Changes in Pancreatic Fat Content Following Diet-Induced Weight Loss
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Population
2.2. Laboratory Methods
2.3. Imaging
2.4. Statistical Analyses
3. Results
3.1. Characteristics of the Study Population at Baseline
3.2. Characteristics of Pancreatic Fat with Blood Biomarkers and Body Fat Volumes
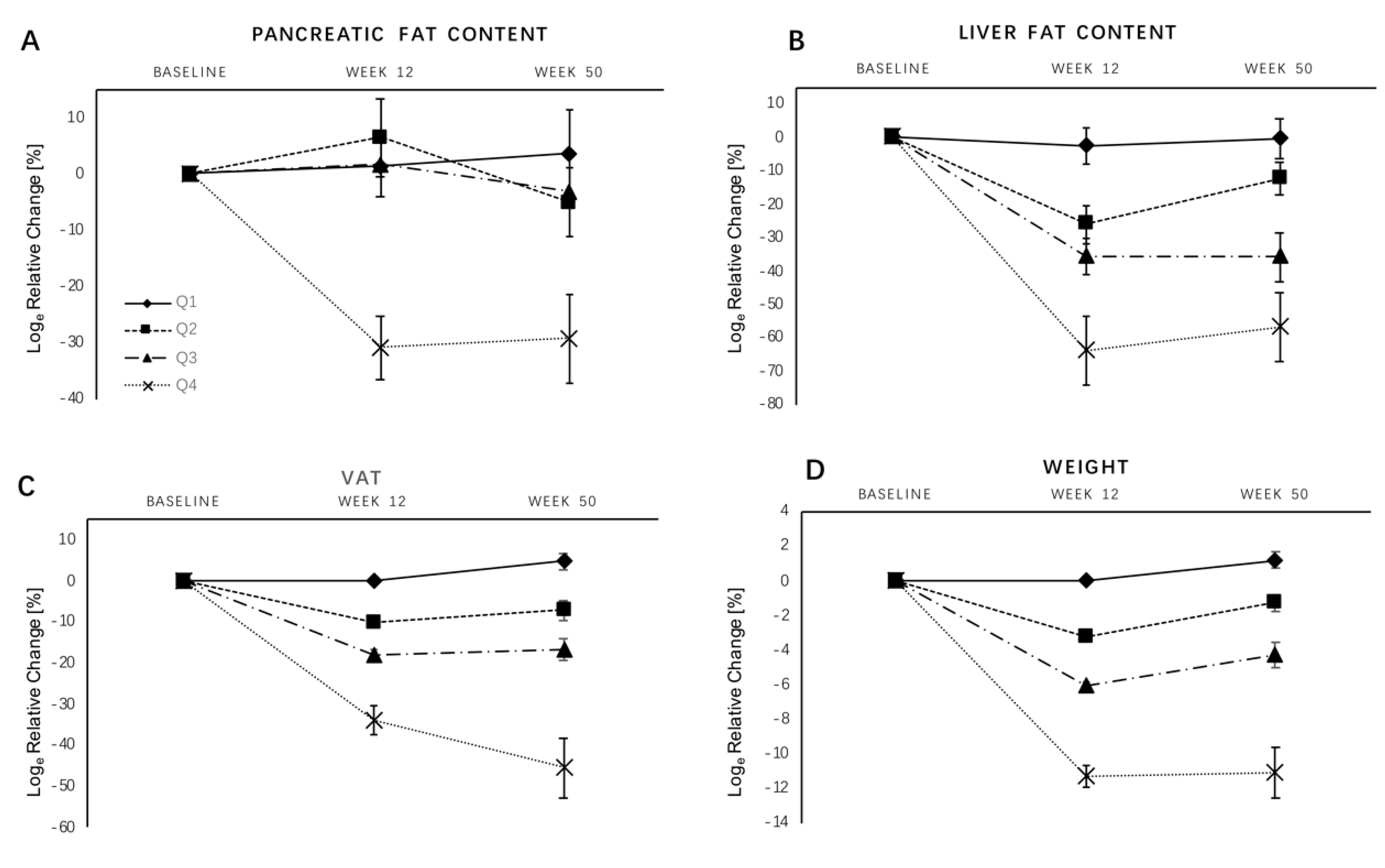
3.3. Effects of Weight Loss on Pancreatic Fat
4. Discussion
Author Contributions
Funding
Conflicts of Interest
References
- Fock, K.M.; Khoo, J. Diet and exercise in management of obesity and overweight: Diet and exercise for weight management. J. Gastroenterol. Hepatol. 2013, 28, 59–63. [Google Scholar] [CrossRef] [PubMed]
- Windarti, N.; Hlaing, S.W.; Kakinaka, M. Obesity Kuznets curve: International evidence. Public Health 2019, 169, 26–35. [Google Scholar] [CrossRef]
- Britton, K.A.; Fox, C.S. Ectopic Fat Depots and Cardiovascular Disease. Circulation 2011, 124, e837–e841. [Google Scholar] [CrossRef]
- Tushuizen, M.E.; Bunck, M.C.; Pouwels, P.J.; Bontemps, S.; van Waesberghe, J.H.T.; Schindhelm, R.K.; Mari, A.; Heine, R.J.; Diamant, M. Pancreatic Fat Content and -Cell Function in Men With and Without Type 2 Diabetes. Diabetes Care 2007, 30, 2916–2921. [Google Scholar] [CrossRef] [PubMed]
- Ou, H.Y.; Wang, C.Y.; Yang, Y.C.; Chen, M.F.; Chang, C.J. The Association between Nonalcoholic Fatty Pancreas Disease and Diabetes. PLoS ONE 2013, 8, e62561. [Google Scholar] [CrossRef]
- Kühn, J.-P.; Berthold, F.; Mayerle, J.; Völzke, H.; Reeder, S.B.; Rathmann, W.; Lerch, M.M.; Hosten, N.; Hegenscheid, K.; Meffert, P.J. Pancreatic Steatosis Demonstrated at MR Imaging in the General Population: Clinical Relevance. Radiology 2015, 276, 129–136. [Google Scholar] [CrossRef]
- Catanzaro, R.; Cuffari, B.; Italia, A.; Marotta, F. Exploring the metabolic syndrome: Nonalcoholic fatty pancreas disease. World J. Gastroenterol. 2016, 22, 7660–7675. [Google Scholar] [CrossRef]
- Sakai, N.S.; Taylor, S.A.; Chouhan, M.D. Obesity, metabolic disease and the pancreas—Quantitative imaging of pancreatic fat. Br. J. Radiol. 2018. [Google Scholar] [CrossRef]
- Smits, M.M.; van Geenen, E.J.M. The clinical significance of pancreatic steatosis. Nat. Rev. Gastroenterol. Hepatol. 2011, 8, 169–177. [Google Scholar] [CrossRef]
