Cordycepin, an Active Constituent of Nutrient Powerhouse and Potential Medicinal Mushroom Cordyceps militaris Linn., Ameliorates Age-Related Testicular Dysfunction in Rats
Abstract
1. Introduction
2. Materials and Methods
2.1. Isolation and Identification of Cordycepin from C. militaris
2.2. Experimental Animals and Design
2.3. Sperm Kinematic Studies
2.4. Histology and Analysis of Spermatogenesis-Related Parameters
2.5. Western Blot Analysis
2.6. Reverse Transcription–Polymerase Chain Reaction (RT–PCR)
2.7. Statistical Analysis
3. Results
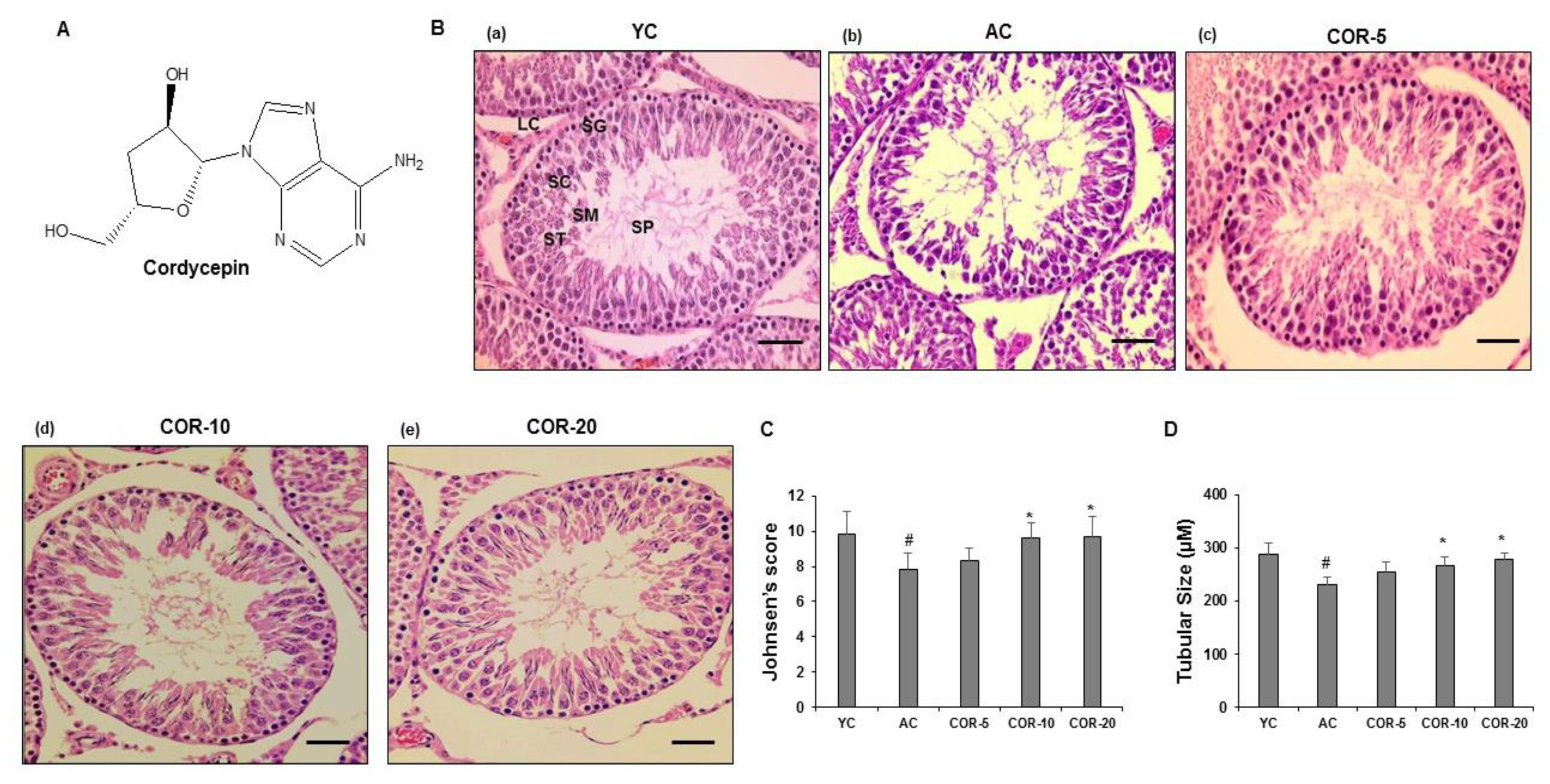
3.1. Interpretation of Cordycepin Structure
3.2. Effect of COR on Sperm Kinematics and Testis Morphology
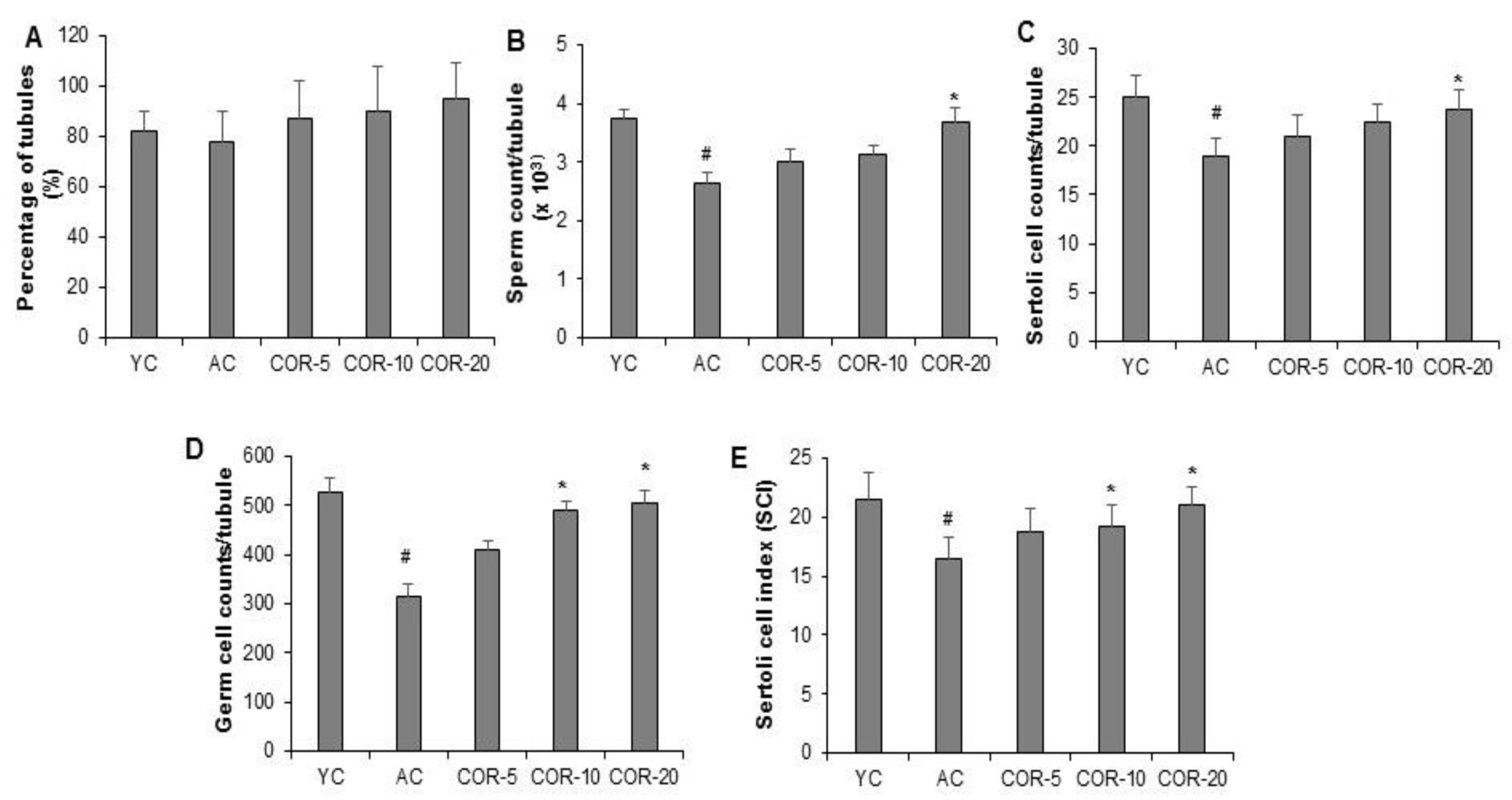
3.3. Effect of COR on Spermatogenesis-Related Histological Parameters
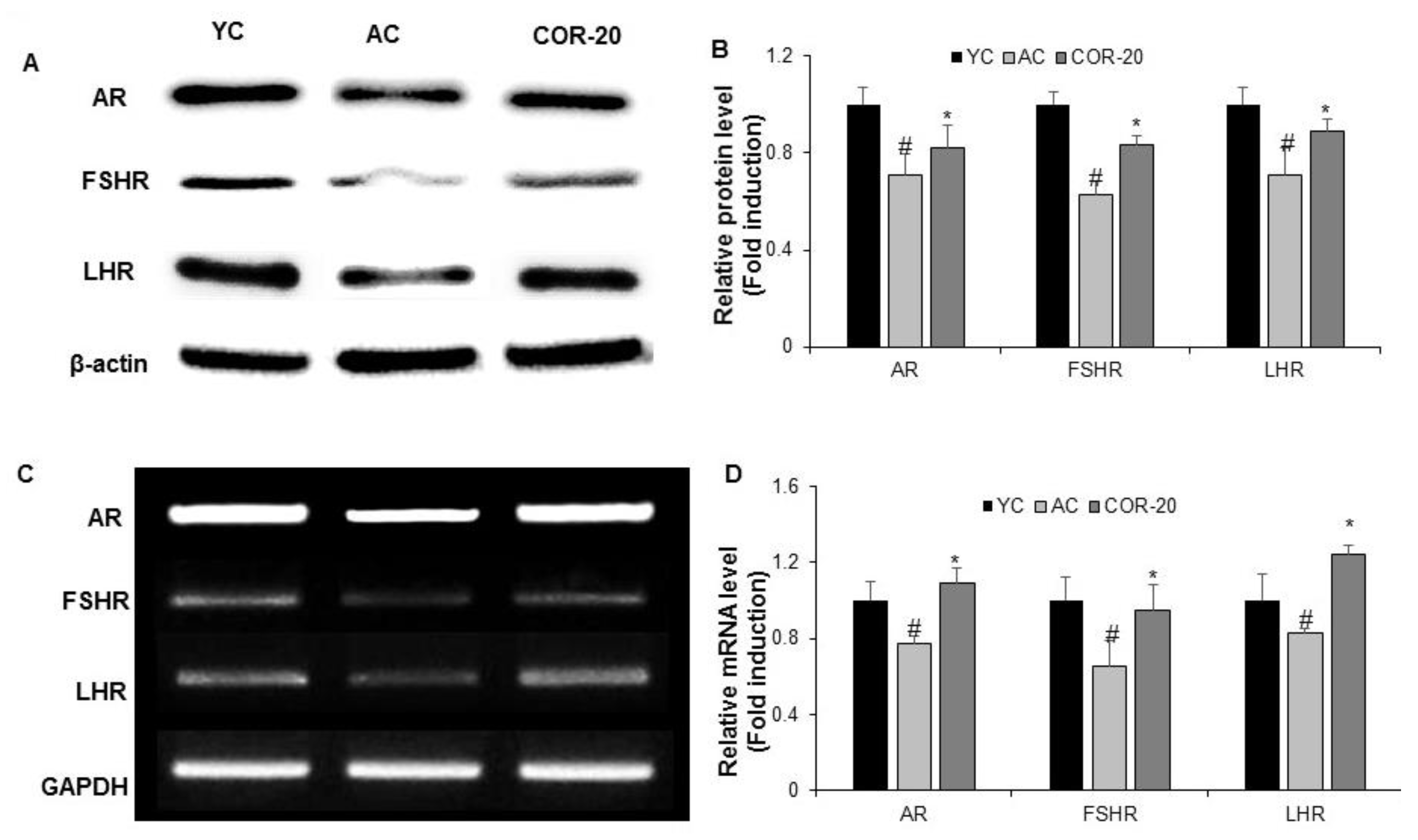
3.4. Effect on Protein and mRNA Levels of Sex Hormone Receptors in Aged Rat Testes
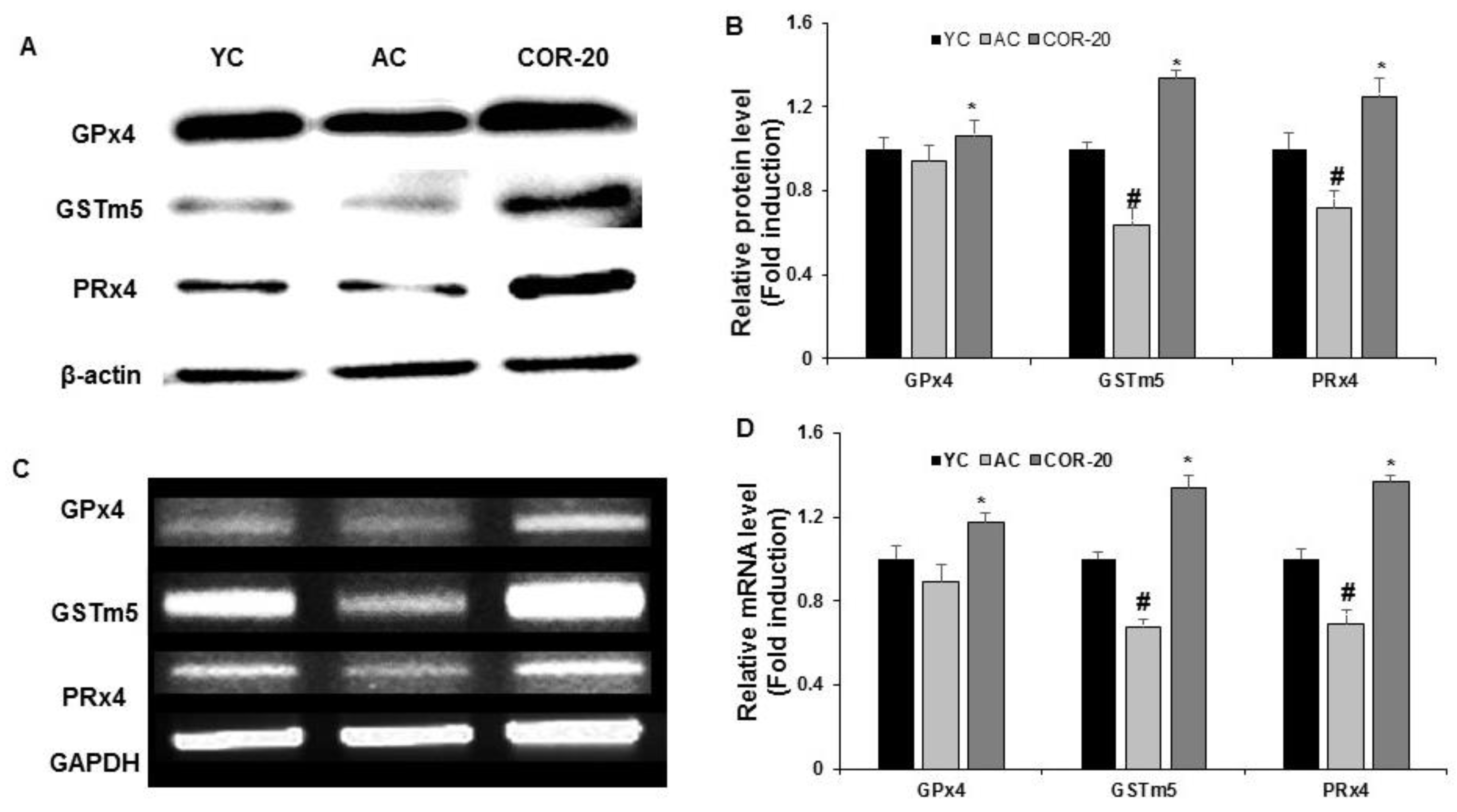
3.5. Effect on the Expression of Antioxidant Enzymes in Aged Rat Testes
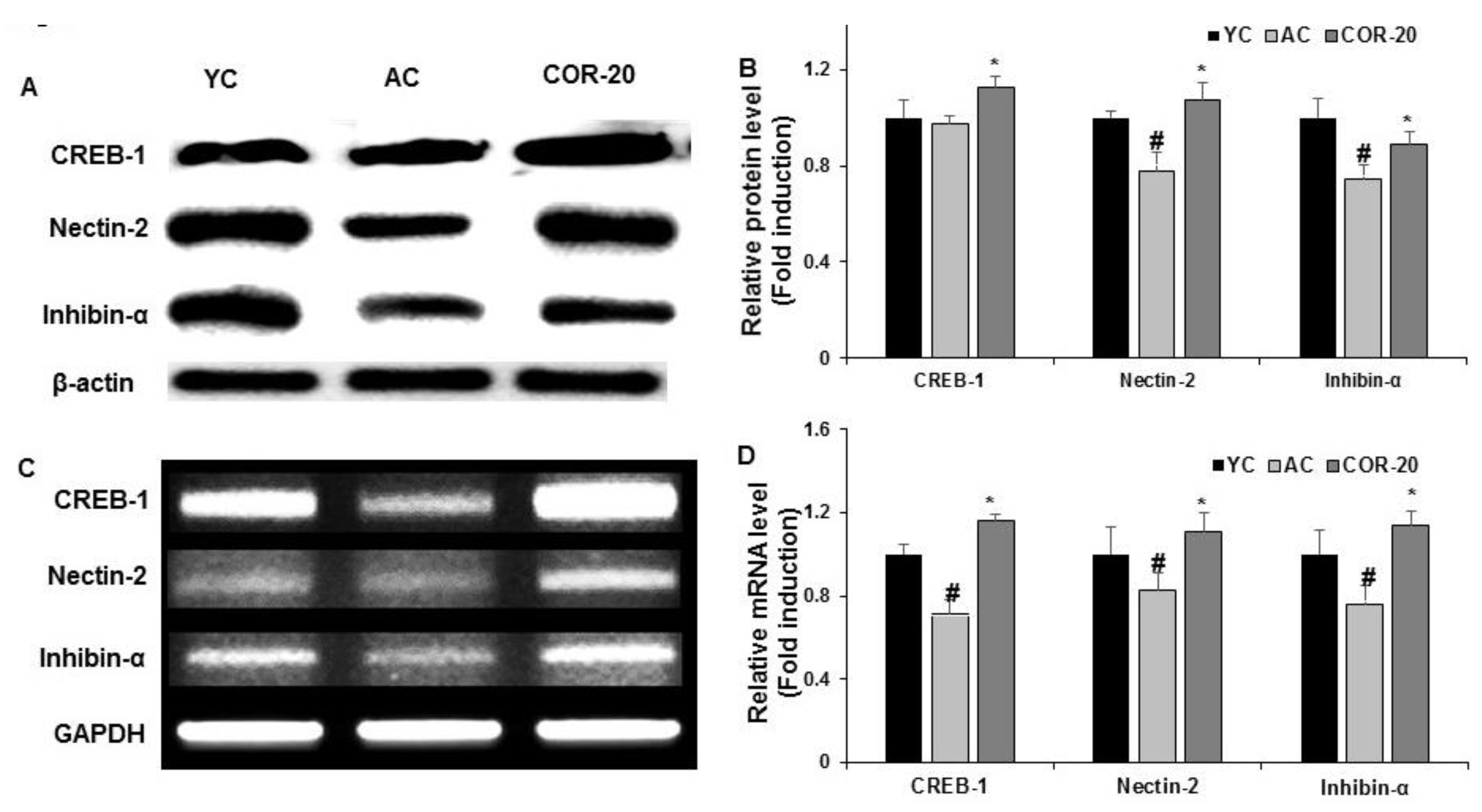
3.6. Effect on Spermatogenesis-Related Transcription Factors Expression in Aged Rat Testes
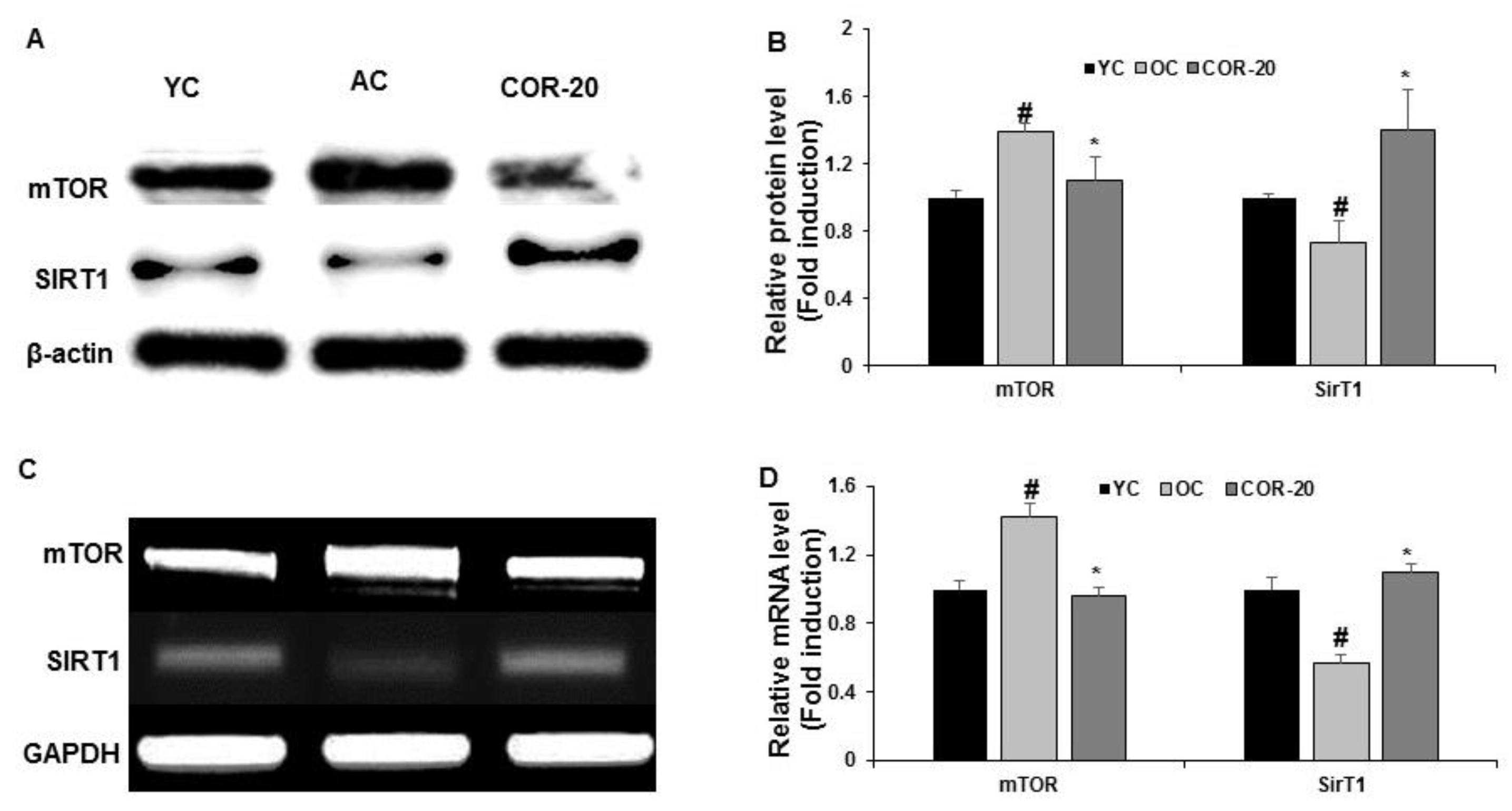
3.7. Effect on mTORC1 and SIRT1 Expression in Aged Rat Testes
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Tenover, J.L. Male hormone replacement therapy including “andropause”. Endocrinol. Metab. Clin. North. Am. 1998, 27, 969–987. [Google Scholar] [CrossRef]
- Nguyen, T.V.; Eisman, J.A.; Kelly, P.J.; Sambrook, P.N. Risk factors for osteoporotic fractures in elderly men. Am. J. Epidemiol. 1996, 144, 255–263. [Google Scholar] [CrossRef]
- Liu, P.Y.; Swerdloff, R.S.; Veldhuis, J.D. Clinical review 171: The rationale, efficacy and safety of androgen therapy in older men: Future research and current practice recommendations. J. Clin. Endocrinol. Metab. 2004, 89, 4789–4796. [Google Scholar] [CrossRef]
- Plas, E.; Berger, P.; Hermann, M.; Pfluger, H. Effects of aging on male fertility? Exp. Gerontol. 2000, 35, 543–551. [Google Scholar] [CrossRef]
