Weight Change and Cardiometabolic Outcomes in Postpartum Women with History of Gestational Diabetes
Abstract
1. Introduction
2. Materials and Methods
2.1. Trial Design
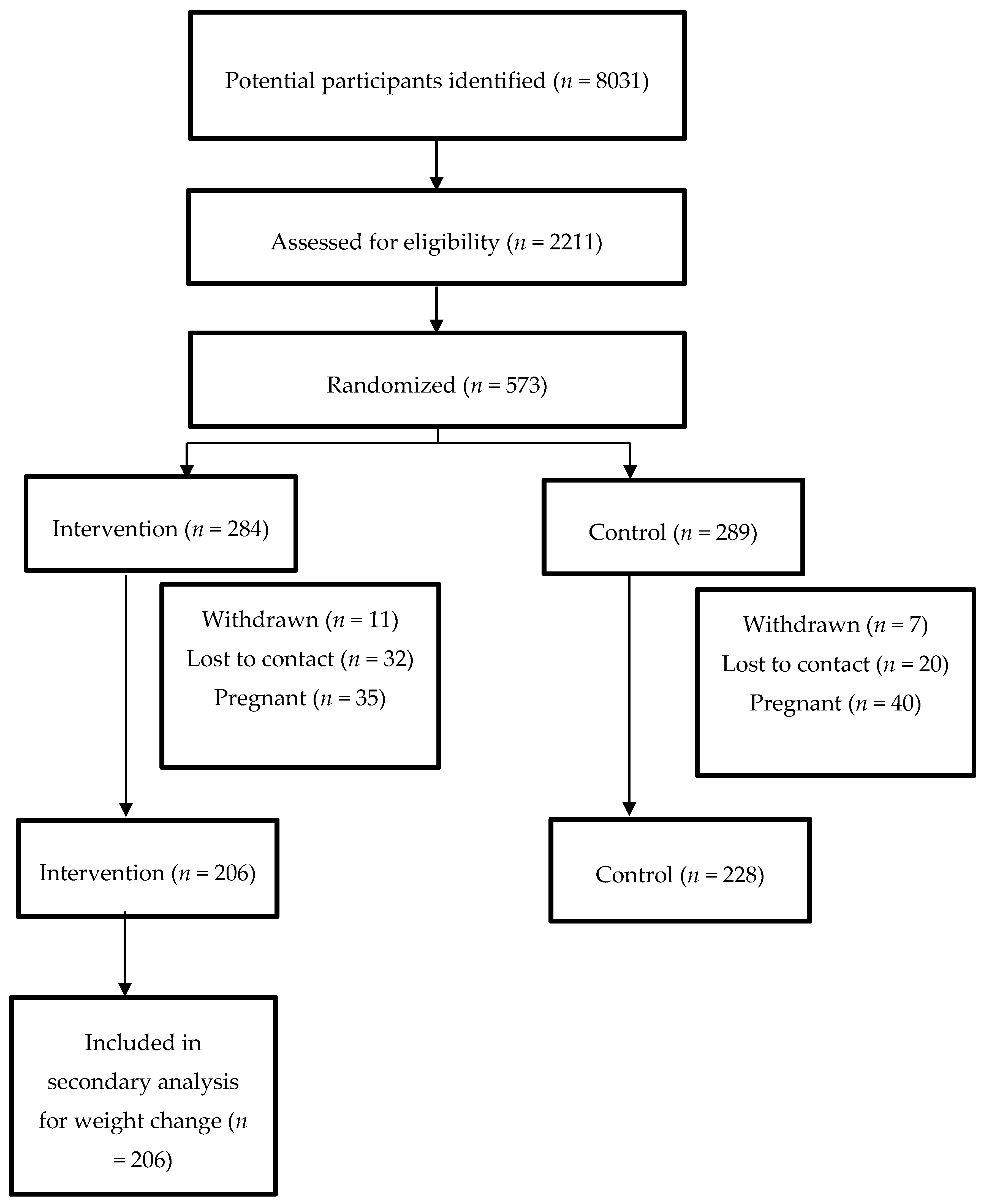
2.2. Participants
2.3. Intervention
2.4. Control
2.5. Measurements
2.6. Randomization
2.7. Statistical Analyses
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Sacks, D.A.; Hadden, D.R.; Maresh, M.; Deerochanawong, C.; Dyer, A.R.; Metzger, B.E.; Lowe, L.P.; Coustan, D.R.; Hod, M.; Oats, J.J.N.; et al. Frequency of gestational diabetes mellitus at collaborating centers based on iadpsg consensus panel–recommended criteria: The hyperglycemia and adverse pregnancy outcome (hapo) study. Diabetes Care 2012, 35, 526–528. [Google Scholar] [CrossRef]
- Brown, F.M.; Wyckoff, J. Application of one-step iadpsg versus two-step diagnostic criteria for gestational diabetes in the real world: Impact on health services, clinical care, and outcomes. Curr. Diabetes Rep. 2017, 17, 85. [Google Scholar] [CrossRef]
- Bellamy, L.; Casas, J.P.; Hingorani, A.D.; Williams, D. Type 2 diabetes mellitus after gestational diabetes: A systematic review and meta-analysis. Lancet 2009, 373, 1773–1779. [Google Scholar] [CrossRef]
- Linne, Y.; Barkeling, B.; Rossner, S. Natural course of gestational diabetes mellitus: Long term follow up of women in the spawn study. BJOG 2002, 109, 1227–1231. [Google Scholar] [CrossRef]
- Liu, H.K.; Zhang, C.P.; Zhang, S.; Wang, L.S.; Leng, J.H.; Liu, D.D.; Fang, H.; Li, W.Q.; Yu, Z.J.; Yang, X.L.; et al. Prepregnancy body mass index and weight change on postpartum diabetes risk among gestational diabetes women. Obesity 2014, 22, 1560–1567. [Google Scholar] [CrossRef] [PubMed]
- Knowler, W.C.; Barrett-Connor, E.; Fowler, S.E.; Hamman, R.F.; Lachin, J.M.; Walker, E.A.; Nathan, D.M. Reduction in the incidence of type 2 diabetes with lifestyle intervention or metformin. N. Engl. J. Med. 2002, 346, 393–403. [Google Scholar] [PubMed]
- Finnish Diabetes Prevention Study Group; Lindstrom, J.; Louheranta, A.; Mannelin, M.; Rastas, M.; Salminen, V.; Eriksson, J.; Uusitupa, M.; Tuomilehto, J. The finnish diabetes prevention study (dps): Lifestyle intervention and 3-year results on diet and physical activity. Diabetes Care 2003, 26, 3230–3236. [Google Scholar] [CrossRef]
