An Oral Glucose Load Decreases Postprandial Extracellular Vesicles in Obese Adults with and without Prediabetes
Abstract
:1. Introduction
2. Materials and Methods
2.1. Subjects
2.2. Metabolic Control
2.3. Body Composition and Aerobic Fitness
2.4. Oral Glucose Tolerance Test (OGTT)
2.5. Arterial Stiffness
2.6. EV Preparation and Characterization
2.7. Clinical Biochemical Analysis
2.8. Statistical Analysis
3. Results
3.1. Subject Characteristics
3.2. Glucose Tolerance
3.3. Insulin Sensitivity and FFA
3.4. Arterial Stiffness, Blood Pressure, and Inflammation
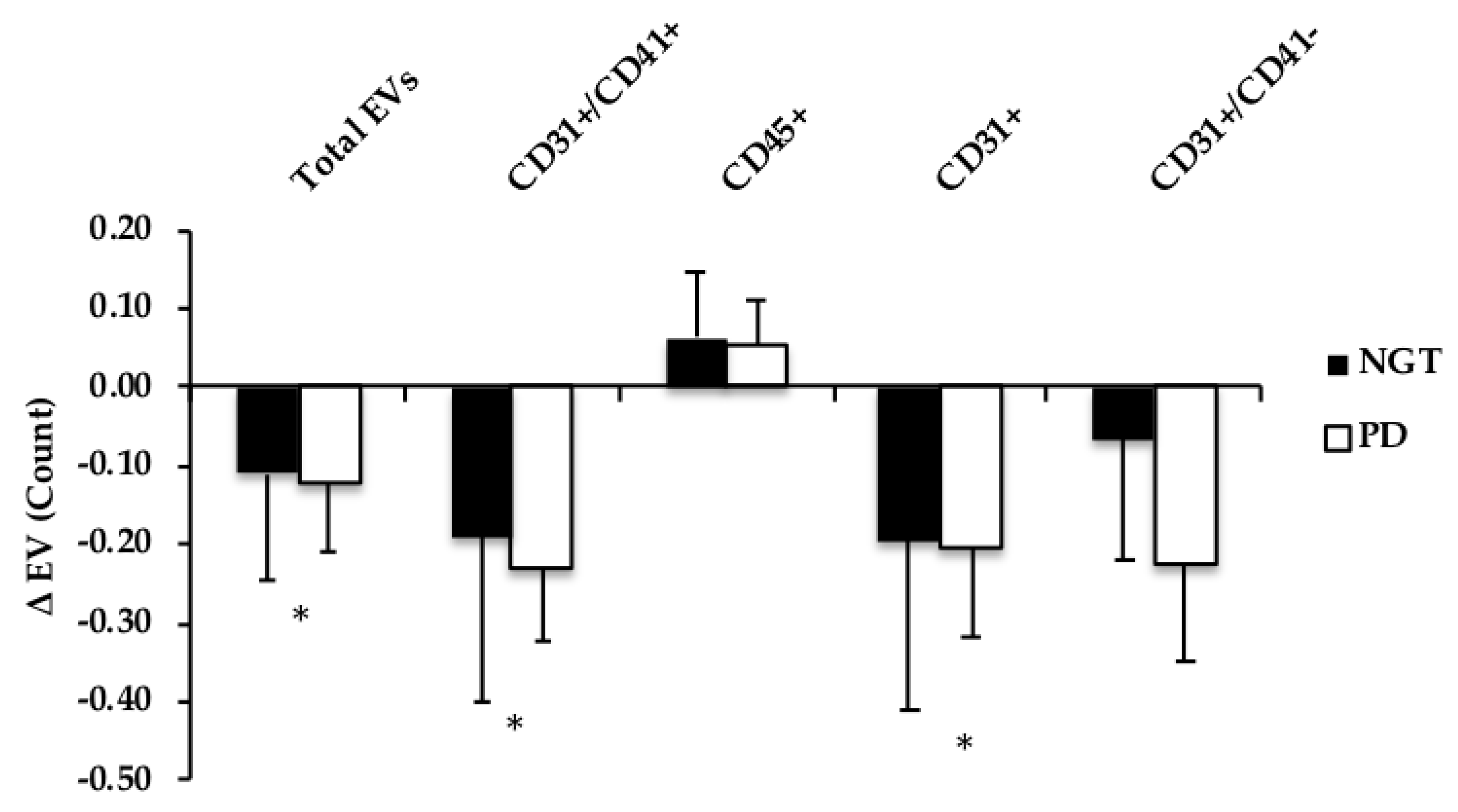
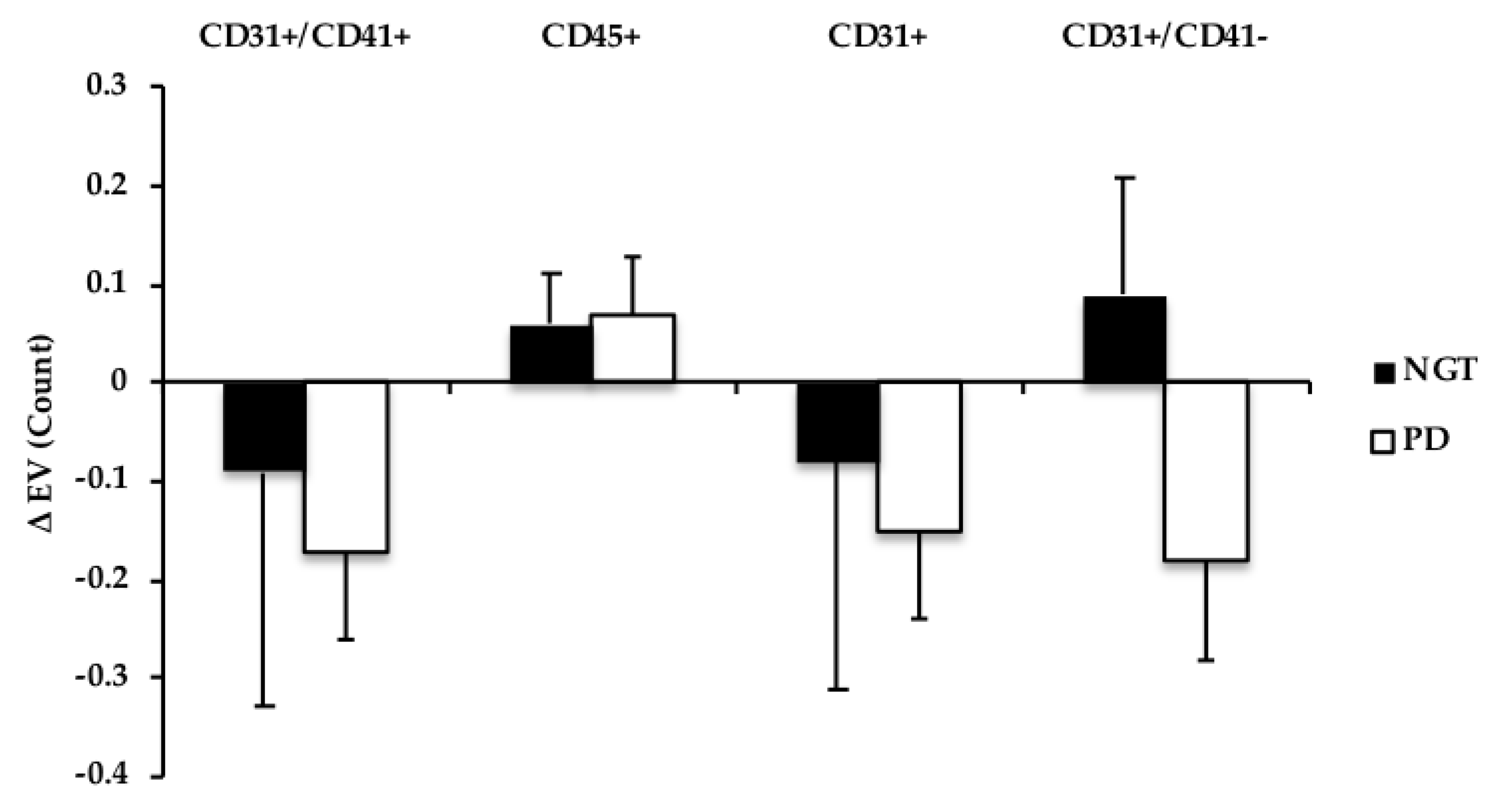
3.5. Extracellular Vesicles
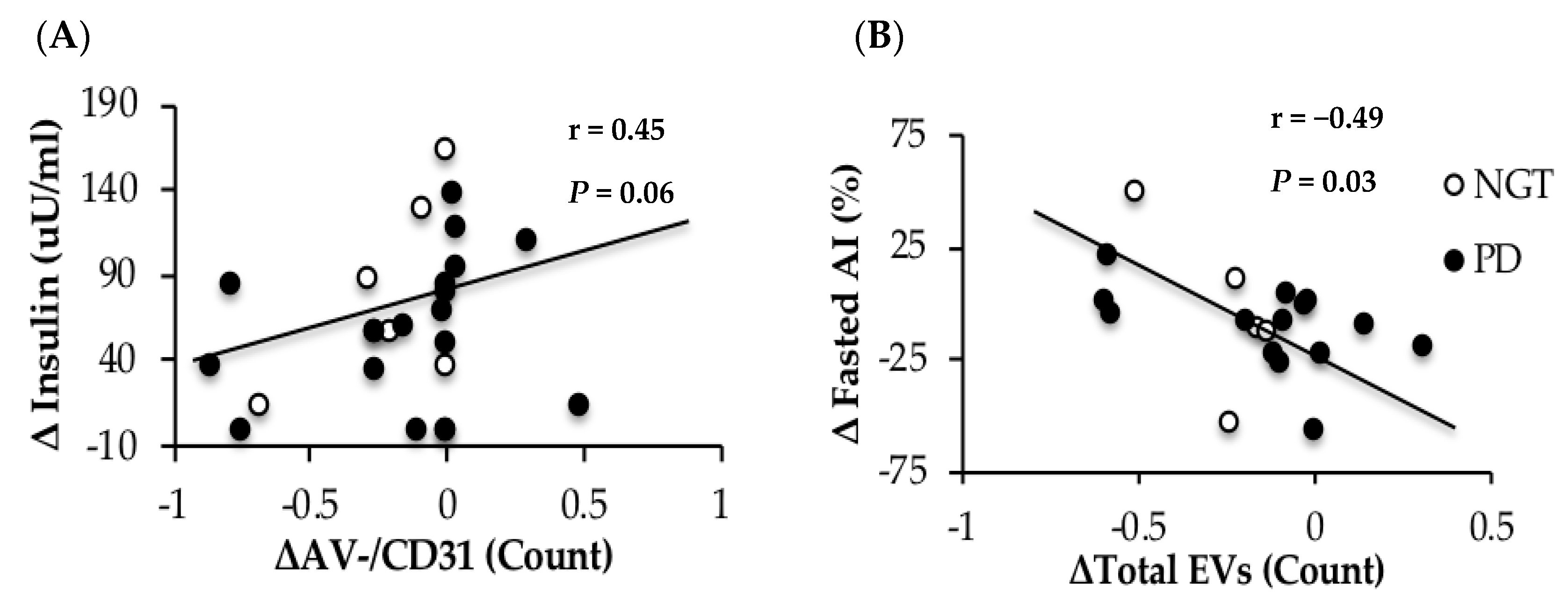
3.6. Correlations
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ogurtsova, K.; da Rocha Fernandes, J.; Huang, Y.; Linnenkamp, U.; Guariguata, L.; Cho, N.; Cavan, D.; Shaw, J.; Makaroff, L. IDF diabetes atlas: Global estimates for the prevalence of diabetes for 2015 and 2040. Diabetes Res. Clin. Pract. 2017, 128, 40–50. [Google Scholar] [CrossRef] [PubMed]
- Ford, E.S.; Zhao, G.; Li, C. Pre-aiabetes and the risk for cardiovascular disease: A systematic review of the evidence. J. Am. Coll. Cardiol. 2010, 55, 1310–1317. [Google Scholar] [CrossRef] [PubMed]
- Bonora, E.; Muggeo, M. Postprandial blood glucose as a risk factor for cardiovascular disease in type ii diabetes: The epidemiological evidence. Diabetologia 2001, 44, 2107–2114. [Google Scholar] [CrossRef] [PubMed]
- Cavalot, F.; Petrelli, A.; Traversa, M.; Bonomo, K.; Fiora, E.; Conti, M.; Anfossi, G.; Costa, G.; Trovati, M. Postprandial blood glucose is a stronger predictor of cardiovascular events than fasting blood glucose in type 2 diabetes mellitus, particularly in women: Lessons from the san luigi gonzaga diabetes study. J. Clin. Endocrinol. Metab. 2006, 91, 813–819. [Google Scholar] [CrossRef] [PubMed]
