Predicting Real-Life Eating Behaviours Using Single School Lunches in Adolescents
Abstract
1. Introduction
2. Materials and Methods
2.1. Subjects
2.2. Experimental Design
2.3. School Lunch Session
2.4. Real-Life Session
2.5. Device and Mobile Application
2.6. Data Analysis
3. Results
3.1. Subjects
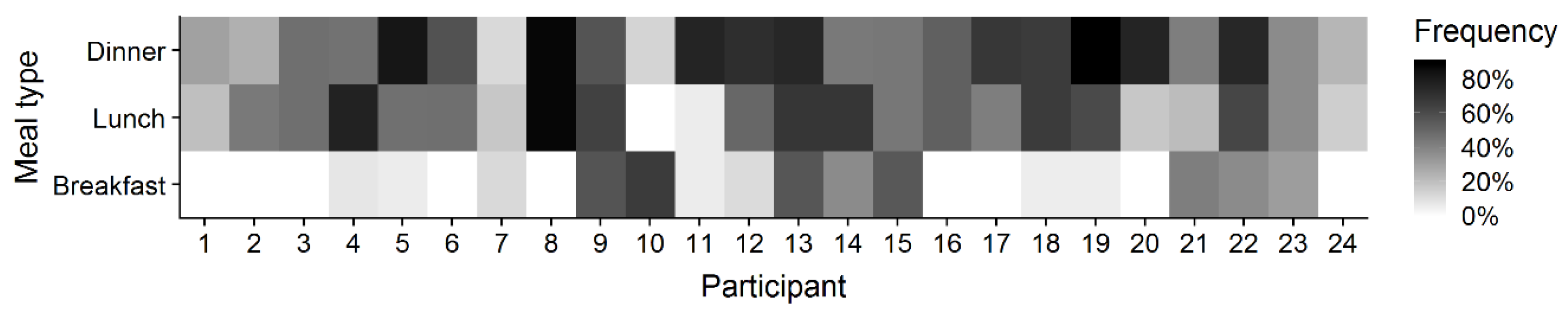
3.2. Recording Frequency
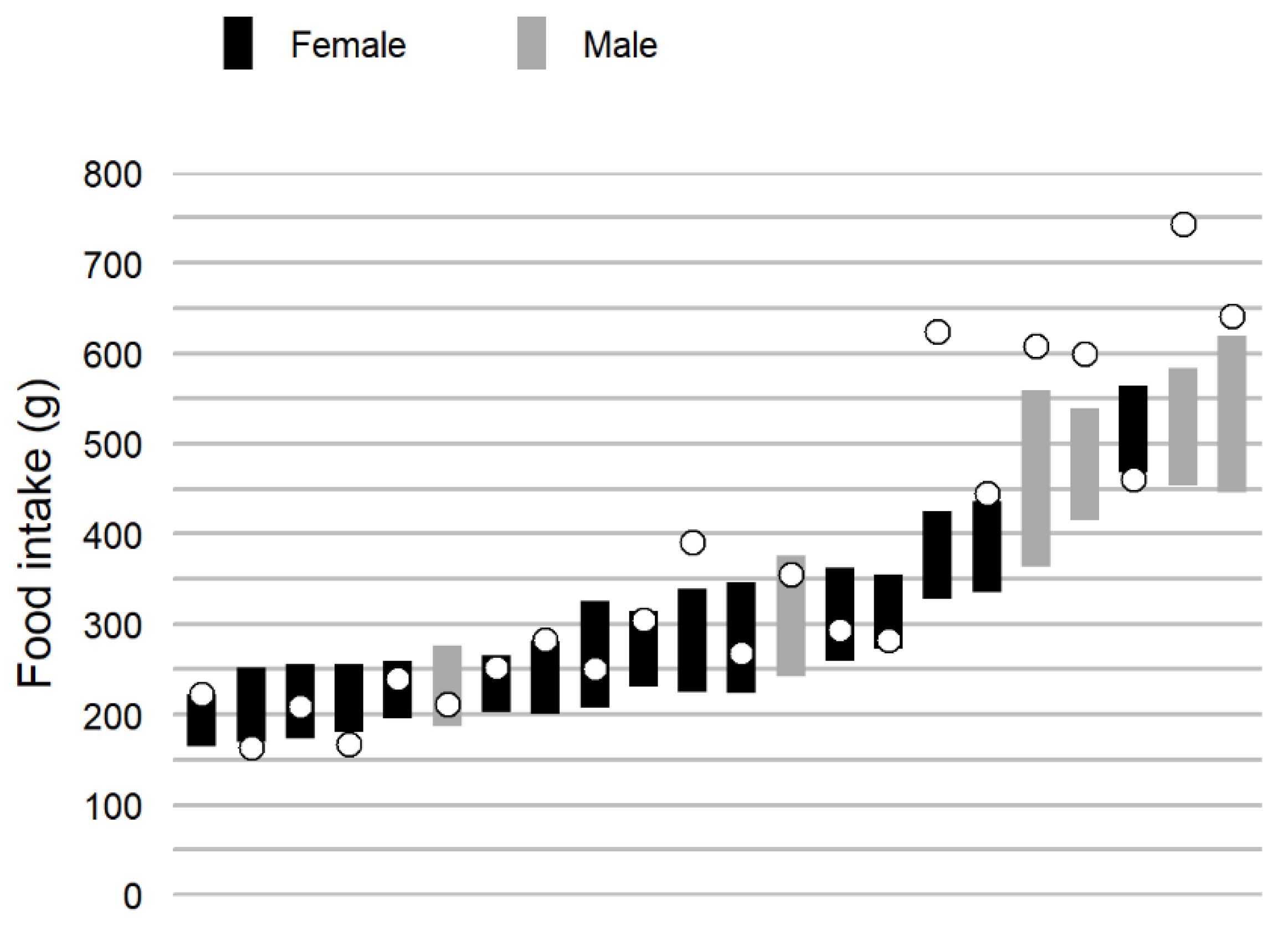
3.3. Real-Life Variance
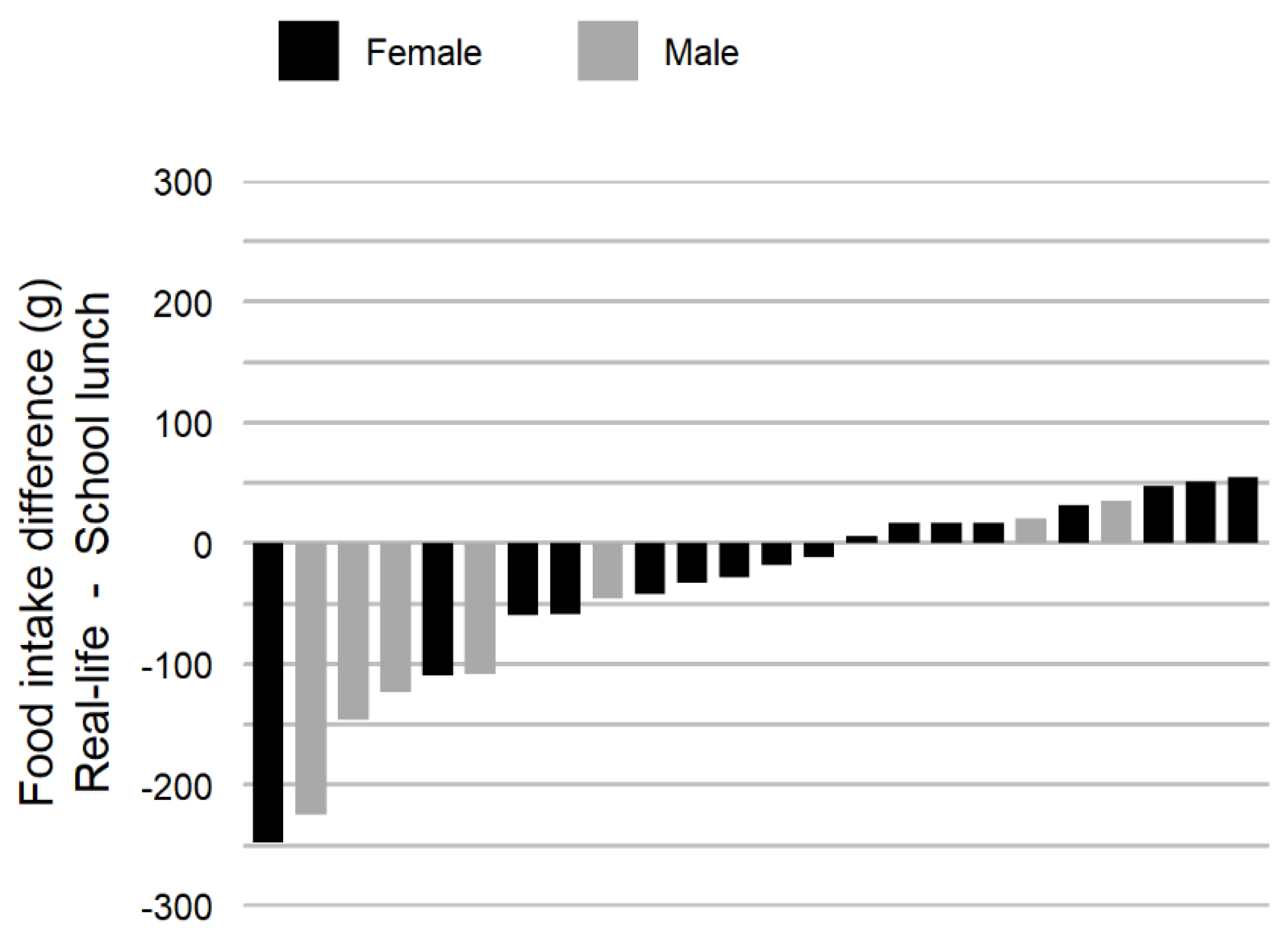
3.4. Comparison of School Lunch and Real-Life Food Intake Weight and Eating Rate
3.5. Agreement between Measures
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- World Health Organization. Report of the Commission on Ending Childhood Obesity; World Health Organization: Geneva, Switzerland, 2016. [Google Scholar]
- Lasschuijt, M.P.; Mars, M.; Stieger, M.; Miquel-Kergoat, S.; de Graaf, C.; Smeets, P. Comparison of oro-sensory exposure duration and intensity manipulations on satiation. Physiol. Behav. 2017, 176, 76–83. [Google Scholar] [CrossRef]
- McCrickerd, K.; Forde, C.G. Sensory influences on food intake control: Moving beyond palatability. Obes. Rev. 2016, 17, 18–29. [Google Scholar] [CrossRef]
- Stroebele, N.; De Castro, J.M. Effect of ambience on food intake and food choice. Nutrition 2004, 20, 821–838. [Google Scholar] [CrossRef]
- Hollands, G.J.; Shemilt, I.; Marteau, T.M.; Jebb, S.A.; Lewis, H.B.; Wei, Y.; Higgins, J.P.; Ogilvie, D. Portion, package or tableware size for changing selection and consumption of food, alcohol and tobacco. Cochrane Database Syst Rev. 2015, 9, CD011045. [Google Scholar] [CrossRef]
- Robinson, E.; Almiron-Roig, E.; Rutters, F.; de Graaf, C.; Forde, C.G.; Tudur Smith, C.; Nolan, S.J.; Jebb, S.A. A systematic review and meta-analysis examining the effect of eating rate on energy intake and hunger. Am. J. Clin. Nutr. 2014, 100, 123–151. [Google Scholar] [CrossRef]
