Lactose Intolerance and Bone Health: The Challenge of Ensuring Adequate Calcium Intake
Abstract
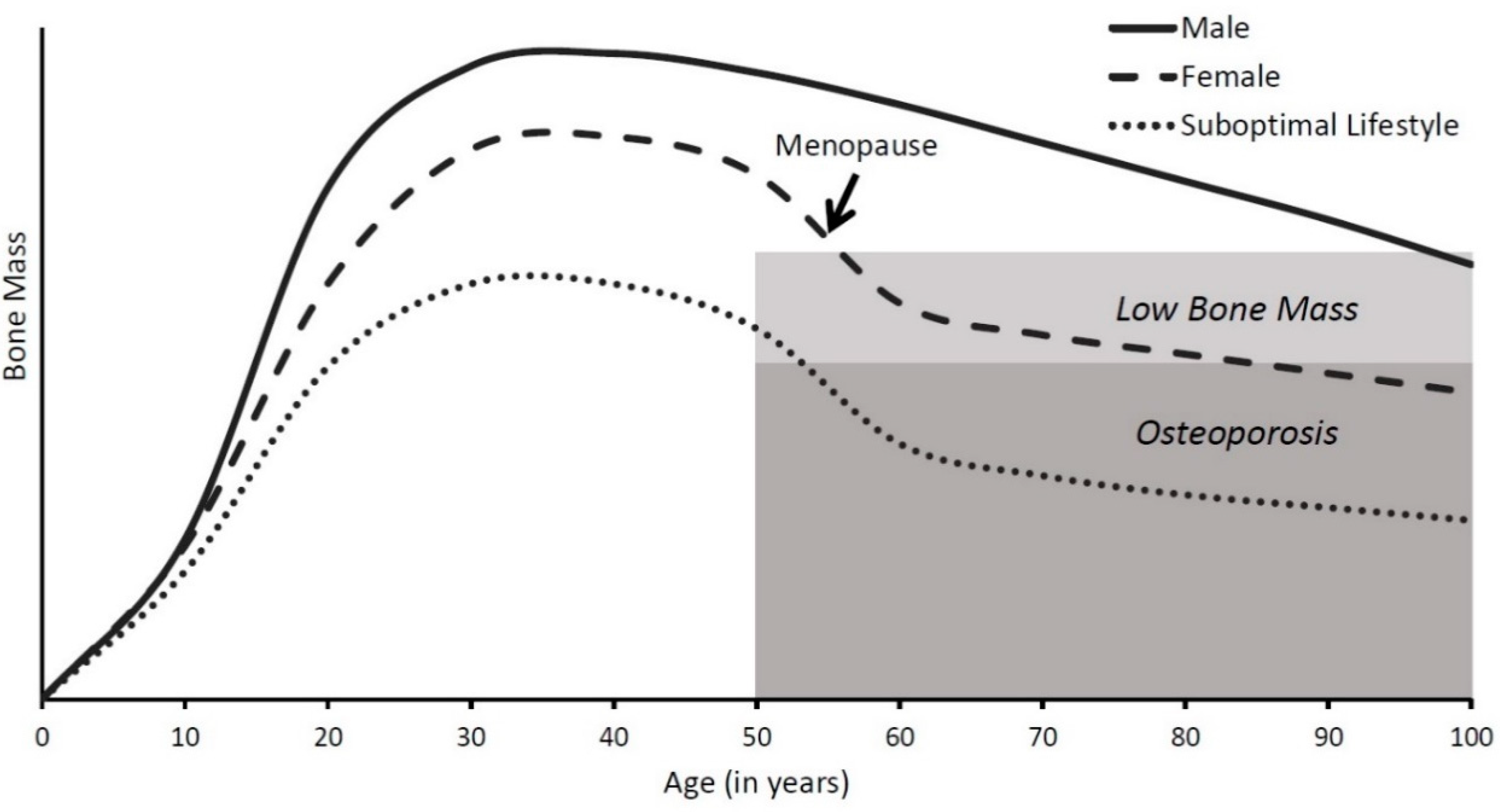
1. Bone Health throughout the Life Span
2. Calcium Requirements
3. Dietary Sources of Calcium
4. Lactose Intolerance and Calcium Bioavailability from Dairy and Alternative Sources
5. Calcium Bioavailability from Alternative Sources and the Impact on Bone Health
6. The Effect of Lactose on Calcium Metabolism and Bone Health in Animal Studies
7. Human Studies of Lactose Intolerance, Dairy Avoidance, and Bone Health
8. Recommendations for Dairy Consumption by Public Health Organizations
8.1. The Majority of People with Lactose Malabsorption Do Not Have Clinical Lactose Intolerance
8.2. Lactose Intolerance Need Not Be an Obstacle to Meeting the Need for Calcium with 3 Servings of Milk and Dairy Products
8.3. Dairy Avoidance May Lead to Deficiencies of Calcium, Vitamin D, and Other Nutrients that Track with Calcium, Which May Predispose Individuals to Decreased Bone Accrual, Osteoporosis, and Other Adverse Health Outcomes
9. Recommendations for Different Age and Racial/Ethnic Groups
10. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| BMC | Bone Mineral Content |
| BMD | Bone Mineral Density |
| DRI | Dietary Reference Intakes |
| IOM | Institute of Medicine |
| IU | International Units |
| NAM | National Academies of Medicine |
| NHANES | National Health And Nutrition Examination Survey |
| NIH | National Institutes of Health |
| NMA | National Medical Association |
| RDA | Recommended Dietary Allowance |
| UL | Upper Limit |
| WIC | Women, Infants, and Children |
References
- Singer, A.; Exuzides, A.; Spangler, L.; O’Malley, C.; Colby, C.; Johnston, K.; Agodoa, I.; Baker, J.; Kagan, R. Burden of illness for osteoporotic fractures compared with other serious diseases among postmenopausal women in the United States. Mayo Clin. Proc. 2015, 90, 53–62. [Google Scholar] [CrossRef] [PubMed]
- Boonen, S.; Autier, P.; Barette, M.; Vanderschueren, D.; Lips, P.; Haentjens, P. Functional outcome and quality of life following hip fracture in elderly women: A prospective controlled study. Osteoporos. Int. 2004, 15, 87–94. [Google Scholar] [PubMed]
- Brenneman, S.K.; Barrett-Connor, E.; Sajjan, S.; Markson, L.E.; Siris, E.S. Impact of recent fracture on health-related quality of life in postmenopausal women. J. Bone Miner. Res. 2006, 21, 809–816. [Google Scholar] [CrossRef] [PubMed]
- Baldan, A.; Tagliati, S.; Saccomandi, D.; Brusaferro, A.; Busoli, L.; Scala, A.; Malaventura, C.; Maggiore, G.; Borgna-Pignatti, C. Assessment of lactose-free diet on the phalangeal bone mineral status in Italian adolescents affected by adult-type hypolactasia. Nutrients 2018, 10, 558. [Google Scholar] [CrossRef]