- O’Reilly, S.L.; Dunbar, J.A.; Versace, V.; Janus, E.; Best, J.D.; Carter, R.; Oats, J.J.; Skinner, T.; Ackland, M.; Phillips, P.A.; et al. Mothers after gestational diabetes in australia (magda): A randomised controlled trial of a postnatal diabetes prevention program. PLoS Med. 2016, 13, e1002092. [Google Scholar] [CrossRef]
- Howells, L.; Musaddaq, B.; McKay, A.J.; Majeed, A. Clinical impact of lifestyle interventions for the prevention of diabetes: An overview of systematic reviews. BMJ Open 2016, 6, e013806. [Google Scholar] [CrossRef] [PubMed]
- Dunkley, A.J.; Bodicoat, D.H.; Greaves, C.J.; Russell, C.; Yates, T.; Davies, M.J.; Khunti, K. Diabetes prevention in the real world: Effectiveness of pragmatic lifestyle interventions for the prevention of type 2 diabetes and of the impact of adherence to guideline recommendations: A systematic review and meta-analysis. Diabetes Care 2014, 37, 922–933. [Google Scholar] [CrossRef]
- Lindstrom, J.; Ilanne-Parikka, P.; Peltonen, M.; Aunola, S.; Eriksson, J.G.; Hemio, K.; Hamalainen, H.; Harkonen, P.; Keinanen-Kiukaanniemi, S.; Laakso, M.; et al. Sustained reduction in the incidence of type 2 diabetes by lifestyle intervention: Follow-up of the finnish diabetes prevention study. Lancet 2006, 368, 1673–1679. [Google Scholar] [CrossRef]
- Goveia, P.; Canon-Montanez, W.; Santos, D.P.; Lopes, G.W.; Ma, R.C.W.; Duncan, B.B.; Ziegelman, P.K.; Schmidt, M.I. Lifestyle intervention for the prevention of diabetes in women with previous gestational diabetes mellitus: A systematic review and meta-analysis. Front. Endocrinol. (Lausanne) 2018, 9, 583. [Google Scholar] [CrossRef]
- Diabetes Prevention Program Research Group. 10-year follow-up of diabetes incidence and weight loss in the diabetes prevention program outcomes study. Lancet 2009, 374, 1677–1686. [Google Scholar] [CrossRef]
- Dutton, G.R.; Kim, Y.; Jacobs, D.R.; Li, X.; Loria, C.M.; Reis, J.P.; Carnethon, M.; Durant, N.H.; Gordon-Larsen, P.; Shikany, J.M.; et al. 25-year weight gain in a racially balanced sample of u.S. Adults: The cardia study. Obesity (Silver Spring) 2016, 24, 1962–1968. [Google Scholar] [CrossRef]
- Adamson, L.; Brown, W.; Byles, J.; Chojenta, C.; Dobson, A.; Fitzgerald, D.; Hockey, R.; Loxton, D.; Powers, J.; Spallek, M.; et al. Women’s Weight: Findings from the Australian Longitudinal Study on Women’s Health.; Australian Longitudinal Study on Women’s Health: Newcastle, Queensland, Australia, 2007. [Google Scholar]