- Ohkuma, T.; Hirakawa, Y.; Nakamura, U.; Kiyohara, Y.; Kitazono, T.; Ninomiya, T. Association between eating rate and obesity: A systematic review and meta-analysis. Int. J. Obes. 2015, 39, 1589–1596. [Google Scholar] [CrossRef] [PubMed]
- Berg, C.; Lappas, G.; Wolk, A.; Strandhagen, E.; Torén, K.; Rosengren, A.; Thelle, D.; Lissner, L. Eating patterns and portion size associated with obesity in a Swedish population. Appetite 2009, 52, 21–26. [Google Scholar] [CrossRef] [PubMed]
- Torbahn, G.; Gellhaus, I.; Koch, B.; von Kries, R.; Obermeier, V.; Holl, R.W.; Fink, K.; van Egmond-Fröhlich, A. Reduction of Portion Size and Eating Rate Is Associated with BMI-SDS Reduction in Overweight and Obese Children and Adolescents: Results on Eating and Nutrition Behaviour from the Observational KgAS Study. Obes Facts 2017, 10, 503–516. [Google Scholar] [CrossRef] [PubMed]
- Ford, A.L.; Bergh, C.; Södersten, P.; Sabin, M.A.; Hollinghurst, S.; Hunt, L.P.; Shield, J.P. Treatment of childhood obesity by retraining eating behaviour: Randomised controlled trial. BMJ 2010, 340, b5388. [Google Scholar] [CrossRef] [PubMed]
- Hubel, R.; Laessle, R.G.; Lehrke, S.; Jass, J. Laboratory measurement of cumulative food intake in humans: Results on reliability. Appetite 2006, 46, 57–62. [Google Scholar] [CrossRef] [PubMed]
- Laessle, R.; Geiermann, L. Reliability of laboratory measurement of human food intake. Appetite 2012, 58, 249–251. [Google Scholar] [CrossRef] [PubMed]
- Barkeling, B.; Rössner, S.; Sjöberg, A. Methodological studies on single meal food intake characteristics in normal weight and obese men and women. Int. J. Obes. Relat. Metab. Disord. J. Int. Assoc. Study Obes. 1995, 19, 284–290. [Google Scholar]