- Heiskanen, M.A.; Motiani, K.K.; Mari, A.; Saunavaara, V.; Eskelinen, J.-J.; Virtanen, K.A.; Koivumäki, M.; Löyttyniemi, E.; Nuutila, P.; Kalliokoski, K.K.; et al. Exercise training decreases pancreatic fat content and improves beta cell function regardless of baseline glucose tolerance: A randomised controlled trial. Diabetologia 2018, 61, 1817–1828. [Google Scholar] [CrossRef]
- Tene, L.; Shelef, I.; Schwarzfuchs, D.; Gepner, Y.; Yaskolka Meir, A.; Tsaban, G.; Zelicha, H.; Bilitzky, A.; Komy, O.; Cohen, N.; et al. The effect of long-term weight-loss intervention strategies on the dynamics of pancreatic-fat and morphology: An MRI RCT study. Clin. Nutr. ESPEN 2018, 24, 82–89. [Google Scholar] [CrossRef] [PubMed]
- Gaborit, B.; Abdesselam, I.; Kober, F.; Jacquier, A.; Ronsin, O.; Emungania, O.; Lesavre, N.; Alessi, M.-C.; Martin, J.C.; Bernard, M.; et al. Ectopic fat storage in the pancreas using 1H-MRS: Importance of diabetic status and modulation with bariatric surgery-induced weight loss. Int. J. Obes. 2015, 39, 480–487. [Google Scholar] [CrossRef] [PubMed]
- Steven, S.; Hollingsworth, K.G.; Small, P.K.; Woodcock, S.A.; Pucci, A.; Aribisala, B.; Al-Mrabeh, A.; Daly, A.K.; Batterham, R.L.; Taylor, R. Weight Loss Decreases Excess Pancreatic Triacylglycerol Specifically in Type 2 Diabetes. Diabetes Care 2016, 39, 158–165. [Google Scholar] [CrossRef]
- Vogt, L.J.; Steveling, A.; Meffert, P.J.; Kromrey, M.-L.; Kessler, R.; Hosten, N.; Krüger, J.; Gärtner, S.; Aghdassi, A.A.; Mayerle, J.; et al. Magnetic Resonance Imaging of Changes in Abdominal Compartments in Obese Diabetics during a Low-Calorie Weight-Loss Program. PLoS ONE 2016, 11, e0153595. [Google Scholar] [CrossRef] [PubMed]
- Heber, S.D.; Hetterich, H.; Lorbeer, R.; Bayerl, C.; Machann, J.; Auweter, S.; Storz, C.; Schlett, C.L.; Nikolaou, K.; Reiser, M.; et al. Pancreatic fat content by magnetic resonance imaging in subjects with prediabetes, diabetes, and controls from a general population without cardiovascular disease. PLoS ONE 2017, 12, e0177154. [Google Scholar] [CrossRef] [PubMed]
- Schübel, R.; Graf, M.E.; Nattenmüller, J.; Nabers, D.; Sookthai, D.; Gruner, L.F.; Johnson, T.; Schlett, C.L.; von Stackelberg, O.; Kirsten, R.; et al. The effects of intermittent calorie restriction on metabolic health: Rationale and study design of the HELENA Trial. Contemp. Clin. Trials 2016, 51, 28–33. [Google Scholar] [CrossRef]
