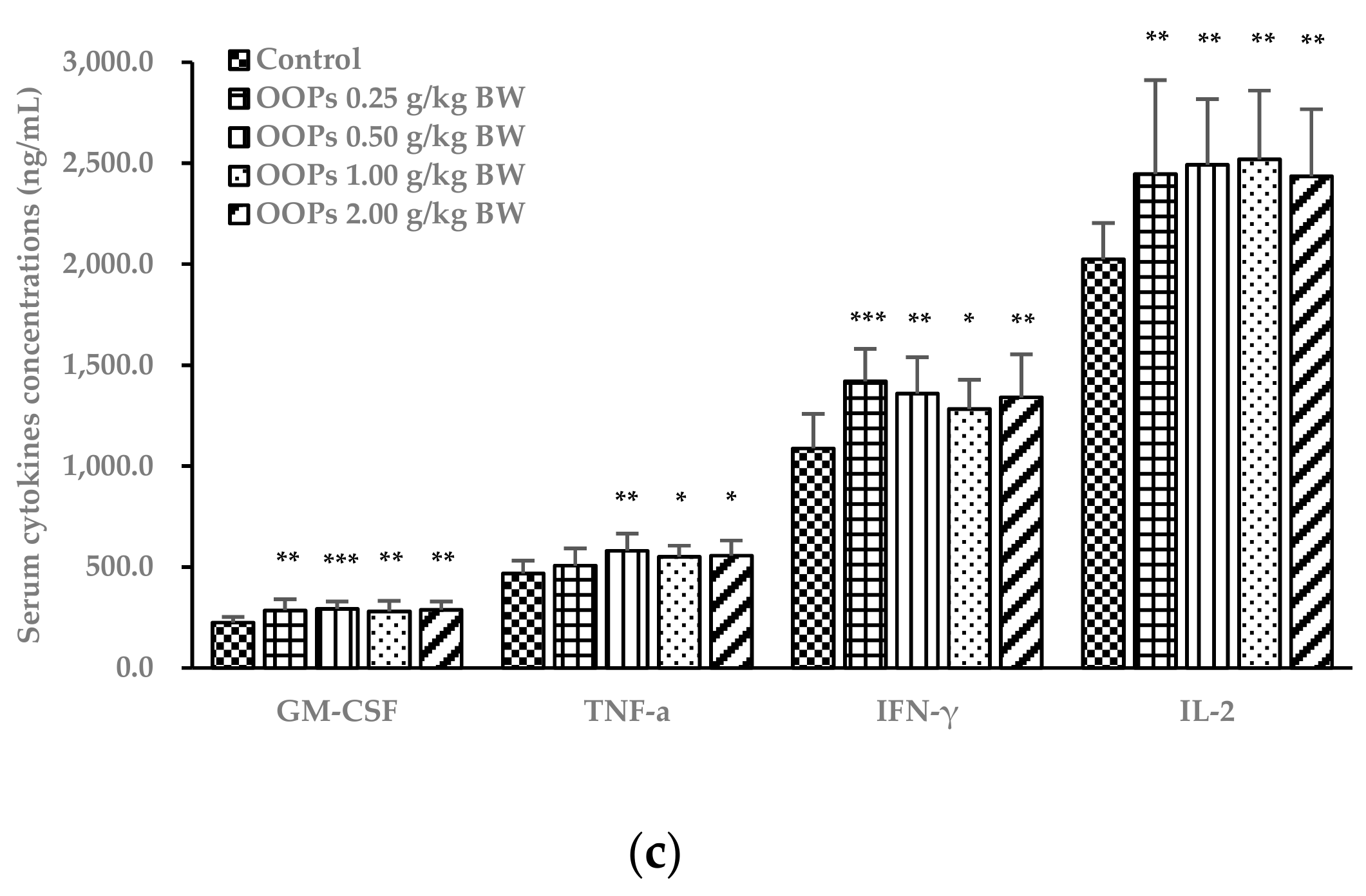
3.7. Effects of OOPs on Cytokine Concentrations in Serum
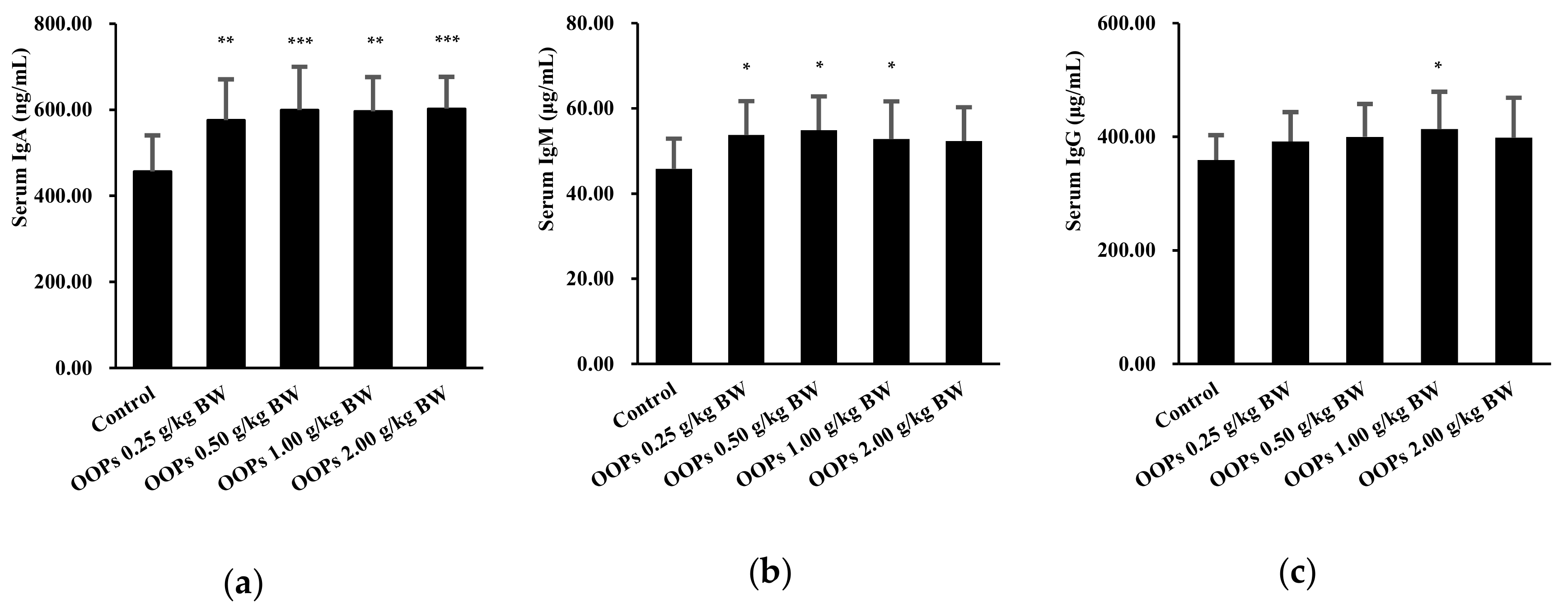
3.8. Effects of OOPs on Serum Immunoglobulin Levels
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Abid, S.; Khajuria, A.; Parvaiz, Q.; Sidiq, T.; Bhatia, A.; Singh, S.; Ahmad, S.; Randhawa, M.K.; Satti, N.K.; Dutt, P. Immunomodulatory studies of a bioactive fraction from the fruit of Prunus cerasus in BALB/c mice. Int. Immunopharmacol. 2012, 12, 626–634. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.K.; He, H.L.; Wang, G.F.; Wu, H.; Zhou, B.C.; Chen, X.L.; Zhang, Y.Z. Oyster (Crassostrea gigas) hydrolysates produced on a plant scale have antitumor activity and immunostimulating effects in BALB/c mice. Mar. Drugs 2010, 8, 255–268. [Google Scholar] [CrossRef] [PubMed]
- Hartmann, R.; Meisel, H. Food-derived peptides with biological activity: From research to food applications. Curr. Opin. Biotechnol. 2007, 18, 163–169. [Google Scholar] [CrossRef]
- Wada, Y.; Lonnerdal, B. Bioactive peptides derived from human milk proteins—Mechanisms of action. J. Nutr. Biochem. 2014, 25, 503–514. [Google Scholar] [CrossRef] [PubMed]
- Shichiri, M.; Ishimaru, S.; Ota, T.; Nishikawa, T.; Isogai, T.; Hirata, Y. Salusins: Newly identified bioactive peptides with hemodynamic and mitogenic activities. Nat. Med. 2003, 9, 1166–1172. [Google Scholar] [CrossRef] [PubMed]
- Hamley, I.W. Small Bioactive Peptides for Biomaterials Design and Therapeutics. Chem. Rev. 2017, 117, 14015–14041. [Google Scholar] [CrossRef] [PubMed]
- Jane, M.; McKay, J.; Pal, S. Effects of daily consumption of psyllium, oat bran and polyGlycopleX on obesity-related disease risk factors: A critical review. Nutrition 2019, 57, 84–91. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Cai, X.; Ma, X.; Jing, L.; Gu, J.; Bao, L.; Li, J.; Xu, M.; Zhang, Z.; Li, Y. Short- and Long-Term Effects of Wholegrain Oat Intake on Weight Management and Glucolipid Metabolism in Overweight Type-2 Diabetics: A Randomized Control Trial. Nutrients 2016, 8, 549. [Google Scholar] [CrossRef]
