Cognitive Training Therapy Improves the Effect of Hypocaloric Treatment on Subjects with Overweight/Obesity: A Randomised Clinical Trial
Abstract
1. Introduction
2. Materials and Methods
2.1. Design
2.2. Participants
2.3. Outcome Measures
2.4. Intervention
2.5. Hypocaloric Dietary Treatment
2.6. Measures
2.6.1. Anthropometric and Clinical Data
2.6.2. Executive Function Assessment
2.7. Statistical Analysis
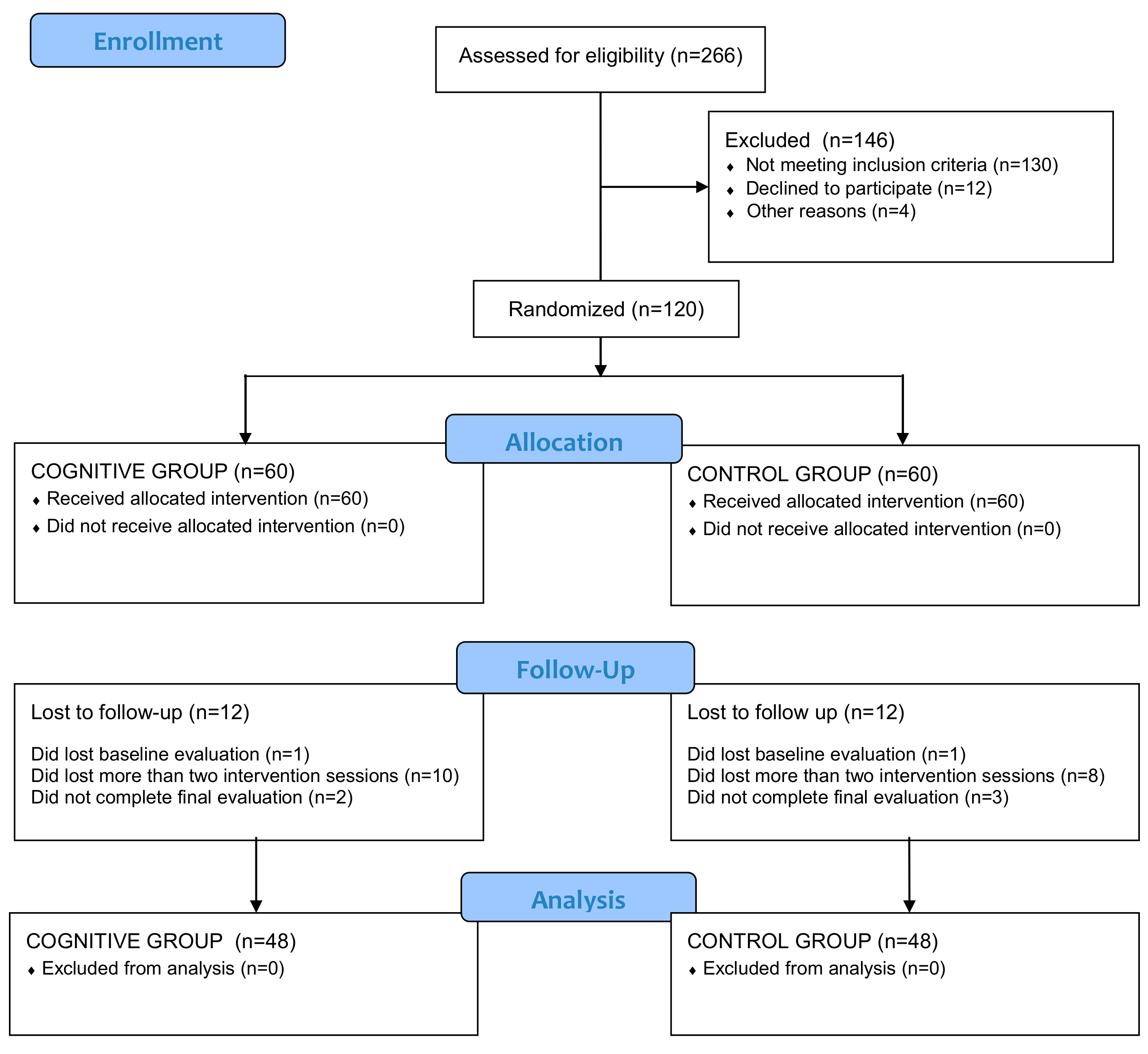
3. Results
3.1. Sample Characteristics
3.2. Efficacy of Cognitive Training on Executive Functions
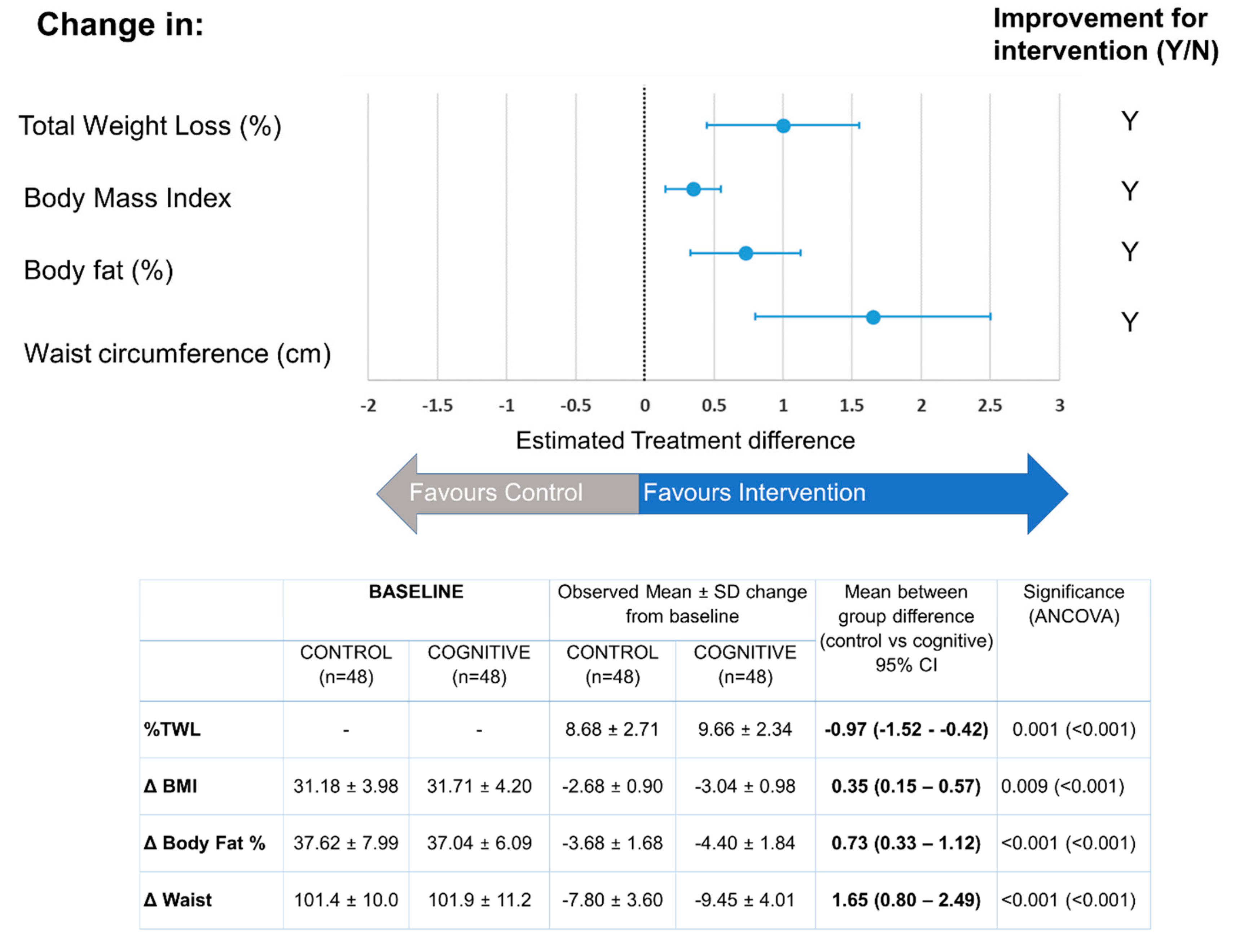
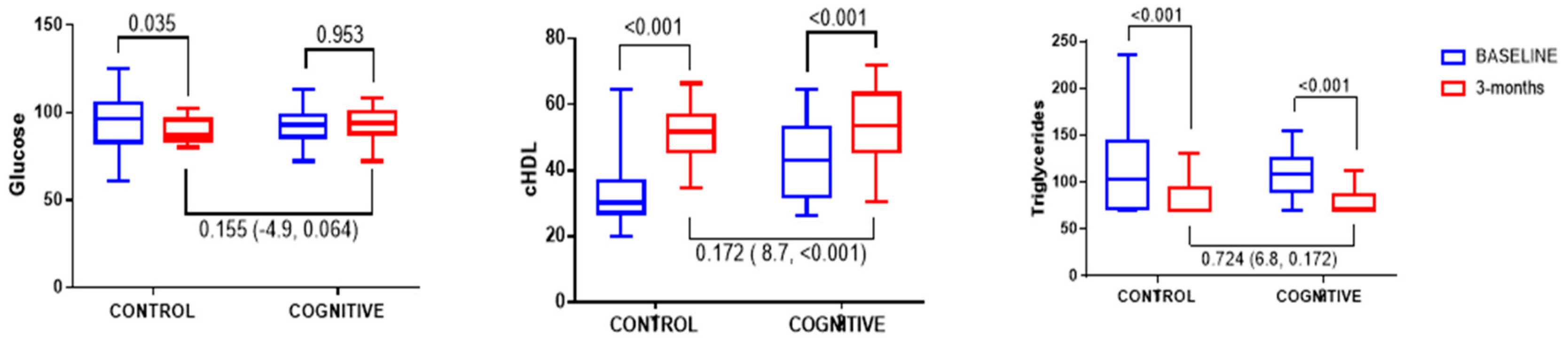
3.3. Efficacy of Cognitive Training on Anthropometrical and Biochemical Parameters
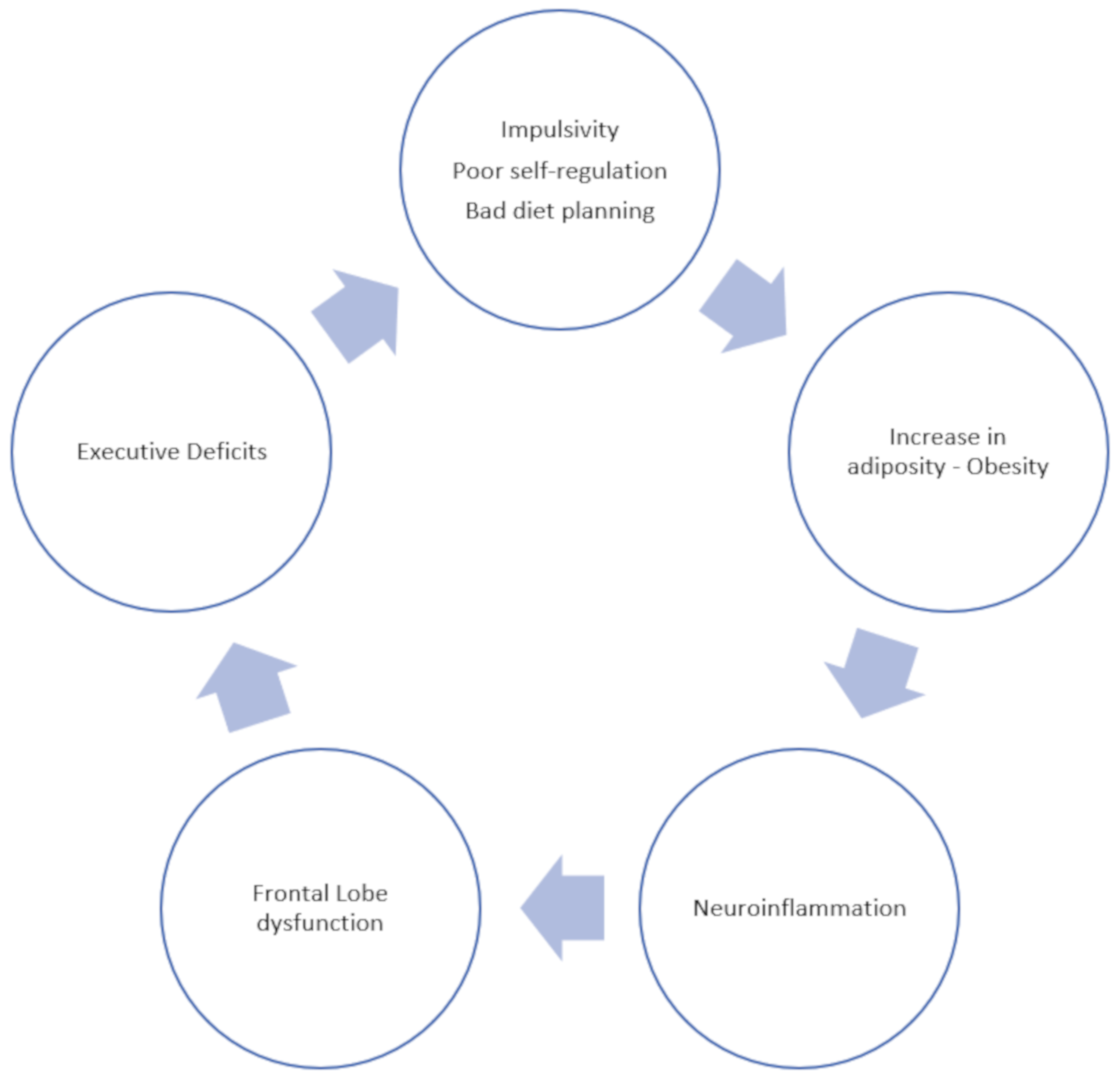
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Smith, E.; Hay, P.; Campbell, L.; Trollor, J.N. A review of the association between obesity and cognitive function across the lifespan: Implications for novel approaches to prevention and treatment. Obes. Rev. 2011, 12, 740–755. [Google Scholar] [CrossRef]
- Gunstad, J.; Paul, R.H.; Cohen, R.A.; Tate, D.F.; Spitznagel, M.B.; Gordon, E. Elevated body mass index is associated with executive dysfunction in otherwise healthy adults. Compr. Psychiatry 2007, 48, 57–61. [Google Scholar] [CrossRef]
- Lokken, K.L.; Boeka, A.G.; Austin, H.M.; Gunstad, J.; Harmon, C.M. Evidence of executive dysfunction in extremely obese adolescents: A pilot study. Surg. Obes. Relat. Dis. 2009, 5, 547–552. [Google Scholar] [CrossRef]
- Coppin, G.; Nolan-Poupart, S.; Jones-Gotman, M.; Small, D.M. Working memory and reward association learning impairments in obesity. Neuropsychologia 2014, 65, 146–155. [Google Scholar] [CrossRef] [PubMed]
- Verdejo-García, A.; Pérez-Expósito, M.; Schmidt-Río-Valle, J.; Fernández-Serrano, M.J.; Cruz, F.; Pérez-García, M.; López-Belmonte, G.; Martín-Matillas, M.; Martín-Lagos, J.A.; Marcos, A.; et al. Selective alterations within executive functions in adolescents with excess weight. Obesity 2010, 18, 1572–1578. [Google Scholar] [CrossRef]