- McCrickerd, K.; Forde, C.G. Consistency of Eating Rate, Oral Processing Behaviours and Energy Intake across Meals. Nutrients 2017, 9, 891. [Google Scholar] [CrossRef] [PubMed]
- Zandian, M.; Ioakimidis, I.; Bergh, C.; Brodin, U.; Södersten, P. Decelerated and linear eaters: Effect of eating rate on food intake and satiety. Physiol. Behav. 2009, 96, 270–275. [Google Scholar] [CrossRef] [PubMed]
- Langlet, B.; Tang Bach, M.; Odegi, D.; Fagerberg, P.; Ioakimidis, I. The Effect of Food Unit Sizes and Meal Serving Occasions on Eating Behaviour Characteristics: Within Person Randomised Crossover Studies on Healthy Women. Nutrients 2018, 10, 880. [Google Scholar] [CrossRef] [PubMed]
- van den Boer, J.H.W.; Kranendonk, J.; van de Wiel, A.; Feskens, E.J.M.; Geelen, A.; Mars, M. Self-reported eating rate is associated with weight status in a Dutch population: A validation study and a cross-sectional study. Int. J. Behav. Nutr. Phys. Act. 2017, 14, 121. [Google Scholar] [CrossRef] [PubMed]
- Petty, A.J.; Melanson, K.J.; Greene, G.W. Self-reported eating rate aligns with laboratory measured eating rate but not with free-living meals. Appetite 2013, 63, 36–41. [Google Scholar] [CrossRef]
- de Castro, J.M. Eating behavior: Lessons from the real world of humans. Nutrition 2000, 16, 800–813. [Google Scholar] [CrossRef]
- Meiselman, H.L. Methodology and theory in human eating research. Appetite 1992, 19, 49–55. [Google Scholar] [CrossRef]
- Langlet, B.; Anvret, A.; Maramis, C.; Moulos, I.; Papapanagiotou, V.; Diou, C.; Lekka, E.; Heimeier, R.; Delopoulos, A.; Ioakimidis, I. Objective measures of eating behaviour in a Swedish high school. Behav. Inf. Technol. 2017, 36, 1005–1013. [Google Scholar] [CrossRef]