- Nomura, S. Dynamic role of microparticles in type 2 diabetes mellitus. Curr. Diabetes Rev. 2009, 5, 245–251. [Google Scholar] [CrossRef]
- Dignat-George, F.; Boulanger, C.M. The many faces of endothelial microparticles. Arterioscler. Thromb. Vasc. Biol. 2011, 31, 27–33. [Google Scholar] [CrossRef]
- Nozaki, T.; Sugiyama, S.; Koga, H.; Sugamura, K.; Ohba, K.; Matsuzawa, Y.; Sumida, H.; Matsui, K.; Jinnouchi, H.; Ogawa, H. Significance of a multiple biomarkers strategy including endothelial dysfunction to improve risk stratification for cardiovascular events in patients at high risk for coronary heart disease. J. Am. Coll. Cardiol. 2009, 54, 601–608. [Google Scholar] [CrossRef]
- Giannella, A.; Radu, C.M.; Franco, L.; Campello, E.; Simioni, P.; Avogaro, A.; Kreutzenberg, S.V.; Ceolotto, G. Circulating levels and characterization of microparticles in patients with different degrees of glucose tolerance. Cardiovasc. Diabetol. 2017, 16, 118. [Google Scholar] [CrossRef]
- Zhang, Y.; Shi, L.; Mei, H.; Zhang, J.; Zhu, Y.; Han, X.; Zhu, D. Inflamed macrophage microvesicles induce insulin resistance in human adipocytes. Nutr. Metab. 2015, 12, 21. [Google Scholar] [CrossRef]
- Kranendonk, M.E.; De Kleijn, D.P.; Kalkhoven, E.; Kanhai, D.A.; Uiterwaal, C.S.; Van der Graaf, Y.; Pasterkamp, G.; Visseren, F.L. Extracellular vesicle markers in relation to obesity and metabolic complications in patients with manifest cardiovascular disease. Cardiovasc. Diabetol. 2014, 13, 37. [Google Scholar] [CrossRef]
- Choi, Y.; Kwon, Y.; Kim, D.; Jeon, J.; Jang, S.C.; Wang, T.; Ban, M.; Kim, M.; Jeon, S.G.; Kim, M. Gut microbe-derived extracellular vesicles induce insulin resistance, thereby impairing glucose metabolism in skeletal muscle. Sci. Rep. 2015, 5, 15878. [Google Scholar] [CrossRef]
