The Influence of Meal Frequency and Timing on Health in Humans: The Role of Fasting
Abstract
“Eat like a king in the morning, a prince at noon, and a peasant at dinner”(Moses ben Maimon or Maimonides. 1135-1404)
1. A Brief Historical Introduction
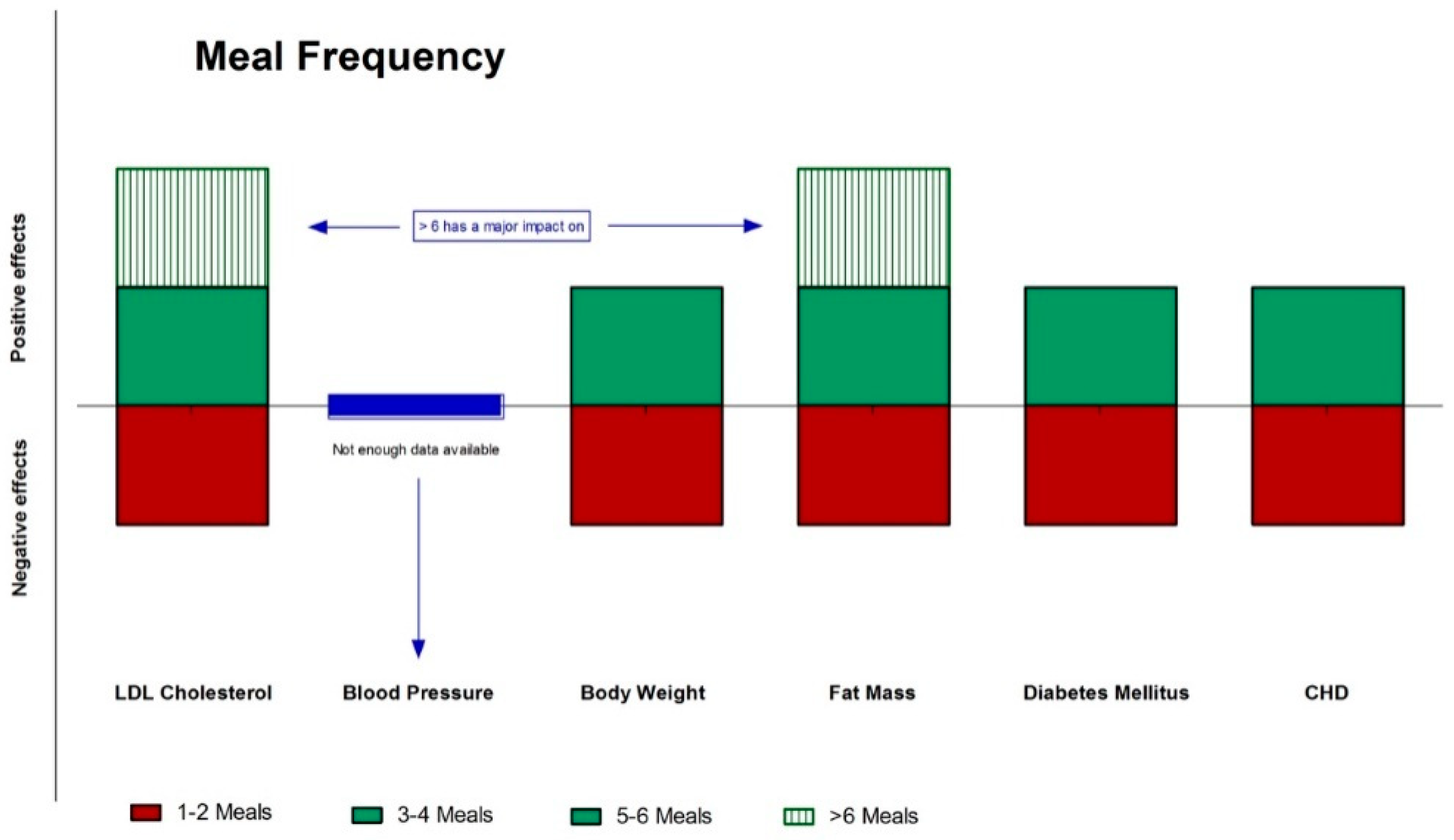
2. Meal Frequency
2.1. Epidemiological Studies about the Effects of Meal Frequency on Cholesterol, Body Weight and Diabetes
2.2. Meal Frequency and Weight Control: One, Two, Three, or More Meals?
2.3. Intervention Studies and Reciprocal Influences of Meal Frequency and Macronutrients
3. Meal Timing
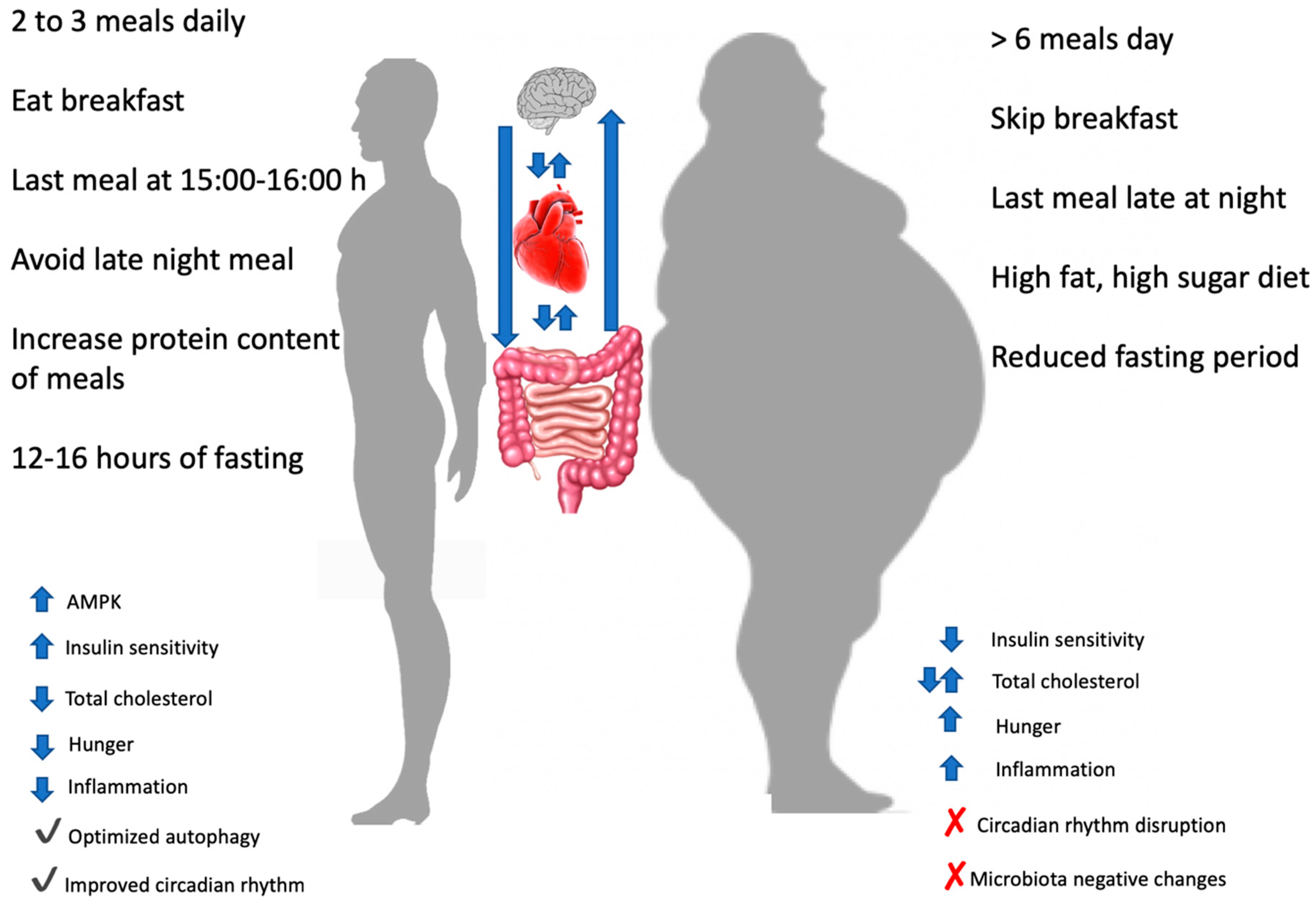