- Vermeulen, A.; Kaufman, J.M. Ageing of the hypothalamo-pituitary-testicular axis in men. Horm. Res. 1995, 43, 25–28. [Google Scholar] [CrossRef]
- Morales, A.; Buvat, J.; Gooren, L.J.; Guay, A.T.; Kaufman, J.M.; Tan, H.M.; Torres, L.O. Endocrine aspects of sexual dysfunction in men. J. Sex. Med. 2004, 1, 69–81. [Google Scholar] [CrossRef]
- Dunkel, L.; Alfthan, H.; Stenman, U.H.; Perheentupa, J. Gonadal control of pulsatile secretion of luteinizing hormone and follicle-stimulating hormone in prepubertal boys evaluated by ultrasensitive time-resolved immunofluorometric assays. J. Clin. Endocrinol. Metab. 1990, 70, 107–114. [Google Scholar] [CrossRef] [PubMed]
- Ryan, M.J.; Dudash, H.J.; Docherty, M.; Geronilla, K.B.; Baker, B.A.; Haff, G.G.; Cutlip, R.G.; Alway, S.E. Aging-Dependent Regulation of Antioxidant Enzymes and Redox Status in Chronically Loaded Rat Dorsiflexor Muscles. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2008, 63, 1015–1026. [Google Scholar] [CrossRef]
- Koeberle, A.; Shindou, H.; Harayama, T.; Yuki, K.; Shimizu, T. Polyunsaturated fatty acids are incorporated into maturating male mouse germ cells by lysophosphatidic acid acyltransferase 3. FASEB J. 2012, 26, 169–180. [Google Scholar] [CrossRef]
- Chauhan, N.S.; Sharma, V.; Dixit, V.K.; Thakur, M. A review on plants used for improvement of sexual performance and virility. Biomed. Res. Int. 2014, 2014, 868062. [Google Scholar] [CrossRef] [PubMed]
- Smith, J.F.; Eisenberg, M.L.; Millstein, S.G.; Nachtigall, R.D.; Shindel, A.W.; Wing, H.; Cedars, M.; Pasch, L.; Katz, P.P. The use of complementary and alternative fertility treatment in couples seeking fertility care: Data from a prospective cohort in the United States. Fertil. Steril. 2010, 93, 2169–2174. [Google Scholar] [CrossRef]
- Bardaweel, S.K.; Shehadeh, M.; Suaifan, G.A.; Kilani, M.V. Complementary and alternative medicine utilization by a sample of infertile couples in Jordan for infertility treatment: Clinics-based survey. BMC Complement. Altern. Med. 2013, 13, 35. [Google Scholar] [CrossRef] [PubMed]