- Kassi, E.; Pervanidou, P.; Kaltsas, G.; Chrousos, G. Metabolic syndrome: Definitions and controversies. BMC Med. 2011, 9. [Google Scholar] [CrossRef]
- Schübel, R.; Nattenmüller, J.; Sookthai, D.; Nonnenmacher, T.; Graf, M.E.; Riedl, L.; Schlett, C.L.; von Stackelberg, O.; Johnson, T.; Nabers, D.; et al. Effects of intermittent and continuous calorie restriction on body weight and metabolism over 50 wk: A randomized controlled trial. Am. J. Clin. Nutr. 2018, 108, 933–945. [Google Scholar] [CrossRef]
- Zhong, X.; Nickel, M.D.; Kannengiesser, S.A.R.; Dale, B.M.; Kiefer, B.; Bashir, M.R. Liver fat quantification using a multi-step adaptive fitting approach with multi-echo GRE imaging: Liver Fat Quantification with Adaptive Fitting. Magn. Reson. Med. 2014, 72, 1353–1365. [Google Scholar] [CrossRef]
- Kühn, T.; Nonnenmacher, T.; Sookthai, D.; Schübel, R.; Quintana Pacheco, D.A.; von Stackelberg, O.; Graf, M.E.; Johnson, T.; Schlett, C.L.; Kirsten, R.; et al. Anthropometric and blood parameters for the prediction of NAFLD among overweight and obese adults. BMC Gastroenterol. 2018, 18. [Google Scholar] [CrossRef]
- Schübel, R.; Nonnenmacher, T.; Sookthai, D.; Gonzalez Maldonado, S.; Sowah, S.; von Stackelberg, O.; Schlett, C.; Grafetstätter, M.; Nabers, D.; Johnson, T.; et al. Similar Weight Loss Induces Greater Improvements in Insulin Sensitivity and Liver Function among Individuals with NAFLD Compared to Individuals without NAFLD. Nutrients 2019, 11, 544. [Google Scholar] [CrossRef] [PubMed]
- Wald, D.; Teucher, B.; Dinkel, J.; Kaaks, R.; Delorme, S.; Boeing, H.; Seidensaal, K.; Meinzer, H.-P.; Heimann, T. Automatic quantification of subcutaneous and visceral adipose tissue from whole-body magnetic resonance images suitable for large cohort studies. J. Magn. Reson. Imaging 2012, 36, 1421–1434. [Google Scholar] [CrossRef]
- Heni, M.; Machann, J.; Staiger, H.; Schwenzer, N.F.; Peter, A.; Schick, F.; Claussen, C.D.; Stefan, N.; Häring, H.-U.; Fritsche, A. Pancreatic fat is negatively associated with insulin secretion in individuals with impaired fasting glucose and/or impaired glucose tolerance: A nuclear magnetic resonance study. Diabetes Metab. Res. Rev. 2010, 26, 200–205. [Google Scholar] [CrossRef]