- Mastronardi, M.L.; Mostefai, H.A.; Meziani, F.; Martinez, M.C.; Asfar, P.; Andriantsitohaina, R. Circulating microparticles from septic shock patients exert differential tissue expression of enzymes related to inflammation and oxidative stress. Crit. Care Med. 2011, 39, 1739–1748. [Google Scholar] [CrossRef]
- Scanu, A.; Molnarfi, N.; Brandt, K.J.; Gruaz, L.; Dayer, J.M.; Burger, D. Stimulated T cells generate microparticles, which mimic cellular contact activation of human monocytes: Differential regulation of pro- and anti-inflammatory cytokine production by high-density lipoproteins. J. Leukoc. Biol. 2008, 83, 921–927. [Google Scholar] [CrossRef]
- Werner, N.; Wassmann, S.; Ahlers, P.; Kosiol, S.; Nickenig, G. Circulating CD31+/annexin V+ apoptotic microparticles correlate with coronary endothelial function in patients with coronary artery disease. Arterioscler. Thromb. Vasc. Biol. 2006, 26, 112–116. [Google Scholar] [CrossRef]
- Mostefai, H.A.; Agouni, A.; Carusio, N.; Mastronardi, M.L.; Heymes, C.; Henrion, D.; Andriantsitohaina, R.; Martinez, M.C. Phosphatidylinositol 3-Kinase and xanthine oxidase regulate nitric oxide and reactive oxygen species productions by apoptotic lymphocyte microparticles in endothelial cells. J. Immunol. 2008, 180, 5028–5035. [Google Scholar] [CrossRef]
- Yun, C.; Jung, K.; Chu, K.; Kim, S.; Ji, K.; Park, H.; Kim, H.; Lee, S.; Lee, S.; Roh, J. Increased circulating endothelial microparticles and carotid atherosclerosis in obstructive sleep apnea. J. Clin. Neurol. 2010, 6, 89–98. [Google Scholar] [CrossRef]
- Esposito, K.; Ciotola, M.; Schisano, B.; Gualdiero, R.; Sardelli, L.; Misso, L.; Giannetti, G.; Giugliano, D. Endothelial microparticles correlate with endothelial dysfunction in obese women. J. Clin. Endocrinol. Metab. 2006, 91, 3676–3679. [Google Scholar] [CrossRef]
- Eichner, N.Z.; Erdbrügger, U.; Malin, S.K. Extracellular vesicles: A novel target for exercise-mediated reductions in type 2 diabetes and cardiovascular disease risk. J. Diabetes Res. 2018, 2018, 7807245. [Google Scholar] [CrossRef]
