Exposure to Vitamin D Fortification Policy in Prenatal Life and the Risk of Childhood Asthma: Results from the D-Tect Study
Abstract
:1. Introduction
2. Methods
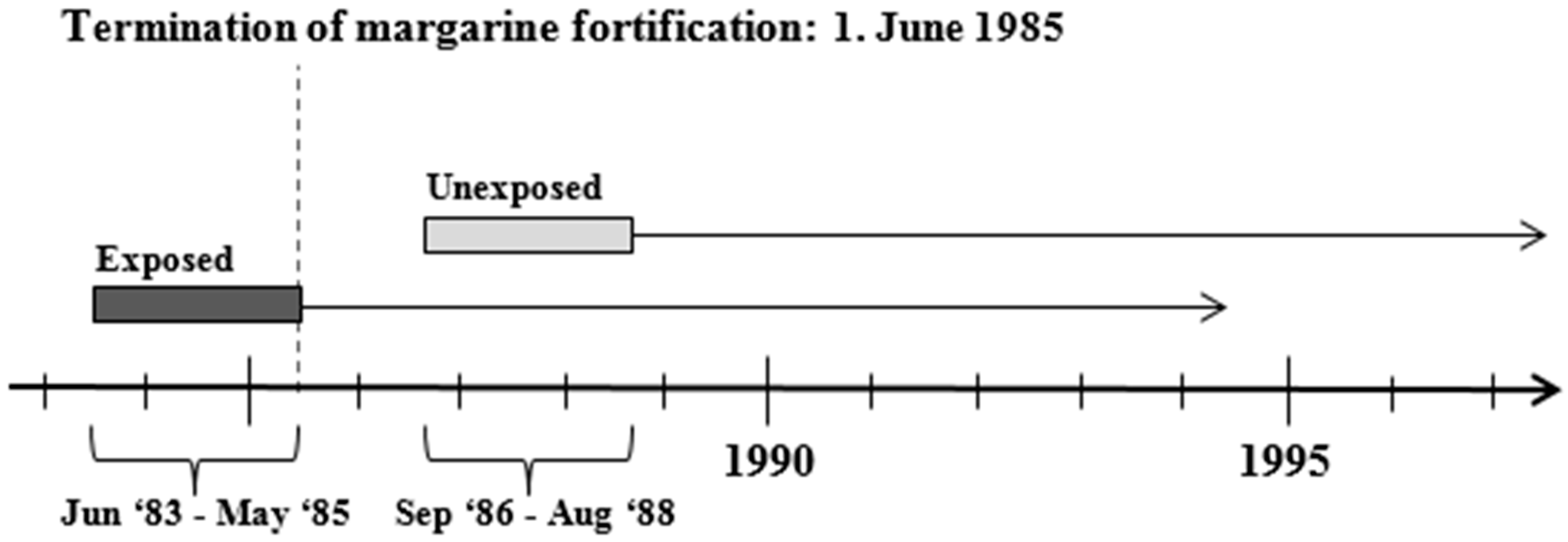
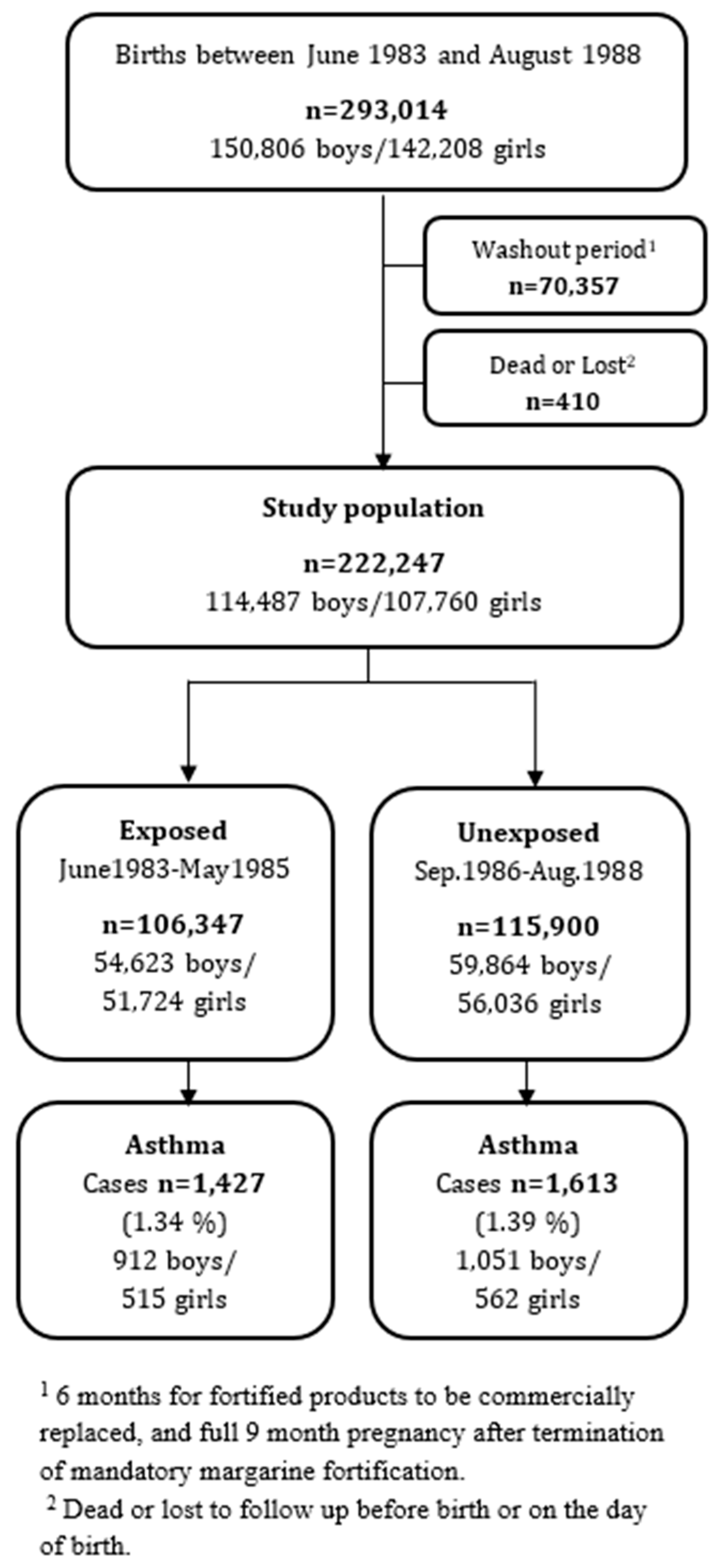
2.1. Study Design
2.2. Definition of Outcome
2.3. Statistical Analysis
3. Results
4. Discussion
Strengths and Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Masoli, M.; Fabian, D.; Holt, S.; Beasley, R. The global burden of asthma: Executive summary of the GINA Dissemination Committee Report. Allergy 2004, 59, 469–478. [Google Scholar] [CrossRef] [PubMed]
- Bush, S.; Kleinert, S.; Pavord, I.D. The asthmas in 2015 and beyond: A Lancet Commission. Lancet 2015, 385, 1273–1275. [Google Scholar] [CrossRef]
- Pedersen, S.E.; Hurd, S.S.; Lemanske, R.F.; Becker, A.; Zar, H.J.; Sly, P.D.; Soto-Quiroz, M.; Wong, G.; Bateman, E.D. Global strategy for the diagnosis and management of asthma in children 5 years and younger. Pediatr. Pulmonol. 2011, 46, 1–17. [Google Scholar] [CrossRef] [PubMed]
- De Luca, G.; Olivieri, F.; Melotti, G.; Aiello, G.; Lubrano, L.; Boner, A.L. Fetal and early postnatal life roots of asthma. J. Matern. Fetal. Neonatal. Med. 2010, 23, 80–83. [Google Scholar] [CrossRef] [PubMed]
- Postma, D.S. Gender Differences in Asthma Development and Progression. Gend. Med. 2007, 4 (Suppl. B), S133–S146. [Google Scholar] [CrossRef]
- Almqvist, C.; Worm, M.; Leynaert, B. Impact of gender on asthma in childhood and adolescence: A GA2LEN review. Allergy 2008, 63, 47–57. [Google Scholar] [CrossRef]
- Pignataro, F.S.; Bonini, M.; Forgione, A.; Melandri, S.; Usmani, O.S. Asthma and gender: The female lung. Pharmacol. Res. 2017, 119, 384–390. [Google Scholar] [CrossRef]
- Wimalawansa, S.J. Non-musculoskeletal benefits of vitamin D. J. Steroid. Biochem. Mol. Biol. 2018, 175, 60–81. [Google Scholar] [CrossRef]
- Larque, E.; Morales, E.; Leis, R.; Blanco-Carnerdo, J.E. Maternal and Foetal Health Implications of Vitamin D Status during Pregnancy. Ann. Nutr. Metab. 2018, 72, 179–192. [Google Scholar] [CrossRef] [PubMed]
