Effects of Fresh Watermelon Consumption on the Acute Satiety Response and Cardiometabolic Risk Factors in Overweight and Obese Adults
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Study Design
2.3. Satiety Questionnaire
2.4. Dietary Assessment and Physical Activity
2.5. Postprandial Glucose and Insulin Response
2.6. Appetite-Regulating Hormones
2.7. C-Reactive Protein
2.8. Serum Lipids
2.9. Thiobarbituric Acid Reactive Substances (TBARS)
2.10. Catalase Activity
2.11. Total Antioxidant Capacity
2.12. Liver Function Markers
2.13. Statistical Analysis
3. Results
3.1. Body Weight and Blood Pressure
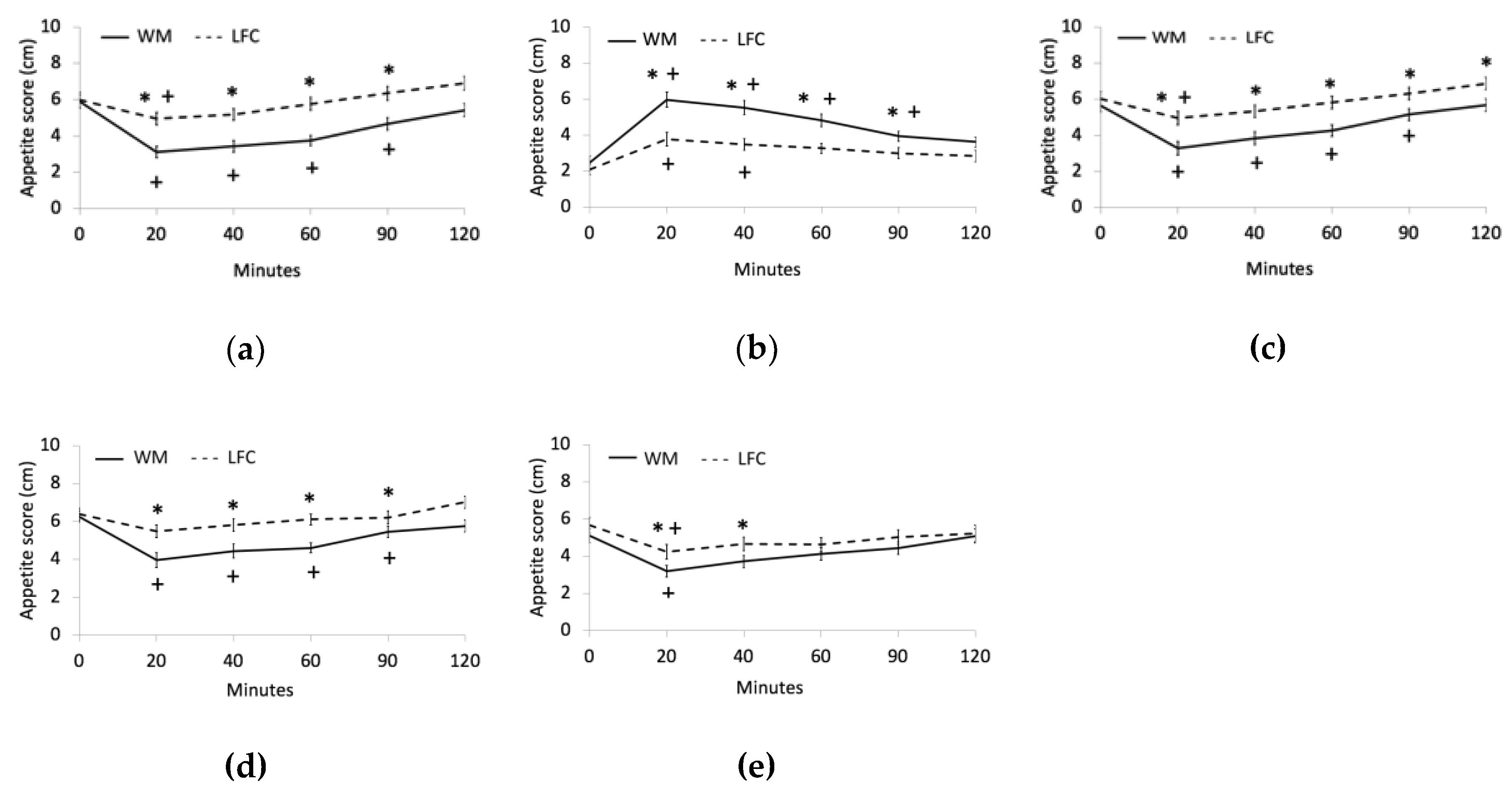
3.2. Appetite
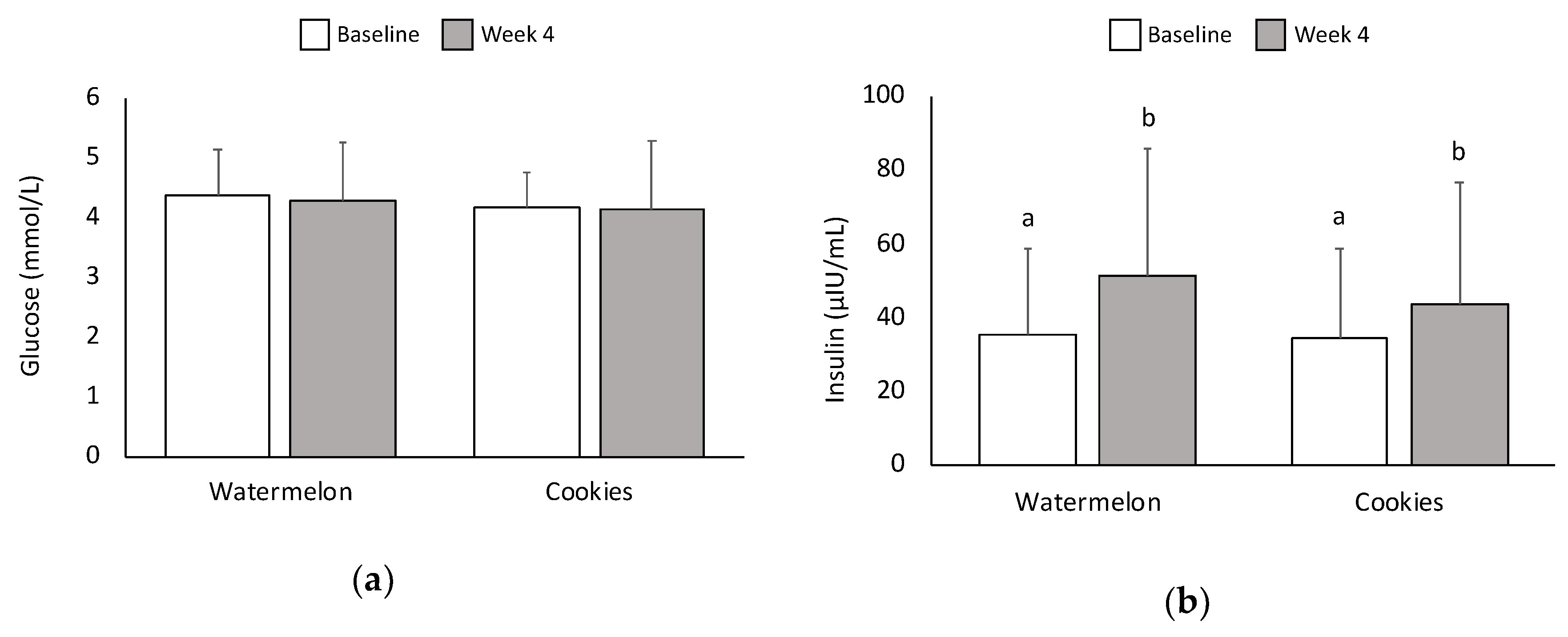
3.3. Glucose and Insulin
3.4. Appetite-Regulating Hormones
3.5. C-Reactive Protein
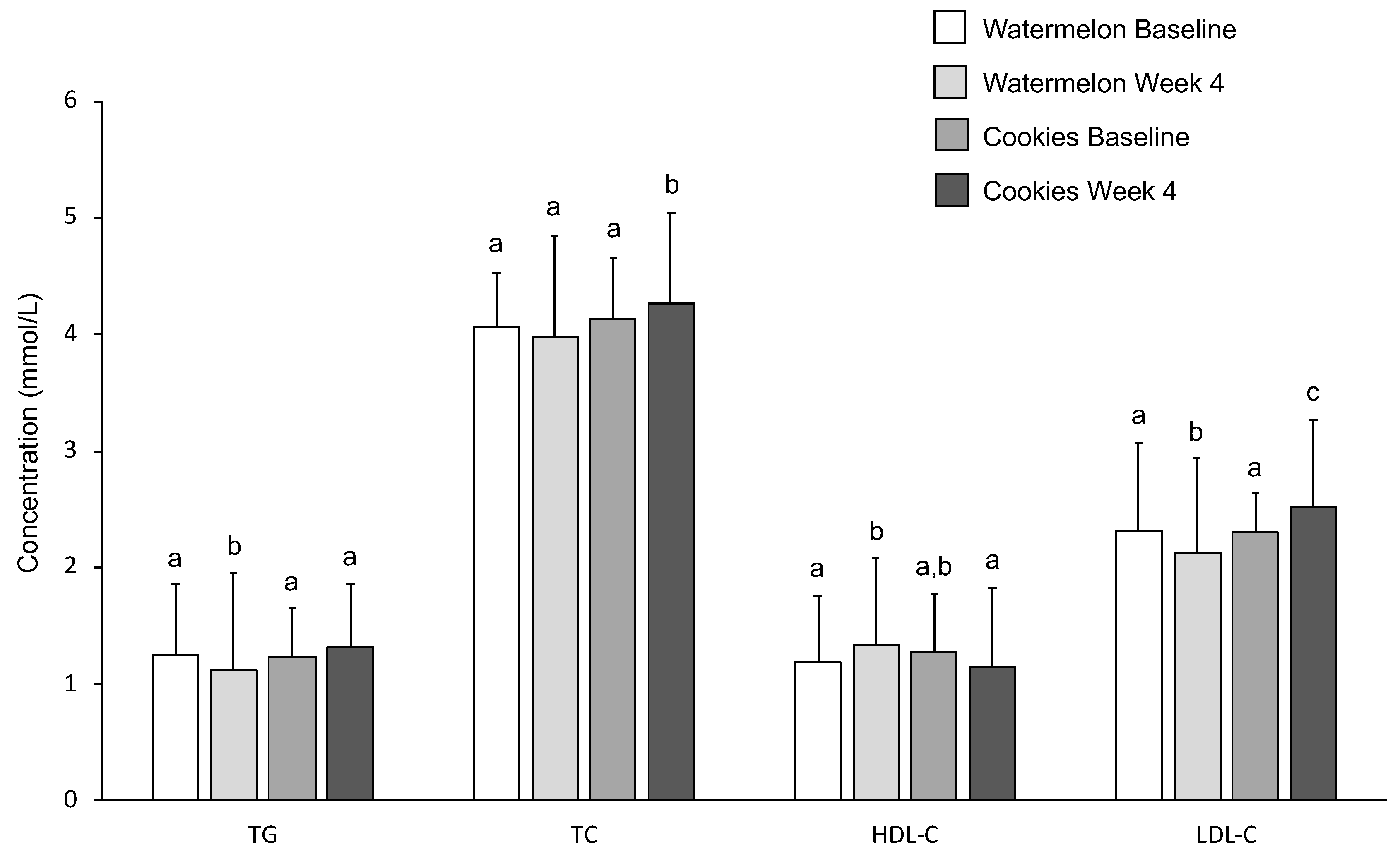
3.6. Serum Lipids
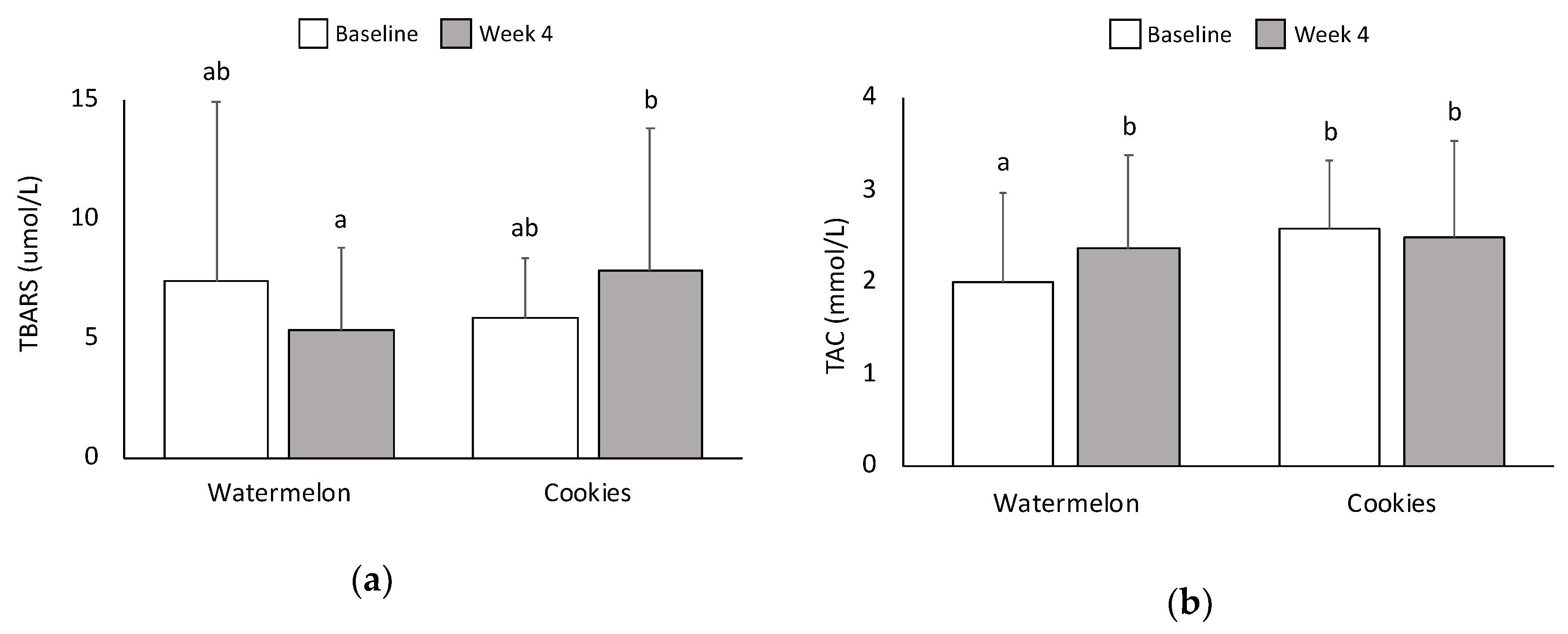