- Jansen, F.; Yang, X.; Franklin, B.S.; Hoelscher, M.; Schmitz, T.; Bedorf, J.; Nickenig, G.; Werner, N. High glucose condition increases NADPH oxidase activity in endothelial microparticles that promote vascular inflammation. Cardiovasc. Res. 2013, 98, 94–106. [Google Scholar] [CrossRef]
- Burger, D.; Turner, M.; Xiao, F.; Munkonda, M.N.; Akbari, S.; Burns, K.D. High glucose increases the formation and pro-oxidative activity of endothelial microparticles. Diabetologia 2017, 60, 1791–1800. [Google Scholar] [CrossRef]
- Francois, M.E.; Myette-Cote, E.; Bammert, T.D.; Durrer, C.; Neudorf, H.; DeSouza, C.A.; Little, J.P. Carbohydrate restriction with postmeal walking effectively mitigates postprandial hyperglycemia and improves endothelial function in type 2 diabetes. Am. J. Physiol. Heart Circ. Physiol. 2017, 314, H105–H113. [Google Scholar] [CrossRef]
- Boulanger, C.M.; Amabile, N.; Guerin, A.P.; Pannier, B.; Leroyer, A.S.; Mallat, C.N.; Tedgui, A.; London, G.M. In vivo shear stress determines circulating levels of endothelial microparticles in end-stage renal disease. Hypertension 2007, 49, 902–908. [Google Scholar] [CrossRef]
- Angelillo-Scherrer, A. Leukocyte-derived microparticles in vascular homeostasis. Circ. Res. 2012, 110, 356–369. [Google Scholar] [CrossRef]
- Connor, D.E.; Exner, T.; Ma, D.D.F.; Joseph, J.E. The majority of circulating platelet-derived microparticles fail to bind annexin v, lack phospholipid-dependent procoagulant activity and demonstrate greater expression of glycoprotein Ib. Thromb. Haemost. 2010, 103, 1044–1052. [Google Scholar]
- Eichner, N.Z.; Gilbertson, N.M.; Gaitan, J.M.; Heiston, E.M.; Musante, L.; LaSalvia, S.; Weltman, A.; Erdbrügger, U.; Malin, S.K. Low cardiorespiratory fitness is associated with higher extracellular vesicle counts in obese adults. Physiol. Rep. 2018, 6, e13701. [Google Scholar] [CrossRef]
