Dietary Diversity Was Positively Associated with Psychological Resilience among Elders: A Population-Based Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Subjects
2.2. Psychological Resilience Assessment
2.3. Assessment of Dietary Diversity
2.4. Covariates
2.5. Statistical Analysis
3. Results
4. Discussion
Author Contributions
Funding
Conflicts of Interest
References
- Nishi, D.; Uehara, R.; Kondo, M.; Matsuoka, Y. Reliability and validity of the Japanese version of the Resilience Scale and its short version. BMC Res. Notes 2010, 3, 310. [Google Scholar] [CrossRef]
- Shi, L.; Sun, J.; Wei, D.; Qiu, J. Recover from the adversity: Functional connectivity basis of psychological resilience. Neuropsychologia 2019, 122, 20–27. [Google Scholar] [CrossRef] [PubMed]
- Fletcher, D.; Sarkar, M. Psychologiacal resilience: A review and critique of definitions, concepts, and theory. Eur. Psychol. 2013, 18, 12–23. [Google Scholar] [CrossRef]
- Smith, P.R. Resilience: Resistance factor for depressive symptom. J. Psychiatr. Ment. Health Nurs. 2009, 16, 829–837. [Google Scholar] [CrossRef] [PubMed]
- Toukhsati, S.R.; Jovanovic, A.; Dehghani, S.; Tran, T.; Tran, A.; Hare, D.L. Low psychological resilience is associated with depression in patients with cardiovascular disease. Eur. J. Cardiovasc. Nurs. 2017, 16, 64–69. [Google Scholar] [CrossRef] [PubMed]
- Smith, J.L.; Hollinger-Smith, L. Savoring resilience and psychological well-being in older adults. Aging Ment. Health 2015, 19, 192–200. [Google Scholar] [CrossRef] [PubMed]
- Jeste, D.V.; Savla, G.N.; Thompson, W.K.; Vahia, I.V.; Glorioso, D.K.; Martin, A.S.; Palmer, B.W.; Rock, D.; Golshan, S.; Kraemer, H.C.; et al. Association between older age and more successful aging: Critical role of resilience and depression. Am. J. Psychiatry 2013, 170, 188–196. [Google Scholar] [CrossRef] [PubMed]
- Gill, T.M.; Robison, J.T.; Tinetti, M.E. Predictors of recovery in activities of daily living among disabled older persons living in the community. J. Gen. Intern. Med. 1997, 12, 757–762. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Wagnild, G. Resilience and successful aging: Comparison among low and high income older adults. J. Gerontol. Nurs. 2003, 29, 42–49. [Google Scholar] [CrossRef] [PubMed]
- Shen, K.; Zeng, Y. The association between resilience and survival among Chinese elderly. In The Handbook of Resilience in Aging: The Key to Successful Aging, 4th ed.; Resnick, B., Roberto, K., Gwyther, L., Eds.; Springer: New York, NY, USA, 2010. [Google Scholar]
- Zeng, Y.; Shen, K. Resilience Significantly Contributes to Exceptional Longevity. Curr. Gerontol. Geriatr. Res. 2010, 2010, 525693. [Google Scholar] [CrossRef]
- Bennett, K.M.; Reyes-Rodriguez, M.F.; Altamar, P.; Soulsby, L.K. Resilience amongst Older Colombians Living in Poverty: An Ecological Approach. J. Cross Cult. Gerontol. 2016, 31, 385–407. [Google Scholar] [CrossRef] [PubMed]