- Papapanagiotou, V.; Diou, C.; Ioakimidis, I.; Sodersten, P.; Delopoulos, A. Automatic analysis of food intake and meal microstructure based on continuous weight measurements. IEEE J. Biomed. Health Inform. 2018, 23, 893–902. [Google Scholar] [CrossRef] [PubMed]
- Esfandiari, M.; Papapanagiotou, V.; Diou, C.; Zandian, M.; Nolstam, J.; Södersten, P.; Bergh, C. Control of Eating Behavior Using a Novel Feedback System. J. Vis. Exp. JoVE 2018. [Google Scholar] [CrossRef]
- Maramis, C.; Diou, C.; Ioakeimidis, I.; Lekka, I.; Dudnik, G.; Mars, M.; Maglaveras, N.; Bergh, C.; Delopoulos, A. Preventing obesity and eating disorders through behavioural modifications: The SPLENDID vision. In Proceedings of the 2014 EAI 4th International Conference on Wireless Mobile Communication and Healthcare (Mobihealth), Athens, Greece, 3–5 November 2014; pp. 7–10. [Google Scholar]
- Papapanagiotou, V.; Diou, C.; Langlet, B.; Ioakimidis, I.; Delopoulos, A. A parametric Probabilistic Context-Free Grammar for food intake analysis based on continuous meal weight measurements. In Proceedings of the 2015 37th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC), Milan, Italy, 25–29 August 2015; pp. 7853–7856. [Google Scholar]
- Diou, C.; Sarafis, I.; Ioakimidis, I.; Delopoulos, A. Data-driven assessments for sensor measurements of eating behavior. In Proceedings of the 2017 IEEE EMBS International Conference on Biomedical Health Informatics (BHI), Orlando, FL, USA, 16–19 February 2017; pp. 129–132. [Google Scholar]
- Ioakimidis, I.; Zandian, M.; Eriksson-Marklund, L.; Bergh, C.; Grigoriadis, A.; Södersten, P. Description of chewing and food intake over the course of a meal. Physiol. Behav. 2011, 104, 761–769. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2018; Available online: http://www.R-project.org/ (accessed on 1 February 2019).