- Matthews, D.; Hosker, J.; Rudenski, A.; Naylor, B.; Treacher, D.; Turner, R. Homeostasis model assessment: Insulin resistance and β-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia 1985, 28, 412–419. [Google Scholar] [CrossRef]
- Matsuda, M.; DeFronzo, R.A. Insulin sensitivity indices obtained from oral glucose tolerance testing: Comparison with the euglycemic insulin clamp. Diabetes Care 1999, 22, 1462–1470. [Google Scholar] [CrossRef]
- Erdbrügger, U.; Rudy, C.K.; Etter, M.E.; Dryden, K.A.; Yeager, M.; Klibanov, A.L.; Lannigan, J. Imaging flow cytometry elucidates limitations of microparticle analysis by conventional flow cytometry. Cytom. Part A 2014, 85, 756–770. [Google Scholar] [CrossRef]
- Wekesa, A.L.; Doyle, L.M.; Fitzmaurice, D.; O’Donovan, O.; Phelan, J.P.; Ross, M.D.; Cross, K.S.; Harrison, M. Influence of a low-carbohydrate diet on endothelial microvesicles in overweight women. Appl. Physiol. Nutr. Metab. 2016, 41, 522–527. [Google Scholar] [CrossRef]
- Malin, S.K.; Braun, B. Effect of metformin on substrate utilization after exercise training in adults with impaired glucose tolerance. Appl. Physiol. Nutr. Metab. 2013, 38, 427–430. [Google Scholar] [CrossRef]
- Mleczko, J.; Ortega, F.J.; Falcon-Perez, J.M.; Wabitsch, M.; Fernandez-Real, J.M.; Mora, S. Extracellular vesicles from hypoxic adipocytes and obese subjects reduce insulin-stimulated glucose uptake. Mol. Nutr. Food Res. 2018, 62, 1700917. [Google Scholar] [CrossRef]
- Freeman, D.W.; Noren Hooten, N.; Eitan, E.; Green, J.; Mode, N.A.; Bodogai, M.; Zhang, Y.; Lehrmann, E.; Zonderman, A.B.; Biragyn, A.; et al. Altered extracellular vesicle concentration, cargo and function in diabetes mellitus. Diabetes 2018, 67, 2377–2388. [Google Scholar] [CrossRef]
- Greenfield, J.R.; Samaras, K.; Chisholm, D.J.; Campbell, L.V. Effect of postprandial insulinemia and insulin resistance on measurement of arterial stiffness (augmentation index). Int. J. Cardiol. 2007, 114, 50–56. [Google Scholar] [CrossRef]