- Ratner, R.E.; Christophi, C.A.; Metzger, B.E.; Dabelea, D.; Bennett, P.H.; Pi-Sunyer, X.; Fowler, S.; Kahn, S.E.; Diabetes Prevention Program Research, G. Prevention of diabetes in women with a history of gestational diabetes: Effects of metformin and lifestyle interventions. J. Clin. Endocrinol. Metab. 2008, 93, 4774–4779. [Google Scholar] [CrossRef]
- Ball, K.; Brown, W.; Crawford, D. Who does not gain weight? Prevalence and predictors of weight maintenance in young women. Int. J. Obes. Relat. Metab. Disord. 2002, 26, 1570–1578. [Google Scholar] [CrossRef] [PubMed]
- Ostbye, T.; Peterson, B.L.; Krause, K.M.; Swamy, G.K.; Lovelady, C.A. Predictors of postpartum weight change among overweight and obese women: Results from the active mothers postpartum study. J. Womens Health 2012, 21, 215–222. [Google Scholar] [CrossRef]
- Carter-Edwards, L.; Ostbye, T.; Bastian, L.A.; Yarnall, K.S.; Krause, K.M.; Simmons, T.J. Barriers to adopting a healthy lifestyle: Insight from postpartum women. BMC Res. Notes 2009, 2, 161. [Google Scholar] [CrossRef] [PubMed]
- Nicklas, J.M.; Zera, C.A.; Seely, E.W.; Abdul-Rahim, Z.S.; Rudloff, N.D.; Levkoff, S.E. Identifying postpartum intervention approaches to prevent type 2 diabetes in women with a history of gestational diabetes. BMC Pregnancy Childbirth 2011, 11, 23. [Google Scholar] [CrossRef] [PubMed]
- Lim, S.S.; Norman, R.J.; Clifton, P.M.; Noakes, M. The effect of comprehensive lifestyle intervention or metformin on obesity in young women. Nutr. Metab. Cardiovasc. Dis. 2011, 21, 261–268. [Google Scholar] [CrossRef]
- Dasgupta, K.; Terkildsen Maindal, H.; Kragelund Nielsen, K.; O‘Reilly, S. Achieving penetration and participation in diabetes after pregnancy prevention interventions following gestational diabetes: A health promotion challenge. Diabetes Res. Clin. Pract. 2018. [Google Scholar] [CrossRef] [PubMed]
- Guo, J.; Chen, J.L.; Whittemore, R.; Whitaker, E. Postpartum lifestyle interventions to prevent type 2 diabetes among women with history of gestational diabetes: A systematic review of randomized clinical trials. J. Womens Health 2016, 25, 38–49. [Google Scholar] [CrossRef] [PubMed]
- Ramlo-Halsted, B.A.; Edelman, S.V. The natural history of type 2 diabetes—Implications for clinical practice. Primary Care 1999, 26, 771–790. [Google Scholar] [CrossRef]
- Gasevic, D.; Frohlich, J.; Mancini, G.B.J.; Lear, S.A. The association between triglyceride to high-density-lipoprotein cholesterol ratio and insulin resistance in a multiethnic primary prevention cohort. Metabol. Clin. Exper. 2012, 61, 583–589. [Google Scholar] [CrossRef]