- Hopkins, W.G. Spreadsheets for Analysis of Validity and Reliability. Sportscience 2017, 21, 36–44. [Google Scholar]
- Hopkins, W.G. Measures of reliability in sports medicine and science. Sports Med. 2000, 30, 1–15. [Google Scholar] [CrossRef]
- Langlet, B.; Fagerberg, P.; Glossner, A.; Ioakimidis, I. Objective quantification of the food proximity effect on grapes, chocolate and cracker consumption in a Swedish high school. A temporal analysis. PLoS ONE 2017, 12, e0182172. [Google Scholar] [CrossRef]
- Ong, J.X.; Ullah, S.; Magarey, A.; Miller, J.; Leslie, E. Relationship between the home environment and fruit and vegetable consumption in children aged 6-12 years: A systematic review. Public Health Nutr. 2017, 20, 464–480. [Google Scholar] [CrossRef]
- Herman, C.P. The social facilitation of eating. A review. Appetite 2015, 86, 61–73. [Google Scholar] [CrossRef]
- Zandian, M.; Ioakimidis, I.; Bergström, J.; Brodin, U.; Bergh, C.; Leon, M.; Shield, J.; Södersten, P. Children eat their school lunch too quickly: An exploratory study of the effect on food intake. BMC Public Health 2012, 12, 351. [Google Scholar] [CrossRef]
- Johnson, F.; Wardle, J. Variety, palatability, and obesity. Adv. Nutr. 2014, 5, 851–859. [Google Scholar] [CrossRef]
- Periwal, V.; Chow, C.C. Patterns in food intake correlate with body mass index. Am. J. Physiol. Endocrinol. Metab. 2006, 291, E929–E936. [Google Scholar] [CrossRef] [PubMed]
- Basiotis, P.P.; Thomas, R.G.; Kelsay, J.L.; Mertz, W. Sources of variation in energy intake by men and women as determined from one year’s daily dietary records. Am. J. Clin. Nutr. 1989, 50, 448–453. [Google Scholar] [CrossRef]
- Basiotis, P.P.; Welsh, S.O.; Cronin, F.J.; Kelsay, J.L.; Mertz, W. Number of days of food intake records required to estimate individual and group nutrient intakes with defined confidence. J. Nutr. 1987, 117, 1638–1641. [Google Scholar] [CrossRef] [PubMed]
- Hebert, J.R.; Clemow, L.; Pbert, L.; Ockene, I.S.; Ockene, J.K. Social desirability bias in dietary self-report may compromise the validity of dietary intake measures. Int. J. Epidemiol. 1995, 24, 389–398. [Google Scholar] [CrossRef]
- Fontana, J.M.; Farooq, M.; Sazonov, E. Automatic ingestion monitor: A novel wearable device for monitoring of ingestive behavior. IEEE Trans. Biomed. Eng. 2014, 61, 1772–1779. [Google Scholar] [CrossRef] [PubMed]
- Sun, M.; Burke, L.E.; Mao, Z.H.; Chen, Y.; Chen, H.C.; Bai, Y.; Li, Y.; Li, C.; Jia, W. eButton: A Wearable Computer for Health Monitoring and Personal Assistance. Proc. Des. Autom. Conf. 2014, 2014, 1–6. [Google Scholar]
- Kyritsis, K.; Tatli, C.L.; Diou, C.; Delopoulos, A. Automated analysis of in meal eating behavior using a commercial wristband IMU sensor. In Proceedings of the 2017 39th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC), Seogwipo, Korea, 11–15 July 2017; pp. 2843–2846. [Google Scholar]
- Wansink, B.; Johnson, K.A. The clean plate club: About 92% of self-served food is eaten. Int. J. Obes. 2005, 39, 371–374. [Google Scholar] [CrossRef]
- GHO|By Category|Prevalence of Overweight among Children and Adolescents, BMI>+1 Standard Deviation above the Median, Crude—Estimates by Country, among Children Aged 10–19 Years [Internet]. WHO. Available online: http://apps.who.int/gho/data/view.main.BMIPLUS1C10-19v?lang=en (accessed on 12 March 2019).