- Kittel, R.; Schmidt, R.; Hilbert, A. Executive functions in adolescents with binge-eating disorder and obesity. Int. J. Eat. Disord. 2017, 50, 933–941. [Google Scholar] [CrossRef] [PubMed]
- Sárvári, M.; Kocsis, P.; Deli, L.; Gajári, D.; Dávid, S.; Pozsgay, Z.; Hegedűs, N.; Tihanyi, K.; Liposits, Z. Ghrelin Modulates the fMRI BOLD Response of Homeostatic and Hedonic Brain Centers Regulating Energy Balance in the Rat. PLoS ONE 2014, 9, e97651. [Google Scholar] [CrossRef] [PubMed]
- Salcedo, I.; Tweedie, D.; Li, Y.; Greig, N.H. Neuroprotective and neurotrophic actions of glucagon-like peptide-1: An emerging opportunity to treat neurodegenerative and cerebrovascular disorders. Br. J. Pharmacol. 2012, 166, 1586–1599. [Google Scholar] [CrossRef] [PubMed]
- Shanley, L.J.; Irving, A.J.; Harvey, J. Leptin enhances NMDA receptor function and modulates hippocampal synaptic plasticity. J. Neurosci. 2001, 21, RC186. [Google Scholar] [CrossRef]
- Winocur, G.; Greenwood, C.E.; Piroli, G.G.; Grillo, C.A.; Reznikov, L.R.; Reagan, L.P.; McEwen, B.S. Memory impairment in Obese Zucker rats: An investigation of cognitive function in an animal model of insulin resistance and obesity. Behav. Neurosci. 2005, 119, 1389–1395. [Google Scholar] [CrossRef]
- Galindo Muñoz, J.S.; Jiménez Rodríguez, D.; Hernández Morante, J.J.; Muñoz, J.S.G.; Rodríguez, D.J.; Hernández Morante, J.J. Diurnal rhythms of plasma GLP-1 levels in normal and overweight/obese subjects: Lack of effect of weight loss. J. Physiol. Biochem. 2015, 71, 17–28. [Google Scholar] [CrossRef] [PubMed]
- Grassi-Oliveira, R.; Honeycutt, J.A.; Holland, F.H.; Ganguly, P.; Brenhouse, H.C. Cognitive impairment effects of early life stress in adolescents can be predicted with early biomarkers: Impacts of sex, experience, and cytokines. Psychoneuroendocrinology 2016, 71, 19–30. [Google Scholar] [CrossRef] [PubMed]
- Espeland, M.A.; Rapp, S.R.; Bray, G.A.; Houston, D.K.; Johnson, K.C.; Kitabchi, A.E.; Hergenroeder, A.L.; Williamson, J.; Jakicic, J.M.; van Dorsten, B.; et al. Long-term impact of behavioral weight loss intervention on cognitive function. J. Gerontol. A Biol. Sci. Med. Sci. 2014, 69, 1101–1108. [Google Scholar] [CrossRef] [PubMed]