- van der Zijl, N.J.; Goossens, G.H.; Moors, C.C.M.; van Raalte, D.H.; Muskiet, M.H.A.; Pouwels, P.J.W.; Blaak, E.E.; Diamant, M. Ectopic Fat Storage in the Pancreas, Liver, and Abdominal Fat Depots: Impact on β-Cell Function in Individuals with Impaired Glucose Metabolism. J. Clin. Endocrinol. Metab. 2011, 96, 459–467. [Google Scholar] [CrossRef]
- Wong, V.W.-S.; Wong, G.L.-H.; Yeung, D.K.-W.; Abrigo, J.M.; Kong, A.P.-S.; Chan, R.S.-M.; Chim, A.M.-L.; Shen, J.; Ho, C.-S.; Woo, J.; et al. Fatty pancreas, insulin resistance, and [beta]-cell function: A population study using fat-water magnetic resonance imaging. Am. J. Gastroenterol. 2014, 109, 589–597. [Google Scholar] [CrossRef] [PubMed]
- Rossi, A.P.; Fantin, F.; Zamboni, G.A.; Mazzali, G.; Zoico, E.; Bambace, C.; Antonioli, A.; Pozzi Mucelli, R.; Zamboni, M. Effect of moderate weight loss on hepatic, pancreatic and visceral lipids in obese subjects. Nutr. Diabetes 2012, 2, e32. [Google Scholar] [CrossRef] [PubMed]
- Fabbrini, E.; Magkos, F. Hepatic Steatosis as a Marker of Metabolic Dysfunction. Nutrients 2015, 7, 4995–5019. [Google Scholar] [CrossRef]
- Fox, C.S.; Massaro, J.M.; Hoffmann, U.; Pou, K.M.; Maurovich-Horvat, P.; Liu, C.-Y.; Vasan, R.S.; Murabito, J.M.; Meigs, J.B.; Cupples, L.A.; et al. Abdominal Visceral and Subcutaneous Adipose Tissue Compartments: Association with Metabolic Risk Factors in the Framingham Heart Study. Circulation 2007, 116, 39–48. [Google Scholar] [CrossRef]
- da Campos, R.M.S.; Masquio, D.C.L.; Corgosinho, F.C.; Caranti, D.A.; de Ganen, A.P.; Tock, L.; Oyama, L.M.; Dâmaso, A.R. Effects of magnitude of visceral adipose tissue reduction: Impact on insulin resistance, hyperleptinemia and cardiometabolic risk in adolescents with obesity after long-term weight-loss therapy. Diabetes Vasc. Dis. Res. 2019. [Google Scholar] [CrossRef]
- Rossi, A.P.; Fantin, F.; Zamboni, G.A.; Mazzali, G.; Rinaldi, C.A.; Giglio, M.D.; Francesco, V.D.; Barillari, M.; Pozzi-Mucelli, R.; Zamboni, M. Predictors of Ectopic Fat Accumulation in Liver and Pancreas in Obese Men and Women. Obesity 2011, 19, 1747–1754. [Google Scholar] [CrossRef]
- Hannukainen, J.C.; Borra, R.; Linderborg, K.; Kallio, H.; Kiss, J.; Lepomäki, V.; Kalliokoski, K.K.; Kujala, U.M.; Kaprio, J.; Heinonen, O.J.; et al. Liver and pancreatic fat content and metabolism in healthy monozygotic twins with discordant physical activity. J. Hepatol. 2011, 54, 545–552. [Google Scholar] [CrossRef]