3.7. Oxidative Stress and Antioxidant Capacity
3.8. Liver Function Markers
4. Discussion and Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Hales, C.; Carroll, M.D.; Fryar, C.D.; Ogden, C.L. Prevalence of Obesity among Adults and Youth: United States, 2015–2016. Available online: https://www.cdc.gov/nchs/products/databriefs/db288.htm (accessed on 26 November 2018).
- Must, A.; McKeown, N.M. The Disease Burden Associated with Overweight and Obesity. In Endotext; De Groot, L.J., Chrousos, G., Dungan, K., Feingold, K.R., Grossman, A., Hershman, J.M., Koch, C., Korbonits, M., McLachlan, R., et al., Eds.; MDText.com, Inc.: South Dartmouth, MA, USA, 2000. [Google Scholar]
- Wing, R.R.; Phelan, S. Long-term weight loss maintenance. Am. J. Clin. Nutr. 2005, 82, 222S–225S. [Google Scholar] [CrossRef] [PubMed]
- Padwal, R.S.; Majumdar, S.R. Drug treatments for obesity: Orlistat, sibutramine, and rimonabant. Lancet 2007, 369, 71–77. [Google Scholar] [CrossRef]
- Piernas, C.; Popkin, B.M. Snacking increased among U.S. adults between 1977 and 2006. J. Nutr. 2010, 140, 325–332. [Google Scholar] [CrossRef] [PubMed]
- Boeing, H.; Bechthold, A.; Bub, A.; Ellinger, S.; Haller, D.; Kroke, A.; Leschik-Bonnet, E.; Müller, M.J.; Oberritter, H.; Schulze, M.; et al. Critical review: Vegetables and fruit in the prevention of chronic diseases. Eur. J. Nutr. 2012, 51, 637–663. [Google Scholar] [CrossRef]
- Rolls, B.J. The relationship between dietary energy density and energy intake. Physiol. Behav. 2009, 97, 609–615. [Google Scholar] [CrossRef]
- Furchner-Evanson, A.; Petrisko, Y.; Howarth, L.; Nemoseck, T.; Kern, M. Type of snack influences satiety responses in adult women. Appetite 2010, 54, 564–569. [Google Scholar] [CrossRef]
- Patel, B.P.; Bellissimo, N.; Luhovyy, B.; Bennett, L.J.; Hurton, E.; Painter, J.E.; Anderson, G.H. An after-school snack of raisins lowers cumulative food intake in young children. J. Food Sci. 2013, 78 (Suppl. 1), A5–A10. [Google Scholar] [CrossRef]
- Farajian, P.; Katsagani, M.; Zampelas, A. Short-term effects of a snack including dried prunes on energy intake and satiety in normal-weight individuals. Eat. Behav. 2010, 11, 201–203. [Google Scholar] [CrossRef]
- Bays, H.; Weiter, K.; Anderson, J. A randomized study of raisins versus alternative snacks on glycemic control and other cardiovascular risk factors in patients with type 2 diabetes mellitus. Phys. Sportsmed. 2015, 43, 37–43. [Google Scholar] [CrossRef]
- Nutrient Database Laboratory (U.S.) and Consumer and Food Economics Institute (U.S.). USDA Nutrient Database for Standard Reference. Available online: https://ndb.nal.usda.gov/ndb/ (accessed on 26 November 2018).