- Horie, N.C.; Serrao, V.T.; Simon, S.S.; Gascon, M.R.P.; dos Santos, A.X.; Zambone, M.A.; del Bigio de Freitas, M.M.; Cunha-Neto, E.; Marques, E.L.; Halpern, A.; et al. Cognitive Effects of Intentional Weight Loss in Elderly Obese Individuals With Mild Cognitive Impairment. J. Clin. Endocrinol. Metab. 2016, 101, 1104–1112. [Google Scholar] [CrossRef] [PubMed]
- Weygandt, M.; Mai, K.; Dommes, E.; Leupelt, V.; Hackmack, K.; Kahnt, T.; Rothemund, Y.; Spranger, J.; Haynes, J.-D. The role of neural impulse control mechanisms for dietary success in obesity. Neuroimage 2013, 83, 669–678. [Google Scholar] [CrossRef] [PubMed]
- Daly, M.; McMinn, D.; Allan, J.L. A bidirectional relationship between physical activity and executive function in older adults. Front. Hum. Neurosci. 2014, 8, 1044. [Google Scholar] [CrossRef]
- Hayes, J.F.; Eichen, D.M.; Barch, D.M.; Wilfley, D.E. Executive function in childhood obesity: Promising intervention strategies to optimize treatment outcomes. Appetite 2018, 124, 10–23. [Google Scholar] [CrossRef]
- Schulz, K.F.; Altman, D.G.; Moher, D.M.; CONSORT Group. CONSORT 2010 Statement: Updated guidelines for reporting parallel group randomised trials. BMJ 2010, 340, 698–702. [Google Scholar] [CrossRef]
- Mayr, S.; Buchner, A.; Erdfelder, E.; Faul, F. A short tutorial of GPower. Tutor. Quant. Methods Psychol. 2007, 3, 51–59. [Google Scholar] [CrossRef]
- Gargallo Fernández Manuel, M.; Breton Lesmes, I.; Basulto Marset, J.; Quiles Izquierdo, J.; Formiguera Sala, X.; Salas-Salvadó, J. Evidence-based nutritional recommendations for the prevention and treatment of overweight and obesity in adults (FESNAD-SEEDO consensus document). The role of diet in obesity treatment (III/III). Nutr. Hosp. 2012, 27, 833–864. [Google Scholar]
- Muñoz, J.S.G.; Cañavate, R.; Hernández, C.M.; Cara-Salmerón, V.; Morante, J.J.H. The association among chronotype, timing of food intake and food preferences depends on body mass status. Eur. J. Clin. Nutr. 2017, 71, 736–742. [Google Scholar] [CrossRef]