- Dias, R.; Santos, R.L.; Sousa, M.F.; Nogueira, M.M.; Torres, B.; Belfort, T.; Dourado, M.C. Resilience of caregivers of people with dementia: A systematic review of biological and psychosocial determinants. Trends Psychiatry Psychother. 2015, 37, 12–19. [Google Scholar] [CrossRef] [PubMed]
- Contador, I.; Fernández-Calvo, B.; Palenzuela, D.L.; Miguéis, S.; Ramos, F. Prediction of burden in family caregivers of patients with dementia: A perspective of optimism based on generalized expectancies of control. Aging Ment. Health 2012, 16, 675–682. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Lansac, V.; Crespo López, M.; Cáceres, R.; Rodríguez-Poyo, M. Resilience in caregivers of patients with dementia: A preliminary study. Rev. Esp. Geriatr. Gerontol. 2012, 47, 102–109. [Google Scholar] [CrossRef] [PubMed]
- Wu, G.; Feder, A.; Cohen, H.; Kim, J.J.; Calderon, S.; Charney, D.S.; Mathé, A.A. Understanding resilience. Front. Behav. Neurosci. 2013, 7, 10. [Google Scholar] [CrossRef]
- Yoshikawa, E.; Nishi, D.; Matsuoka, Y.J. Association between regular physical exercise and depressive symptoms mediated through social support and resilience in Japanese company workers: A cross-sectional study. BMC Public Health 2016, 16, 553. [Google Scholar] [CrossRef] [PubMed]
- Tiainen, A.M.; Männistö, S.; Lahti, M.; Blomstedt, P.A.; Lahti, J.; Perälä, M.M.; Räikkönen, K.; Kajantie, E.; Eriksson, J.G. Personality and dietary intake -findings in the Helsinki birth cohort study. PLoS ONE 2013, 8, e68284. [Google Scholar] [CrossRef]
- Ward, L.; Pasinetti, G.M. Recommendations for development of botanical polyphenols as “Natural Drugs” for promotion of resilience against stress-induced depression and cognitive impairment. Neuromol. Med. 2016, 18, 487–495. [Google Scholar] [CrossRef]
- Bonaccio, M.; Di Castelnuovo, A.; Costanzo, S.; Pounis, G.; Persichillo, M.; Cerletti, C.; Donati, M.B.; de Gaetano, G.; Iacoviello, L. Mediterranean-type diet is associated with higher psychological resilience in a general adult population: Findings from the Moli-sani study. Eur. J. Clin. Nutr. 2018, 72, 154–160. [Google Scholar] [CrossRef]
- Lutz, L.J.; Gaffney-Stomberg, E.; Williams, K.W.; McGraw, S.M.; Niro, P.J.; Karl, J.P.; Cable, S.J.; Cropper, T.L.; McClung, J.P. Adherence to the Dietary Guidelines for Americans Is Associated with Psychological Resilience in Young Adults: A Cross-Sectional Study. J. Acad. Nutr. Diet. 2017, 117, 396–403. [Google Scholar] [CrossRef]
- Ruel, M.T. Operationalizing Dietary Diversity: A Review of Measurement Issues and Research Priorities. J. Nutr. 2003, 133, 3911S–3926S. [Google Scholar] [CrossRef]
- Oldewage-Theron, W.H.; Kruger, R. Food Variety and Dietary Diversity as Indicators of the Dietary Adequacy and Health Status of an Elderly Population in Sharpeville, South Africa. J. Nutr. Elder. 2008, 27, 101–133. [Google Scholar] [CrossRef]