- Chi, A.; Wildfire, J.; McLoughlin, R.; Wood, R.A.; Bloomberg, G.R.; Kattan, M.; Gregen, P.; Gold, D.R.; Witter, F.; Chen, T.; et al. Umbilical cord plasma 25-hydroxyvitamin D concentration and immune function at birth: The Urban Environment and Childhood Asthma study. Clin. Exp. Allergy 2011, 41, 842–850. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Wilson, R.; Bennet, E.; Zosky, R. Identification of vitamin D sensitive pathways during lung development. Respir Res. 2016, 17, 47. [Google Scholar] [CrossRef] [PubMed]
- Kho, A.T.; Sharma, S.; Qiu, W.; Gaedigk, R.; Klanderman, B.; Niu, S.; Anderson, C.; Leeder, J.S.; Weiss, S.T.; Tantisira, K.G. Vitamin D related genes in lung development and asthma pathogenesis. BMC Med. Genom. 2013, 6, 47. [Google Scholar] [CrossRef]
- Hollis, B.; Wagner, C. Nutritional vitamin D status during pregnancy: Reason for concern. CMAJ 2006, 174, 1287–1290. [Google Scholar] [CrossRef]
- Nurmatov, U.; Devereux, G.; Sheikh, A. Nutrients and foods for the primary prevention of asthma and allergy: Systematic review and meta-analysis. J. Allergy Clin. Immunol. 2011, 127, 724–733. [Google Scholar] [CrossRef] [PubMed]
- Beckhaus, A.A.; Garcia-Marcos, L.; Forno, E.; Pacheco-Gonzalez, R.M.; Celedón, J.C.; Castro-Rodriguez, J.A. Maternal nutrition during pregnancy and risk of asthma, wheeze, and atopic diseases during childhood: A systematic review and meta-analysis. Allergy 2015, 70, 1588–1604. [Google Scholar] [CrossRef] [PubMed]
- Feng, H.; Xun, P.; Pike, K.; Wills, A.K.; Chawes, B.L.; Bisgaard, H.; Cai, W.; Wan, Y.; He, K. In utero exposure to 25-hydroxyvitamin D and risk of childhood asthma, wheeze, and respiratory tract infections: A meta-analysis of birth cohort studies. J. Allergy Clin. Immunol. 2017, 139, 1508–1517. [Google Scholar] [CrossRef]
- Song, H.; Yang, L.; Jia, C. Maternal vitamin D status during pregnancy and risk of childhood asthma: A meta-analysis of prospective studies. Mol. Nutr. Food Res. 2017, 61, 1600657. [Google Scholar] [CrossRef] [PubMed]