- Lobo, A.; Saz, P.; Marcos, G.; Día, J.L.; de la Cámara, C.; Ventura, T.; Morales Asín, F.; Fernando Pascual, L.; Montañés, J.A.; Aznar, S. Revalidation and standardization of the cognition mini-exam (first Spanish version of the Mini-Mental Status Examination) in the general geriatric population. Med. Clin. 1999, 112, 767–774. [Google Scholar]
- Deitel, M.; Greenstein, R.J. Recommendations for Reporting Weight Loss. Obes. Surg. 2003, 13, 159–160. [Google Scholar] [CrossRef]
- Uchida, S.; Kawashima, R. Reading and solving arithmetic problems improves cognitive functions of normal aged people: A randomized controlled study. Age 2008, 30, 21–29. [Google Scholar] [CrossRef] [PubMed]
- Kawashima, R.; Okita, K.; Yamazaki, R.; Tajima, N.; Yoshida, H.; Taira, M.; Iwata, K.; Sasaki, T.; Maeyama, K.; Usui, N.; et al. Reading aloud and arithmetic calculation improve frontal function of people with dementia. J. Gerontol. A Biol. Sci. Med. Sci. 2005, 60, 380–384. [Google Scholar] [CrossRef] [PubMed]
- Nouchi, R.; Taki, Y.; Takeuchi, H.; Hashizume, H.; Nozawa, T.; Kambara, T.; Sekiguchi, A.; Miyauchi, C.M.; Kotozaki, Y.; Nouchi, H.; et al. Brain training game boosts executive functions, working memory and processing speed in the young adults: A randomized controlled trial. PLoS ONE 2013, 8, e55518. [Google Scholar] [CrossRef] [PubMed]
- McDougall, S.; House, B. Brain training in older adults: Evidence of transfer to memory span performance and pseudo-Matthew effects. Neuropsychol. Dev. Cogn. B Aging. Neuropsychol. Cogn. 2012, 19, 195–221. [Google Scholar] [CrossRef] [PubMed]
- Salas-Salvadó, J.; Rubio, M.A.; Barbany, M.; Moreno, B.; Grupo Colaborativo de la SEEDO. SEEDO 2007 Consensus for the evaluation of overweight and obesity and the establishment of therapeutic intervention criteria. Med. Cliín. 2007, 128, 184–196. [Google Scholar] [CrossRef]
- Renteria, L.; Li, S.T.; Pliskin, N.H. Reliability and Validity of the Spanish Language Wechsler Adult Intelligence Scale (3rd Edition) in a Sample of American, Urban, Spanish-Speaking Hispanics. Clin. Neuropsychol. 2008, 22, 455–470. [Google Scholar] [CrossRef]