- Barnes, T.L.; French, S.A.; Harnack, L.J.; Mitchell, N.R.; Wolfson, J. Snacking behaviors, diet quality, and body mass index in a community sample of working adults. J. Acad. Nutr. Diet. 2015, 115, 1117–1123. [Google Scholar] [CrossRef]
- U.S. Department of Health and Human Services and U.S. Department of Agriculture. 2015–2020 Dietary Guidelines for Americans, 8th ed.; 2015. Available online: https://health.gov/dietaryguidelines/2015/resources/2015-2020_dietary_guidelines.pdf (accessed on 26 November 2018).
- Perkins-Veazie, P.; Davis, A.; Collins, J.K. Watermelon: From dessert to functional food. Isr. J. Plant Sci. 2012, 60, 395–402. [Google Scholar]
- Story, E.N.; Kopec, R.E.; Schwartz, S.J.; Harris, G.K. An Update on the Health Effects of Tomato Lycopene. Annu. Rev. Food Sci. Technol. 2010, 1, 189–210. [Google Scholar] [CrossRef]
- Figueroa, A.; Wong, A.; Jaime, S.J.; Gonzales, J.U. Influence of l-citrulline and watermelon supplementation on vascular function and exercise performance. Curr. Opin. Clin. Nutr. Metab. Care 2017, 20, 92–98. [Google Scholar] [CrossRef] [PubMed]
- Poduri, A.; Rateri, D.L.; Saha, S.K.; Saha, S.; Daugherty, A. Citrullus lanatus “sentinel” (watermelon) extract reduces atherosclerosis in LDL receptor-deficient mice. J. Nutr. Biochem. 2013, 24, 882–886. [Google Scholar] [CrossRef] [PubMed]
- Hong, M.Y.; Hartig, N.; Kaufman, K.; Hooshmand, S.; Figueroa, A.; Kern, M. Watermelon consumption improves inflammation and antioxidant capacity in rats fed an atherogenic diet. Nutr. Res. 2015, 35, 251–258. [Google Scholar] [CrossRef] [PubMed]
- Hong, M.Y.; Beidler, J.; Hooshmand, S.; Figueroa, A.; Kern, M. Watermelon and l-arginine consumption improve serum lipid profile and reduce inflammation and oxidative stress by altering gene expression in rats fed an atherogenic diet. Nutr. Res. 2018, 58, 46–54. [Google Scholar] [CrossRef] [PubMed]
- Massa, N.M.; Silva, A.S.; de Oliveira, C.V.; Costa, M.J.; Persuhn, D.C.; Barbosa, C.V.; Gonçalves, M.D. Supplementation with Watermelon Extract Reduces Total Cholesterol and LDL Cholesterol in Adults with Dyslipidemia under the Influence of the MTHFR C677T Polymorphism. J. Am. Coll. Nutr. 2016, 35, 514–520. [Google Scholar] [CrossRef] [PubMed]
- Figueroa, A.; Sanchez-Gonzalez, M.A.; Wong, A.; Arjmandi, B.H. Watermelon extract supplementation reduces ankle blood pressure and carotid augmentation index in obese adults with prehypertension or hypertension. Am. J. Hypertens. 2012, 25, 640–643. [Google Scholar] [CrossRef]
- Flint, A.; Raben, A.; Blundell, J.E.; Astrup, A. Reproducibility, power and validity of visual analogue scales in assessment of appetite sensations in single test meal studies. Int. J. Obes. Relat. Metab. Disord. J. Int. Assoc. Study Obes. 2000, 24, 38–48. [Google Scholar] [CrossRef]
- Sallis, J.F.; Haskell, W.L.; Wood, P.D.; Fortmann, S.P.; Rogers, T.; Blair, S.N.; Paffenbarger, R.S., Jr. Physical activity assessment methodology in the Five-City Project. Am. J. Epidemiol. 1985, 121, 91–106. [Google Scholar] [CrossRef]