- Matkovic, V.; Jelic, T.; Wardlaw, G.M.; Ilich, J.Z.; Goel, P.K.; Wright, J.K.; Andon, M.B.; Smith, K.T.; Heaney, R.P. Timing of peak bone mass in Caucasian females and its implication for the prevention of osteoporosis. Inference from a cross-sectional model. J. Clin. Investig. 1994, 93, 799–808. [Google Scholar] [CrossRef]
- Teegarden, D.; Proulx, W.R.; Martin, B.R.; Zhao, J.; McCabe, G.P.; Lyle, R.M.; Peacock, M.; Slemenda, C.; Johnston, C.C.; Weaver, C.M. Peak bone mass in young women. J. Bone Miner. Res. 1995, 10, 711–715. [Google Scholar] [CrossRef]
- Office of the Surgeon General (US). Bone Health and Osteoporosis: A Report of the Surgeon General; Basics of Bone in Health and Disease; US Department of Health and Human Services, Office of the Surgeon General: Washington, DC, USA, 2004.
- Chrischilles, E.A.; Butler, C.D.; Davis, C.S.; Wallace, R.B. A model of lifetime osteoporosis impact. Arch. Intern. Med. 1991, 151, 2026–2032. [Google Scholar] [CrossRef] [PubMed]
- Kenny, A.M.; Prestwood, K.M. Osteoporosis. Pathogenesis, diagnosis, and treatment in older adults. Rheum Dis Clin. N. Am. 2000, 26, 569–591. [Google Scholar] [CrossRef]
- Greenspan, S.L.; Maitland, L.A.; Myers, E.R.; Krasnow, M.B.; Kido, T.H. Femoral bone loss progresses with age: A longitudinal study in women over age 65. J. Bone Miner. Res. 1994, 9, 1959–1965. [Google Scholar] [CrossRef] [PubMed]
- Hannan, M.T.; Felson, D.T.; Anderson, J.J. Bone mineral density in elderly men and women: Results from the Framingham osteoporosis study. J. Bone Miner. Res. 1992, 7, 547–553. [Google Scholar] [CrossRef]
- Institute of Medicine (US) Committee. Dietary Reference Intakes for Calcium and Vitamin D; National Academies Press (US): Washington, DC, USA, 2011.
- Bailey, R.L.; Gahche, J.J.; Miller, P.E.; Thomas, P.R.; Dwyer, J.T. Why US adults use dietary supplements. JAMA Intern. Med. 2013, 173, 355–361. [Google Scholar] [CrossRef]
- Marston, R.M.; Welsh, S.O. Nutrient content of the US food supply. Nat. Food Rev. 1984, 25, 7–13. [Google Scholar]
- Buchowski, M.S. Chapter 1 Calcium in the context of dietary sources and metabolism. In Calcium: Chemistry, Analysis, Function and Effects; The Royal Society of Chemistry: London, UK, 2016; pp. 3–20. [Google Scholar] [CrossRef]
- Bailey, R.L.; Dodd, K.W.; Goldman, J.A.; Gahche, J.J.; Dwyer, J.T.; Moshfegh, A.J.; Sempos, C.T.; Picciano, M.F. Estimation of total usual calcium and vitamin D intakes in the United States. J. Nutr. 2010, 140, 817–822. [Google Scholar] [CrossRef] [PubMed]
- Weaver, C.M.; Plawecki, K.L. Dietary calcium: Adequacy of a vegetarian diet. Am. J. Clin. Nutr. 1994, 59, 1238S–1241S. [Google Scholar] [CrossRef]
- United States Department of Agriculture (USDA). National Nutrient Database. Available online: https://ndb.nal.usda.gov/ndb/ (accessed on 24 February 2019).