- Steinborn, M.B.; Langner, R.; Flehmig, H.C.; Huestegge, L. Methodology of performance scoring in the d2 sustained-attention test: Cumulative-reliability functions and practical guidelines. Psychol. Assess. 2018, 30, 339–357. [Google Scholar] [CrossRef]
- Chiu, E.-C.; Koh, C.-L.; Tsai, C.-Y.; Lu, W.-S.; Sheu, C.-F.; Hsueh, I.-P.; Hsieh, C.-L. Practice effects and test–re-test reliability of the Five Digit Test in patients with stroke over four serial assessments. Brain Inj. 2014, 28, 1726–1733. [Google Scholar] [CrossRef]
- Sánchez-Benavides, G.; Peña-Casanova, J.; Casals-Coll, M.; Gramunt, N.; Manero, R.M.; Puig-Pijoan, A.; Aguilar, M.; Robles, A.; Antúnez, C.; Frank-García, A.; et al. One-Year Reference Norms of Cognitive Change in Spanish Old Adults: Data from the NEURONORMA Sample. Arch. Clin. Neuropsychol. 2016, 31, 378–388. [Google Scholar] [CrossRef]
- Mueller, S.T.; Piper, B.J. The Psychology Experiment Building Language (PEBL) and PEBL Test Battery. J. Neurosci. Methods 2014, 222, 250–259. [Google Scholar] [CrossRef]
- Lewis, J.A.; Rok, J.; Lewis, J.; Towers, M. Statistical principles for clinical trials (ICH E9) An introductory note on an international guideline. Stat. Med. 1993, 18, 1655–1658. [Google Scholar] [CrossRef]
- Prickett, C.; Brennan, L.; Stolwyk, R. Examining the relationship between obesity and cognitive function: A systematic literature review. Obes. Res. Clin. Pract. 2015, 9, 93–113. [Google Scholar] [CrossRef]
- Smith, E.; Whittingham, C. Cognitive remediation therapy plus behavioural weight loss compared to behavioural weight loss alone for obesity: Study protocol for a randomised controlled trial. Trials 2017, 18, 42. [Google Scholar] [CrossRef]
- Nouchi, R.; Taki, Y.; Takeuchi, H.; Hashizume, H.; Akitsuki, Y.; Shigemune, Y.; Sekiguchi, A.; Kotozaki, Y.; Tsukiura, T.; Yomogida, Y.; et al. Brain Training Game Improves Executive Functions and Processing Speed in the Elderly: A Randomized Controlled Trial. PLoS ONE 2012, 7, e29676. [Google Scholar] [CrossRef]