- Wang, P.; Yang, J.; Yerke, A.; Sang, S. Avenacosides: Metabolism, and potential use as exposure biomarkers of oat intake. Mol. Nutr. Food Res. 2017, 61, 1700196. [Google Scholar] [CrossRef] [PubMed]
- Su, R. Study on the Development of Oat Industry in China—Evidence of Inner Mongolia. Ph.D. Thesis, Inner Mongolia Agricultural University, Hohhot, China, 2013. [Google Scholar]
- Chu, Y.-F. Oats Nutrition and Technology; Wiley/Blackwell: Chichester, West Sussex, UK; Hoboken, NJ, USA, 2014. [Google Scholar]
- Welch, R.W. The Oat Crop: Production and Utilization, 1st ed.; Chapman & Hall: London, UK; New York, NY, USA, 1995. [Google Scholar]
- Jing, X.; Yang, C.; Zhang, L. Characterization and Analysis of Protein Structures in Oat Bran. J. Food Sci. 2016, 81, C2337–C2343. [Google Scholar] [CrossRef]
- Grundy, M.M.; Fardet, A.; Tosh, S.M.; Rich, G.T.; Wilde, P.J. Processing of oat: The impact on oat’s cholesterol lowering effect. Food Funct. 2018, 9, 1328–1343. [Google Scholar] [CrossRef] [PubMed]
- Yu, G.; Wang, F.; Zhang, B.; Fan, J. In vitro inhibition of platelet aggregation by peptides derived from oat (Avena sativa L.), highland barley (Hordeum vulgare Linn. var. nudum Hook. f.), and buckwheat (Fagopyrum esculentum Moench) proteins. Food Chem. 2016, 194, 577–586. [Google Scholar] [CrossRef]
- Tong, L.T.; Guo, L.; Zhou, X.; Qiu, J.; Liu, L.; Zhong, K.; Zhou, S. Effects of dietary oat proteins on cholesterol metabolism of hypercholesterolaemic hamsters. J. Sci. Food Agric. 2016, 96, 1396–1401. [Google Scholar] [CrossRef]
- Comino, I.; Bernardo, D.; Bancel, E.; de Lourdes Moreno, M.; Sanchez, B.; Barro, F.; Suligoj, T.; Ciclitira, P.J.; Cebolla, A.; Knight, S.C.; et al. Identification and molecular characterization of oat peptides implicated on coeliac immune response. Food Nutr. Res. 2016, 60, 30324. [Google Scholar] [CrossRef]
- Wang, F.; Yu, G.; Zhang, Y.; Zhang, B.; Fan, J. Dipeptidyl Peptidase IV Inhibitory Peptides Derived from Oat (Avena sativa L.), Buckwheat (Fagopyrum esculentum), and Highland Barley (Hordeum vulgare trifurcatum (L.) Trofim) Proteins. J. Agric. Food Chem. 2015, 63, 9543–9549. [Google Scholar] [CrossRef]