- Pacheco-Gonzalez, R.M.; Garcia-Marcos, L.; Morales, E. Prenatal vitamin D status and respiratory and allergic outcomes in childhood: A meta-analysis of observational studies. Pediatr. Allergy Immunol. 2018, 29, 243–253. [Google Scholar] [CrossRef]
- Chawes, B.L.; Bønnelykke, K.; Stokholm, J.; Vissing, N.H.; Bjarnadóttir, E.; Schoos, A.M.; Wolsk, H.M.; Pedersen, T.M.; Vinding, R.K.; Thorsteinsdóttir, S.; et al. Effect of vitamin d3 supplementation during pregnancy on risk of persistent wheeze in the offspring: A randomized clinical trial. JAMA 2016, 315, 353–361. [Google Scholar] [CrossRef]
- Litonjua, A.A.; Carey, V.J.; Laranjo, N.; Harshfield, B.J.; McElrath, T.F.; O’Connor, G.T.; Sandel, M.; Iverson, R.E.; Lee-Paritz, A.; Strunk, R.C.; et al. Effect of prenatal supplementation with vitamin d on asthma or recurrent wheezing in offspring by age 3 years: The vdaart randomized clinical trial. JAMA 2016, 315, 362–370. [Google Scholar] [CrossRef]
- Wolsk, H.M.; Chawes, B.L.; Litonjua, A.A.; Hollis, B.W.; Waage, J.; Stokholm, J.; Bønnelykke, K.; Bisgaard, H.; Weiss, S.T. Prenatal vitamin D supplementation reduces risk of asthma/recurrent wheeze in early childhood: A combined analysis of two randomized controlled trials. PLoS ONE 2017, 12, e0186657. [Google Scholar] [CrossRef]
- Nordic Council of Ministers. The fortification of foods with vitamins and minerals. In Tilsætning af Vitaminer og Mineraler til Levnedsmidler; Nordic Council of Ministers: Copenhagen, Denmark, 1989. [Google Scholar]
- Jacobsen, R.; Abrahamsen, B.; Bauerek, M.; Holst, C.; Jensen, C.B.; Knop, J.; Raymond, K.; Rasmussen, L.B.; Stougaard, M.; Sørensen, T.I.A.; et al. The influence of early exposure to vitamin D for development of diseases later in life. Bmc Public Health 2013, 13, 515. [Google Scholar] [CrossRef]
- Pedersen, C.B. The Danish Civil Registration System. Scand. J. Public Health 2011, 39 (Suppl. 7), 22–25. [Google Scholar] [CrossRef] [PubMed]