- Lawrence, N.S.; O’Sullivan, J.; Parslow, D.; Javaid, M.; Adams, R.C.; Chambers, C.D.; Kos, K.; Verbruggen, F. Training response inhibition to food is associated with weight loss and reduced energy intake. Appetite 2015, 95, 17–28. [Google Scholar] [CrossRef]
- Stice, E.; Lawrence, N.S.; Kemps, E.; Veling, H. Training motor responses to food: A novel treatment for obesity targeting implicit processes. Clin. Psychol. Rev. 2016, 49, 16–27. [Google Scholar] [CrossRef]
- Verbeken, S.; Braet, C.; Goossens, L.; van der Oord, S. Executive function training with game elements for obese children: A novel treatment to enhance self-regulatory abilities for weight-control. Behav. Res. Ther. 2013, 51, 290–299. [Google Scholar] [CrossRef]
- Boeka, A.; Lokken, K. Neuropsychological performance of a clinical sample of extremely obese individuals. Arch. Clin. Neuropsychol. 2008, 23, 467–474. [Google Scholar] [CrossRef] [PubMed]
- Cheke, L.G.; Bonnici, H.M.; Clayton, N.S.; Simons, J.S. Obesity and insulin resistance are associated with reduced activity in core memory regions of the brain. Neuropsychologia 2017, 96, 137–149. [Google Scholar] [CrossRef]
- Cook, R.L.; O’Dwyer, N.J.; Donges, C.E.; Parker, H.M.; Cheng, H.L.; Steinbeck, K.S.; Cox, E.P.; Franklin, J.L.; Garg, M.L.; Rooney, K.B.; et al. Relationship between Obesity and Cognitive Function in Young Women: The Food, Mood and Mind Study. J. Obes. 2017, 2017, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Brooks, S.J.; Wiemerslage, L.; Burch, K.; Maiorana, S.; Cocolas, E.; Schiöth, H.; Kamaloodien, K.; Stein, D. The impact of cognitive training in substance use disorder: The effect of working memory training on impulse control in methamphetamine users. Psychopharmacology 2017, 234, 1911–1921. [Google Scholar] [CrossRef]
- Sweeting, A.N.; Caterson, I.D. Approaches to obesity management. Intern. Med. J. 2017, 47, 734–739. [Google Scholar] [CrossRef] [PubMed]
- Medic, N.; Ziauddeen, H.; Ersche, K.D.; Farooqi, I.S.; Bullmore, E.T.; Nathan, P.J.; Ronan, L.; Fletcher, P.C. Increased body mass index is associated with specific regional alterations in brain structure. Int. J. Obes. 2016, 40, 1177–1182. [Google Scholar] [CrossRef] [PubMed]