- Weaver, C.M.; Martin, B.R.; Nakatsu, C.H.; Armstrong, A.P.; Clavijo, A.; McCabe, L.D.; McCabe, G.P.; Duignan, S.; Schoterman, M.H.; van den Heuvel, E.G. Galactooligosaccharides improve mineral absorption and bone properties in growing rats through gut fermentation. J. Agric. Food Chem. 2011, 59, 6501–6510. [Google Scholar] [CrossRef] [PubMed]
- Abrams, S.A.; Griffin, I.J.; Davila, P.M. Calcium and zinc absorption from lactose-containing and lactose-free infant formulas. Am. J. Clin. Nutr. 2002, 76, 442–446. [Google Scholar] [CrossRef] [PubMed]
- Zittermann, A.; Bock, P.; Drummer, C.; Scheld, K.; Heer, M.; Stehle, P. Lactose does not enhance calcium bioavailability in lactose-tolerant, healthy adults. Am. J. Clin. Nutr. 2000, 71, 931–936. [Google Scholar] [CrossRef] [PubMed]
- Brink, E.J.; van Beresteijn, E.C.; Dekker, P.R.; Beynen, A.C. Urinary excretion of magnesium and calcium as an index of absorption is not affected by lactose intake in healthy adults. Br. J. Nutr. 1993, 69, 863–870. [Google Scholar] [CrossRef]
- Smith, T.M.; Kolars, J.C.; Savaiano, D.A.; Levitt, M.D. Absorption of calcium from milk and yogurt. Am. J. Clin. Nutr. 1985, 42, 1197–1200. [Google Scholar] [CrossRef] [PubMed]
- Tremaine, W.J.; Newcomer, A.D.; Riggs, B.L.; McGill, D.B. Calcium absorption from milk in lactase-deficient and lactase-sufficient adults. Dig. Dis. Sci. 1986, 31, 376–378. [Google Scholar] [CrossRef] [PubMed]
- Griessen, M.; Cochet, B.; Infante, F.; Jung, A.; Bartholdi, P.; Donath, A.; Loizeau, E.; Courvoisier, B. Calcium absorption from milk in lactase-deficient subjects. Am. J. Clin. Nutr. 1989, 49, 377–384. [Google Scholar] [CrossRef] [PubMed]
- Nickel, K.P.; Martin, B.R.; Smith, D.L.; Smith, J.B.; Miller, G.D.; Weaver, C.M. Calcium bioavailability from bovine milk and dairy products in premenopausal women using intrinsic and extrinsic labeling techniques. J. Nutr. 1996, 126, 1406–1411. [Google Scholar] [CrossRef] [PubMed]
- Horowitz, M.; Wishart, J.; Mundy, L.; Nordin, B.E. Lactose and calcium absorption in postmenopausal osteoporosis. Arch. Intern. Med. 1987, 147, 534–536. [Google Scholar] [CrossRef] [PubMed]
- Schuette, S.A.; Yasillo, N.J.; Thompson, C.M. The effect of carbohydrates in milk on the absorption of calcium by postmenopausal women. J. Am. Coll. Nutr. 1991, 10, 132–139. [Google Scholar] [CrossRef] [PubMed]
- Obermayer-Pietsch, B.M.; Gugatschka, M.; Reitter, S.; Plank, W.; Strele, A.; Walter, D.; Bonelli, C.; Goessler, W.; Dobnig, H.; Hogenauer, C.; et al. Adult-type hypolactasia and calcium availability: Decreased calcium intake or impaired calcium absorption? Osteoporos. Int. 2007, 18, 445–451. [Google Scholar] [CrossRef] [PubMed]
- Vandenplas, Y. Lactose intolerance. Asia Pac. J. Clin. Nutr. 2015, 24, S9–S13. [Google Scholar] [PubMed]
- McCarthy, K.S.; Parker, M.; Ameerally, A.; Drake, S.L.; Drake, M.A. Drivers of choice for fluid milk versus plant-based alternatives: What are consumer perceptions of fluid milk? J. Dairy Sci. 2017, 100, 6125–6138. [Google Scholar] [CrossRef] [PubMed]
- Singhal, S.; Baker, R.D.; Baker, S.S. A comparison of the nutritional value of cow’s milk and nondairy beverages. J. Pediatr. Gastroenterol. Nutr. 2017, 64, 799–805. [Google Scholar] [CrossRef] [PubMed]
- Zegler, J. Dairy and Nondiary Milk-US-2013. Available online: http://store.mintel.com/dairy-and-non-dairy-milk-us-april-2013 (accessed on 19 February 2019).
- Marrapodi, A. The Dairy and Nondairy Beverage Market. Available online: http://www.naturalproductsinsider.com/articles/2016/02/the-dairy-and-nondairy-beverage-market.aspx (accessed on 19 February 2019).