- Yin, Z.; Fei, Z.; Qiu, C.; Brasher, M.S.; Kraus, V.B.; Zhao, W.; Shi, X.; Zeng, Y. Dietary Diversity and Cognitive Function among Elderly People: A Population-Based Study. J. Nutr. Health Aging 2017, 21, 1089–1094. [Google Scholar] [CrossRef]
- Dawson, S.L.; Dash, S.R.; Jacka, F.N. The importance of diet and gut health to the treatment and prevention of mental disorders. Int. Rev. Neurobiol. 2016, 131, 325–346. [Google Scholar] [CrossRef]
- MacLeod, S.; Musich, S.; Hawkins, K.; Alsgaard, K.; Wicker, E.R. The impact of resilience among older adults. Geriatr. Nurs. 2016, 37, 266–272. [Google Scholar] [CrossRef]
- Zeng, Y. Towards Deeper Research and Better Policy for Healthy Aging—Using the Unique Data of Chinese Longitudinal Healthy Longevity Survey. China Econ. J. 2012, 5, 131–149. [Google Scholar] [CrossRef]
- Connor, K.M.; Davidson, J.R. Development of a new resilience scale: The Connor-Davidson Resilience Scale (CD-RISC). Depress Anxiety 2003, 18, 76–82. [Google Scholar] [CrossRef]
- Kennedy, G.; Ballard, T.; Dop, M.C. Guidelines for Measuring Household and Individual Dietary Diversity; FAO UN: Rome, Italy, 2013. [Google Scholar]
- Yin, Z.; Shi, X.; Kraus, V.B.; Fitzgerald, S.M.; Qian, H.; Xu, J.; Zhai, Y.; Sereny, M.D.; Zeng, Y. High normal plasma triglycerides are associated with preserved cognitive function in Chinese oldest old. Age Ageing 2012, 41, 600–606. [Google Scholar] [CrossRef]
- Katz, S.; Ford, A.B.; Moscowitz, R.W.; JACKSON, B.A.; JAFFE, M.W. The index of ADL: A standardized measure of biological and psychosocial function. JAMA 1963, 185, 914–919. [Google Scholar] [CrossRef]
- Trichopoulou, A.; Bamia, C.; Trichopoulos, D. Anatomy of health effects of Mediterranean diet: Greek EPIC prospective cohort study. BMJ 2009, 338, b2337. [Google Scholar] [CrossRef]
- Mirmiran, P.; Azadbakht, L.; Azizi, F. Dietary diversity within food groups: An indicator of specific nutrient adequacy in Teheranian women. J. Am. Coll. Nutr. 2006, 25, 354–361. [Google Scholar] [CrossRef]
- Ma, L.; Chang, H.; Liu, Y.; Hsieh, H.L.; Lo, L.; Lin, M.; Lu, K. The relationship between health-promoting behaviors and resilience in patients with chronic kidney disease. Sci. World J. 2013, 2013, 124973. [Google Scholar] [CrossRef]
- Narmaki, E.; Siassi, F.; Fariba, K.; Qorbani, M.; Shiraseb, F.; Ataie-Jafari, A.; Sotoudeh, G. Dietary diversity as a proxy measure of blood antioxidant status in women. Nutrition 2015, 31, 722–726. [Google Scholar] [CrossRef]
- Du, J.; Zhu, M.; Bao, H.; Li, B.; Dong, Y.; Xiao, C.; Zhang, G.; Henter, I.; Rudorfer, M.; Vitiello, B. The Role of Nutrients in Protecting Mitochondrial Function and Neurotransmitter Signaling: Implications for the Treatment of Depression, PTSD, and Suicidal Behaviors. Crit. Rev. Food Sci. Nutr. 2016, 56, 2560–2578. [Google Scholar] [CrossRef]