- Kim-Dorner, S.J.; Deuster, P.A.; Zeno, S.A.; Remaley, A.T.; Poth, M. Should triglycerides and the triglycerides to high-density lipoprotein cholesterol ratio be used as surrogates for insulin resistance? Metabol. Clin. Exper. 2010, 59, 299–304. [Google Scholar] [CrossRef]
- Shih, S.T.F.; Davis-Lameloise, N.; Janus, E.D.; Wildey, C.; Versace, V.L.; Hagger, V.; Asproloupos, D.; O’Reilly, S.; Phillips, P.A.; Ackland, M.; et al. Mothers after gestational diabetes in australia diabetes prevention program (magda-dpp) post-natal intervention: Study protocol for a randomized controlled trial. Trials 2013, 14. [Google Scholar] [CrossRef]
- Dunbar, J.A.; Hernan, A.L.; Janus, E.D.; Vartiainen, E.; Laatikainen, T.; Versace, V.L.; Reynolds, J.; Best, J.D.; Skinner, T.C.; O’Reilly, S.L.; et al. Challenges of diabetes prevention in the real world: Results and lessons from the melbourne diabetes prevention study. BMJ Open Diabetes Res. Care 2015, 3. [Google Scholar] [CrossRef]
- Laatikainen, T.; Dunbar, J.A.; Chapman, A.; Kilkkinen, A.; Vartiainen, E.; Heistaro, S.; Philpot, B.; Absetz, P.; Bunker, S.; O‘Neil, A.; et al. Prevention of type 2 diabetes by lifestyle intervention in an australian primary health care setting: Greater green triangle (ggt) diabetes prevention project. BMC Public Health 2007, 7, 249. [Google Scholar] [CrossRef]
- Tuomilehto, J.; Lindström, J.; Eriksson, J.G.; Valle, T.T.; Hämäläinen, H.; Ilanne-Parikka, P.; Keinänen-Kiukaanniemi, S.; Laakso, M.; Louheranta, A.; Rastas, M.; et al. Prevention of type 2 diabetes mellitus by changes in lifestyle among subjects with impaired glucose tolerance. N. Engl. J. Med. 2001, 344, 1343–1350. [Google Scholar] [CrossRef]
- Nankervis, A.; McIntyre, H.D.; Moses, R.; Ross, G.P.; Callaway, L.; Porter, C.; Jeffries, W.; Boorman, C.; De Vries, B.; McElduff, A. ADIPS Consensus Guidelines for the Testing and Diagnosis of Gestational Diabetes Mellitus in Australia; The Australasian Diabetes in Pregnancy Society: Sydney, Australia, 2014. [Google Scholar]
- Tolonen, H.; Kuulasmaa, K.; Laatikainen, T.; Wolf, H.; Project, E.H.R.M. Recommendations for Indicators, International Collaboration, Protocol and Manual Operations Chronic Disease Risk Factor Surveys; Finnish National Public Health Institute: Helsinki, Finland, 2002. [Google Scholar]