- Friedewald, W.T.; Levy, R.I.; Fredrickson, D.S. Estimation of the concentration of low-density lipoprotein cholesterol in plasma, without use of the preparative ultracentrifuge. Clin. Chem. 1972, 18, 499–502. [Google Scholar]
- Kaliora, A.C.; Kokkinos, A.; Diolintzi, A.; Stoupaki, M.; Gioxari, A.; Kanellos, P.T.; Dedoussis, G.V.Z.; Vlachogiannakos, J.; Revenas, C.; Ladas, S.D.; et al. The effect of minimal dietary changes with raisins in NAFLD patients with non-significant fibrosis: A randomized controlled intervention. Food Funct. 2016, 7, 4533–4544. [Google Scholar] [CrossRef]
- Kanellos, P.T.; Kaliora, A.C.; Tentolouris, N.K.; Argiana, V.; Perrea, D.; Kalogeropoulos, N.; Kountouri, A.M.; Karathanos, V.T. A pilot, randomized controlled trial to examine the health outcomes of raisin consumption in patients with diabetes. Nutrition 2014, 30, 358–364. [Google Scholar] [CrossRef]
- Wu, G.; Collins, J.K.; Perkins-Veazie, P.; Siddiq, M.; Dolan, K.D.; Kelly, K.A.; Heaps, C.L.; Meininger, C.J. Dietary supplementation with watermelon pomace juice enhances arginine availability and ameliorates the metabolic syndrome in Zucker diabetic fatty rats. J. Nutr. 2007, 137, 2680–2685. [Google Scholar] [CrossRef] [PubMed]
- Figueroa, A.; Sanchez-Gonzalez, M.A.; Perkins-Veazie, P.M.; Arjmandi, B.H. Effects of watermelon supplementation on aortic blood pressure and wave reflection in individuals with prehypertension: A pilot study. Am. J. Hypertens. 2011, 24, 40–44. [Google Scholar] [CrossRef] [PubMed]
- Figueroa, A.; Wong, A.; Hooshmand, S.; Sanchez-Gonzalez, M.A. Effects of watermelon supplementation on arterial stiffness and wave reflection amplitude in postmenopausal women. Menopause 2013, 20, 573–577. [Google Scholar] [CrossRef]
- Figueroa, A.; Wong, A.; Kalfon, R. Effects of Watermelon Supplementation on Aortic Hemodynamic Responses to the Cold Pressor Test in Obese Hypertensive Adults. Am. J. Hypertens. 2013, 27, 899–906. [Google Scholar] [CrossRef]
- Shanely, R.A.; Nieman, D.C.; Perkins-Veazie, P.; Henson, D.A.; Meaney, M.P.; Knab, A.M.; Cialdell-Kam, L. Comparison of Watermelon and Carbohydrate Beverage on Exercise-Induced Alterations in Systemic Inflammation, Immune Dysfunction, and Plasma Antioxidant Capacity. Nutrients 2016, 8, 518. [Google Scholar] [CrossRef]
- Flood-Obbagy, J.E.; Rolls, B.J. The effect of fruit in different forms on energy intake and satiety at a meal. Appetite 2009, 52, 416–422. [Google Scholar] [CrossRef]
- Burton-Freeman, B. Dietary fiber and energy regulation. J. Nutr. 2000, 130, 272S–275S. [Google Scholar] [CrossRef]
- Howarth, L.; Petrisko, Y.; Furchner-Evanson, A.; Nemoseck, T.; Kern, M. Snack selection influences nutrient intake, triglycerides, and bowel habits of adult women: A pilot study. J. Am. Diet. Assoc. 2010, 110, 1322–1327. [Google Scholar] [CrossRef] [PubMed]
- Żurakowski, A.; Zahorska-Markiewicz, B.; Olszanecka-Glinianowicz, M.; Kocelak, P. Effect of Meal Volume on Hunger and Satiety in Obese Subjects: Volume of meal and satiety. EJIFCC 2006, 17, 167–176. [Google Scholar] [PubMed]