- Gadde, K.M.; Apolzan, J.W.; Berthoud, H.-R. Pharmacotherapy for Patients with Obesity. Clin. Chem. 2018, 64, 118–129. [Google Scholar] [CrossRef] [PubMed]
- Van der Donk, M.L.A.; Hiemstra-Beernink, A.-C.; Tjeenk-Kalff, A.C.; van der Leij, A.V.; Lindauer, R.J. Interventions to improve executive functioning and working memory in school-aged children with AD(H)D: A randomised controlled trial and stepped-care approach. BMC Psychiatry 2013, 13, 23. [Google Scholar] [CrossRef]
- Vance, D.E.; Fazeli, P.L.; Cheatwood, J.; Nicholson, C.; Morrison, S.; Moneyham, L.D. Targeting HIV-Related Neurocognitive Impairments with Cognitive Training Strategies: Insights from the Cognitive Aging Literature. Curr. Top. Behav. Neurosci. 2019. [Google Scholar] [CrossRef]
| CONTROL (n = 48) | COGNITIVE (n = 48) | Sig. (t-test, χ-test) | |
|---|---|---|---|
| Age (years) | 46 ± 7 | 44 ± 10 | 0.376 |
| BMI (kg/m2) | 31.18 ± 3.98 | 31.71 ± 4.20 | 0.643 |
| Waist | 101.4 ± 10.0 | 101.9 ± 11.2 | 0.856 |
| Body fat (%) | 37.62 ± 7.99 | 37.04 ± 6.09 | 0.764 |
| SBP (mmHg) | 130 ± 13 | 118 ± 16 | 0.220 |
| DBP (mmHg) | 79 ± 7 | 71 ± 9 | 0.120 |
| FPG (mg/dl) | 101 ± 23 | 93 ± 10 | 0.089 |
| Triglycerides (mg/dl) | 117 ± 49 | 107 ± 27 | 0.144 |
| cHDL (mg/dl) | 34 ± 11 | 43 ± 11 | 0.015 |
| Cognitive variables | |||
| Working memory (Z-L-N score) | 0.80 ± 1.06 | 1.04 ± 1.02 | 0.433 |
| Selective attention (Z-Total d2 test) | 0.23 ± 1.52 | 0.04 ± 1.34 | 0.656 |
| Z-Focusing Index (d2 test) | −0.66 ± 1.53 | −0.70 ± 1.27 | 0.913 |
| Z-Inhibition (5-digit test) | 0.26 ± 1.10 | 0.17 ± 0.78 | 0.997 |
| Z-Flexibility (5-digit test) | −0.21 ± 1.36 | −0.42 ± 1.01 | 0.598 |
| Task planning (Z score 30 min) | 1.90 ± 0.69 | 1.96 ± 1.11 | 0.884 |
| Decision making (Z-IGT score) | −0.82 ± 1.49 | −0.31 ± 1.39 | 0.223 |
| BASELINE | Observed Mean ± SD Change from Baseline | Mean between Group Difference (Control vs. Cognitive) 95% CI | Significance (ANCOVA) | |||
|---|---|---|---|---|---|---|
| CONTROL (n = 48) | COGNITIVE (n = 48) | CONTROL (n = 48) | COGNITIVE (n = 48) | |||