- Liu, H.K.; Wang, L.S.; Zhang, S.; Leng, J.; Li, N.; Li, W.Q.; Wang, J.; Tian, H.G.; Qi, L.; Yang, X.L.; et al. One-year weight losses in the tianjin gestational diabetes mellitus prevention programme: A randomized clinical trial. Diabetes Obes. Metabol. 2018, 20, 1246–1255. [Google Scholar] [CrossRef]
- Peacock, A.S.; Bogossian, F.E.; Wilkinson, S.A.; Gibbons, K.S.; Kim, C.; McIntyre, H.D. A randomised controlled trial to delay or prevent type 2 diabetes after gestational diabetes: Walking for exercise and nutrition to prevent diabetes for you. Int. J. Endocrinol. 2015. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Y.; Manson, J.E.; Yuan, C.; Liang, M.H.; Grodstein, F.; Stampfer, M.J.; Willett, W.C.; Hu, F.B. Associations of weight gain from early to middle adulthood with major health outcomes later in life. JAMA 2017, 318, 255–269. [Google Scholar] [CrossRef] [PubMed]
- Pennings, N.; Jaber, J.; Ahiawodzi, P. Ten-year weight gain is associated with elevated fasting insulin levels and precedes glucose elevation. Diabetes-Metab. Res. 2018, 34, e2986. [Google Scholar] [CrossRef] [PubMed]
- Sotos-Prieto, M.; Bhupathiraju, S.N.; Mattei, J.; Fung, T.T.; Li, Y.; Pan, A.; Willett, W.C.; Rimm, E.B.; Hu, F.B. Association of changes in diet quality with total and cause-specific mortality. N. Engl. J. Med. 2017, 377, 143–153. [Google Scholar] [CrossRef] [PubMed]
- Kramer, C.K.; Campbell, S.; Retnakaran, R. Gestational diabetes and the risk of cardiovascular disease in women: A systematic review and meta-analysis. Diabetologia 2019. [Google Scholar] [CrossRef]
- Lu, M.Q.; Wan, Y.; Yang, B.; Huggins, C.E.; Li, D. Effects of low-fat compared with high-fat diet on cardiometabolic indicators in people with overweight and obesity without overt metabolic disturbance: A systematic review and meta-analysis of randomised controlled trials. Brit. J. Nutr. 2018, 119, 96–108. [Google Scholar] [CrossRef]
- Dansinger, M.; Williams, P.T.; Superko, H.R.; Asztalos, B.F.; Schaefer, E.J. Effects of weight change on hdl-cholesterol and its subfractions in over 28,000 men and women. J. Clin. Lipidol. 2018. [Google Scholar] [CrossRef]
- Zwald, M.L.; Akinbami, L.J.; Fakhouri, T.H.; Fryar, C.D. Prevalence of low high-density lipoprotein cholesterol among adults, by physical activity: United States, 2011–2014. NCHS Data Brief. 2017, 276, 1–8. [Google Scholar]
- Kim, C.; Newton, K.M.; Knopp, R.H. Gestational diabetes and the incidence of type 2 diabetes—A systematic review. Diabetes Care 2002, 25, 1862–1868. [Google Scholar] [CrossRef]