- Schuster, M.J.; Wang, X.; Hawkins, T.; Painter, J.E. Comparison of the nutrient content of cow’s milk and nondairy milk alternatives: what’s the difference? Nutr. Today 2018, 53, 153–159. [Google Scholar] [CrossRef]
- Makinen, O.E.; Wanhalinna, V.; Zannini, E.; Arendt, E.K. Foods for special dietary needs: Non-dairy plant-based milk substitutes and fermented dairy-type products. Crit. Rev. Food Sci. Nutr. 2016, 56, 339–349. [Google Scholar] [CrossRef] [PubMed]
- Weaver, C.M. Calcium bioavailability and its relation to osteoporosis. Proc. Soc. Exp. Biol. Med. 1992, 200, 157–160. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Martin, B.R.; Weaver, C.M. Calcium bioavailability of calcium carbonate fortified soymilk is equivalent to cow’s milk in young women. J. Nutr. 2005, 135, 2379–2382. [Google Scholar] [CrossRef] [PubMed]
- Tang, A.L.; Walker, K.Z.; Wilcox, G.; Strauss, B.J.; Ashton, J.F.; Stojanovska, L. Calcium absorption in Australian osteopenic post-menopausal women: An acute comparative study of fortified soymilk to cows’ milk. Asia Pac. J. Clin. Nutr. 2010, 19, 243–249. [Google Scholar] [PubMed]
- Gonnelli, S.; Campagna, M.S.; Montagnani, A.; Caffarelli, C.; Cadirni, A.; Giorgi, G.; Nuti, R. Calcium bioavailability from a new calcium-fortified orange beverage, compared with milk, in healthy volunteers. Int. J. Vitam. Nutr. Res. 2007, 77, 249–254. [Google Scholar] [CrossRef] [PubMed]
- Heaney, R.P.; Rafferty, K.; Dowell, M.S.; Bierman, J. Calcium fortification systems differ in bioavailability. J. Am. Diet. Assoc. 2005, 105, 807–809. [Google Scholar] [CrossRef] [PubMed]
- Heaney, R.P. Factors influencing the measurement of bioavailability, taking calcium as a model. J. Nutr. 2001, 131, 1344S–1348S. [Google Scholar] [CrossRef] [PubMed]
- Bonjour, J.P. Dietary protein: An essential nutrient for bone health. J. Am. Coll. Nutr. 2005, 24, 526S–536S. [Google Scholar] [CrossRef] [PubMed]
- Goulding, A.; Rockell, J.E.; Black, R.E.; Grant, A.M.; Jones, I.E.; Williams, S.M. Children who avoid drinking cow’s milk are at increased risk for prepubertal bone fractures. J. Am. Diet. Assoc. 2004, 104, 250–253. [Google Scholar] [CrossRef] [PubMed]
- Rizzoli, R.; Biver, E.; Bonjour, J.P.; Coxam, V.; Goltzman, D.; Kanis, J.A.; Lappe, J.; Rejnmark, L.; Sahni, S.; Weaver, C.; et al. Benefits and safety of dietary protein for bone health-an expert consensus paper endorsed by the European Society for Clinical and Economical Aspects of Osteopororosis, Osteoarthritis, and Musculoskeletal Diseases and by the International Osteoporosis Foundation. Osteoporos. Int. 2018, 29, 1933–1948. [Google Scholar]
- Gui, J.C.; Brasic, J.R.; Liu, X.D.; Gong, G.Y.; Zhang, G.M.; Liu, C.J.; Gao, G.Q. Bone mineral density in postmenopausal Chinese women treated with calcium fortification in soymilk and cow’s milk. Osteoporos. Int. 2012, 23, 1563–1570. [Google Scholar] [CrossRef] [PubMed]
- Matthews, V.L.; Knutsen, S.F.; Beeson, W.L.; Fraser, G.E. Soy milk and dairy consumption is independently associated with ultrasound attenuation of the heel bone among postmenopausal women: The Adventist Health Study-2. Nutr. Res. 2011, 31, 766–775. [Google Scholar] [CrossRef]
- Andres, A.; Casey, P.H.; Cleves, M.A.; Badger, T.M. Body fat and bone mineral content of infants fed breast milk, cow’s milk formula, or soy formula during the first year of life. J. Pediatr. 2013, 163, 49–54. [Google Scholar] [CrossRef]
- Kukuruzovic, R.H.; Brewster, D.R. Milk formulas in acute gastroenteritis and malnutrition: A randomized trial. J. Paediatr. Child Health 2002, 38, 571–577. [Google Scholar] [CrossRef] [PubMed]
- Buchowski, M.S.; Miller, D.D. Lactose, calcium source and age affect calcium bioavailability in rats. J. Nutr. 1991, 121, 1746–1754. [Google Scholar] [CrossRef] [PubMed]
- Armbrecht, H.J. Age and the effects of lactose on calcium and phosphorus uptake by rat small intestine. Nutr. Res. 1987, 7, 1169–1177. [Google Scholar] [CrossRef]
- Heijnen, A.M.; Brink, E.J.; Lemmens, A.G.; Beynen, A.C. Ileal pH and apparent absorption of magnesium in rats fed on diets containing either lactose or lactulose. Br. J. Nutr. 1993, 70, 747–756. [Google Scholar] [CrossRef]
- Hamalainen, M.M.; Knuuttila, M.; Svanberg, M.; Koskinen, T. Comparison of the effect of gluconate, lactose, and xylitol on bone recalcification in calcium-deficient rats. Bone 1990, 11, 429–438. [Google Scholar] [CrossRef]
- Shortt, C.; Flynn, A. Effect of dietary lactose on salt-mediated changes in mineral metabolism and bone composition in the rat. Br. J. Nutr. 1991, 66, 73–81. [Google Scholar] [CrossRef] [PubMed]