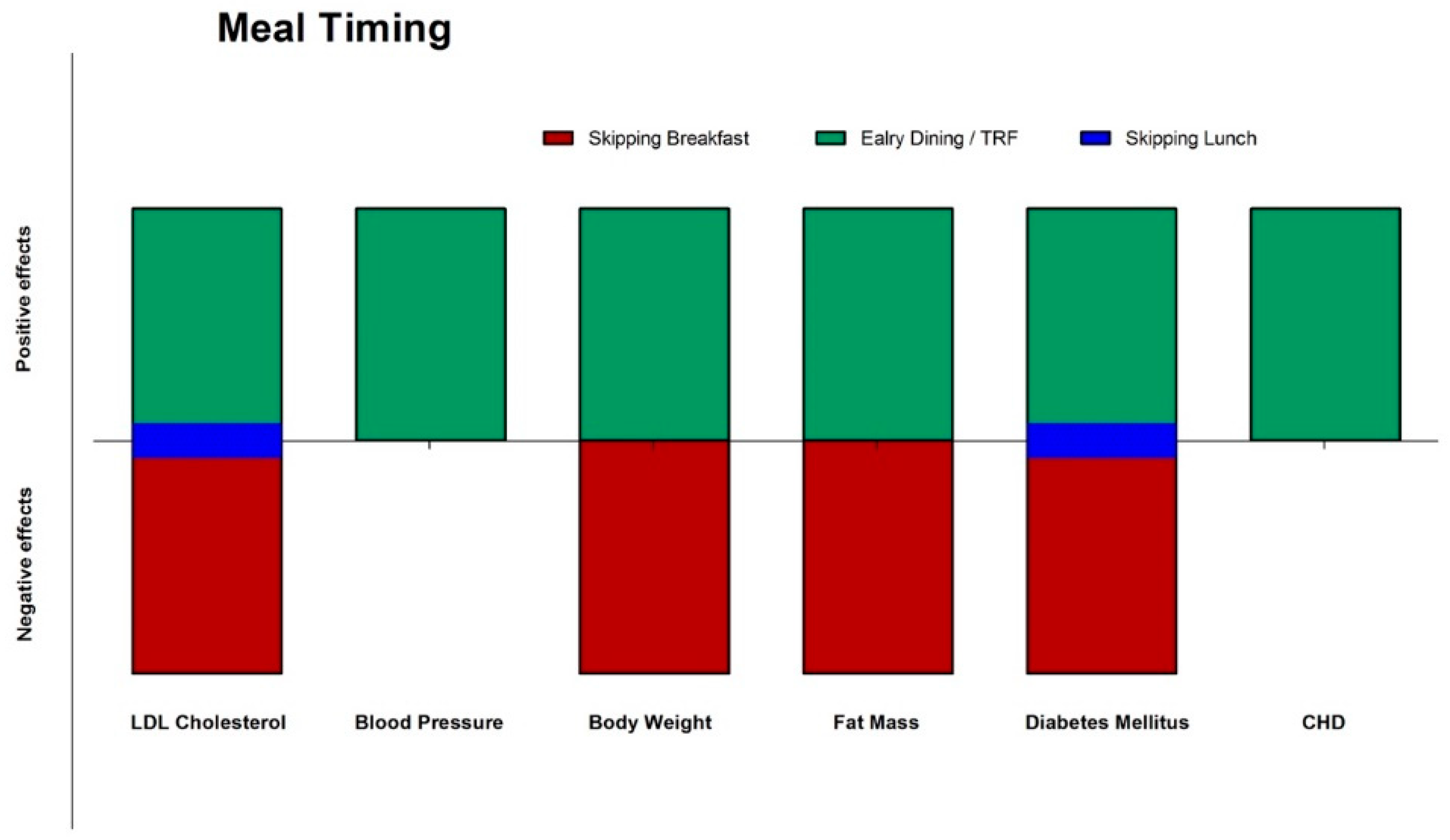
3.1. Epidemiological Data on Meal Timing: Breakfast or Not Breakfast, This Is The Question
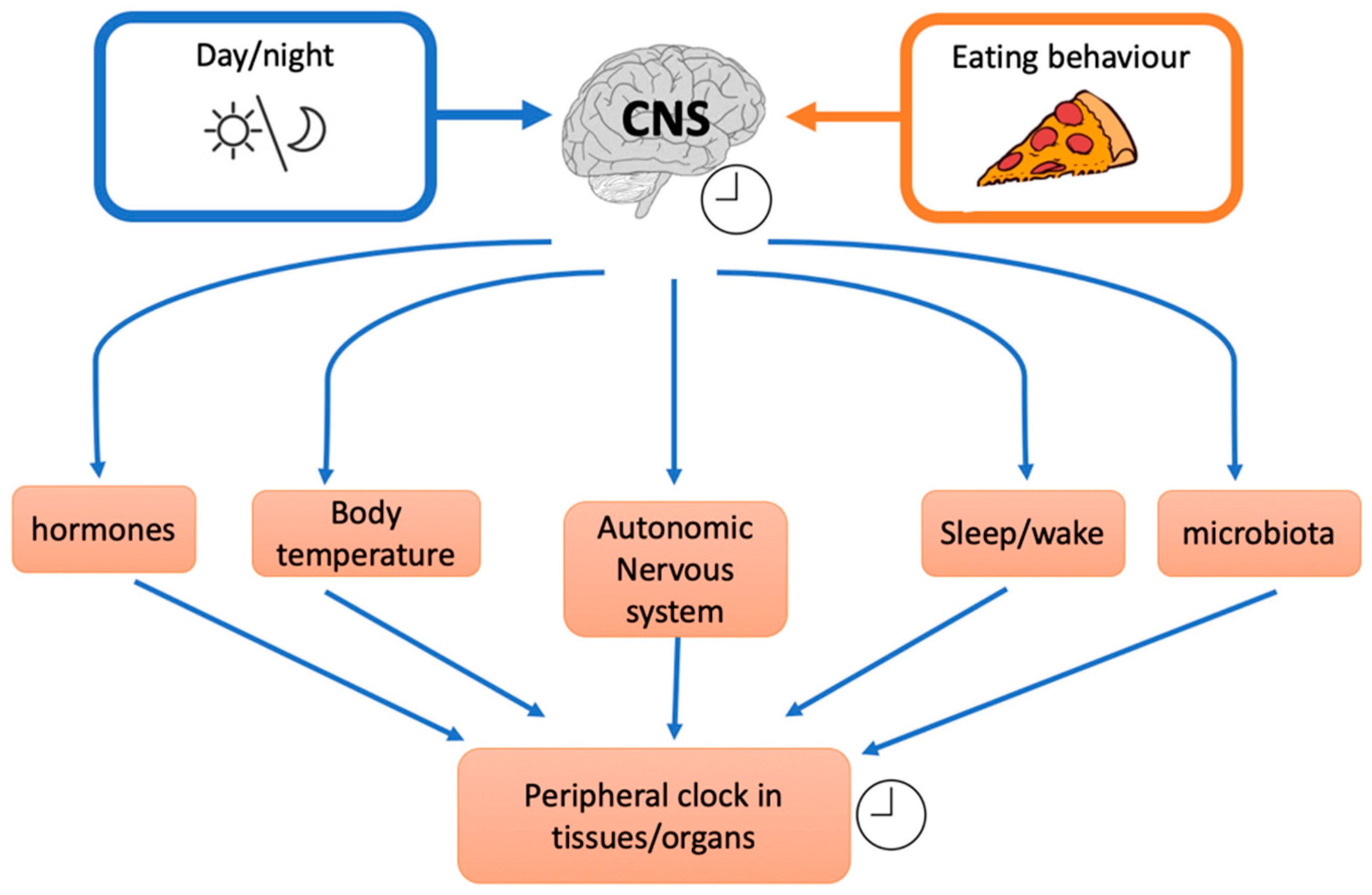
3.2. Intervention Studies and Meal Timing: Inner Clock Mechanisms
- (1)
- (2)
- (3)
- Modification of resting energy expenditure: feeding time may affect energy expenditure/basal thermogenesis as core body temperature is controlled by circadian clocks. For example, Rev-erbα is a cellular circadian clock that controls the rhythmic expression of uncoupling protein 1 (UcP1), a fundamental factor for brown adipose tissue thermogenesis [83].