- Xu, C.; Lv, J.; You, S.; Zhao, Q.; Chen, X.; Hu, X. Supplementation with oat protein ameliorates exercise-induced fatigue in mice. Food Funct. 2013, 4, 303–309. [Google Scholar] [CrossRef]
- Shen, W.; Matsui, T. Current knowledge of intestinal absorption of bioactive peptides. Food Funct. 2017, 8, 4306–4314. [Google Scholar] [CrossRef]
- Sheng, X.; Yan, J.; Meng, Y.; Kang, Y.; Han, Z.; Tai, G.; Zhou, Y.; Cheng, H. Immunomodulatory effects of Hericium erinaceus derived polysaccharides are mediated by intestinal immunology. Food Funct. 2017, 8, 1020–1027. [Google Scholar] [CrossRef] [PubMed]
- He, L.X.; Ren, J.W.; Liu, R.; Chen, Q.H.; Zhao, J.; Wu, X.; Zhang, Z.F.; Wang, J.B.; Pettinato, G.; Li, Y. Ginseng (Panax ginseng Meyer) oligopeptides regulate innate and adaptive immune responses in mice via increased macrophage phagocytosis capacity, NK cell activity and Th cells secretion. Food Funct. 2017, 8, 3523–3532. [Google Scholar] [CrossRef]
- Tan, S.Y.; Siow, P.C.; Peh, E.; Henry, C.J. Influence of rice, pea and oat proteins in attenuating glycemic response of sugar-sweetened beverages. Eur. J. Nutr. 2017, 57, 2795–2803. [Google Scholar] [CrossRef] [PubMed]
- Gimenez, M.J.; Real, A.; Garcia-Molina, M.D.; Sousa, C.; Barro, F. Characterization of celiac disease related oat proteins: Bases for the development of high quality oat varieties suitable for celiac patients. Sci. Rep. 2017, 7, 42588. [Google Scholar] [CrossRef]
- Bleakley, S.; Hayes, M.; O’Shea, N.; Gallagher, E.; Lafarga, T. Predicted Release and Analysis of Novel ACE-I, Renin, and DPP-IV Inhibitory Peptides from Common Oat (Avena sativa) Protein Hydrolysates Using in Silico Analysis. Foods 2017, 6, 108. [Google Scholar] [CrossRef]
- Du, Y.; Esfandi, R.; Willmore, W.G.; Tsopmo, A. Antioxidant Activity of Oat Proteins Derived Peptides in Stressed Hepatic HepG2 Cells. Antioxidants (Basel) 2016, 5, 39. [Google Scholar] [CrossRef]