- Pendergast, F.J.; Livingstone, K.M.; Worsley, A.; McNaughton, S.A. Correlates of meal skipping in young adults: A systematic review. Int. J. Behav. Nutr. Phys. Act. 2016, 13, 125. [Google Scholar] [CrossRef] [PubMed]
- Williams, R.A.; Roe, L.S.; Rolls, B.J. Assessment of satiety depends on the energy density and portion size of the test meal. Obesity 2014, 22, 318–324. [Google Scholar] [CrossRef]
- Robinson, E.; Nolan, S.; Tudur-Smith, C.; Boyland, E.J.; Harrold, J.A.; Halford, J.C.G. The not so clean plate club: Food self-served won’t always result in food eaten. Int. J. Obes. 2005, 39, 376. [Google Scholar] [CrossRef]
- Rippin, H.L.; Hutchinson, J.; Jewell, J.; Breda, J.J.; Cade, J.E. Portion Size of Energy-Dense Foods among French and UK Adults by BMI Status. Nutrients 2018, 11, 12. [Google Scholar] [CrossRef] [PubMed]
- Kong, F.; Tan, J. DietCam: Regular Shape Food Recognition with a Camera Phone. In Proceedings of the 2011 International Conference on Body Sensor Networks, Dallas, TX, USA, 23–25 May 2011; pp. 127–132. [Google Scholar]
| Total (n = 24) | Male (n = 7) | Female (n = 17) | |
|---|---|---|---|
| Age, y | 16.8 (0.7) | 17.2 (0.5) | 16.6 (0.7) |
| Weight, kg | 62.2 (14.4) | 75.7 (10.1) | 56.6 (12.1) |
| Height, cm | 168.1 (10.3) | 181.3 (6.8) | 162.7 (5.3) |
| BMI, kg/m2 | 21.9 (4.1) | 23.3 (4.7) | 21.3 (3.8) |
| Real-Life m | School Lunch | Diff. ± 95% CI | p-Value | Correlation | |
|---|---|---|---|---|---|
| Food intake weight, g | 327.4 (110.6) | 367.4 (167.2) | −40.0 ± 35 | 0.027 * | 0.92 |
| Eating rate, g/min | 33.5 (14.8) | 27.7 (13.3) | 5.8 ± 4.3 | 0.010 * | 0.75 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Langlet, B.; Fagerberg, P.; Delopoulos, A.; Papapanagiotou, V.; Diou, C.; Maramis, C.; Maglaveras, N.; Anvret, A.; Ioakimidis, I. Predicting Real-Life Eating Behaviours Using Single School Lunches in Adolescents. Nutrients 2019, 11, 672. https://doi.org/10.3390/nu11030672
Langlet B, Fagerberg P, Delopoulos A, Papapanagiotou V, Diou C, Maramis C, Maglaveras N, Anvret A, Ioakimidis I. Predicting Real-Life Eating Behaviours Using Single School Lunches in Adolescents. Nutrients. 2019; 11(3):672. https://doi.org/10.3390/nu11030672
Chicago/Turabian StyleLanglet, Billy, Petter Fagerberg, Anastasios Delopoulos, Vasileios Papapanagiotou, Christos Diou, Christos Maramis, Nikolaos Maglaveras, Anna Anvret, and Ioannis Ioakimidis. 2019. "Predicting Real-Life Eating Behaviours Using Single School Lunches in Adolescents" Nutrients 11, no. 3: 672. https://doi.org/10.3390/nu11030672
APA StyleLanglet, B., Fagerberg, P., Delopoulos, A., Papapanagiotou, V., Diou, C., Maramis, C., Maglaveras, N., Anvret, A., & Ioakimidis, I. (2019). Predicting Real-Life Eating Behaviours Using Single School Lunches in Adolescents. Nutrients, 11(3), 672. https://doi.org/10.3390/nu11030672