- (4)
- (5)
- Circadian clocks influence also insulin resistance through glucose absorption, muscle, fat tissue, and liver insulin sensitivity [86] and food intake or nutritional challenge influence, in turn, circadian clock. Indeed shift workers, transcontinental travelers and people with irregular work schedules often show gastrointestinal symptoms as alterations in bowel habits, constipation, and diarrhoea. These examples indicate that some intestinal functions are rhythmically regulated and that their disruptions lead to health disorders. It was demonstrated that Clock (a peripheral cellular clock) regulates nutrient absorption through the expression of many nutrient transport proteins in the intestine e.g., GLUT2, GLUT5, and Pept1 (a major protein involved in the transport of small peptides from the intestinal lumen to intestinal epithelial cells). However, other external factors could influence the internal clock. For example, NAD+ (nicotinamide adenine dinucleotide) levels are influenced by nutritional status and/or physical activity. NAD+ influences the SIRT1-dependent deacetylase that activates, through deacetylation, the clock genes BMAL1 (brain-muscle-arnt-Like-protein 1) and PER2 (Period gene 2). Nicotinamide phosphoribosyltransferase (NAMPT) a downstream of BMAL1, has an oscillatory behaviour, therefore modulating the intracellular concentration of NAD+. Thus, in a feedback loop, NAD+ concentration regulates SIRT1 that modulates nuclear factors such as PPARγ (peroxisome proliferator-activated receptor gamma) and cofactors as PGC-1α (peroxisome proliferator-activated receptor gamma coactivator 1-alpha) with many effects on different tissues e.g., on hepatic glucose homeostasis (PGC-1α) or adipose tissue lipid mobilization (PPARγ). In general, a regular availability of food (regular meal timing) influences the release, from the gut, of different signals. It has been suggested that signals coming from intestine inform the dorsomedial hypothalamus (DMH) about food availability. Thus, DMH might influence other tissue and regulate food anticipation, digestion, and absorption. Thus, even though circadian genes expressed by gut play an important role, there is some evidence that food, per se, is an important regulator of food entrainment through Clock activity.
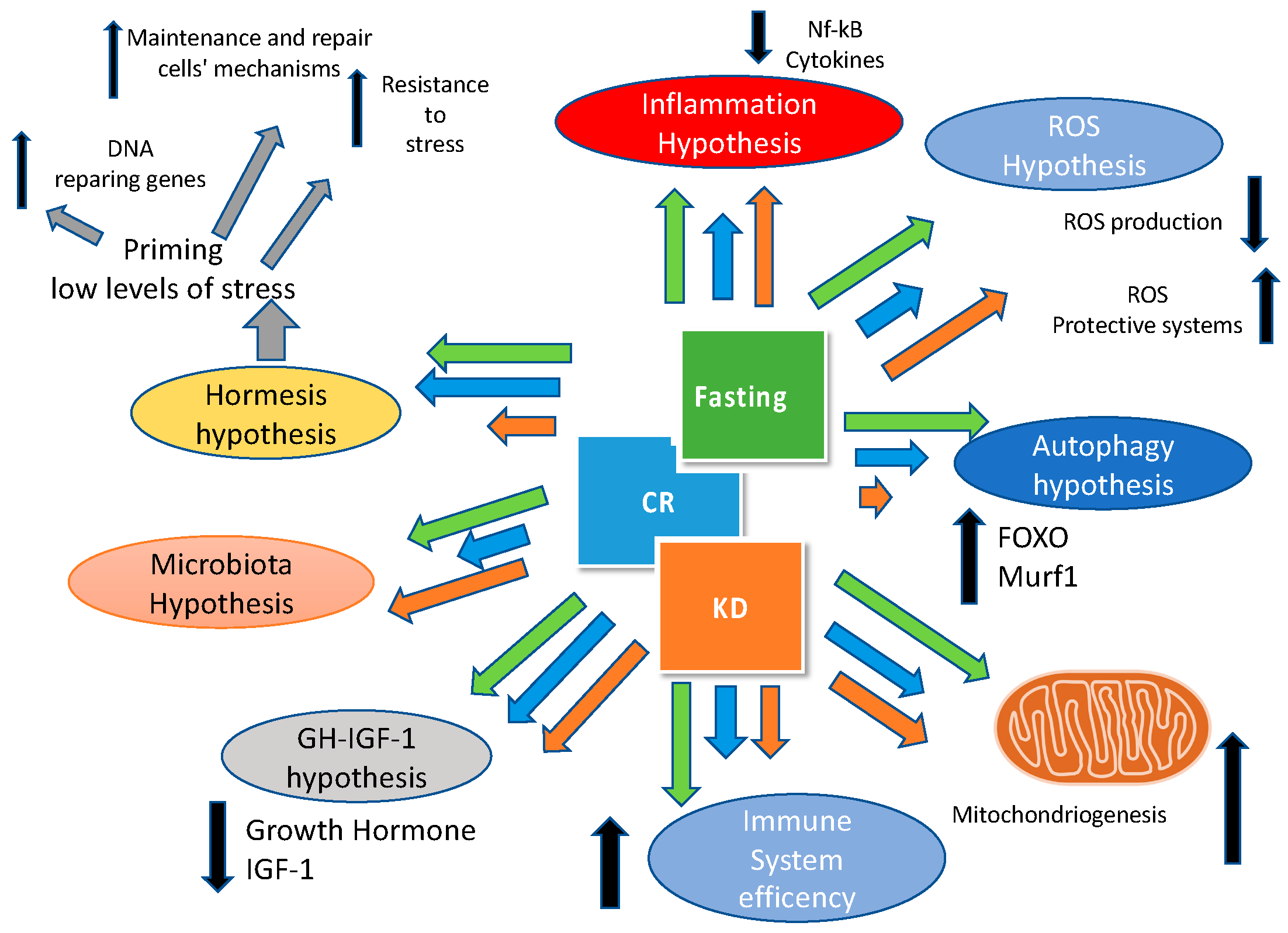
4. Reducing Meal Frequency: The Case for Time Restricted Feeding
The Importance of Fasting: What’s New?