- Kopalli, S.R.; Cha, K.M.; Jeong, M.S.; Lee, S.H.; Sung, J.H.; Seo, S.K.; Kim, S.K. Pectinase-treated Panax ginseng ameliorates hydrogen peroxide-induced oxidative stress in GC-2 sperm cells and modulates testicular gene expression in aged rats. J. Ginseng. Res. 2016, 40, 185–195. [Google Scholar] [CrossRef] [PubMed]
- Kopalli, S.R.; Cha, K.M.; Ryu, J.H.; Lee, S.H.; Jeong, M.S.; Hwang, S.Y.; Lee, Y.J.; Song, H.W.; Kim, S.N.; Kim, J.C.; et al. Korean red ginseng improves testicular ineffectiveness in aging rats by modulating spermatogenesis-related molecules. Exp. Gerontol. 2017, 90, 26–33. [Google Scholar] [CrossRef]
- Das, S.K.; Masuda, M.; Sakurai, A.; Sakakibara, M. Medicinal uses of the mushroom Cordyceps militaris: Current state and prospects. Fitoterapia 2010, 81, 961–968. [Google Scholar] [CrossRef] [PubMed]
- Ying, J.; Mao, X.; Ma, Q.; Wen, H. Icons of Medicinal Mushroom from China; Science Press Beijing: Beijing, China, 1987; pp. 151–155. [Google Scholar]
- Song, C.H.; Jeon, Y.; Yang, B.K.; Ra, K.S.; Sung, J.M. Anti-complementary activity of exo-polymers produced from submerged mycelial cultures of higher fungi with particular reference to Cordyceps militaris. J. Microbiol. Biotechnol. 1998, 8, 536–539. [Google Scholar]
- Mizuno, T. Medicinal Effects and Utilization of Cordyceps (Fr.) Link (Ascomycetes) and Isaria Fr. (Mitosporic Fungi) Chinese Caterpillar Fungi, “Tochukaso” (Review). Int. J. Med. Mush. 1999, 1, 251–261. [Google Scholar] [CrossRef]
- Ng, T.B.; Wang, H.X. Pharmacological actions of Cordyceps, a prized folk medicine. J. Pharm. Pharmacol. 2005, 57, 1509–1519. [Google Scholar] [CrossRef]
- Tuli, H.S.; Sandhu, S.S.; Sharma, A.K. Pharmacological and therapeutic potential of Cordyceps with special reference to Cordycepin. 3 Biotech. 2014, 4, 1–12. [Google Scholar] [CrossRef]
- Chen, Y.C.; Chen, Y.H.; Pan, B.S.; Chang, M.M.; Huang, B.M. Functional study of Cordyceps sinensis and cordycepin in male reproduction: A review. J. Food Drug Anal. 2017, 25, 197–205. [Google Scholar] [CrossRef]