- Valderhaug, T.G.; Sharma, A.; Kravdal, G.; Ronningen, R.; Nermoen, I. The usage of fasting glucose and glycated hemoglobin for the identification of unknown type 2 diabetes in high risk patients with morbid obesity. Scand. J. Clin. Lab. Investig. 2017, 77, 505–512. [Google Scholar] [CrossRef]
- Moon, J.H.; Kwak, S.H.; Jung, H.S.; Choi, S.H.; Lim, S.; Cho, Y.M.; Park, K.S.; Jang, H.C.; Cho, N.H. Weight gain and progression to type 2 diabetes in women with a history of gestational diabetes mellitus. J. Clin. Endocrinol. Metabol. 2015, 100, 3548–3555. [Google Scholar] [CrossRef] [PubMed]
- Andajani-Sutjahjo, S.; Ball, K.; Warren, N.; Inglis, V.; Crawford, D. Perceived personal, social and environmental barriers to weight maintenance among young women: A community survey. Int. J. Behav. Nutr. Phys. Act. 2004, 1, 15. [Google Scholar] [CrossRef] [PubMed]
| Weight Loss >2 kg (n = 74) | Weight Stable ±2 kg (n = 74) | Weight Gain >2 kg (n = 58) | p-Value | |
|---|---|---|---|---|
| Age | ||||
| ≤24 | 2 (2.7) | 1 (1.4) | 2 (3.4) | 0.422 |
| 25–29 | 10 (13.5) | 6 (8.2) | 8 (13.8) | |
| 30–34 | 27 (36.5) | 26 (35.6) | 18 (31.0) | |
| 35–39 | 21 (28.4) | 27 (37.0) | 12 (20.7) | |
| ≥40 | 14 (18.9) | 13 (17.8) | 18 (31.0) | |
| BMI | ||||
| <20 | 4 (5.6) | 8 (11.0) | 1 (1.7) | 0.061 |
| 20 to <25 | 21 (29.2) | 26 (35.6) | 14 (24.1) | |
| 25 to <30 | 22 (30.6) | 13 (17.8) | 12 (20.7) | |
| >30 | 25 (34.7) | 26 (35.6) | 31 (53.4) | |
| PHQ | ||||
| Minimal depression | 43 (58.1) | 50 (68.5) | 40 (69.0) | 0.127 |
| Mild depression (0–9) | 24 (32.4) | 17 (23.3) | 16 (27.6) | |
| Moderate depression (10–19) | 7 (9.5) | 3 (4.1) | 2 (3.4) | |
| Moderately severe depression (20–27) | 3 (4.1) | |||
| Work Situation | ||||
| Full time | 13 (17.6) | 12 (16.2) | 10 (17.2) | 0.913 |
| Part time | 13 (17.6) | 12 (16.2) | 14 (24.1) | |
| Casual | 3 (4.1) | 2 (2.7) | 2 (3.4) | |
| Home duties | 34 (45.9) | 36 (48.6) | 22 (37.9) | |
| Unemployed | 2 (2.7) | 2 (2.7) | ||
| Other | 9 (12.2) | 9 (12.2) | 10 (17.2) | |
| Smoking Status | ||||
| No | 72 (97.3) | 72 (97.3) | 54 (93.1) | 0.291 |
| Yes | 2 (2.7) | 1 (1.4) | 4 (6.9) | |
| Family Income | ||||
| Low | 19 (25.7) | 21 (28.4) | 9 (15.5) | 0.124 |
| Medium | 34 (45.9) | 26 (35.1) | 28 (48.3) | |
| High | 21 (28.4) | 24 (32.4) | 21 (36.2) | |
| Highest Level Education | ||||
| Primary | 1 (1.4) | 2 (2.7) | 0.083 | |
| Secondary | 7 (9.5) | 9 (12.2) | 8 (13.8) | |