- Santiago-Lopez, L.; Hernandez-Mendoza, A.; Vallejo-Cordoba, B.; Mata-Haro, V.; Gonzalez-Cordova, A.F. Food-derived immunomodulatory peptides. J. Sci. Food Agric. 2016, 96, 3631–3641. [Google Scholar] [CrossRef]
- Iwasaki, A.; Medzhitov, R. Control of adaptive immunity by the innate immune system. Nat. Immunol. 2015, 16, 343–353. [Google Scholar] [CrossRef]
- Constant, S.L.; Bottomly, K. Induction of Th1 and Th2 CD4+ T cell responses: The alternative approaches. Annu. Rev. Immunol. 1997, 15, 297–322. [Google Scholar] [CrossRef]
- Liao, W.; Lin, J.X.; Wang, L.; Li, P.; Leonard, W.J. Modulation of cytokine receptors by IL-2 broadly regulates differentiation into helper T cell lineages. Nat. Immunol. 2011, 12, 551–559. [Google Scholar] [CrossRef]
- Cerutti, A.; Chen, K.; Chorny, A. Immunoglobulin responses at the mucosal interface. Annu. Rev. Immunol. 2011, 29, 273. [Google Scholar] [CrossRef]
- Nimmerjahn, F.; Ravetch, J.V. Antibody-mediated modulation of immune responses. Immunol. Rev. 2010, 236, 265–275. [Google Scholar] [CrossRef]
- Chalamaiah, M.; Hemalatha, R.; Jyothirmayi, T.; Diwan, P.V.; Kumar, P.U.; Nimgulkar, C.; Kumar, B.D. Immunomodulatory effects of protein hydrolysates from rohu (Labeo rohita) egg (roe) in BALB/c mice. Food Res. Int. 2014, 62, 1054–1061. [Google Scholar] [CrossRef]
| Amino Acid | Composition of OOPs (g/100g) |
|---|---|
| Alanine | 4.43 |
| Arginine | 4.08 |
| Aspartate | 5.62 |
| Cysteine | 1.89 |
| Glutamate | 13.61 |
| Glycine | 4.24 |
| Histidine | 1.46 |
| Isoleucine | 2.47 |
| Leucine | 4.66 |
| Lysine | 2.82 |
| Methionine | 1.26 |
| Phenylalanine | 3.30 |
| Proline | 3.67 |
| Serine | 3.45 |
| Threonine | 2.73 |
| Tyrosine | 2.72 |
| Valine | 3.72 |
| Groups | Initial Body Weight (g, N = 40) | Final Body Weight (g, N = 40) | Index of Spleen (mg/g, N = 10) | Index of Thymus (mg/g, N = 10) |
|---|---|---|---|---|
| Control | 18.43 ± 0.94 | 19.56 ± 0.82 | 3.65 ± 0.30 | 1.87 ± 0.27 |
| OOPs 0.25 g/kg BW | 19.41 ± 1.11 | 20.33 ± 0.75 | 3.48 ± 0.58 | 2.05 ± 0.52 |