| Working Memory | 0.80 ± 1.06 | 1.04 ± 1.02 | 0.23 ± 0.79 | 0.55 ± 0.69 * | −0.32 (−0.50 – −0.51) | 0.001 (0.003) |
| Selective Attention | 0.23 ± 1.52 | 0.04 ± 1.34 | 0.69 ± 0.60 * | 0.54 ± 0.69 * | 0.15 (−0.02 – 0.31) | 0.076 (0.054) |
| Cognitive Flexibility | −0.21 ± 1.36 | −0.42 ± 1.01 | 0.37 ± 0.70 * | 0.41 ± 0.79 * | −0.03 (−0.15 – 0.22) | 0.693 (0.270) |
| Task Planning | 1.90 ± 0.69 | 1.96 ± 1.11 | 0.70 ± 0.66 * | 0.96 ± 0.83 * | −0.25 (−0.44 – −0.06) | 0.009 (0.003) |
| Decision making | −0.82 ± 1.49 | −0.31 ± 1.39 | 1.13 ± 2.21 | 1.23 ± 2.62 * | −0.09 (−0.70 – 0.52) | 0.776 (0.603) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Galindo Muñoz, J.S.; Morillas-Ruiz, J.M.; Gómez Gallego, M.; Díaz Soler, I.; Barberá Ortega, M.d.C.; Martínez, C.M.; Hernández Morante, J.J. Cognitive Training Therapy Improves the Effect of Hypocaloric Treatment on Subjects with Overweight/Obesity: A Randomised Clinical Trial. Nutrients 2019, 11, 925. https://doi.org/10.3390/nu11040925
Galindo Muñoz JS, Morillas-Ruiz JM, Gómez Gallego M, Díaz Soler I, Barberá Ortega MdC, Martínez CM, Hernández Morante JJ. Cognitive Training Therapy Improves the Effect of Hypocaloric Treatment on Subjects with Overweight/Obesity: A Randomised Clinical Trial. Nutrients. 2019; 11(4):925. https://doi.org/10.3390/nu11040925
Chicago/Turabian StyleGalindo Muñoz, Joaquín S., Juana María Morillas-Ruiz, María Gómez Gallego, Inmaculada Díaz Soler, María del Carmen Barberá Ortega, Carlos M. Martínez, and Juan José Hernández Morante. 2019. "Cognitive Training Therapy Improves the Effect of Hypocaloric Treatment on Subjects with Overweight/Obesity: A Randomised Clinical Trial" Nutrients 11, no. 4: 925. https://doi.org/10.3390/nu11040925
APA StyleGalindo Muñoz, J. S., Morillas-Ruiz, J. M., Gómez Gallego, M., Díaz Soler, I., Barberá Ortega, M. d. C., Martínez, C. M., & Hernández Morante, J. J. (2019). Cognitive Training Therapy Improves the Effect of Hypocaloric Treatment on Subjects with Overweight/Obesity: A Randomised Clinical Trial. Nutrients, 11(4), 925. https://doi.org/10.3390/nu11040925