- Cho, H.J.; Cho, J.Y.; Rhee, M.H.; Kim, H.S.; Lee, H.S.; Park, H.J. Inhibitory effects of cordycepin (3’-deoxyadenosine), a component of Cordyceps militaris, on human platelet aggregation induced by thapsigargin. J. Microbiol. Biotechnol. 2007, 17, 1134–1138. [Google Scholar] [PubMed]
- Chen, L.S.; Stellrecht, C.M.; Gandhi, V. RNA-directed agent, cordycepin, induces cell death in multiple myeloma cells. Br. J. Haematol. 2008, 140, 682–691. [Google Scholar] [CrossRef]
- Yun, Y.; Shin, H.; Seung, L.; Sung, K.; Chong, L.; Nam, J.H.; Kyung, J.K. Anti-diabetic Effects of CCCA, CMKSS, and Cordycepin from Cordyceps militaris and the Immune Responses in Streptozotocin-induced Diabetic Mice. Nat. Prod. Sci. 2003, 9, 291–298. [Google Scholar]
- Chang, W.; Lim, S.; Song, H.; Song, B.W.; Kim, H.J.; Cha, M.J.; Sung, J.M.; Kim, T.W.; Hwang, K.C. Cordycepin inhibits vascular smooth muscle cell proliferation. Eur. J. Pharmacol. 2008, 597, 64–69. [Google Scholar] [CrossRef] [PubMed]
- Chang, Y.; Jeng, K.C.; Huang, K.F.; Lee, Y.C.; Hou, C.W.; Chen, K.H.; Cheng, F.Y.; Liao, J.W.; Chen, Y.S. Effect of Cordyceps militaris supplementation on sperm production, sperm motility and hormones in Sprague-Dawley rats. Am. J. Chin. Med. 2008, 36, 849–859. [Google Scholar] [CrossRef] [PubMed]
- Ramesh, T.; Yoo, S.K.; Kim, S.W.; Hwang, S.Y.; Sohn, S.H.; Kim, I.W.; Kim, S.K. Cordycepin (3’-deoxyadenosine) attenuates age-related oxidative stress and ameliorates antioxidant capacity in rats. Exp. Gerontol. 2012, 47, 979–987. [Google Scholar] [CrossRef] [PubMed]
- Won, Y.J.; Kim, B.K.; Shin, Y.K.; Jung, S.H.; Yoo, S.K.; Hwang, S.Y.; Sung, J.H.; Kim, S.K. Pectinase-treated Panax ginseng extract (GINST) rescues testicular dysfunction in aged rats via redox-modulating proteins. Exp. Gerontol. 2014, 53, 57–66. [Google Scholar] [CrossRef] [PubMed]
- Johnson, S.C.; Rabinovitch, P.S.; Kaeberlein, M. mTOR is a key modulator of ageing and age-related disease. Nature 2013, 493, 338–345. [Google Scholar] [CrossRef] [PubMed]
- Rato, L.; Alves, M.G.; Silva, B.M.; Sousa, M.; Oliveira, P.F. Sirtuins: Novel Players in Male Reproductive Health. Curr. Med. Chem. 2016, 23, 1084–1099. [Google Scholar] [CrossRef] [PubMed]