- Covarrubias, Y.; Fowler, K.J.; Mamidipalli, A.; Hamilton, G.; Wolfson, T.; Leinhard, O.D.; Jacobsen, G.; Horgan, S.; Schwimmer, J.B.; Reeder, S.B.; et al. Pilot study on longitudinal change in pancreatic proton density fat fraction during a weight-loss surgery program in adults with obesity. J. Magn. Reson. Imaging 2019. [Google Scholar] [CrossRef] [PubMed]
- Saisho, Y. Pancreas Volume and Fat Deposition in Diabetes and Normal Physiology: Consideration of the Interplay Between Endocrine and Exocrine Pancreas. Rev. Diabet. Stud. 2016, 13, 132–147. [Google Scholar] [CrossRef] [PubMed]
- Idilman, I.S.; Tuzun, A.; Savas, B.; Elhan, A.H.; Celik, A.; Idilman, R.; Karcaaltincaba, M. Quantification of liver, pancreas, kidney, and vertebral body MRI-PDFF in non-alcoholic fatty liver disease. Abdom. Imaging 2015, 40, 1512–1519. [Google Scholar] [CrossRef] [PubMed]
- Wildman-Tobriner, B.; Middleton, M.M.; Moylan, C.A.; Rossi, S.; Flores, O.; Chang, Z.A.; Abdelmalek, M.F.; Sirlin, C.B.; Bashir, M.R. Association Between Magnetic Resonance Imaging–Proton Density Fat Fraction and Liver Histology Features in Patients with Nonalcoholic Fatty Liver Disease or Nonalcoholic Steatohepatitis. Gastroenterology 2018, 155, 1428–1435. [Google Scholar] [CrossRef] [PubMed]
- Tang, A.; Desai, A.; Hamilton, G.; Wolfson, T.; Gamst, A.; Lam, J.; Clark, L.; Hooker, J.; Chavez, T.; Ang, B.D.; et al. Accuracy of MR Imaging–estimated Proton Density Fat Fraction for Classification of Dichotomized Histologic Steatosis Grades in Nonalcoholic Fatty Liver Disease. Radiology 2015, 274, 416–425. [Google Scholar] [CrossRef]
- Pinnick, K.E.; Collins, S.C.; Londos, C.; Gauguier, D.; Clark, A.; Fielding, B.A. Pancreatic Ectopic Fat Is Characterized by Adipocyte Infiltration and Altered Lipid Composition. Obesity 2008, 16, 522–530. [Google Scholar] [CrossRef] [PubMed]
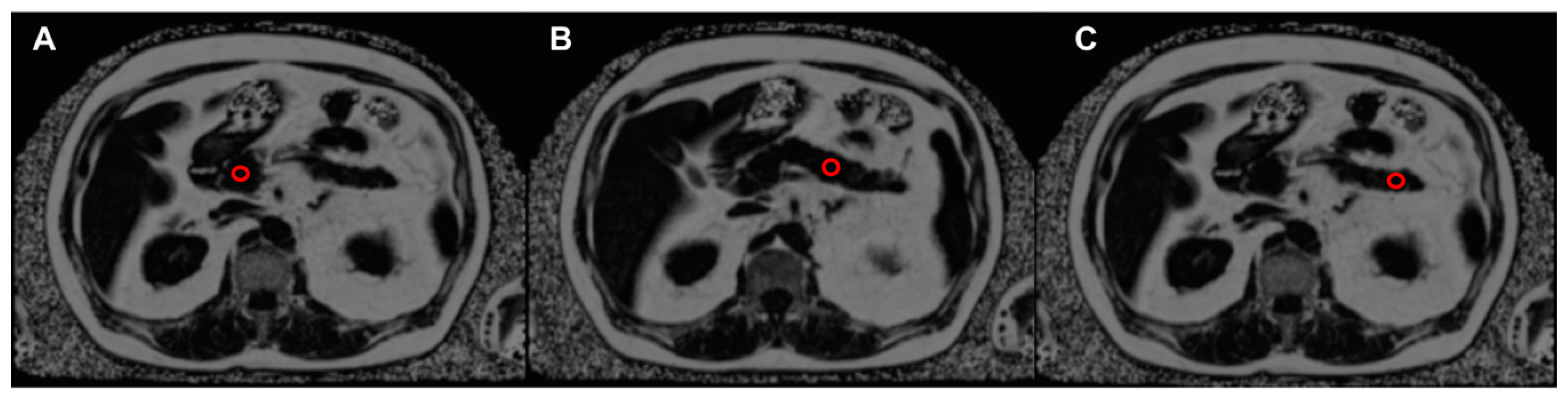
| Q1 | Q2 | Q3 | Q4 | |
|---|---|---|---|---|
| ≤2% | >2% to ≤4.5% | >4.5% to ≤7.5% | >7.5% | |
| n = 35 | n = 34 | n = 35 | n = 33 | |
| Women [n(%)] | 20 (57) | 15 (44) | 19 (54) | 18 (55) |
| Age [year] | 51.0 ± 6.3 | 51.2 ± 8.3 | 51.2 ± 7.8 | 47.4 ± 8.3 |
| Weight [kg] | 94.3 ± 15.8 | 94.0 ± 14.0 | 93.3 ± 15.5 | 95.0 ± 14.2 |
| Height [cm] | 171.2 ± 10.5 | 173.8 ± 9.8 | 173.4 ± 10.7 | 173.3 ± 7.9 |
| BMI [kg/m2] | 32.1 ± 4.1 | 31.1 ± 3.7 | 30.9 ± 3.4 | 31.6 ± 3.7 |
| Waist circumference [cm] | 105.6 ± 12.3 | 105.5 ± 10.7 | 102.4 ± 11.4 | 103.9 ± 11.8 |