| OOPs 0.5 g/kg BW | 19.50 ± 1.13 | 19.72 ± 1.27 | 3.67 ± 0.35 | 2.09 ± 0.59 |
| OOPs 1.0 g/kg BW | 18.78 ± 0.72 | 20.08 ± 1.17 | 3.65 ± 0.42 | 2.27 ± 0.50 |
| OOPs 2.0 g/kg BW | 19.23 ± 0.95 | 20.56 ± 1.21 | 3.52 ± 0.34 | 1.92 ± 0.50 |
| Groups. | CD3+ (%) | CD4+ CD8− (%) | CD4− CD8+ (%) | CD4+ CD8−/CD4− CD8+ (%) | CD4+ CD25+ (%) |
|---|---|---|---|---|---|
| Control | 40.50±4.69 | 58.77 ± 1.47 | 34.32 ± 2.64 | 1.72 ± 0.11 | 7.67 ± 1.05 |
| OOPs 0.25 g/kg BW | 42.51 ± 4.51 | 59.96 ± 1.04 | 34.18 ± 3.18 | 1.77 ± 0.18 | 7.91 ± 1.87 |
| OOPs 0.5 g/kg BW | 42.67 ± 4.73 | 59.9 ± 2.19 | 34.39 ± 4.32 | 1.77 ± 0.29 | 8.83 ± 1.54 |
| OOPs 1.0 g/kg BW | 45.33 ± 2.81 * | 60.59 ± 1.29 ** | 33.22 ± 2.55 | 1.84 ± 0.18 | 8.69 ± 1.48 |
| OOPs 2.0 g/kg BW | 45.63 ± 3.95 ** | 60.68 ± 1.11 ** | 31.97 ± 2.73 | 1.91 ± 0.19 | 8.63 ± 1.33 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mao, R.; Wu, L.; Zhu, N.; Liu, X.; Liu, R.; Li, Y. Naked Oat (Avena nuda L.) Oligopeptides: Immunomodulatory Effects on Innate and Adaptive Immunity in Mice via Cytokine Secretion, Antibody Production, and Th Cells Stimulation. Nutrients 2019, 11, 927. https://doi.org/10.3390/nu11040927
Mao R, Wu L, Zhu N, Liu X, Liu R, Li Y. Naked Oat (Avena nuda L.) Oligopeptides: Immunomodulatory Effects on Innate and Adaptive Immunity in Mice via Cytokine Secretion, Antibody Production, and Th Cells Stimulation. Nutrients. 2019; 11(4):927. https://doi.org/10.3390/nu11040927
Chicago/Turabian StyleMao, Ruixue, Lan Wu, Na Zhu, Xinran Liu, Rui Liu, and Yong Li. 2019. "Naked Oat (Avena nuda L.) Oligopeptides: Immunomodulatory Effects on Innate and Adaptive Immunity in Mice via Cytokine Secretion, Antibody Production, and Th Cells Stimulation" Nutrients 11, no. 4: 927. https://doi.org/10.3390/nu11040927
APA StyleMao, R., Wu, L., Zhu, N., Liu, X., Liu, R., & Li, Y. (2019). Naked Oat (Avena nuda L.) Oligopeptides: Immunomodulatory Effects on Innate and Adaptive Immunity in Mice via Cytokine Secretion, Antibody Production, and Th Cells Stimulation. Nutrients, 11(4), 927. https://doi.org/10.3390/nu11040927