- Haidl, G.; Jung, A.; Schill, W.B. Ageing and sperm function. Hum. Reprod. 1996, 11, 558–560. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Kidd, S.A.; Eskenazi, B.; Wyrobek, A.J. Effects of male age on semen quality and fertility: A review of the literature. Fertil. Steril. 2001, 75, 237–248. [Google Scholar] [CrossRef]
- Santiago, J.; Silva, J.V.; Alves, M.G.; Oliveira, P.F.; Fardilha, M. Testicular Aging: An Overview of Ultrastructural, Cellular, and Molecular Alterations. J. Gerontol. A Biol. Sci. Med. Sci. 2018, gly082. [Google Scholar] [CrossRef]
- Shirai, M.; Yamanaka, M.; Shiina, H.; Igawa, M.; Fujime, M.; Lue, T.F.; Dahiya, R. Downregulation of androgen, estrogen and progesterone receptor genes and protein is involved in aging-related erectile dysfunction. Int. J. Impot. Res. 2003, 15, 391–396. [Google Scholar] [CrossRef][Green Version]
- Hiort, O.; Holterhus, P.M. Androgen insensitivity and male infertility. Int. J. Androl. 2003, 26, 16–20. [Google Scholar] [CrossRef]
- Haji, M.; Kato, K.I.; Nawata, H.; Ibayashi, H. Age-related changes in the concentrations of cytosol receptors for sex steroid hormones in the hypothalamus and pituitary gland of the rat. Brain Res. 1981, 204, 373–386. [Google Scholar] [CrossRef]
- Heckert, L.L.; Griswold, M.D. The expression of the follicle-stimulating hormone receptor in spermatogenesis. Recent Prog. Horm. Res. 2002, 57, 129–148. [Google Scholar] [CrossRef]
- Don, J.; Stelzer, G. The expanding family of CREB/CREM transcription factors that are involved with spermatogenesis. Mol. Cell Endocrinol. 2002, 187, 115–124. [Google Scholar] [CrossRef]
- Mueller, S.; Rosenquist, T.A.; Takai, Y.; Bronson, R.A.; Wimmer, E. Loss of nectin-2 at Sertoli-spermatid junctions leads to male infertility and correlates with severe spermatozoan head and midpiece malformation, impaired binding to the zona pellucida, and oocyte penetration. Biol. Reprod. 2003, 69, 1330–1340. [Google Scholar] [CrossRef]
- Cai, K.; Hua, G.; Ahmad, S.; Liang, A.; Han, L.; Wu, C.; Yang, F.; Yang, L. Action mechanism of inhibin alpha-subunit on the development of Sertoli cells and first wave of spermatogenesis in mice. PLoS ONE 2011, 6, e25585. [Google Scholar] [CrossRef]