| Systolic blood pressure (mmHg) | 139.6 ± 11.0 | 132.2 ± 14.0 | 136.6 ± 14.4 | 140.0 ± 21.9 |
| Diastolic blood pressure (mmHg) | 90.1 ± 8.1 | 86.0 ± 81 | 87.3 ± 7.7 | 86.9 ± 9.8 |
| Metabolic syndrome [n(%)] | 9 (25.7) | 3 (8.8) | 5 (14.3) | 5 (15.2) |
| Fat depositions | ||||
| VAT [L] | 5.3 ± 2.2 | 5.0 ± 2.2 | 4.8 ± 2.0 | 4.7 ± 2.0 |
| SAT [L] | 13.1 ± 4.6 | 11.2 ± 2.8 | 12.1 ± 3.9 | 12.9 ± 4.0 |
| Pancreas fat content [%] | 10.3 ± 5.1 | 7.7 ± 4.1 | 10.1 ± 11.2 | 8.6 ± 4.2 |
| Liver fat content [%] | 7.1 ± 4.4 | 8.8 ± 7.8 | 7.9 ± 6.5 | 7.4 ± 4.9 |
| Glucose metabolism | ||||
| Glucose [mg/dL] | 93.4 ± 8.0 | 93.2 ± 6.8 | 94.8 ± 6.8 | 91.8 ± 8.0 |
| Insulin [mU/L] | 14.7 ± 7.8 | 12.6 ± 7.4 | 10.8 ± 5.1 | 11.2 ± 5.4 |
| HbA1c [%] | 5.5 ± 0.4 | 5.5 ± 0.3 | 5.5 ± 0.3 | 5.4 ± 0.3 |
| HOMA-IR | 3.4 ± 1.9 | 3.0 ± 1.8 | 2.6 ± 1.2 | 2.6 ± 1.3 |
| IGF-1 [ng/mL] | 114.6 ± 34.3 | 124.3 ± 34.1 | 111.3 ± 33.3 | 105.1 ± 27.0 |
| Liver function tests | ||||
| ALT [U/L] | 25.1 ± 7.2 | 31.5 ± 14.1 | 26.9 ± 12.1 | 24.4 ± 9.7 |
| AST [U/L] | 21.8 ± 4.0 | 25.8 ± 6.9 | 22.5 ± 4.0 | 22.3 ± 5.0 |
| GGT [U/L] | 29.7 ± 13.9 | 26.3 ± 16.1 | 30.4 ± 19.8 | 24.3 ± 12.1 |
| Lipid metabolism | ||||
| Triglycerides [mg/dL] | 139.4 ± 64.9 | 136.1 ± 89.3 | 143.9 ± 93.2 | 108.3 ± 53.5 |
| Cholesterol [mg/dL] | 211.4 ± 34.1 | 202.1 ± 35.9 | 214.4 ± 36.0 | 203.3 ± 34.5 |
| HDL [mg/dL] | 54.0 ± 15.0 | 52.6 ± 14.3 | 56.8 ± 13.7 | 52.9 ± 14.9 |
| LDL [mg/dL] | 129.5 ± 26.0 | 120.7 ± 25.0 | 128.8 ± 26.5 | 128.7 ± 29.5 |
| Inflammation | ||||
| CRP [mg/pL] | 7.0 ± 8.6 | 4.3 ± 5.4 | 3.7 ± 2.8 | 3.8 ± 3.8 |
| IFNγ [ng/μL] | 16.6 ± 15.8 | 16.7 ± 24.8 | 17.2 ± 16.6 | 11.6 ± 8.9 |
| TNFα [ng/μL] | 4.2 ± 2.7 | 4.4 ± 2.8 | 4.9 ± 2.7 | 4.1 ± 2.5 |
| IL-6 [ng/μL] | 2.0 ± 1.7 | 1.8 ± 3.4 | 1.3 ± 0.8 | 1.3 ± 1.1 |
| IL-8 [ng/μL] | 10.7 ± 4.4 | 14.0 ± 23.0 | 10.0 ± 4.7 | 10.6 ± 5.2 |
| Adipokines | ||||
| Adiponectin [ng/mL] | 15.7 ± 8.3 | 18.1 ± 11.4 | 17.0 ± 11.2 | 19.4 ± 13.2 |
| Leptin [ng/mL] | 29.7 ± 25.1 | 20.4 ± 19.9 | 21.2 ± 15.1 | 29.4 ± 29.0 |
| Resistin [ng/mL] | 5.6 ± 2.5 | 5.5 ± 2.0 | 5.3 ± 1.5 | 6.4 ± 3.3 |
| Pancreatic Fat Content | VAT | Liver Fat Content | ||||
|---|---|---|---|---|---|---|
| rho | p-Value | rho | p-Value | rho | p-Value | |
| BMI | 0.269 | 0.002 * | 0.600 | <0.001 * | 0.364 | <0.001 * |
| Weight | 0.292 | 0.001 * | 0.667 | <0.001 * | 0.399 | <0.001 * |