- Rolls, B.J.; Bell, E.A.; Waugh, B.A. Increasing the volume of a food by incorporating air affects satiety in men. Am. J. Clin. Nutr. 2000, 72, 361–368. [Google Scholar] [CrossRef] [PubMed]
- Sørensen, L.B.; Møller, P.; Flint, A.; Martens, M.; Raben, A. Effect of sensory perception of foods on appetite and food intake: A review of studies on humans. Int. J. Obes. 2003, 27, 1152–1166. [Google Scholar] [CrossRef] [PubMed]
- Esfahani, A.; Lam, J.; Kendall, C.W.C. Acute effects of raisin consumption on glucose and insulin reponses in healthy individuals. J. Nutr. Sci. 2014, 3, e1. [Google Scholar] [CrossRef] [PubMed]
- Sansone, K.; Kern, M.; Hong, M.Y.; Liu, C.; Hooshmand, S. Acute Effects of Dried Apple Consumption on Metabolic and Cognitive Responses in Healthy Individuals. J. Med. Food 2018, 21, 1158–1164. [Google Scholar] [CrossRef]
- Byrne, H.K.; Kim, Y.; Hertzler, S.R.; Watt, C.A.; Mattern, C.O. Glycemic and insulinemic responses to different preexercise snacks in participants with impaired fasting glucose. Int. J. Sport Nutr. Exerc. Metab. 2011, 21, 1–10. [Google Scholar] [CrossRef]
- Nilsson, A.C.; Ostman, E.M.; Holst, J.J.; Björck, I.M.E. Including indigestible carbohydrates in the evening meal of healthy subjects improves glucose tolerance, lowers inflammatory markers, and increases satiety after a subsequent standardized breakfast. J. Nutr. 2008, 138, 732–739. [Google Scholar] [CrossRef]
- Moore, M.C.; Cherrington, A.D.; Mann, S.L.; Davis, S.N. Acute fructose administration decreases the glycemic response to an oral glucose tolerance test in normal adults. J. Clin. Endocrinol. Metab. 2000, 85, 4515–4519. [Google Scholar] [CrossRef]
- Moore, M.C.; Davis, S.N.; Mann, S.L.; Cherrington, A.D. Acute fructose administration improves oral glucose tolerance in adults with type 2 diabetes. Diabetes Care 2001, 24, 1882–1887. [Google Scholar] [CrossRef]
- Sievenpiper, J.L.; Chiavaroli, L.; de Souza, R.J.; Mirrahimi, A.; Cozma, A.I.; Ha, V.; Wang, D.D.; Yu, M.E.; Carleton, A.J.; Beyene, J.; et al. ‘Catalytic’ doses of fructose may benefit glycaemic control without harming cardiometabolic risk factors: A small meta-analysis of randomised controlled feeding trials. Br. J. Nutr. 2012, 108, 418–423. [Google Scholar] [CrossRef]
- Jazet, I.M.; Pijl, H.; Meinders, A.E. Adipose tissue as an endocrine organ: Impact on insulin resistance. Neth. J. Med. 2003, 61, 194–212. [Google Scholar]
- English, P.J.; Coughlin, S.R.; Hayden, K.; Malik, I.A.; Wilding, J.P.H. Plasma adiponectin increases postprandially in obese, but not in lean, subjects. Obes. Res. 2003, 11, 839–844. [Google Scholar] [CrossRef]
- Massa, N.M.L.; Silva, A.S.; Toscano, L.T.; Silva, J.D.R.; Persuhn, D.C.; Gonçalves, M.D.C.R. Watermelon extract reduces blood pressure but does not change sympathovagal balance in prehypertensive and hypertensive subjects. Blood 2016, 25, 244–248. [Google Scholar]
- Kuhn, K.; Harris, P.; Cunningham, G.; Robbins, I.; Summar, M.; Christman, B. Oral citrulline effectively elevates plasma arginine levels for 24 h in normal volunteers. Circ. AHA Sci. Sess. 2006, II, 1339. [Google Scholar]