5. Meal Frequency and Timing: The Microbiota Connection
6.Concluding Thoughts
Author Contributions
Funding
Conflicts of Interest
References
- Flandrin, J.-L.; Montanari, M. Storia dell’alimentazione; Laterza: Bari, Italy, 2003. [Google Scholar]
- Carroll, A. Three Squares: The Invention of the American Meal; Basic Books (AZ): New York, NY, USA, 2013. [Google Scholar]
- Affinita, A.; Catalani, L.; Cecchetto, G.; De Lorenzo, G.; Dilillo, D.; Donegani, G.; Fransos, L.; Lucidi, F.; Mameli, C.; Manna, E.; et al. Breakfast: A multidisciplinary approach. Ital. J. Pediatr. 2013, 39, 44. [Google Scholar] [CrossRef]
- Albala, K. Food in Early Modern Europe; Greenwood Publishing Group: Santa Barbara, CA, USA, 2003. [Google Scholar]
- Dolve, M.A. Three Meals a Day; SDSU: Brookings, SD, USA, 1925. [Google Scholar]
- Mattson, M.P.; Allison, D.B.; Fontana, L.; Harvie, M.; Longo, V.D.; Malaisse, W.J.; Mosley, M.; Notterpek, L.; Ravussin, E.; Scheer, F.A.; et al. Meal frequency and timing in health and disease. Proc. Natl. Acad. Sci. USA 2014, 111, 16647–16653. [Google Scholar] [CrossRef]
- Potter, C.; Griggs, R.L.; Brunstrom, J.M.; Rogers, P.J. Breaking the fast: Meal patterns and beliefs about healthy eating style are associated with adherence to intermittent fasting diets. Appetite 2018, 133, 32–39. [Google Scholar] [CrossRef]
- Gwinup, G.; Byron, R.C.; Roush, W.H.; Kruger, F.A.; Hamwi, G.J. Effect of nibbling versus gorging on serum lipids in man. Am. J. Clin. Nutr. 1963, 13, 209–213. [Google Scholar] [CrossRef]
- Fabry, P.; Hejl, Z.; Fodor, J.; Braun, T.; Zvolankova, K. The frequency of meals. Its relation to overweight, hypercholesterolaemia, and decreased glucose-tolerance. Lancet 1964, 2, 614–615. [Google Scholar] [CrossRef]
- Fabry, P.; Fodor, J.; Hejl, Z.; Geizerova, H.; Balcarova, O. Meal frequency and ischaemic heart-disease. Lancet 1968, 2, 190–191. [Google Scholar] [CrossRef]
- Edelstein, S.L.; Barrett-Connor, E.L.; Wingard, D.L.; Cohn, B.A. Increased meal frequency associated with decreased cholesterol concentrations; rancho bernardo, ca, 1984–1987. Am. J. Clin. Nutr. 1992, 55, 664–669. [Google Scholar] [CrossRef]
- Jenkins, D.J.; Wolever, T.M.; Vuksan, V.; Brighenti, F.; Cunnane, S.C.; Rao, A.V.; Jenkins, A.L.; Buckley, G.; Patten, R.; Singer, W.; et al. Nibbling versus gorging: Metabolic advantages of increased meal frequency. N. Engl. J. Med. 1989, 321, 929–934. [Google Scholar] [CrossRef]
- Titan, S.M.; Bingham, S.; Welch, A.; Luben, R.; Oakes, S.; Day, N.; Khaw, K.T. Frequency of eating and concentrations of serum cholesterol in the norfolk population of the european prospective investigation into cancer (epic-norfolk): Cross sectional study. BMJ 2001, 323, 1286–1288. [Google Scholar] [CrossRef]
- Ma, Y.; Bertone, E.R.; Stanek, E.J., 3rd; Reed, G.W.; Hebert, J.R.; Cohen, N.L.; Merriam, P.A.; Ockene, I.S. Association between eating patterns and obesity in a free-living us adult population. Am. J. Epidemiol. 2003, 158, 85–92. [Google Scholar] [CrossRef]
- Holmback, I.; Ericson, U.; Gullberg, B.; Wirfalt, E. A high eating frequency is associated with an overall healthy lifestyle in middle-aged men and women and reduced likelihood of general and central obesity in men. Br. J. Nutr. 2010, 104, 1065–1073. [Google Scholar] [CrossRef]
- Mekary, R.A.; Giovannucci, E.; Willett, W.C.; van Dam, R.M.; Hu, F.B. Eating patterns and type 2 diabetes risk in men: Breakfast omission, eating frequency, and snacking. Am. J. Clin. Nutr. 2012, 95, 1182–1189. [Google Scholar] [CrossRef]
- Mekary, R.A.; Giovannucci, E.; Cahill, L.; Willett, W.C.; van Dam, R.M.; Hu, F.B. Eating patterns and type 2 diabetes risk in older women: Breakfast consumption and eating frequency. Am. J. Clin. Nutr. 2013, 98, 436–443. [Google Scholar] [CrossRef]
- Cahill, L.E.; Chiuve, S.E.; Mekary, R.A.; Jensen, M.K.; Flint, A.J.; Hu, F.B.; Rimm, E.B. Prospective study of breakfast eating and incident coronary heart disease in a cohort of male us health professionals. Circulation 2013, 128, 337–343. [Google Scholar] [CrossRef]
- St-Onge, M.P.; Ard, J.; Baskin, M.L.; Chiuve, S.E.; Johnson, H.M.; Kris-Etherton, P.; Varady, K.; American Heart Association Obesity Committee of the Council onLifestyle and Cardiometabolic Health; Council on Cardiovascular Disease in the Young; Council on Clinical Cardiology; et al. Meal timing and frequency: Implications for cardiovascular disease prevention: A scientific statement from the american heart association. Circulation 2017, 135, e96–e121. [Google Scholar] [CrossRef]
- Mann, J. Meal frequency and plasma lipids and lipoproteins. Br. J. Nutr. 1997, 77, S83–S90. [Google Scholar] [CrossRef]
- Collaborators, G.B.D.O.; Afshin, A.; Forouzanfar, M.H.; Reitsma, M.B.; Sur, P.; Estep, K.; Lee, A.; Marczak, L.; Mokdad, A.H.; Moradi-Lakeh, M.; et al. Health effects of overweight and obesity in 195 countries over 25 years. N. Engl. J. Med. 2017, 377, 13–27. [Google Scholar] [CrossRef]
- Obesity and Overweight, Fact Sheet N 311, Updated March 2013. Available online: https://www.who.int/news-room/fact-sheets/detail/obesity-and-overweight (accessed on 11 February 2019).