- Lynge, E.; Sandegaard, J.L.; Rebolj, M. The Danish National Patient Register. Scand. J. Public Health 2011, 39 (Suppl. 7), 30–33. [Google Scholar] [CrossRef] [PubMed]
- Juul, A.; Teilmann, G.; Scheike, T.; Hertel, N.T.; Holm, K.; Laursen, E.M.; Main, K.M.; Skakkebæk, N.E. Pubertal development in Danish children: Comparison of recent European and US data. Int. J. Androl. 2006, 29, 247–255. [Google Scholar] [CrossRef] [PubMed]
- Moth, G.; Vedsted, P.; Schiøtz, P.O. National registry diagnoses agree with medical records on hospitalized asthmatic children. Acta Pædiatr. 2007, 96, 1470–1473. [Google Scholar] [CrossRef] [PubMed]
- Therneau, T.M.; Grambsch, P.M. Modeling Survival Data: Extending the Cox Model; Statistics for Biology and Health; Springer: New York, NY, USA, 2000; Volume XIV, p. 350. [Google Scholar]
- Just, J.; Saint Pierre, P.; Amat, F.; Gouvis-Echraghi, R.; Lambert-Guillemot, N.; Guiddir, T.; Annesi Maesano, I. What lessons can be learned about asthma phenotypes in children from cohort studies? Pediatric Allergy Immunol. 2015, 26, 300–305. [Google Scholar] [CrossRef] [PubMed]
- Erkkola, M.; Kaila, M.; Nwaru, B.I.; Kronberg-Kippilä, C.; Ahonen, S.; Nevalainen, J.; Veijola, R.; Pekkanen, J.; Ilonen, J.; Simell, O.; et al. Maternal vitamin D intake during pregnancy is inversely associated with asthma and allergic rhinitis in 5-year-old children. Clin. Exp. Allergy 2009, 39, 875–882. [Google Scholar] [CrossRef] [PubMed]
- Camargo, C.A.; Rifas-Shiman, S.L.; Litonjua, A.A.; Rich-Edwards, J.W.; Weiss, S.T.; Gold, D.R.; Kleinman, K.; Gillman, M.W. Maternal intake of vitamin D during pregnancy and risk of recurrent wheeze in children at 3 y of age. Am. J. Clin. Nutr. 2007, 85, 788–795. [Google Scholar] [CrossRef] [PubMed]
- Devereux, G.; Litonjua, A.A.; Turner, S.W.; Craig, L.C.; McNeill, G.; Martindale, S.; Helms, P.J.; Seaton, A.; Weiss, S.T. Maternal vitamin D intake during pregnancy and early childhood wheezing. Am. J. Clin. Nutr. 2007, 85, 853–859. [Google Scholar] [CrossRef] [PubMed]