- Perna, L.; Mielck, A.; Lacruz, M.E.; Emeny, R.T.; Holle, R.; Breitfelder, A.; Ladwig, K.H. Socioeconomic position, resilience, and health behaviour among elderly people. Int. J. Public Health 2012, 57, 341–349. [Google Scholar] [CrossRef]
- Dowlati, Y.; Herrmann, N.; Swardfager, W.; Liu, H.; Sham, L.; Reim, E.K.; Lanctôt, K.L. A meta-analysis of cytokines in major depression. Biol. Psychiatry 2010, 67, 446–547. [Google Scholar] [CrossRef]
- Hamarat, E.; Thompson, D.; Steele, D.; Matheny, K.; Simons, C. Age differences in coping resources and satisfaction with life among middle-aged, young-old, and oldest-old adults. J. Genet. Psychol. 2002, 163, 360–367. [Google Scholar] [CrossRef]
- Van Kessel, G. The ability of older people to overcome adversity: A review of the resilience concept. Geriatr. Nurs. 2013, 34, 122–127. [Google Scholar] [CrossRef]
- Carstensen, L.L.; Fung, H.; Charles, S.T. Socioemotional selectivity theory and the regulation of emotion in the second half of life. Motiv. Emot. 2003, 27, 103–123. [Google Scholar] [CrossRef]
| Characteristics | Total Sample | SRS Status | ||
|---|---|---|---|---|
| Low | High | p Value | ||
| No. of subjects | 8158 | 3555 | 4603 | |
| Age (years), mean (SD) | 84.90 (10.84) | 86.76(10.67) | 83.45 (10.75) | <0.001 |
| Female | 4318 (52.93) | 2142 (60.25) | 2176 (47.27) | <0.001 |
| Education years | ||||
| 0 | 4499 (55.15) | 2342 (65.88) | 2157 (46.86) | <0.001 |
| 1–6 | 2652 (32.51) | 944 (26.55) | 1708 (37.11) | |
| >6 | 1007 (12.34) | 269 (7.57) | 738 (16.03) | |
| Married | 3394 (41.60) | 1164 (32.74) | 2230 (48.45) | <0.001 |
| Technical occupation history | 1591 (19.50) | 457 (12.86) | 1134 (24.64) | <0.001 |
| Smoking | 1553 (19.04) | 556 (15.64) | 997 (21.68) | <0.001 |
| Alcohol drinking | 1431 (17.54) | 479 (13.47) | 952(20.68) | <0.001 |
| Physical activities | 4536 (55.60) | 1597 (44.92) | 2939 (63.85) | <0.001 |
| Leisure activities | 6057 (74.25) | 2235 (62.87) | 3822 (83.03) | <0.001 |
| Social activities | 4614 (56.56) | 1763(49.59) | 2851 (61.94) | <0.001 |
| Stroke | 1098 (13.46) | 486 (13.67) | 612 (13.30) | 0.62 |
| Diabetes | 1054 (12.92) | 429 (12.07) | 625 (13.58) | 0.04 |
| ADL disability | 1710 (21.36) | 992 (28.44) | 718 (15.89) | <0.001 |
| Hearing decline | 3467 (42.6)2 | 1754 (49.46) | 1713 (37.34) | <0.001 |
| DDS, mean (SD) | 4.48 (1.99) | 3.95(1.95) | 4.90 (1.93) | <0.001 |
| Poor DD | 4056 (49.72) | 2163(60.84) | 1893(41.13) | <0.001 |
| SRS | 1593 (3.06) | 13.11 (1.87) | 18.11 (1.75) | <0.001 |
| DD | Model 1 | Model 2 | Model 3 |
|---|---|---|---|
| β coefficient (95% CI) of SRS | |||
| Good | 0.00 (reference) | 0.00 (reference) | 0.00 (reference) |
| Poor | −1.08 (−1.21, −0.95) ** | −0.92 (−1.06, −0.79) ** | −0.94 (−1.07, −0.81) ** |