- Moser, R.L.; Peo, E.R., Jr.; Crenshaw, T.D.; Cunningham, P.J. Effect of dietary lactose on gain, feed conversion, blood, bone and intestinal parameters in postweaning rats and swine. J. Anim. Sci. 1980, 51, 89–99. [Google Scholar] [CrossRef]
- Miller, S.C.; Miller, M.A.; Omura, T.H. Dietary lactose improves endochondral growth and bone development and mineralization in rats fed a vitamin D-deficient diet. J. Nutr. 1988, 118, 72–77. [Google Scholar] [CrossRef]
- Au, W.Y.; Raisz, L.G. Restoration of parathyroid responsiveness in vitamin D-deficient rats by parenteral calcium or dietary lactose. J. Clin. Investig. 1967, 46, 1572–1578. [Google Scholar] [CrossRef] [PubMed]
- Schaafsma, G.; Visser, W.J.; Dekker, P.R.; Van Schaik, M. Effect of dietary calcium supplementation with lactose on bone in vitamin D-deficient rats. Bone 1987, 8, 357–362. [Google Scholar] [CrossRef]
- Greger, J.L.; Gutkowski, C.M.; Khazen, R.R. Interactions of lactose with calcium, magnesium and zinc in rats. J. Nutr. 1989, 119, 1691–1697. [Google Scholar] [CrossRef] [PubMed]
- Weaver, C.M.; Janle, E.; Martin, B.; Browne, S.; Guiden, H.; Lachcik, P.; Lee, W.H. Dairy versus calcium carbonate in promoting peak bone mass and bone maintenance during subsequent calcium deficiency. J. Bone Miner. Res. 2009, 24, 1411–1419. [Google Scholar] [CrossRef]
- Huncharek, M.; Muscat, J.; Kupelnick, B. Impact of dairy products and dietary calcium on bone-mineral content in children: Results of a meta-analysis. Bone 2008, 43, 312–321. [Google Scholar] [CrossRef]
- Black, R.E.; Williams, S.M.; Jones, I.E.; Goulding, A. Children who avoid drinking cow milk have low dietary calcium intakes and poor bone health. Am. J. Clin. Nutr. 2002, 76, 675–680. [Google Scholar] [CrossRef] [PubMed]
- Parsons, T.J.; van Dusseldorp, M.; van der Vliet, M.; van de Werken, K.; Schaafsma, G.; van Staveren, W.A. Reduced bone mass in Dutch adolescents fed a macrobiotic diet in early life. J. Bone Miner. Res. 1997, 12, 1486–1494. [Google Scholar] [CrossRef]
- Rockell, J.E.; Williams, S.M.; Taylor, R.W.; Grant, A.M.; Jones, I.E.; Goulding, A. Two-year changes in bone and body composition in young children with a history of prolonged milk avoidance. Osteoporos. Int. 2005, 16, 1016–1023. [Google Scholar] [CrossRef]
- Suchy, F.J.; Brannon, P.M.; Carpenter, T.O.; Fernandez, J.R.; Gilsanz, V.; Gould, J.B.; Hall, K.; Hui, S.L.; Lupton, J.; Mennella, J.; et al. NIH consensus development conference statement: Lactose intolerance and health. NIH Consens. State Sci. Statements 2010, 27, 1–27. [Google Scholar]
- Du, X.Q.; Greenfield, H.; Fraser, D.R.; Ge, K.Y.; Liu, Z.H.; He, W. Milk consumption and bone mineral content in Chinese adolescent girls. Bone 2002, 30, 521–528. [Google Scholar] [CrossRef]
- Matlik, L.; Savaiano, D.; McCabe, G.; VanLoan, M.; Blue, C.L.; Boushey, C.J. Perceived milk intolerance is related to bone mineral content in 10- to 13-year-old female adolescents. Pediatrics 2007, 120, e669–e677. [Google Scholar] [CrossRef] [PubMed]
- Lau, E.M.; Lynn, H.; Chan, Y.H.; Lau, W.; Woo, J. Benefits of milk powder supplementation on bone accretion in Chinese children. Osteoporos. Int. 2004, 15, 654–658. [Google Scholar] [CrossRef] [PubMed]
- Gibbons, M.J.; Gilchrist, N.L.; Frampton, C.; Maguire, P.; Reilly, P.H.; March, R.L.; Wall, C.R. The effects of a high calcium dairy food on bone health in pre-pubertal children in New Zealand. Asia Pac. J. Clin. Nutr. 2004, 13, 341–347. [Google Scholar] [PubMed]
- Bonjour, J.P.; Carrie, A.L.; Ferrari, S.; Clavien, H.; Slosman, D.; Theintz, G.; Rizzoli, R. Calcium-enriched foods and bone mass growth in prepubertal girls: A randomized, double-blind, placebo-controlled trial. J. Clin. Investig. 1997, 99, 1287–1294. [Google Scholar] [CrossRef] [PubMed]
- Segal, E.; Dvorkin, L.; Lavy, A.; Rozen, G.S.; Yaniv, I.; Raz, B.; Tamir, A.; Ish-Shalom, S. Bone density in axial and appendicular skeleton in patients with lactose intolerance: Influence of calcium intake and vitamin D status. J. Am. Coll. Nutr. 2003, 22, 201–207. [Google Scholar] [CrossRef]
- Corazza, G.R.; Benati, G.; Di Sario, A.; Tarozzi, C.; Strocchi, A.; Passeri, M.; Gasbarrini, G. Lactose intolerance and bone mass in postmenopausal Italian women. Br. J. Nutr. 1995, 73, 479–487. [Google Scholar] [CrossRef]
- Laaksonen, M.M.; Impivaara, O.; Sievanen, H.; Viikari, J.S.; Lehtimaki, T.J.; Lamberg-Allardt, C.J.; Karkkainen, M.U.; Valimaki, M.; Heikkinen, J.; Kroger, L.M.; et al. Associations of genetic lactase non-persistence and sex with bone loss in young adulthood. Bone 2009, 44, 1003–1009. [Google Scholar] [CrossRef]