- Vuillermin, P.J.; Macia, L.; Nanan, R.; Tang, M.L.; Collier, F.; Brix, S. The maternal microbiome during pregnancy and allergic disease in the offspring. Semin Immunopathol. 2017, 39, 669–675. [Google Scholar] [CrossRef]
- Camargo, C.A.; Ingham, T.; Wickens, K.; Thadhani, R.; Silvers, K.M.; Epton, M.J.; Town, G.I.; Pattemore, P.K.; Espinola, J.A.; Crane, J. Cord-blood 25-hydroxyvitamin D levels and risk of respiratory infection, wheezing, and asthma. Pediatrics 2011, 127, e180–e187. [Google Scholar] [CrossRef] [PubMed]
- Chawes, B.L.; Bonnelykke, K.; Jensen, P.F.; Schoos, A.M.; Heickendorff, L.; Bisgaard, H. Cord blood 25(OH)-vitamin D deficiency and childhood asthma, allergy and eczema: The COPSAC2000 birth cohort study. PLoS ONE 2014, 9, e99856. [Google Scholar] [CrossRef]
- Gazibara, T.; den Dekker, H.T.; de Jongste, J.C.; McGrath, J.J.; Eyles, D.W.; Burne, T.H.; Reiss, I.K.; Franco, O.H.; Tiemeier, H.; Jaddoe, V.W.; et al. Associations of maternal and fetal 25-hydroxyvitamin D levels with childhood lung function and asthma: The Generation R Study. Clin. Exp. Allergy 2016, 46, 337–346. [Google Scholar] [CrossRef] [PubMed]
- Gopal-Kothandapani, J.S.; Evans, L.F.; Walsh, J.S.; Gossiel, F.; Rigby, A.S.; Eastell, R.; Bishop, N.J. Effect of vitamin D supplementation on free and total vitamin D: A comparison of Asians and Caucasians. Clin. Endocrinol. 2019, 90, 222–231. [Google Scholar] [CrossRef]
- Bikle, D.D.; Malmstroem, S.; Schwartz, J. Current Controversies: Are Free Vitamin Metabolite Levels a More Accurate Assessment of Vitamin D Status than Total Levels? Endocrinol. Metab. Clin. North Am. 2017, 46, 901–918. [Google Scholar] [CrossRef]
- Jacobsen, R.; Hypponen, E.; Sorensen, T.I.; Vaag, A.A.; Heitmann, B.L. Gestational and Early Infancy Exposure to Margarine Fortified with Vitamin D through a National Danish Programme and the Risk of Type 1 Diabetes: The D-Tect Study. PloS ONE 2015, 10, e0128631. [Google Scholar] [CrossRef]
- Nordic Nutrition Recommendations 2012; Nordic Council of Ministers: Copenhagen, Denmark, 2004; Volume 5.