- Koh-Banerjee, P.; Wang, Y.; Hu, F.B.; Spiegelman, D.; Willett, W.C.; Rimm, E.B. Changes in body weight and body fat distribution as risk factors for clinical diabetes in us men. Am. J. Epidemiol. 2004, 159, 1150–1159. [Google Scholar] [CrossRef]
- Thompson, W.G.; Cook, D.A.; Clark, M.M.; Bardia, A.; Levine, J.A. Treatment of obesity. Mayo Clin. Proc. 2007, 82, 93–101. [Google Scholar] [CrossRef]
- Paoli, A.; Moro, T.; Marcolin, G.; Neri, M.; Bianco, A.; Palma, A.; Grimaldi, K. High-intensity interval resistance training (hirt) influences resting energy expenditure and respiratory ratio in non-dieting individuals. J. Transl. Med. 2012, 10, 237. [Google Scholar] [CrossRef]
- Garaulet, M.; Gomez-Abellan, P. Timing of food intake and obesity: A novel association. Physiol. Behav. 2014, 134, 44–50. [Google Scholar] [CrossRef]
- Kulovitz, M.G.; Kravitz, L.R.; Mermier, C.; Gibson, A.L.; Conn, C.A.; Kolkmeyer, D.; Kerksick, C.M. Potential role of meal frequency as a strategy for weight loss and health in overweight or obese adults. Nutrition 2014, 30, 386–392. [Google Scholar] [CrossRef] [PubMed]
- Kahleova, H.; Lloren, J.I.; Mashchak, A.; Hill, M.; Fraser, G.E. Meal frequency and timing are associated with changes in body mass index in adventist health study 2. J. Nutr. 2017, 147, 1722–1728. [Google Scholar] [CrossRef]
- Jones, P.J.; Leitch, C.A.; Pederson, R.A. Meal-frequency effects on plasma hormone concentrations and cholesterol synthesis in humans. Am. J. Clin. Nutr. 1993, 57, 868–874. [Google Scholar] [CrossRef] [PubMed]
- McGrath, S.A.; Gibney, M.J. The effects of altered frequency of eating on plasma lipids in free-living healthy males on normal self-selected diets. Eur. J. Clin. Nutr. 1994, 48, 402–407. [Google Scholar]
- Keast, D.R.; Nicklas, T.A.; O’Neil, C.E. Snacking is associated with reduced risk of overweight and reduced abdominal obesity in adolescents: National health and nutrition examination survey (nhanes) 1999–2004. Am. J. Clin. Nutr. 2010, 92, 428–435. [Google Scholar] [CrossRef]
- Van der Heijden, A.A.; Hu, F.B.; Rimm, E.B.; van Dam, R.M. A prospective study of breakfast consumption and weight gain among U.S. Men. Obesity 2007, 15, 2463–2469. [Google Scholar] [CrossRef]
- Howarth, N.C.; Huang, T.T.; Roberts, S.B.; Lin, B.H.; McCrory, M.A. Eating patterns and dietary composition in relation to BMI in younger and older adults. Int. J. Obes. (Lond.) 2007, 31, 675–684. [Google Scholar] [CrossRef]
- Taylor, M.A.; Garrow, J.S. Compared with nibbling, neither gorging nor a morning fast affect short-term energy balance in obese patients in a chamber calorimeter. Int. J. Obes. Relat. Metab. Disord. 2001, 25, 519–528. [Google Scholar] [CrossRef] [PubMed]
- Romon, M.; Edme, J.L.; Boulenguez, C.; Lescroart, J.L.; Frimat, P. Circadian variation of diet-induced thermogenesis. Am. J. Clin. Nutr. 1993, 57, 476–480. [Google Scholar] [CrossRef]
- Bo, S.; Fadda, M.; Castiglione, A.; Ciccone, G.; De Francesco, A.; Fedele, D.; Guggino, A.; Parasiliti Caprino, M.; Ferrara, S.; VezioBoggio, M.; et al. Is the timing of caloric intake associated with variation in diet-induced thermogenesis and in the metabolic pattern? A randomized cross-over study. Int. J. Obes. (Lond.) 2015, 39, 1689–1695. [Google Scholar] [CrossRef] [PubMed]
- Morris, C.J.; Garcia, J.I.; Myers, S.; Yang, J.N.; Trienekens, N.; Scheer, F.A. The human circadian system has a dominating role in causing the morning/evening difference in diet-induced thermogenesis. Obesity 2015, 23, 2053–2058. [Google Scholar] [CrossRef] [PubMed]
- Weststrate, J.A.; Weys, P.J.; Poortvliet, E.J.; Deurenberg, P.; Hautvast, J.G. Diurnal variation in postabsorptive resting metabolic rate and diet-induced thermogenesis. Am. J. Clin. Nutr. 1989, 50, 908–914. [Google Scholar] [CrossRef]
- Kanaley, J.A.; Heden, T.D.; Liu, Y.; Fairchild, T.J. Alteration of postprandial glucose and insulin concentrations with meal frequency and composition. Br. J. Nutr. 2014, 112, 1484–1493. [Google Scholar] [CrossRef] [PubMed]
- Ohkawara, K.; Cornier, M.A.; Kohrt, W.M.; Melanson, E.L. Effects of increased meal frequency on fat oxidation and perceived hunger. Obesity 2013, 21, 336–343. [Google Scholar] [CrossRef] [PubMed]
- Paoli, A.; Bosco, G.; Camporesi, E.M.; Mangar, D. Ketosis, ketogenic diet and food intake control: A complex relationship. Front. Psychol. 2015, 6, 27. [Google Scholar] [CrossRef]
- Alhussain, M.H.; Macdonald, I.A.; Taylor, M.A. Irregular meal-pattern effects on energy expenditure, metabolism, and appetite regulation: A randomized controlled trial in healthy normal-weight women. Am. J. Clin. Nutr. 2016, 104, 21–32. [Google Scholar] [CrossRef]
- Lecerf, J.M.; de Lorgeril, M. Dietary cholesterol: From physiology to cardiovascular risk. Br. J. Nutr. 2011, 106, 6–14. [Google Scholar] [CrossRef]
- Nayor, M.; Vasan, R.S. Recent update to the us cholesterol treatment guidelines: A comparison with international guidelines. Circulation 2016, 133, 1795–1806. [Google Scholar] [CrossRef]
- Dietschy, J.M.; Brown, M.S. Effect of alterations of the specific activity of the intracellular acetyl coa pool on apparent rates of hepatic cholesterogenesis. J. Lipid Res. 1974, 15, 508–516. [Google Scholar]
- Ness, G.C.; Zhao, Z.; Wiggins, L. Insulin and glucagon modulate hepatic 3-hydroxy-3-methylglutaryl-coenzyme a reductase activity by affecting immunoreactive protein levels. J. Biol. Chem. 1994, 269, 29168–29172. [Google Scholar]
- Trapani, L.; Pallottini, V. Hypercholesterolemia and 3-hydroxy 3-methylglutaryl coenzyme a reductase regulation during ageing. Sci. World J. 2009, 9, 564–574. [Google Scholar] [CrossRef] [PubMed]
- Dong, X.Y.; Tang, S.Q. Insulin-induced gene: A new regulator in lipid metabolism. Peptides 2010, 31, 2145–2150. [Google Scholar] [CrossRef]
- Paoli, A. Ketogenic diet for obesity: Friend or foe? Int. J. Environ. Res. Public Health 2014, 11, 2092–2107. [Google Scholar] [CrossRef]
- Paoli, A.; Rubini, A.; Volek, J.S.; Grimaldi, K.A. Beyond weight loss: A review of the therapeutic uses of very-low-carbohydrate (ketogenic) diets. Eur. J. Clin. Nutr. 2013, 67, 789–796. [Google Scholar] [CrossRef]