| Odds ratio (95% CI) of low SRS status | |||
| Good | 1.00 (reference) | 1.00 (reference) | 1.00 (reference) |
| Poor | 1.93 (1.76, 2.13) ** | 1.80 (1.63, 1.98) ** | 1.83 (1.66, 2.01) ** |
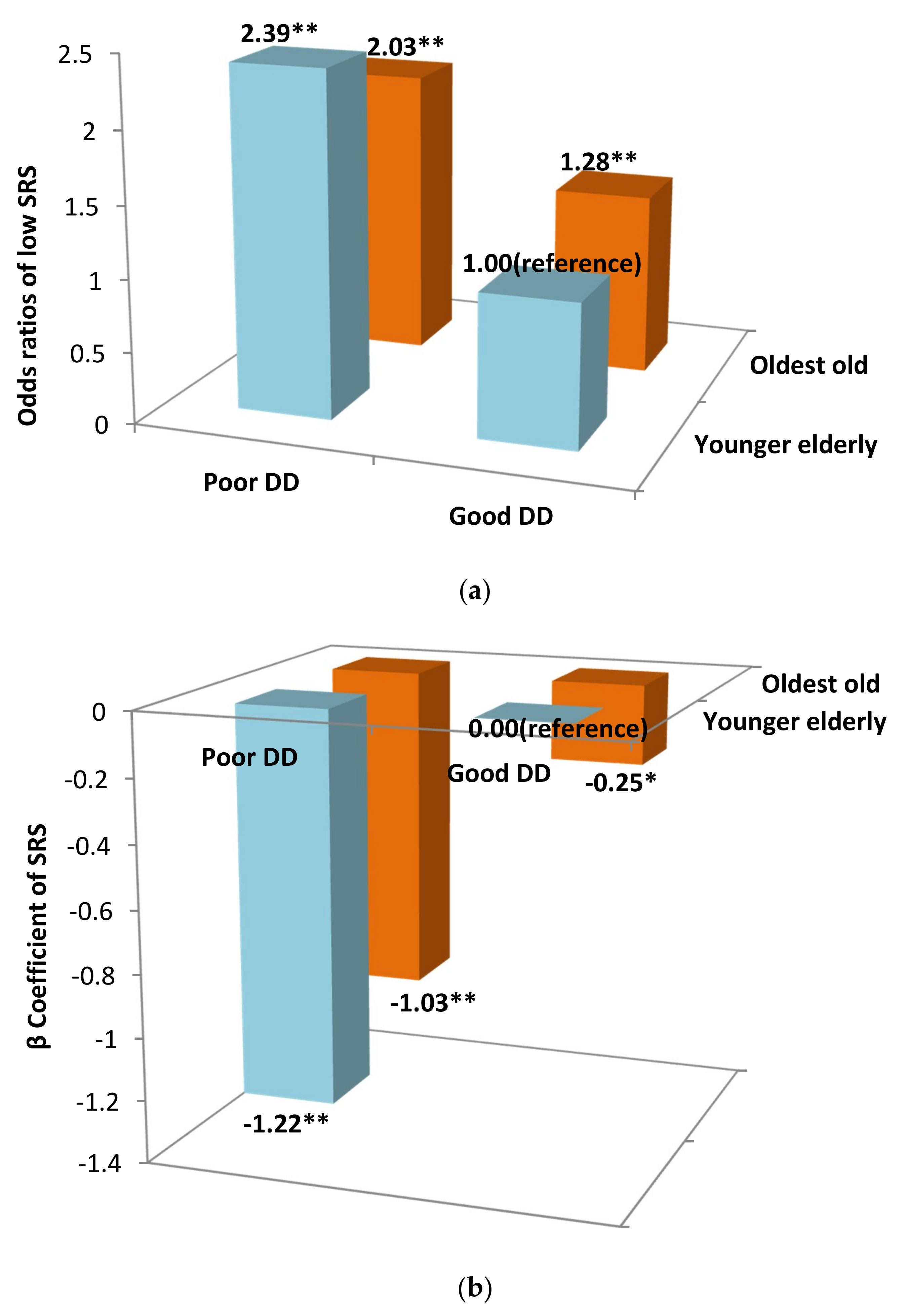
| DD | Younger Elderly (n = 3023) | Oldest Old (n = 5135) | ||
|---|---|---|---|---|
| Model 1 | Full model | Model 1 | Full model | |
| β coefficient (95% CI) of SRS | ||||
| Good | 0.00 (reference) | 0.00 (reference) | 0.00 (reference) | 0.00 (reference) |
| Poor | −1.33 (−1.53, −1.12) ** | −1.14 (−1.35, −0.94) ** | −0.94 (−1.10, −0.77) ** | −0.80 (−0.97, −0.64) ** |
| OR (95% CI) of Low SRS status | ||||
| Good | 1.00 (reference) | 1.00 (reference) | 1.00 (reference) | 1.00 (reference) |
| Poor | 2.48 (2.11, 2.92) ** | 2.32 (1.96, 2.74) ** | 1.71 (1.52, 1.92) ** | 1.61 (1.43, 1.82) ** |
| Food Group | β Coefficients (95% CI) | p Value |
|---|---|---|
| Meat | 0.418 (0.267, 0.570) | <0.001 |
| Bean | 0.263 (0.129, 0.397) | 0.001 |
| Vegetables | 0.489 (0.287, 0.692) | <0.001 |
| Fruits | 0.558 (0.420, 0.695) | <0.001 |
| Egg | 0.304 (0.157, 0.451) | <0.001 |
| Fish | 0.039 (−0.097, 0.175) | 0.574 |
| Milk | 0.298 (0.154, 0.442) | <0.001 |
| Tea | 0.058 (−0.081, 0.198) | 0.412 |
| Nut | 0.302 (0.109, 0.495) | 0.002 |
| DDS | β Coefficients (95% CI) | Change |
|---|---|---|
| Total DDS | 0.290 (0.257, 0.323) | |
| Minus meat | 0.275 (0.242, 0.308) | −5.17% |
| Minus beans | 0.288 (0.255, 0.322) | −0.70% |
| Minus vegetables | 0.263 (0.231, 0.294) | −9.31% |
| Minus fruits | 0.264 (0.230, 0.297) | −8.97% |
| Minus egg | 0.285 (0.252, 0.319) | −1.72% |
| Minus fish | 0.302 (0.269, 0.336) | 4.13% |
| Minus milk | 0.277 (0.244, 0.310) | −4.48% |
| Minus tea | 0.286 (0.255, 0.319) | −1.38% |
| Minus nuts | 0.270 (0.239, 0.302) | −6.90% |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yin, Z.; Brasher, M.S.; Kraus, V.B.; Lv, Y.; Shi, X.; Zeng, Y. Dietary Diversity Was Positively Associated with Psychological Resilience among Elders: A Population-Based Study. Nutrients 2019, 11, 650. https://doi.org/10.3390/nu11030650
Yin Z, Brasher MS, Kraus VB, Lv Y, Shi X, Zeng Y. Dietary Diversity Was Positively Associated with Psychological Resilience among Elders: A Population-Based Study. Nutrients. 2019; 11(3):650. https://doi.org/10.3390/nu11030650
Chicago/Turabian StyleYin, Zhaoxue, Melanie Sereny Brasher, Virginia B. Kraus, Yuebin Lv, Xiaoming Shi, and Yi Zeng. 2019. "Dietary Diversity Was Positively Associated with Psychological Resilience among Elders: A Population-Based Study" Nutrients 11, no. 3: 650. https://doi.org/10.3390/nu11030650
APA StyleYin, Z., Brasher, M. S., Kraus, V. B., Lv, Y., Shi, X., & Zeng, Y. (2019). Dietary Diversity Was Positively Associated with Psychological Resilience among Elders: A Population-Based Study. Nutrients, 11(3), 650. https://doi.org/10.3390/nu11030650