- Stevenson, D.K.; Verter, J.; Fanaroff, A.A.; Oh, W.; Ehrenkranz, R.A.; Shankaran, S.; Donovan, E.F.; Wright, L.L.; Lemons, J.A.; Tyson, J.E. Sex differences in outcomes of very low birthweight infants: The newborn male disadvantage. Arch. Dis. Child. Fetal Neonatal Ed. 2000, 83, F182–F185. [Google Scholar] [CrossRef]
- Just, J.; Bourgoin-Heck, M.; Amat, F. Clinical phenotypes in asthma during childhood. Clin. Exp. Allergy 2017, 47, 848–855. [Google Scholar] [CrossRef]
- Guilbert, T.W.; Mauger, D.T.; Lemanske, R.F. Childhood asthma-predictive phenotype. J. Allergy Clin. Immunol. Pract. 2014, 2, 664–670. [Google Scholar] [CrossRef]
- Martinez, F.D.; Wright, A.L.; Taussig, L.M.; Holberg, C.J.; Halonen, M.; Morgan, W.J. Asthma and wheezing in the first six years of life. The Group Health Medical Associates. N. Engl. J. Med. 1995, 332, 133–138. [Google Scholar] [CrossRef]
- Torday, J.S.; Nielsen, H.C.; Fencl Mde, M.; Avery, M.E. Sex differences in fetal lung maturation. Am. Rev. Respir. Dis. 1981, 123, 205–208. [Google Scholar]
- Zosky, G.R.; Hart, P.H.; Whitehouse, A.J.; Kusel, M.M.; Ang, W.; Foong, R.E.; Chen, L.; Holt, P.G.; Sly, P.D.; Hall, G.L. Vitamin D deficiency at 16 to 20 weeks’ gestation is associated with impaired lung function and asthma at 6 years of age. Ann. Am. Thorac. Soc. 2014, 11, 571–577. [Google Scholar] [CrossRef]
- Pedersen, C.B.; Gotzsche, H.; Moller, J.O.; Mortensen, P.B. The Danish Civil Registration System. A cohort of eight million persons. Dan Med. Bull. 2006, 53, 441–449. [Google Scholar]
- Schmidt, M.; Schmidt, S.A.; Sandegaard, J.L.; Ehrenstein, V.; Pedersen, L.; Sorensen, H.T. The Danish National Patient Registry: A review of content, data quality, and research potential. Clin. Epidemiol. 2015, 7, 449–490. [Google Scholar] [CrossRef] [PubMed]
- Olsen, J.; Frische, G.; Poulsen, A.O.; Kirchheiner, H. Changing smoking, drinking, and eating behaviour among pregnant women in Denmark. Evaluation of a health campaign in a local region. Scand. J. Soc. Med. 1989, 17, 277–280. [Google Scholar] [CrossRef] [PubMed]
- Bendixen, H.; Holst, C.; Sorensen, T.I.; Raben, A.; Bartels, E.M.; Astrup, A. Major increase in prevalence of overweight and obesity between 1987 and 2001 among Danish adults. Obes. Res. 2004, 12, 1464–1472. [Google Scholar] [CrossRef] [PubMed]
- Ministry of Treasury. Tre Reformer af Personskatterne 1987–2002; Ministry of Treasury: Copenhagen, Denmark, 2001.