- Paoli, A.; Cenci, L.; Grimaldi, K.A. Effect of ketogenic mediterranean diet with phytoextracts and low carbohydrates/high-protein meals on weight, cardiovascular risk factors, body composition and diet compliance in italian council employees. Nutr. J. 2011, 10, 112. [Google Scholar] [CrossRef]
- Sutherland, W.H.; de Jong, S.A.; Walker, R.J. Effect of dietary cholesterol and fat on cell cholesterol transfer to postprandial plasma in hyperlipidemic men. Lipids 2007, 42, 901–911. [Google Scholar] [CrossRef] [PubMed]
- Paoli, A.; Moro, T.; Bosco, G.; Bianco, A.; Grimaldi, K.A.; Camporesi, E.; Mangar, D. Effects of n-3 polyunsaturated fatty acids (omega-3) supplementation on some cardiovascular risk factors with a ketogenic mediterranean diet. Mar. Drugs 2015, 13, 996–1009. [Google Scholar] [CrossRef]
- Chapelot, D. The role of snacking in energy balance: A biobehavioral approach. J. Nutr. 2011, 141, 158–162. [Google Scholar] [CrossRef] [PubMed]
- Leidy, H.J.; Armstrong, C.L.; Tang, M.; Mattes, R.D.; Campbell, W.W. The influence of higher protein intake and greater eating frequency on appetite control in overweight and obese men. Obesity 2010, 18, 1725–1732. [Google Scholar] [CrossRef]
- Belinova, L.; Kahleova, H.; Malinska, H.; Topolcan, O.; Windrichova, J.; Oliyarnyk, O.; Kazdova, L.; Hill, M.; Pelikanova, T. The effect of meal frequency in a reduced-energy regimen on the gastrointestinal and appetite hormones in patients with type 2 diabetes: A randomised crossover study. PLoS ONE 2017, 12, e0174820. [Google Scholar] [CrossRef]
- McCrory, M.A.; Campbell, W.W. Effects of eating frequency, snacking, and breakfast skipping on energy regulation: Symposium overview. J. Nutr. 2011, 141, 144–147. [Google Scholar] [CrossRef] [PubMed]
- Nas, A.; Mirza, N.; Hagele, F.; Kahlhofer, J.; Keller, J.; Rising, R.; Kufer, T.A.; Bosy-Westphal, A. Impact of breakfast skipping compared with dinner skipping on regulation of energy balance and metabolic risk. Am. J. Clin. Nutr. 2017, 105, 1351–1361. [Google Scholar] [CrossRef]
- Almoosawi, S.; Vingeliene, S.; Karagounis, L.G.; Pot, G.K. Chrono-nutrition: A review of current evidence from observational studies on global trends in time-of-day of energy intake and its association with obesity. Proc. Nutr. Soc. 2016, 75, 487–500. [Google Scholar] [CrossRef] [PubMed]
- Leech, R.M.; Timperio, A.; Livingstone, K.M.; Worsley, A.; McNaughton, S.A. Temporal eating patterns: Associations with nutrient intakes, diet quality, and measures of adiposity. Am. J. Clin. Nutr. 2017, 106, 1121–1130. [Google Scholar] [CrossRef]
- Jakubowicz, D.; Barnea, M.; Wainstein, J.; Froy, O. High caloric intake at breakfast vs. Dinner differentially influences weight loss of overweight and obese women. Obesity 2013, 21, 2504–2512. [Google Scholar] [CrossRef] [PubMed]
- Dhurandhar, E.J.; Dawson, J.; Alcorn, A.; Larsen, L.H.; Thomas, E.A.; Cardel, M.; Bourland, A.C.; Astrup, A.; St-Onge, M.-P.; Hill, J.O.; et al. The effectiveness of breakfast recommendations on weight loss: A randomized controlled trial. Am. J. Clin. Nutr. 2014, 100, 507–513. [Google Scholar] [CrossRef]
- Uzhova, I.; Fuster, V.; Fernandez-Ortiz, A.; Ordovas, J.M.; Sanz, J.; Fernandez-Friera, L.; Lopez-Melgar, B.; Mendiguren, J.M.; Ibanez, B.; Bueno, H.; et al. The importance of breakfast in atherosclerosis disease: Insights from the pesa study. J. Am. Coll. Cardiol. 2017, 70, 1833–1842. [Google Scholar] [CrossRef]
- Betts, J.A.; Chowdhury, E.A.; Gonzalez, J.T.; Richardson, J.D.; Tsintzas, K.; Thompson, D. Is breakfast the most important meal of the day? Proc. Nutr. Soc. 2016, 75, 464–474. [Google Scholar] [CrossRef] [PubMed]
- Chowdhury, E.A.; Richardson, J.D.; Holman, G.D.; Tsintzas, K.; Thompson, D.; Betts, J.A. The causal role of breakfast in energy balance and health: A randomized controlled trial in obese adults. Am. J. Clin. Nutr. 2016, 103, 747–756. [Google Scholar] [CrossRef]
- Betts, J.A.; Richardson, J.D.; Chowdhury, E.A.; Holman, G.D.; Tsintzas, K.; Thompson, D. The causal role of breakfast in energy balance and health: A randomized controlled trial in lean adults. Am. J. Clin. Nutr. 2014, 100, 539–547. [Google Scholar] [CrossRef] [PubMed]
- Fong, M.; Caterson, I.D.; Madigan, C.D. Are large dinners associated with excess weight, and does eating a smaller dinner achieve greater weight loss? A systematic review and meta-analysis. Br. J. Nutr. 2017, 118, 616–628. [Google Scholar] [CrossRef]
- Pavlovski, I.; Evans, J.A.; Mistlberger, R.E. Feeding time entrains the olfactory bulb circadian clock in anosmic per2::Luc mice. Neuroscience 2018, 393, 175–184. [Google Scholar] [CrossRef]
- Laermans, J.; Broers, C.; Beckers, K.; Vancleef, L.; Steensels, S.; Thijs, T.; Tack, J.; Depoortere, I. Shifting the circadian rhythm of feeding in mice induces gastrointestinal, metabolic and immune alterations which are influenced by ghrelin and the core clock gene bmal1. PLoS ONE 2014, 9, e110176. [Google Scholar] [CrossRef]
- Bouchard-Cannon, P.; Cheng, H.Y. Scheduled feeding alters the timing of the suprachiasmatic nucleus circadian clock in dexras1-deficient mice. Chronobiol. Int. 2012, 29, 965–981. [Google Scholar] [CrossRef] [PubMed]
- Vollmers, C.; Gill, S.; DiTacchio, L.; Pulivarthy, S.R.; Le, H.D.; Panda, S. Time of feeding and the intrinsic circadian clock drive rhythms in hepatic gene expression. Proc. Natl. Acad. Sci. USA 2009, 106, 21453–21458. [Google Scholar] [CrossRef]
- Albrecht, U. The circadian clock, metabolism and obesity. Obes. Rev. 2017, 18, 25–33. [Google Scholar] [CrossRef]
- Scheer, F.A.; Kalsbeek, A.; Buijs, R.M. Cardiovascular control by the suprachiasmatic nucleus: Neural and neuroendocrine mechanisms in human and rat. Biol. Chem. 2003, 384, 697–709. [Google Scholar] [CrossRef]
- Kim, P.; Oster, H.; Lehnert, H.; Schmid, S.M.; Salamat, N.; Barclay, J.L.; Maronde, E.; Inder, W.; Rawashdeh, O. Coupling the circadian clock to homeostasis: The role of period in timing physiology. Endocr. Rev. 2019, 40, 66–95. [Google Scholar] [CrossRef] [PubMed]