- Kim, C.-H.; Park, M.-K.; Kim, S.-K.; Cho, Y.-H. Antioxidant capacity and anti-inflammatory activity of lycopene in watermelon. Int. J. Food Sci. Technol. 2014, 49, 2083–2091. [Google Scholar] [CrossRef]
- Edwards, A.J.; Vinyard, B.T.; Wiley, E.R.; Brown, E.D.; Collins, J.K.; Perkins-Veazie, P.; Baker, R.A.; Clevidence, B.A. Consumption of watermelon juice increases plasma concentrations of lycopene and beta-carotene in humans. J. Nutr. 2003, 133, 1043–1050. [Google Scholar] [CrossRef]
- Wink, D.A.; Miranda, K.M.; Espey, M.G.; Pluta, R.M.; Hewett, S.J.; Colton, C.; Vitek, M.; Feelisch, M.; Grisham, M.B. Mechanisms of the Antioxidant Effects of Nitric Oxide. Antioxid. Redox Signal. 2001, 3, 203–213. [Google Scholar] [CrossRef]
| Measurements | Watermelon (n = 33) | Cookies (n = 33) | ||
|---|---|---|---|---|
| Baseline | Week 4 | Baseline | Week 4 | |
| Body Weight (kg) | 89.4 ± 15 a | 88.9 ± 16 b | 89.3 ± 16 a | 89.9 ± 16 c |
| BMI | 30.5 ± 3.5 a | 30.4 ± 3.7 b | 30.5 ± 3.7 a | 30.7 ± 3.8 c |
| SBP (mm Hg) | 127 ± 15 a,b | 125 ± 14 a | 124 ± 14 a | 129 ± 14 b |
| DBP (mm Hg) | 79.9 ± 7.2 a | 79.6 ± 9.7 a | 77.2 ± 9.0b | 81.2 ± 10 a |
| W/H ratio | 0.850 ± 0.06 a,b | 0.845 ± 0.07 a | 0.847 ± 0.07 a,b | 0.857 ± 0.07 b |
| Body Fat (%) | 37.8 ± 8.2 a | 38.0 ± 8.5 a | 37.7 ± 8.2 a | 38.2 ± 7.9 b |
| Watermelon (n = 33) | Cookies (n = 33) | |||
|---|---|---|---|---|
| Pre | Post | Pre | Post | |
| Leptin (ng/mL) | 3.65 ± 2.02 a | 3.28 ± 2.01 b | 3.71 ± 1.96 a | 3.42 ± 2.07 a,b |
| Ghrelin (pg/mL) | 414 ± 246 a | 520 ± 356 b | 424 ± 250 a | 511 ± 352 a,b |
| Adiponectin (μg/mL) | 9.93 ± 5.81 | 10.66 ± 5.08 | 9.20 ± 5.63 | 8.41 ± 6.33 |
| CCK (pg/mL) | 465 ± 242 | 514 ± 170 | 495 ± 178 | 542 ± 207 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lum, T.; Connolly, M.; Marx, A.; Beidler, J.; Hooshmand, S.; Kern, M.; Liu, C.; Hong, M.Y. Effects of Fresh Watermelon Consumption on the Acute Satiety Response and Cardiometabolic Risk Factors in Overweight and Obese Adults. Nutrients 2019, 11, 595. https://doi.org/10.3390/nu11030595
Lum T, Connolly M, Marx A, Beidler J, Hooshmand S, Kern M, Liu C, Hong MY. Effects of Fresh Watermelon Consumption on the Acute Satiety Response and Cardiometabolic Risk Factors in Overweight and Obese Adults. Nutrients. 2019; 11(3):595. https://doi.org/10.3390/nu11030595
Chicago/Turabian StyleLum, Tiffany, Megan Connolly, Amanda Marx, Joshua Beidler, Shirin Hooshmand, Mark Kern, Changqi Liu, and Mee Young Hong. 2019. "Effects of Fresh Watermelon Consumption on the Acute Satiety Response and Cardiometabolic Risk Factors in Overweight and Obese Adults" Nutrients 11, no. 3: 595. https://doi.org/10.3390/nu11030595
APA StyleLum, T., Connolly, M., Marx, A., Beidler, J., Hooshmand, S., Kern, M., Liu, C., & Hong, M. Y. (2019). Effects of Fresh Watermelon Consumption on the Acute Satiety Response and Cardiometabolic Risk Factors in Overweight and Obese Adults. Nutrients, 11(3), 595. https://doi.org/10.3390/nu11030595