| Certificate Level | 10 (13.5) | 5 (6.8) | 5 (8.6) | |
| Diploma Level | 4 (5.4) | 10 (13.5) | 11 (19.0) | |
| Bachelor Degree | 37 (50.0) | 30 (40.5) | 22 (37.9) | |
| Master Degree | 14 (18.9) | 15 (20.3) | 6 (10.3) | |
| Doctoral Degree | 1 (1.4) | 3 (5.2) | ||
| Other | 2 (2.7) | 3 (5.2) | ||
| Time since Childbirth | ||||
| 0–26 weeks | 31 (43.7) | 39 (52.7) | 33 (56.9) | 0.578 |
| 27–52 weeks | 28 (39.4) | 20 (27.0) | 15 (25.9) | |
| 53–104 weeks | 11 (15.5) | 14 (18.9) | 10 (17.2) | |
| >104 weeks | 1 (1.4) | 1 (1.4) | ||
| Marital Status | ||||
| Married | 62 (83.8) | 53 (71.6) | 47 (81.0) | 0.48 |
| Single | 4 (5.4) | 4 (5.4) | 2 (3.4) | |
| Widowed | ||||
| De facto | 7 (9.5) | 15 (20.3) | 9 (15.5) | |
| Divorced | 1 (1.4) | |||
| Separated | 1 (1.4) | |||
| Breastfeeding Initiated | ||||
| No | 12 (16.2) | 13 (17.6) | 7 (12.1) | 0.621 |
| Yes | 62 (83.8) | 60 (81.1) | 51 (87.9) | |
| Parity | ||||
| 1 child | 32 (43.2) | 28 (38.4) | 24 (41.4) | 0.976 |
| 2 children | 27 (36.5) | 30 (41.1) | 23 (39.7) | |
| ≥3 children | 15 (20.3) | 15 (20.5) | 11 (19.0) | |
| Total Sessions Attended | ||||
| 0 | 19 (25.7) | 19 (25.7) | 14 (24.1) | 0.795 |
| 1 | 12 (16.2) | 8 (10.8) | 7 (12.1) | |
| 2 | 6 (8.1) | 6 (8.1) | 7 (12.1) | |
| 3 | 5 (6.8) | 6 (8.1) | 2 (3.4) | |
| 4 | 4 (5.4) | 2 (2.7) | 6 (10.3) | |
| 5 | 12 (16.2) | 10 (13.5) | 7 (12.1) | |
| 6 | 11 (14.9) | 11 (14.9) | 9 (15.5) | |
| 7 | 4 (5.4) | 7 (9.5) | 5 (8.6) | |
| 8 | 1 (1.4) | 5 (6.8) | 1 (1.7) | |
| Weight Loss >2 kg (n = 74) | Weight Stability ±2 kg (n = 74) | Weight Gain >2 kg (n = 58) | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Baseline | 12 Months | Change | p-Value 1 | Baseline | 12 Months | Change | p-Value 1 | Baseline | 12 Months | Change | p-Value 1 | p-Value 2 | p-Value 3 | |
| BMI, kg/m2 | 28.4 ± 5.8 | 26.5 ± 5.6 | −1.9 ± 1.1 a | <0.001 | 28.0 ± 7.5 | 28.1 ± 7.5 | 0.1 ± 0.5 b | 0.191 | 31.5 ± 7.8 | 33.4 ± 8.2 | 1.9 ± 1.0 c | <0.001 | <0.001 | <0.001 |
| Weight, kg | 73.5 ± 18.6 | 68.7 ± 17.9 | −4.9 ± 2.9 a | <0.001 | 73.5 ± 20.8 | 73.7 ± 20.8 | 0.2 ± 1.2 b | 0.153 | 83.5 ± 21.4 | 88.7 ± 23.0 | 5.2 ± 3.0 c | <0.001 | <0.001 | <0.001 |
| Waist, cm | 89.2 ± 12.1 | 83.7 ± 12.1 | −5.4 ± 5.1 a | <0.001 | 90.6 ± 15.2 | 88.8 ± 15.5 | −1.8 ± 4.6 b | 0.001 | 96.5 ± 15.6 | 97.9 ± 16.1 | 1.4 ± 4.6 c | 0.026 | <0.001 | <0.001 |
| FPG, mmol/L | 4.8 ± 0.5 | 4.9 ± 0.5 | 0.1 ± 0.4 a | 0.074 | 4.8 ± 0.6 | 5.0 ± 0.7 | 0.2 ± 0.5 ab | 0.001 | 4.8 ± 0.5 | 5.1 ± 0.7 | 0.3 ± 0.6 bc | 0.001 | 0.101 | 0.076 |