- Kim, J.S.; Song, M.S.; Seo, H.S.; Yang, M.; Kim, S.H.; Kim, J.C.; Kim, H.; Saito, T.R.; Shin, T.; Moon, C. Immunohistochemical analysis of cAMP response element-binding protein in mouse testis during postnatal development and spermatogenesis. Histochem. Cell Biol. 2009, 131, 501–507. [Google Scholar] [CrossRef]
- Shi, Y.; Venkataraman, S.L.; Dodson, G.E.; Mabb, A.M.; LeBlanc, S.; Tibbetts, R.S. Direct regulation of CREB transcriptional activity by ATM in response to genotoxic stress. Proc. Natl. Acad. Sci. USA 2004, 101, 5898–5903. [Google Scholar] [CrossRef] [PubMed]
- Smith, L.B. Nonclassical Testosterone Signaling: A New Pathway Controlling Spermatogenesis? Biol. Reprod. 2016, 94, 1–2. [Google Scholar] [CrossRef] [PubMed]
- Morrison, M.E.; Racaniello, V.R. Molecular cloning and expression of a murine homolog of the human poliovirus receptor gene. J. Virol. 1992, 66, 2807–2813. [Google Scholar] [PubMed]
- Welt, C.; Sidis, Y.; Keutmann, H.; Schneyer, A. Activins, inhibins, and follistatins: From endocrinology to signaling. A paradigm for the new millennium. Exp. Biol. Med. (Maywood) 2002, 227, 724–752. [Google Scholar] [CrossRef]
- Iuchi, Y.; Okada, F.; Tsunoda, S.; Kibe, N.; Shirasawa, N.; Ikawa, M.; Okabe, M.; Ikeda, Y.; Fujii, J. Peroxiredoxin 4 knockout results in elevated spermatogenic cell death via oxidative stress. Biochem. J. 2009, 419, 149–158. [Google Scholar] [CrossRef]
- Rao, A.V.; Shaha, C. Role of glutathione S-transferases in oxidative stress-induced male germ cell apoptosis. Free Radic. Biol. Med. 2000, 29, 1015–1027. [Google Scholar] [CrossRef]
- Hemachand, T.; Gopalakrishnan, B.; Salunke, D.M.; Totey, S.M.; Shaha, C. Sperm plasma-membrane-associated glutathione S-transferases as gamete recognition molecules. J. Cell Sci. 2002, 115, 2053–2065. [Google Scholar]
- Ursini, F.; Heim, S.; Kiess, M.; Maiorino, M.; Roveri, A.; Wissing, J.; Flohe, L. Dual function of the selenoprotein PHGPx during sperm maturation. Science 1999, 285, 1393–1396. [Google Scholar] [CrossRef]