- Jensen, A. Kuren der rystede Danmark. Available online: https://www.berlingske.dk/samfund/kuren-der-rystede-danmark (accessed on 23 April 2019).
| Admissions | Time at Risk (Years) | Rate per 100,000 Years at Risk | Admissions | Time at Risk (Years) | Rate per 100,000 Years at Risk | HR | (95% CI) | Adjusted † HR | (95% CI) | p for Interaction with Month of Birth | |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Exposed | Unexposed | ||||||||||
| All | 1427 | 938,797 | 152.0 | 1613 | 1,022,110 | 157.8 | 0.96 | (0.90–1.03) | 0.96 | (0.90–1.04) | 0.28 |
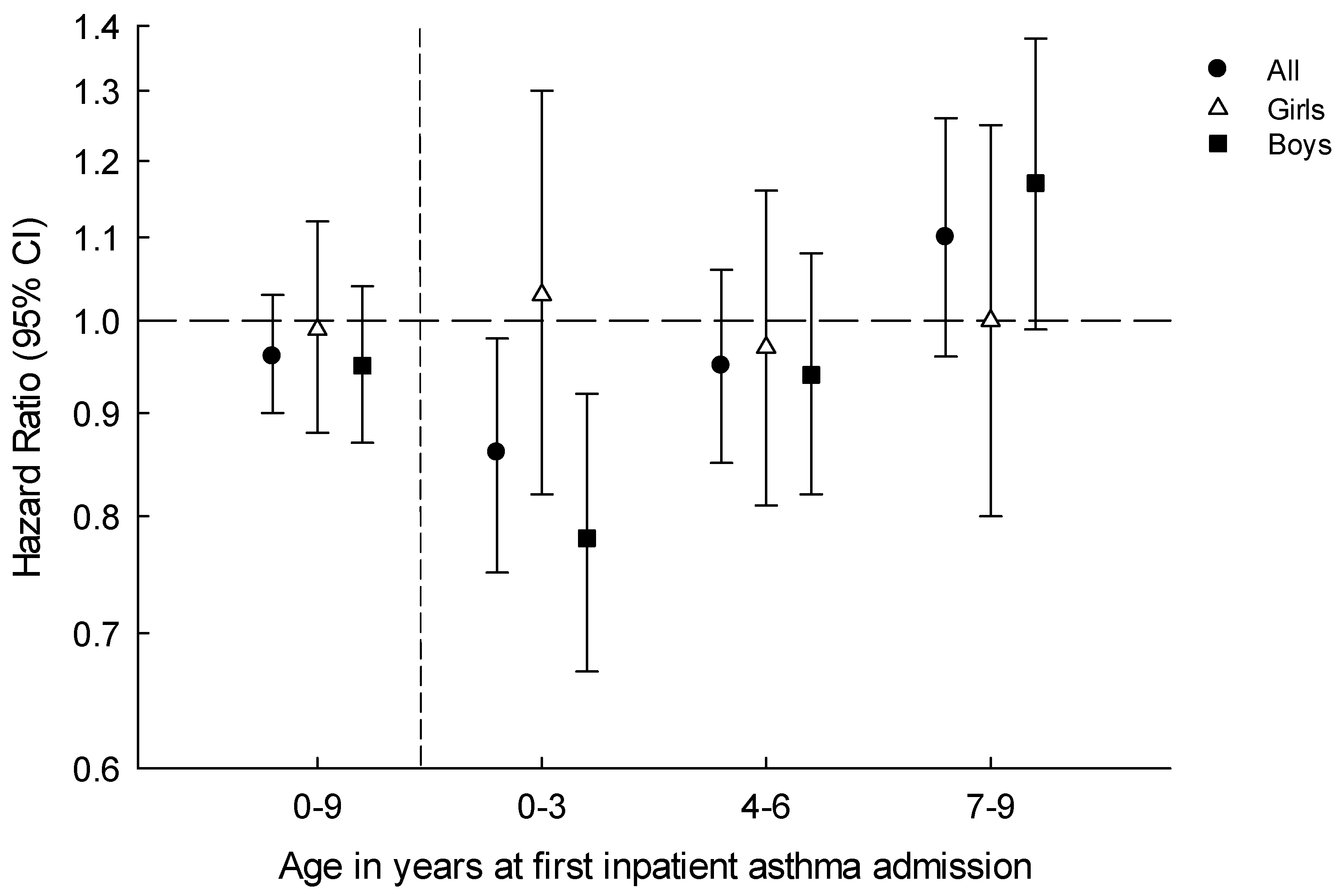
| 0–3 years | 393 | 315,879 | 124.4 | 498 | 343,928 | 144.8 | 0.86 | (0.75–0.98) | 0.86 | (0.75–0.98) | 0.63 |
| 4–6 years | 596 | 312,640 | 190.6 | 682 | 340,339 | 200.4 | 0.95 | (0.85–1.06) | 0.95 | (0.85–1.06) | 0.02 |
| 7–9 years | 438 | 310,278 | 141.2 | 433 | 337,843 | 128.2 | 1.10 | (0.96–1.26) | 1.10 | (0.96–1.26) | 0.78 |
| Girls | 515 | 457,866 | 112.5 | 562 | 496,107 | 113.3 | 0.99 | (0.88–1.12) | 0.99 | (0.88–1.12) | 0.30 |
| 0–3 years | 146 | 153,849 | 94.9 | 153 | 166,624 | 91.8 | 1.03 | (0.82–1.30) | 1.03 | (0.82–1.30) | 0.98 |
| 4–6 years | 221 | 152,494 | 144.9 | 248 | 165,223 | 150.1 | 0.97 | (0.81–1.16) | 0.97 | (0.81–1.16) | 0.14 |
| 7–9 years | 148 | 151,523 | 97.7 | 161 | 164,259 | 98.0 | 1.00 | (0.80–1.25) | 1.00 | (0.80–1.24) | 0.41 |
| Boys | 912 | 480,931 | 189.6 | 1,051 | 526,003 | 199.8 | 0.95 | (0.87–1.04) | 0.95 | (0.87–1.04) | 0.50 |
| 0–3 years | 247 | 177,304 | 194.6 | 345 | 162,030 | 152.4 | 0.78 | (0.67–0.92) | 0.78 | (0.67–0.92) | 0.24 |