| Waist circumference | 0.250 | 0.004 * | 0.671 | <0.001 * | 0.477 | <0.001 * |
| Systolic blood pressure (mmHg) | 0.053 | 0.760 | 0.223 | 0.009 | 0.151 | 0.080 |
| Diastolic blood pressure (mmHg) | 0.027 | 0.535 | 0.324 | <0.001 * | 0.208 | 0.015 |
| Fat depositions | ||||||
| VAT | 0.411 | <0.001 * | 1.000 | 0.586 | <0.001 * | |
| SAT | 0.261 | 0.003 * | 0.536 | <0.001 * | 0.360 | <0.001 * |
| Pancreatic fat content | 1.000 | 0.411 | <0.001 * | 0.195 | 0.026 | |
| Liver fat content | 0.195 | 0.026 | 0.586 | <0.001 * | 1.000 | |
| Glucose metabolism | ||||||
| Glucose | 0.086 | 0.328 | 0.260 | 0.003 * | 0.255 | 0.003 * |
| Insulin | 0.081 | 0.361 | 0.481 | <0.001 * | 0.454 | <0.001 * |
| HbA1c | 0.039 | 0.658 | 0.269 | 0.002 * | 0.332 | <0.001 * |
| HOMA-IR | 0.084 | 0.340 | 0.482 | <0.001 * | 0.463 | <0.001 * |
| IGF-1β | −0.040 | 0.655 | −0.023 | 0.799 | −0.039 | 0.658 |
| Liver function tests | ||||||
| ALT | 0.152 | 0.084 | 0.317 | <0.001 * | 0.548 | <0.001 * |
| AST | 0.076 | 0.389 | −0.033 | 0.706 | 0.197 | 0.025 |
| GGT | 0.060 | 0.500 | 0.257 | 0.003 * | 0.221 | 0.011 |
| Lipid metabolism | ||||||
| Triglycerides | 0.102 | 0.250 | 0.381 | <0.001 * | 0.337 | <0.001 * |
| Cholesterol | −0.033 | 0.706 | 0.137 | 0.120 | 0.099 | 0.262 |
| HDL | −0.178 | 0.042 | −0.370 | <0.001 * | −0.278 | 0.001 * |
| LDL | −0.004 | 0.960 | 0.168 | 0.055 | 0.087 | 0.324 |
| Inflammation | ||||||
| CRP | 0.151 | 0.086 | 0.384 | <0.001 * | 0.145 | 0.100 |
| IFNγ | 0.015 | 0.870 | 0.006 | 0.943 | −0.002 | 0.980 |
| TNFα | −0.008 | 0.932 | 0.019 | 0.830 | −0.003 | 0.972 |
| IL-6 | 0.030 | 0.736 | 0.197 | 0.025 | 0.118 | 0.180 |
| IL-8 | 0.024 | 0.790 | 0.064 | 0.468 | 0.077 | 0.381 |
| Adipokines | ||||||
| Adiponectin | 0.067 | 0.450 | −0.015 | 0.866 | −0.036 | 0.683 |
| Leptin | 0.062 | 0.482 | 0.344 | <0.001 * | 0.165 | 0.060 |
| Resistin | 0.080 | 0.366 | 0.111 | 0.211 | −0.017 | 0.850 |
| Baseline | Week 12 | Loge relative Change (Baseline–Week 12) | p-Value | Week 50 | Loge Relative Change (Baseline–Week 50) | p-Value | ||
|---|---|---|---|---|---|---|---|---|
| Mean ± SD | Mean ± SD | Mean ± SEM | Mean ± SD | Mean ± SEM | ||||
| Weight [kg] | Q1 | 94.3 ± 15.8 | 94.3 ± 15.8 | 0.0 ± 0.2 | <0.01 * | 95.5 ± 16.3 | 1.2 ± 0.5 | <0.01 * |
| Q2 | 94.0 ± 14.0 | 91.0 ± 13.6 | −3.2 ± 0.1 | 93.5 ± 13.5 | −1.3 ± 0.5 | |||
| Q3 | 93.3 ± 15.5 | 87.8 ± 14.6 | −6.1 ± 0.2 | 89.3 ± 15.8 | −4.3 ± 0.7 | |||