- Coussens, M.; Maresh, J.G.; Yanagimachi, R.; Maeda, G.; Allsopp, R. Sirt1 deficiency attenuates spermatogenesis and germ cell function. PLoS ONE 2008, 3, e1571. [Google Scholar] [CrossRef]
- Jesus, T.T.; Oliveira, P.F.; Sousa, M.; Cheng, C.Y.; Alves, M.G. Mammalian target of rapamycin (mTOR): A central regulator of male fertility? Crit. Rev. Biochem. Mol. Biol. 2017, 52, 235–253. [Google Scholar] [CrossRef]
| Peroxiredoxin (PRx4) | Forward: 5′-CTG ACT GAC TAT CGT GGG AAA TAC T-3′ Reverse: 5′-GAT CTG GGA TTA TTG TTT CAC TAC C-3′ |
| Glutathione S-transferase mu 5 (GSTm5) | Forward: 5′-TAT GCT CCT GGA GTT TAC TGA TAC C-3′ Reverse: 5′-AGA CGT CAT AAG TGA GAA AAT CCA C-3′ |
| Glutathione peroxidase (GPx4) | Forward: 5′-GCA AAA CCG ACG TAA ACT ACA CT-3′ Reverse: 5′-CGT TCT TAT CAA TGA GAA ACT TGG T-3′ |
| Inhibin-α | Forward: 5′-AGG AAG GCC TCT TCA CTT ATG TAT T-3′ Reverse: 5′-CTC TTG GAA GGA GAT ATT GAG AGC-3′ |
| Androgen receptor (AR) | Forward: 5′-CTG GAC TAC CTG GAT CTC TA-3′ Reverse: 5′-CCT GGG CTG TAG TTT TAT TG-3′ |
| Follicle-stimulating hormone receptor (FSHR) | Forward: 5′-GGA CTG AGT TTT GAA AGT GT-3′ Reverse: 5′-TTC CAT AAC TGG GTT CAT CA-3′ |
| Luteinizing hormone receptor (LHR) | Forward: 5′-CTA TCT CCC TGT CAA AGT AA-3′ Reverse: 5′-TTT GTA CTT CTT CAA ATC CA-3′ |
| Nectin-2 | Forward: 5′-AGT GAC CTG GCT CAG AGT CA-3′ Reverse: 5′-TAG GTA CCA GTT GTC ATC AT-3′ |
| Glyceraldehyde-3-phosphate dehydrogenase (GAPDH) | Forward: 5′-AAC TTT GGC ATT GTG GAA GGG C-3′ Reverse: 5′-ACA CAT TGG GGG TAG GAA CAC G-3′ |
| Cyclic adenosine monophosphate (cAMP) response-element binding protein-1 (CREB-1) | Forward: 5′-ACT GGC TTG GCA CAA CCA GA-3′ Reverse: 5′- GGC AGA AGT CTC TTC ATG ATT-3′ |
| Mammalian target of rapamycin complex 1 (mTORC1) | Forward: 5′-GACAACAGCCAGGGCCGCAT-3′ Reverse: 5′-ACGCTGCCTTTCTCGACGGC-3′ |
| Sirtuin 1 (SIRT-1) | Forward: 5′-GCTGGGGTTTCTGTCTCCTG-3′ Reverse: 5′-GACACAGAGACGGCTGGAAC-3′ |
| Parameters | YC | AC | COR-5 | COR-10 | COR-20 |
|---|---|---|---|---|---|
| Motility (%) | 74.70 ± 8.80 | 34.60 ± 8.70# | 40.40 ± 18.40 | 59.70 ± 6.50* | 67.00 ± 5.90** |