| 4–6 years | 375 | 175,116 | 247.8 | 434 | 160,146 | 234.2 | 0.94 | (0.82–1.08) | 0.95 | (0.82–1.09) | 0.10 |
| 7–9 years | 290 | 173,583 | 156.7 | 272 | 158,755 | 182.7 | 1.17 | (0.99–1.38) | 1.16 | (0.99–1.37) | 0.71 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Thorsteinsdottir, F.; Maslova, E.; Jacobsen, R.; Frederiksen, P.; Keller, A.; Backer, V.; Heitmann, B.L. Exposure to Vitamin D Fortification Policy in Prenatal Life and the Risk of Childhood Asthma: Results from the D-Tect Study. Nutrients 2019, 11, 924. https://doi.org/10.3390/nu11040924
Thorsteinsdottir F, Maslova E, Jacobsen R, Frederiksen P, Keller A, Backer V, Heitmann BL. Exposure to Vitamin D Fortification Policy in Prenatal Life and the Risk of Childhood Asthma: Results from the D-Tect Study. Nutrients. 2019; 11(4):924. https://doi.org/10.3390/nu11040924
Chicago/Turabian StyleThorsteinsdottir, Fanney, Ekaterina Maslova, Ramune Jacobsen, Peder Frederiksen, Amélie Keller, Vibeke Backer, and Berit Lilienthal Heitmann. 2019. "Exposure to Vitamin D Fortification Policy in Prenatal Life and the Risk of Childhood Asthma: Results from the D-Tect Study" Nutrients 11, no. 4: 924. https://doi.org/10.3390/nu11040924
APA StyleThorsteinsdottir, F., Maslova, E., Jacobsen, R., Frederiksen, P., Keller, A., Backer, V., & Heitmann, B. L. (2019). Exposure to Vitamin D Fortification Policy in Prenatal Life and the Risk of Childhood Asthma: Results from the D-Tect Study. Nutrients, 11(4), 924. https://doi.org/10.3390/nu11040924