- Hallkvist, O.M.; Johansson, J.; Nordstrom, A.; Nordstrom, P.; Hult, A. Dairy product intake and bone properties in 70-year-old men and women. Arch. Osteoporos. 2018, 13, 9. [Google Scholar] [CrossRef]
- Honkanen, R.; Pulkkinen, P.; Jarvinen, R.; Kroger, H.; Lindstedt, K.; Tuppurainen, M.; Uusitupa, M. Does lactose intolerance predispose to low bone density? A population-based study of perimenopausal Finnish women. Bone 1996, 19, 23–28. [Google Scholar] [CrossRef]
- Obermayer-Pietsch, B.M.; Bonelli, C.M.; Walter, D.E.; Kuhn, R.J.; Fahrleitner-Pammer, A.; Berghold, A.; Goessler, W.; Stepan, V.; Dobnig, H.; Leb, G.; et al. Genetic predisposition for adult lactose intolerance and relation to diet, bone density, and bone fractures. J. Bone Miner. Res. 2004, 19, 42–47. [Google Scholar] [CrossRef]
- Goulding, A.; Taylor, R.W.; Keil, D.; Gold, E.; Lewis-Barned, N.J.; Williams, S.M. Lactose malabsorption and rate of bone loss in older women. Age Ageing 1999, 28, 175–180. [Google Scholar] [CrossRef] [PubMed]
- Di Stefano, M.; Veneto, G.; Malservisi, S.; Cecchetti, L.; Minguzzi, L.; Strocchi, A.; Corazza, G.R. Lactose malabsorption and intolerance and peak bone mass. Gastroenterology 2002, 122, 1793–1799. [Google Scholar] [CrossRef]
- Chiu, J.F.; Lan, S.J.; Yang, C.Y.; Wang, P.W.; Yao, W.J.; Su, L.H.; Hsieh, C.C. Long-term vegetarian diet and bone mineral density in postmenopausal Taiwanese women. Calcif. Tissue Int. 1997, 60, 245–249. [Google Scholar] [CrossRef] [PubMed]
- Szilagyi, A.; Ishayek, N. Lactose intolerance, dairy avoidance, and treatment options. Nutrients 2018, 10, 1994. [Google Scholar] [CrossRef]
- Turner, L.W.; Hunt, S.; Kendrick, O.; Eddy, J. Dairy-product intake and hip fracture among older women: Issues for health behavior. Psychol. Rep. 1999, 85, 423–430. [Google Scholar] [CrossRef]
- Johnell, O.; Gullberg, B.; Kanis, J.A.; Allander, E.; Elffors, L.; Dequeker, J.; Dilsen, G.; Gennari, C.; Lopes Vaz, A.; Lyritis, G.; et al. Risk factors for hip fracture in European women: The MEDOS Study. Mediterranean Osteoporosis Study. J. Bone Miner. Res. 1995, 10, 1802–1815. [Google Scholar] [CrossRef] [PubMed]
- Enattah, N.S.; Sulkava, R.; Halonen, P.; Kontula, K.; Jarvela, I. Genetic variant of lactase-persistent C/T-13910 is associated with bone fractures in very old age. J. Am. Geriatr. Soc. 2005, 53, 79–82. [Google Scholar] [CrossRef] [PubMed]
- Enattah, N.; Pekkarinen, T.; Valimaki, M.J.; Loyttyniemi, E.; Jarvela, I. Genetically defined adult-type hypolactasia and self-reported lactose intolerance as risk factors of osteoporosis in Finnish postmenopausal women. Eur. J. Clin. Nutr. 2005, 59, 1105–1111. [Google Scholar] [CrossRef] [PubMed]
- Feskanich, D.; Meyer, H.E.; Fung, T.T.; Bischoff-Ferrari, H.A.; Willett, W.C. Milk and other dairy foods and risk of hip fracture in men and women. Osteoporos. Int. 2018, 29, 385–396. [Google Scholar] [CrossRef]
- Bischoff-Ferrari, H.A.; Dawson-Hughes, B.; Baron, J.A.; Kanis, J.A.; Orav, E.J.; Staehelin, H.B.; Kiel, D.P.; Burckhardt, P.; Henschkowski, J.; Spiegelman, D.; et al. Milk intake and risk of hip fracture in men and women: A meta-analysis of prospective cohort studies. J. Bone Miner. Res. 2011, 26, 833–839. [Google Scholar] [CrossRef]
- Benetou, V.; Orfanos, P.; Zylis, D.; Sieri, S.; Contiero, P.; Tumino, R.; Giurdanella, M.C.; Peeters, P.H.; Linseisen, J.; Nieters, A.; et al. Diet and hip fractures among elderly Europeans in the EPIC cohort. Eur. J. Clin. Nutr. 2011, 65, 132–139. [Google Scholar] [CrossRef]
- Sahni, S.; Mangano, K.M.; Tucker, K.L.; Kiel, D.P.; Casey, V.A.; Hannan, M.T. Protective association of milk intake on the risk of hip fracture: Results from the Framingham Original Cohort. J. Bone Miner. Res. 2014, 29, 1756–1762. [Google Scholar] [CrossRef] [PubMed]
- Kalkwarf, H.J.; Khoury, J.C.; Lanphear, B.P. Milk intake during childhood and adolescence, adult bone density, and osteoporotic fractures in US women. Am. J. Clin. Nutr. 2003, 77, 257–265. [Google Scholar] [CrossRef]
- Feskanich, D.; Bischoff-Ferrari, H.A.; Frazier, A.L.; Willett, W.C. Milk consumption during teenage years and risk of hip fractures in older adults. JAMA Pediatr. 2014, 168, 54–60. [Google Scholar] [CrossRef]
- U.S. Department of Health and Human Services; U.S. Department of Agriculture. 2015–2020 Dietary Guidelines for Americans, 8th ed. Available online: https://health.gov/dietaryguidelines/2015/guidelines/ (accessed on 19 February 2019).