| Progressive (%) | 33.70 ± 4.20 | 12.70 ± 3.90# | 15.40 ± 7.40 | 22.20 ± 2.20* | 29.20 ± 2.10** |
| VAP (µM/s) | 220.30 ± 32.80 | 155.30 ± 14.60# | 179.90 ± 17.60 | 188.00 ± 11.40* | 209.70 ± 9.30** |
| VSL (µM/s) | 158.20 ±27.50 | 113.40 ± 12.60# | 129.10 ± 12.20 | 134.30 ± 13.40* | 145.10 ± 21.50** |
| VCL (µM/s) | 268.30 ± 25.50 | 270.50 ± 80.70 | 265.50 ± 38.90 | 278.70 ± 28.70 | 334.80 ± 51.20 |
| LIN (%) | 58.70 ± 6.20 | 32.70 ± 9.50# | 34.40 ± 1.90 | 38.70 ± 0.70 | 45.60 ± 1.60* |
| STR (%) | 71.50 ± 4.30 | 72.50± 11.90 | 71.50 ± 1.20 | 71.80 ± 0.80 | 69.10± 2.30 |
| WOB (%) | 81.90 ± 6.70 | 59.80 ± 11.80# | 63.70 ± 1.60 | 68.30 ± 0.70* | 72.70 ± 1.10** |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kopalli, S.R.; Cha, K.-M.; Lee, S.-H.; Hwang, S.-Y.; Lee, Y.-J.; Koppula, S.; Kim, S.-K. Cordycepin, an Active Constituent of Nutrient Powerhouse and Potential Medicinal Mushroom Cordyceps militaris Linn., Ameliorates Age-Related Testicular Dysfunction in Rats. Nutrients 2019, 11, 906. https://doi.org/10.3390/nu11040906
Kopalli SR, Cha K-M, Lee S-H, Hwang S-Y, Lee Y-J, Koppula S, Kim S-K. Cordycepin, an Active Constituent of Nutrient Powerhouse and Potential Medicinal Mushroom Cordyceps militaris Linn., Ameliorates Age-Related Testicular Dysfunction in Rats. Nutrients. 2019; 11(4):906. https://doi.org/10.3390/nu11040906
Chicago/Turabian StyleKopalli, Spandana Rajendra, Kyu-Min Cha, Sang-Ho Lee, Seock-Yeon Hwang, Young-Joo Lee, Sushruta Koppula, and Si-Kwan Kim. 2019. "Cordycepin, an Active Constituent of Nutrient Powerhouse and Potential Medicinal Mushroom Cordyceps militaris Linn., Ameliorates Age-Related Testicular Dysfunction in Rats" Nutrients 11, no. 4: 906. https://doi.org/10.3390/nu11040906
APA StyleKopalli, S. R., Cha, K.-M., Lee, S.-H., Hwang, S.-Y., Lee, Y.-J., Koppula, S., & Kim, S.-K. (2019). Cordycepin, an Active Constituent of Nutrient Powerhouse and Potential Medicinal Mushroom Cordyceps militaris Linn., Ameliorates Age-Related Testicular Dysfunction in Rats. Nutrients, 11(4), 906. https://doi.org/10.3390/nu11040906