- McHill, A.W.; Phillips, A.J.; Czeisler, C.A.; Keating, L.; Yee, K.; Barger, L.K.; Garaulet, M.; Scheer, F.A.; Klerman, E.B. Later circadian timing of food intake is associated with increased body fat. Am. J. Clin. Nutr. 2017, 106, 1213–1219. [Google Scholar] [CrossRef] [PubMed]
- Poggiogalle, E.; Jamshed, H.; Peterson, C.M. Circadian regulation of glucose, lipid, and energy metabolism in humans. Metabolism 2018, 84, 11–27. [Google Scholar] [CrossRef] [PubMed]
- Mazzoccoli, G.; Pazienza, V.; Vinciguerra, M. Clock genes and clock-controlled genes in the regulation of metabolic rhythms. Chronobiol. Int. 2012, 29, 227–251. [Google Scholar] [CrossRef]
- Wehrens, S.M.T.; Christou, S.; Isherwood, C.; Middleton, B.; Gibbs, M.A.; Archer, S.N.; Skene, D.J.; Johnston, J.D. Meal timing regulates the human circadian system. Curr. Biol. 2017, 27, 1768–1775. [Google Scholar] [CrossRef]
- Spiegel, K.; Tasali, E.; Penev, P.; Van Cauter, E. Brief communication: Sleep curtailment in healthy young men is associated with decreased leptin levels, elevated ghrelin levels, and increased hunger and appetite. Ann. Intern. Med. 2004, 141, 846–850. [Google Scholar] [CrossRef] [PubMed]
- Maroni, M.J.; Capri, K.M.; Cushman, A.V.; Monteiro De Pina, I.K.; Chasse, M.H.; Seggio, J.A. Constant light alters serum hormone levels related to thyroid function in male cd-1 mice. Chronobiol. Int. 2018, 35, 1456–1463. [Google Scholar] [CrossRef]
- Garaulet, M.; Vera, B.; Bonnet-Rubio, G.; Gomez-Abellan, P.; Lee, Y.C.; Ordovas, J.M. Lunch eating predicts weight-loss effectiveness in carriers of the common allele at perilipin1: The ontime (obesity, nutrigenetics, timing, mediterranean) study. Am. J. Clin. Nutr. 2016, 104, 1160–1166. [Google Scholar] [CrossRef]
- Garaulet, M.; Esteban Tardido, A.; Lee, Y.C.; Smith, C.E.; Parnell, L.D.; Ordovas, J.M. Sirt1 and clock 3111t> c combined genotype is associated with evening preference and weight loss resistance in a behavioral therapy treatment for obesity. Int. J. Obes. (Lond.) 2012, 36, 1436–1441. [Google Scholar] [CrossRef]
- Chaix, A.; Lin, T.; Le, H.D.; Chang, M.W.; Panda, S. Time-restricted feeding prevents obesity and metabolic syndrome in mice lacking a circadian clock. Cell Metab. 2018, 29, 303–319. [Google Scholar] [CrossRef] [PubMed]
- Fukuda, Y.; Morita, T. Effects of the light-dark cycle on diurnal rhythms of diet-induced thermogenesis in humans. Chronobiol. Int. 2017, 34, 1465–1472. [Google Scholar] [CrossRef] [PubMed]
- Oike, H.; Oishi, K.; Kobori, M. Nutrients, clock genes, and chrononutrition. Curr. Nutr. Rep. 2014, 3, 204–212. [Google Scholar] [CrossRef] [PubMed]
- Stenvers, D.J.; Scheer, F.; Schrauwen, P.; la Fleur, S.E.; Kalsbeek, A. Circadian clocks and insulin resistance. Nat. Rev. Endocrinol. 2018, 15, 75–89. [Google Scholar] [CrossRef] [PubMed]
- Carlson, O.; Martin, B.; Stote, K.S.; Golden, E.; Maudsley, S.; Najjar, S.S.; Ferrucci, L.; Ingram, D.K.; Longo, D.L.; Rumpler, W.V.; et al. Impact of reduced meal frequency without caloric restriction on glucose regulation in healthy, normal-weight middle-aged men and women. Metabolism 2007, 56, 1729–1734. [Google Scholar] [CrossRef] [PubMed]
- Stote, K.S.; Baer, D.J.; Spears, K.; Paul, D.R.; Harris, G.K.; Rumpler, W.V.; Strycula, P.; Najjar, S.S.; Ferrucci, L.; Ingram, D.K.; et al. A controlled trial of reduced meal frequency without caloric restriction in healthy, normal-weight, middle-aged adults. Am. J. Clin. Nutr. 2007, 85, 981–988. [Google Scholar] [CrossRef]
- Rothschild, J.; Hoddy, K.K.; Jambazian, P.; Varady, K.A. Time-restricted feeding and risk of metabolic disease: A review of human and animal studies. Nutr. Rev. 2014, 72, 308–318. [Google Scholar] [CrossRef]
- Longo, V.D.; Mattson, M.P. Fasting: Molecular mechanisms and clinical applications. Cell Metab. 2014, 19, 181–192. [Google Scholar] [CrossRef]
- Noyan, H.; El-Mounayri, O.; Isserlin, R.; Arab, S.; Momen, A.; Cheng, H.S.; Wu, J.; Afroze, T.; Li, R.K.; Fish, J.E.; et al. Cardioprotective signature of short-term caloric restriction. PLoS ONE 2015, 10, e0130658. [Google Scholar] [CrossRef] [PubMed]
- Fontana, L.; Villareal, D.T.; Das, S.K.; Smith, S.R.; Meydani, S.N.; Pittas, A.G.; Klein, S.; Bhapkar, M.; Rochon, J.; Ravussin, E.; et al. Effects of 2-year calorie restriction on circulating levels of igf-1, igf-binding proteins and cortisol in nonobese men and women: A randomized clinical trial. Aging Cell 2016, 15, 22–27. [Google Scholar] [CrossRef]
- Most, J.; Tosti, V.; Redman, L.M.; Fontana, L. Calorie restriction in humans: An update. Ageing Res. Rev. 2017, 39, 36–45. [Google Scholar] [CrossRef] [PubMed]
- Acosta-Rodriguez, V.A.; de Groot, M.H.M.; Rijo-Ferreira, F.; Green, C.B.; Takahashi, J.S. Mice under caloric restriction self-impose a temporal restriction of food intake as revealed by an automated feeder system. Cell Metab. 2017, 26, 267–277. [Google Scholar] [CrossRef]
- Castellini, M.A.; Rea, L.D. The biochemistry of natural fasting at its limits. Experientia 1992, 48, 575–582. [Google Scholar] [CrossRef] [PubMed]
- Paoli, A.; Bianco, A.; Grimaldi, K.A. The ketogenic diet and sport: A possible marriage? Exerc. Sport Sci. Rev. 2015, 43, 153–162. [Google Scholar] [CrossRef] [PubMed]
- Paoli, A.; Bianco, A.; Grimaldi, K.A.; Lodi, A.; Bosco, G. Long term successful weight loss with a combination biphasic ketogenic mediterranean diet and mediterranean diet maintenance protocol. Nutrients 2013, 5, 5205–5217. [Google Scholar] [CrossRef] [PubMed]
- Veech, R.L.; Bradshaw, P.C.; Clarke, K.; Curtis, W.; Pawlosky, R.; King, M.T. Ketone bodies mimic the life span extending properties of caloric restriction. IUBMB Life 2017, 69, 305–314. [Google Scholar] [CrossRef] [PubMed]
- Tinsley, G.M.; Bounty, P.M.L. Effects of intermittent fasting on body composition and clinical health markers in humans. Nutr. Rev. 2015, 73, 661–674. [Google Scholar] [CrossRef]
- Moro, T.; Tinsley, G.; Bianco, A.; Marcolin, G.; Pacelli, Q.F.; Battaglia, G.; Palma, A.; Gentil, P.; Neri, M.; Paoli, A. Effects of eight weeks of time-restricted feeding (16/8) on basal metabolism, maximal strength, body composition, inflammation, and cardiovascular risk factors in resistance-trained males. J. Transl. Med. 2016, 14, 290. [Google Scholar] [CrossRef]