| Q4 | 95.0 ± 14.2 | 84.9 ± 12.8 | −11.3 ± 0.6 | 85.3 ± 14.0 | −11.1 ± 1.5 | |||
| VAT [l] | Q1 | 5.3 ± 2.2 | 5.2 ± 2.1 | −0.1 ± 0.9 | <0.01 * | 5.6 ± 2.3 | 4.9 ± 2.0 | <0.01 * |
| Q2 | 5.0 ± 2.2 | 4.5 ± 2.0 | −10.0 ± 1.3 | 4.8 ± 2.2 | −7.1 ± 2.5 | |||
| Q3 | 4.8 ± 2.0 | 4.1 ± 1.8 | −18.1 ± 1.3 | 4.1 ± 1.9 | −16.7 ± 2.6 | |||
| Q4 | 4.7 ± 2.0 | 3.4 ± 1.6 | −33.9 ± 3.4 | 3.2 ± 1.9 | −45.4 ± 7.3 | |||
| Pancreatic fat content [%] | Q1 | 10.3 ± 5.1 | 10.6 ± 5.1 | 1.3 ± 6.7 | <0.01 * (0.07 a) | 10.1 ± 4.0 | 3.6 ± 5.9 | 0.01 * (0.37 b) |
| Q2 | 7.7 ± 4.1 | 9.1 ± 6.1 | 6.5 ± 6.9 | 8.5 ± 6.7 | −5.1 ± 6.1 | |||
| Q3 | 10.1 ± 11.2 | 10.3 ± 12.2 | 1.8 ± 5.4 | 9.7 ± 11.4 | −3.0 ± 7.9 | |||
| Q4 | 8.6 ± 4.2 | 6.8 ± 4.3 | −30.8 ± 5.7 | 6.5 ± 3.0 | −29.2 ± 7.8 | |||
| Liver fat content [%] | Q1 | 7.1 ± 4.4 | 6.7 ± 4.1 | −2.6 ± 5.4 | <0.01 * | 7.9 ± 5.8 | −0.3 ± 6.1 | <0.01 * |
| Q2 | 8.8 ± 7.8 | 7.2 ± 6.2 | −25.9 ± 5.7 | 8.3 ± 7.3 | −12.3 ± 4.8 | |||
| Q3 | 7.9 ± 6.5 | 5.3 ± 4.2 | −35.8 ± 5.5 | 5.7 ± 5.6 | −35.9 ± 7.3 | |||
| Q4 | 7.4 ± 4.9 | 3.5 ± 1.7 | −63.9 ± 10.3 | 3.6 ± 1.9 | −56.9 ± 10.5 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jiang, Y.; Spurny, M.; Schübel, R.; Nonnenmacher, T.; Schlett, C.L.; von Stackelberg, O.; Ulrich, C.M.; Kaaks, R.; Kauczor, H.-U.; Kühn, T.; et al. Changes in Pancreatic Fat Content Following Diet-Induced Weight Loss. Nutrients 2019, 11, 912. https://doi.org/10.3390/nu11040912
Jiang Y, Spurny M, Schübel R, Nonnenmacher T, Schlett CL, von Stackelberg O, Ulrich CM, Kaaks R, Kauczor H-U, Kühn T, et al. Changes in Pancreatic Fat Content Following Diet-Induced Weight Loss. Nutrients. 2019; 11(4):912. https://doi.org/10.3390/nu11040912
Chicago/Turabian StyleJiang, Yixin, Manuela Spurny, Ruth Schübel, Tobias Nonnenmacher, Christopher L. Schlett, Oyunbileg von Stackelberg, Cornelia M. Ulrich, Rudolf Kaaks, Hans-Ulrich Kauczor, Tilman Kühn, and et al. 2019. "Changes in Pancreatic Fat Content Following Diet-Induced Weight Loss" Nutrients 11, no. 4: 912. https://doi.org/10.3390/nu11040912
APA StyleJiang, Y., Spurny, M., Schübel, R., Nonnenmacher, T., Schlett, C. L., von Stackelberg, O., Ulrich, C. M., Kaaks, R., Kauczor, H.-U., Kühn, T., & Nattenmüller, J. (2019). Changes in Pancreatic Fat Content Following Diet-Induced Weight Loss. Nutrients, 11(4), 912. https://doi.org/10.3390/nu11040912