| HbA1c, % | 5.30 ± 0.4 | 5.18 ± 0.4 | −0.12 ± 0.4 a | 0.004 | 5.40 ± 0.4 | 5.34 ± 0.5 | −0.06 ± 0.5 ab | 0.292 | 5.33 ± 0.4 | 5.35 ± 0.4 | 0.02 ± 0.4 bc | 0.658 | 0.144 | 0.141 |
| Tchol, mmol/L | 5.2 ± 0.9 | 4.7 ± 0.9 | −0.6 ± 0.8 a | <0.001 | 5.1 ± 0.9 | 4.8 ± 0.8 | −0.2 ± 0.7 b | 0.007 | 5.2 ± 1.0 | 4.9 ± 0.8 | −0.2 ± 0.6 b | 0.005 | 0.005 | 0.004 |
| LDL, mmol/L | 3.2 ± 0.9 | 2.8 ± 0.8 | −0.4 ± 0.6 a | <0.001 | 3.1 ± 0.8 | 2.9 ± 0.7 | −0.2 ± 0.6 b | 0.044 | 3.0 ± 0.9 | 2.9 ± 0.8 | −0.1 ± 0.7 b | 0.238 | 0.017 | 0.014 |
| HDL, mmol/L | 1.5 ± 0.3 | 1.4 ± 0.3 | −0.1 ± 0.3 a | 0.014 | 1.4 ± 0.4 | 1.3 ± 0.4 | −0.1 ± 0.2 a | 0.014 | 1.5 ± 0.3 | 1.3 ± 0.3 | −0.2 ± 0.3 a | <0.001 | 0.884 | 0.891 |
| TG, mmol/L | 1.3 ± 0.7 | 1.1 ± 0.5 | −0.2 ± 0.5 a | 0.001 | 1.3 ± 0.8 | 1.3 ± 0.6 | 0 ± 0.6 b | 0.984 | 1.2 ± 0.6 | 1.4 ± 0.7 | 0.3 ± 0.5 c | <0.001 | <0.001 | <0.001 |
| TG/HDL | 1.0 ± 0.7 | 0.9 ± 0.6 | −0.1 ± 0.4 a | 0.017 | 1.1 ± 0.9 | 1.1 ± 0.8 | 0 ± 0.6 a | 0.625 | 0.9 ± 0.7 | 1.2 ± 0.8 | 0.4 ± 0.7 b | <0.001 | <0.001 | <0.001 |
| SBP, mmHg | 111.4 ± 11.5 | 112.0 ± 10.6 | 0.7 ± 9.5 a | 0.536 | 113.1 ± 14.4 | 112.0 ± 13.2 | −1.1 ± 9.5 a | 0.338 | 113.7 ± 12.8 | 114.9 ± 10.5 | 1.2 ± 12.3 a | 0.460 | 0.407 | 0.298 |
| DBP, mmHg | 70.4 ± 9.3 | 72.3 ± 9.5 | 2 ± 8.5 a | 0.052 | 71.1 ± 10.6 | 71.8 ± 11.3 | 0.7 ± 7.3 a | 0.422 | 72.0 ± 8.6 | 73.4 ± 8.4 | 1.5 ± 7.9 a | 0.161 | 0.614 | 0.620 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lim, S.; Versace, V.L.; O’Reilly, S.; Janus, E.; Dunbar, J. Weight Change and Cardiometabolic Outcomes in Postpartum Women with History of Gestational Diabetes. Nutrients 2019, 11, 922. https://doi.org/10.3390/nu11040922
Lim S, Versace VL, O’Reilly S, Janus E, Dunbar J. Weight Change and Cardiometabolic Outcomes in Postpartum Women with History of Gestational Diabetes. Nutrients. 2019; 11(4):922. https://doi.org/10.3390/nu11040922
Chicago/Turabian StyleLim, Siew, Vincent L. Versace, Sharleen O’Reilly, Edward Janus, and James Dunbar. 2019. "Weight Change and Cardiometabolic Outcomes in Postpartum Women with History of Gestational Diabetes" Nutrients 11, no. 4: 922. https://doi.org/10.3390/nu11040922
APA StyleLim, S., Versace, V. L., O’Reilly, S., Janus, E., & Dunbar, J. (2019). Weight Change and Cardiometabolic Outcomes in Postpartum Women with History of Gestational Diabetes. Nutrients, 11(4), 922. https://doi.org/10.3390/nu11040922