- Savaiano, D. Lactose intolerance: A self-fulfilling prophecy leading to osteoporosis? Nutr. Rev. 2003, 61, 221–223. [Google Scholar] [PubMed]
- Heaney, R.P. Dairy intake, dietary adequacy, and lactose intolerance. Adv. Nutr. 2013, 4, 151–156. [Google Scholar] [CrossRef]
- Nicklas, T.A.; Qu, H.; Hughes, S.O.; Wagner, S.E.; Foushee, H.R.; Shewchuk, R.M. Prevalence of Self-reported Lactose Intolerance in a Multiethnic Sample of Adults. Nutr. Today 2009, 44, 222–227. [Google Scholar] [CrossRef]
- Suarez, F.L.; Savaiano, D.A.; Levitt, M.D. A comparison of symptoms after the consumption of milk or lactose-hydrolyzed milk by people with self-reported severe lactose intolerance. N. Engl. J. Med. 1995, 333, 1–4. [Google Scholar] [CrossRef]
- Hertzler, S.R.; Savaiano, D.A. Colonic adaptation to daily lactose feeding in lactose maldigesters reduces lactose intolerance. Am. J. Clin. Nutr. 1996, 64, 232–236. [Google Scholar] [CrossRef] [PubMed]
- Miller, G.D.; Jarvis, J.K.; Council, N.D.; McBean, L.D. Handbook of Dairy Foods and Nutrition; CRC Press: Boca Raton, FL, USA, 2002. [Google Scholar]
- Weaver, C.M. How sound is the science behind the dietary recommendations for dairy? Am. J. Clin. Nutr. 2014, 99, 1217S–1222S. [Google Scholar] [CrossRef]
- Nicklas, T.A.; O’Neil, C.E.; Fulgoni, V.L., 3rd. The role of dairy in meeting the recommendations for shortfall nutrients in the American diet. J. Am. Coll. Nutr. 2009, 28, 73S–81S. [Google Scholar] [CrossRef] [PubMed]
- Imamura, F.; Fretts, A.; Marklund, M.; Ardisson Korat, A.V.; Yang, W.S.; Lankinen, M.; Qureshi, W.; Helmer, C.; Chen, T.A.; Wong, K.; et al. Fatty acid biomarkers of dairy fat consumption and incidence of type 2 diabetes: A pooled analysis of prospective cohort studies. PLoS Med. 2018, 15, e1002670. [Google Scholar] [CrossRef] [PubMed]
- Mozaffarian, D.; Wu, J.H.Y. Flavonoids, dairy foods, and cardiovascular and metabolic health: A review of emerging biologic pathways. Circ. Res. 2018, 122, 369–384. [Google Scholar] [CrossRef] [PubMed]
- Stallings, V.A.; Oddleifson, N.W.; Negrini, B.Y.; Zemel, B.S.; Wellens, R. Bone mineral content and dietary calcium intake in children prescribed a low-lactose diet. J. Pediatr. Gastroenterol. Nutr. 1994, 18, 440–445. [Google Scholar] [CrossRef]
- World Cancer Research Fund; American Institute for Cancer Research. Food, Nutrition, Physical Activity, and the Prevention of Cancer: A Global Perspective; The American Institute for Cancer Research: Washington, DC, USA, 2007. [Google Scholar]
- McGuire, S. Institute of Medicine. 2009. School Meals: Building Blocks for Healthy Children; The National Academies Press: Washington, DC, USA, 2011; Volume 2, pp. 64–65. [Google Scholar]
- Institute of Medicine. WIC Food Packages: Time for a Change; The National Academies Press: Washington, DC, USA, 2006; p. 432. [Google Scholar] [CrossRef]
- Heyman, M.B. Lactose intolerance in infants, children, and adolescents. Pediatrics 2006, 118, 1279–1286. [Google Scholar] [CrossRef]
- National Medical Association. Lactose intolerance and African Americans: Implications for the consumption of appropriate intake levels of key nutrients. J. Natl. Med. Assoc. 2009, 101, 5S–23S. [Google Scholar] [CrossRef]
- Weaver, C.M.; Heaney, R.P. Food sources, supplements, and bioavailability. In Calcium in Human Health; Weaver, C.M., Heaney, R.P., Eds.; Humana Press: Totowa, NJ, USA, 2006; pp. 129–142. [Google Scholar] [CrossRef]
- Lee, G.J.; Birken, C.S.; Parkin, P.C.; Lebovic, G.; Chen, Y.; L’Abbe, M.R.; Maguire, J.L. Consumption of non-cow’s milk beverages and serum vitamin D levels in early childhood. CMAJ 2014, 186, 1287–1293. [Google Scholar] [CrossRef]
- EFSA Panel on Dietetic Products, Nutrition and Allergies (NDA). Scientific opinion on lactose thresholds in lactose intolerance and galactosaemia. EFSA J. 2010, 8, 1777. [Google Scholar] [CrossRef]