- Tinsley, G.M.; Forsse, J.S.; Butler, N.K.; Paoli, A.; Bane, A.A.; La Bounty, P.M.; Morgan, G.B.; Grandjean, P.W. Time-restricted feeding in young men performing resistance training: A randomized controlled trial. Eur. J. Sport Sci. 2016, 17, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Kul, S.; Savas, E.; Ozturk, Z.A.; Karadag, G. Does ramadan fasting alter body weight and blood lipids and fasting blood glucose in a healthy population? A meta-analysis. J. Relig. Health 2014, 53, 929–942. [Google Scholar] [CrossRef] [PubMed]
- Mammucari, C.; Schiaffino, S.; Sandri, M. Downstream of akt: Foxo3 and mtor in the regulation of autophagy in skeletal muscle. Autophagy 2008, 4, 524–526. [Google Scholar] [CrossRef]
- Gatica, D.; Chiong, M.; Lavandero, S.; Klionsky, D.J. Molecular mechanisms of autophagy in the cardiovascular system. Circ. Res. 2015, 116, 456–467. [Google Scholar] [CrossRef] [PubMed]
- Godar, R.J.; Ma, X.; Liu, H.; Murphy, J.T.; Weinheimer, C.J.; Kovacs, A.; Crosby, S.D.; Saftig, P.; Diwan, A. Repetitive stimulation of autophagy-lysosome machinery by intermittent fasting preconditions the myocardium to ischemia-reperfusion injury. Autophagy 2015, 11, 1537–1560. [Google Scholar] [CrossRef]
- Ahmet, I.; Wan, R.; Mattson, M.P.; Lakatta, E.G.; Talan, M. Cardioprotection by intermittent fasting in rats. Circulation 2005, 112, 3115–3121. [Google Scholar] [CrossRef] [PubMed]
- Soeters, M.R.; Soeters, P.B.; Schooneman, M.G.; Houten, S.M.; Romijn, J.A. Adaptive reciprocity of lipid and glucose metabolism in human short-term starvation. Am. J. Physiol. 2012, 303, E1397–E1407. [Google Scholar] [CrossRef] [PubMed]
- Mager, D.E.; Wan, R.; Brown, M.; Cheng, A.; Wareski, P.; Abernethy, D.R.; Mattson, M.P. Caloric restriction and intermittent fasting alter spectral measures of heart rate and blood pressure variability in rats. FASEB J. 2006, 20, 631–637. [Google Scholar] [CrossRef]
- Hatori, M.; Vollmers, C.; Zarrinpar, A.; DiTacchio, L.; Bushong, E.A.; Gill, S.; Leblanc, M.; Chaix, A.; Joens, M.; Fitzpatrick, J.A.; et al. Time-restricted feeding without reducing caloric intake prevents metabolic diseases in mice fed a high-fat diet. Cell Metab. 2012, 15, 848–860. [Google Scholar] [CrossRef]
- Albenberg, L.G.; Wu, G.D. Diet and the intestinal microbiome: associations, functions, and implications for health and disease. Gastroenterology 2014, 146, 1564–1572. [Google Scholar] [CrossRef]
- Karbach, S.H.; Schonfelder, T.; Brandao, I.; Wilms, E.; Hormann, N.; Jackel, S.; Schuler, R.; Finger, S.; Knorr, M.; Lagrange, J.; et al. Gut microbiota promote angiotensin ii-induced arterial hypertension and vascular dysfunction. J. Am. Heart Assoc. 2016, 5, e003698. [Google Scholar] [CrossRef]
- Li, J.; Zhao, F.; Wang, Y.; Chen, J.; Tao, J.; Tian, G.; Wu, S.; Liu, W.; Cui, Q.; Geng, B.; et al. Gut microbiota dysbiosis contributes to the development of hypertension. Microbiome 2017, 5, 14. [Google Scholar] [CrossRef] [PubMed]
- Koeth, R.A.; Levison, B.S.; Culley, M.K.; Buffa, J.A.; Wang, Z.; Gregory, J.C.; Org, E.; Wu, Y.; Li, L.; Smith, J.D.; et al. Gamma-butyrobetaine is a proatherogenic intermediate in gut microbial metabolism of l-carnitine to tmao. Cell Metab. 2014, 20, 799–812. [Google Scholar] [CrossRef] [PubMed]
- Zhu, W.; Gregory, J.C.; Org, E.; Buffa, J.A.; Gupta, N.; Wang, Z.; Li, L.; Fu, X.; Wu, Y.; Mehrabian, M.; et al. Gut microbial metabolite tmao enhances platelet hyperreactivity and thrombosis risk. Cell 2016, 165, 111–124. [Google Scholar] [CrossRef]
- Asher, G.; Sassone-Corsi, P. Time for food: the intimate interplay between nutrition, metabolism, and the circadian clock. Cell 2015, 161, 84–92. [Google Scholar] [CrossRef] [PubMed]
- Voigt, R.M.; Forsyth, C.B.; Green, S.J.; Mutlu, E.; Engen, P.; Vitaterna, M.H.; Turek, F.W.; Keshavarzian, A. Circadian disorganization alters intestinal microbiota. PLoS ONE 2014, 9, e97500. [Google Scholar] [CrossRef] [PubMed]
- Schoenfeld, B.J.; Aragon, A.A.; Krieger, J.W. Effects of meal frequency on weight loss and body composition: a meta-analysis. Nutr. Rev. 2015, 73, 69–82. [Google Scholar] [CrossRef] [PubMed]
- Thaiss, C.A.; Zeevi, D.; Levy, M.; Zilberman-Schapira, G.; Suez, J.; Tengeler, A.C.; Abramson, L.; Katz, M.N.; Korem, T.; Zmora, N.; et al. Transkingdom control of microbiota diurnal oscillations promotes metabolic homeostasis. Cell 2014, 159, 514–529. [Google Scholar] [CrossRef]
- Phillips, M.L. Circadian rhythms: Of owls, larks and alarm clocks. Nature 2009, 458, 142–144. [Google Scholar] [CrossRef]
- Putilov, A.A. Owls, larks, swifts, woodcocks and they are not alone: A historical review of methodology for multidimensional self-assessment of individual differences in sleep-wake pattern. Chronobiol. Int. 2017, 34, 426–437. [Google Scholar] [CrossRef] [PubMed]
- Maukonen, M.; Kanerva, N.; Partonen, T.; Kronholm, E.; Tapanainen, H.; Kontto, J.; Männistö, S. Chronotype differences in timing of energy and macronutrient intakes:A population-based study in adults. Obesity (Silver Spring) 2017, 25, 608–615. [Google Scholar] [CrossRef] [PubMed]
- Tahara, Y.; Shibata, S. Entrainment of the mouse circadian clock: Effects of stress, exercise, and nutrition. Free Radic. Biol. Med. 2018, 119, 129–138. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Paoli, A.; Tinsley, G.; Bianco, A.; Moro, T. The Influence of Meal Frequency and Timing on Health in Humans: The Role of Fasting. Nutrients 2019, 11, 719. https://doi.org/10.3390/nu11040719
Paoli A, Tinsley G, Bianco A, Moro T. The Influence of Meal Frequency and Timing on Health in Humans: The Role of Fasting. Nutrients. 2019; 11(4):719. https://doi.org/10.3390/nu11040719
Chicago/Turabian StylePaoli, Antonio, Grant Tinsley, Antonino Bianco, and Tatiana Moro. 2019. "The Influence of Meal Frequency and Timing on Health in Humans: The Role of Fasting" Nutrients 11, no. 4: 719. https://doi.org/10.3390/nu11040719
APA StylePaoli, A., Tinsley, G., Bianco, A., & Moro, T. (2019). The Influence of Meal Frequency and Timing on Health in Humans: The Role of Fasting. Nutrients, 11(4), 719. https://doi.org/10.3390/nu11040719