| Age | RDA 1 | UL 2 | |
|---|---|---|---|
| Female | Male | ||
| 1–3 y | 700 | 700 | 2500 |
| 4–8 y | 1000 | 1000 | 2500 |
| 9–18 y | 1300 | 1300 | 3000 |
| 19–50 y | 1000 | 1000 | 2500 |
| 51–70 y | 1200 | 1000 | 2000 |
| >70 y | 1200 | 1200 | 2000 |
| Food | mg Ca/100 g | mg Ca/Serving |
|---|---|---|
| Milk | ||
| Skim | 122 | 299 |
| 1% | 125 | 305 |
| 2% | 120 | 293 |
| Whole | 113 | 276 |
| Yogurt | ||
| Greek | 100 | 150 |
| Plain | 121 | 275 |
| Vanilla | 171 | 291 |
| Cheese | ||
| American | 789 | 150 |
| Cheddar | 893 | 250 |
| Colby | 685 | 194 |
| Cottage Cheese | 111 | 125 |
| Goat | 140 | 40 |
| Mozzarella | 505 | 143 |
| Provolone | 756 | 214 |
| Swiss | 890 | 252 |
| Snacks | ||
| Milk Chocolate | 189 | 47 |
| Vanilla Ice Cream | 128 | 84 |
| Dairy Substitutes | ||
| Almond Milk 1 | 188 | 451 |
| Cashew Milk 1 | 127 | 451 |
| Coconut Milk 1 | 188 | 451 |
| Hemp Milk 1 | 208 | 499 |
| Rice Milk 1 | 118 | 283 |
| Soy Milk 1 | 123 | 299 |
| Tofu Yogurt | 118 | 309 |
| Food | mg Ca/100 g | mg Ca/Serving |
|---|---|---|
| Fruits and Vegetables | ||
| Arugula | 160 | 16 |
| Beet Greens | 117 | 44 |
| Broccoli | 47 | 43 |
| Brussel Sprouts | 26 | 25 |
| Cabbage, Bok Choy | 105 | 74 |
| Cabbage, Chinese | 77 | 59 |
| Cabbage, Green | 40 | 36 |
| Cauliflower | 22 | 24 |
| Collards | 232 | 84 |
| Kale | 254 | 53 |
| Kohlrabi | 24 | 32 |
| Mustard Greens | 115 | 64 |
| Orange Juice 1 | 140 | 349 |
| Spinach | 99 | 30 |
| Tofu, nigari 2 | 345 | 421 |
| Tofu, plain | 31 | 26 |
| Turnip Greens | 190 | 104 |
| Meats and Fish | ||
| Beef | 18 | 20 |
| Canned Pink Salmon | 215 | 183 |
| Canned Sockeye Salmon | 198 | 168 |
| Herring | 83 | 71 |
| Oyster | 44 | 37 |
| Trout | 43 | 37 |
| Turkey | 19 | 21 |
| Walleye | 110 | 175 |
| Nuts, Beans, and Seeds | ||
| Almonds | 269 | 76 |
| Beans, Black | 105 | 40 |
| Beans, Pinto | 111 | 40 |
| Beans, Red | 130 | 60 |
| Beans, Soya | 62 | 81 |
| Beans, White | 49 | 64 |
| Hazelnuts | 114 | 32 |
| Sesame Seeds | 975 | 88 |
| Sunflower Seeds | 78 | 36 |
| Water | Cow’s Milk | Dairy Substitute Beverages | |||||
|---|---|---|---|---|---|---|---|
| Bottled | Skim 1 | Whole 2 | Soy 3 | Rice 3 | Almond 3 | Coconut 3 | |
| Energy (kcal) | 0 | 81 | 149 | 104 | 113 | 91 | 70 |
| Macronutrients (g) | |||||||
| Protein | 0 | 8.1 | 7.7 | 6.3 | 0.7 | 1.0 | 0 |
| Fat | 0 | 0.2 | 7.9 | 3.6 | 2.3 | 2.5 | 4.5 |
| Carbohydrates | 0 | 12 | 12 | 12 | 22 | 16 | 8 |
| Minerals (mg) | |||||||
| Calcium | 24 | 293 | 276 | 299 | 283 | 451 | 101 |
| Magnesium | 5 | 26 | 24 | 36 | 26 | 17 | 41 |
| Phosphorus | 0 | 242 | 205 | 104 | 134 | 19 | - |
| Potassium | 0 | 347 | 322 | 296 | 65 | 120 | 41 |
| Sodium | 5 | 101 | 105 | 114 | 94 | 151 | 0 |
| Vitamins (IU) | |||||||
| Vitamin A | 0 | 490 | 395 | 450 | 499 | 499 | 499 |
| Vitamin D | 0 | 113 | 124 | 104 | 101 | 101 | 120 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hodges, J.K.; Cao, S.; Cladis, D.P.; Weaver, C.M. Lactose Intolerance and Bone Health: The Challenge of Ensuring Adequate Calcium Intake. Nutrients 2019, 11, 718. https://doi.org/10.3390/nu11040718
Hodges JK, Cao S, Cladis DP, Weaver CM. Lactose Intolerance and Bone Health: The Challenge of Ensuring Adequate Calcium Intake. Nutrients. 2019; 11(4):718. https://doi.org/10.3390/nu11040718
Chicago/Turabian StyleHodges, Joanna K., Sisi Cao, Dennis P. Cladis, and Connie M. Weaver. 2019. "Lactose Intolerance and Bone Health: The Challenge of Ensuring Adequate Calcium Intake" Nutrients 11, no. 4: 718. https://doi.org/10.3390/nu11040718
APA StyleHodges, J. K., Cao, S., Cladis, D. P., & Weaver, C. M. (2019). Lactose Intolerance and Bone Health: The Challenge of Ensuring Adequate Calcium Intake. Nutrients, 11(4), 718. https://doi.org/10.3390/nu11040718