- Horn, P.; Cortese-Krott, M.M.; Amabile, N.; Hundsdorfer, C.; Kroncke, K.D.; Kelm, M.; Heiss, C. Circulating microparticles carry a functional endothelial nitric oxide synthase that is decreased in patients with endothelial dysfunction. J. Am. Heart Assoc. 2012, 2, e003764. [Google Scholar] [CrossRef]
- Nomura, S.; Nakamura, T.; Cone, J.; Tandon, N.N.; Kambayashi, J. Cytometric analysis of high shear-induced platelet microparticles and effect of cytokines on microparticle generation. Cytometry 2000, 40, 173–181. [Google Scholar] [CrossRef]
- Hirsova, P.; Ibrahim, S.H.; Krishnan, A.; Verma, V.K.; Bronk, S.F.; Werneburg, N.W.; Charlton, M.R.; Shah, V.H.; Malhi, H.; Gores, G.J. Lipid-induced signaling causes release of inflammatory extracellular vesicles from hepatocytes. Gastroenterology 2016, 150, 956–967. [Google Scholar] [CrossRef]
- Tschritter, O.; Fritsche, A.; Shirkavand, F.; Machicao, F.; Haring, H.; Stumvoll, M. Assessing the shape of the glucose curve during an oral glucose tolerance test. Diabetes Care 2003, 26, 1026–1033. [Google Scholar] [CrossRef]
- Colombo, M.; Raposo, G.; Théry, C. Biogenesis, secretion, and intercellular interactions of exosomes and other extracellular vesicles. Annu. Rev. Cell Dev. Biol. 2014, 30, 255–289. [Google Scholar] [CrossRef]
| NGT | Prediabetes | P-Value | |
|---|---|---|---|
| N (M/F) | 8 (1/7) | 17 (4/13) | - |
| Age (year) | 50.1 ± 5.1 | 53.5 ± 4.1 | 0.63 |
| Body weight (kg) | 90.3 ± 2.9 | 98.1 ± 3.0 | 0.34 |
| Body Mass Index (kg/m2) | 33.2 ± 1.4 | 32.1 ± 1.6 | 0.67 |
| Body Fat (%) | 42.3 ± 2.9 | 44.1 ± 5.3 | 0.64 |
| Fat Free Mass (kg) | 53.9 ± 1.9 | 53.7 ± 2.9 | 0.95 |
| VO2peak (L/min) | 2.1 ± 0.2 | 1.8 ± 0.1 | 0.10 |
| VO2peak (mL/kg/min) | 23.4 ± 1.8 | 18.6 ± 0.9 * | 0.01 |
| HDL (mg/dL) | 49.7 ± 3.0 | 52.8 ± 5.9 | 0.71 |
| LDL (mg/dL) | 111.3 ± 7.0 | 131.7 ± 15.8 | 0.37 |
| Triglycerides (mg/dL) | 103.0 ± 22.2 | 136.7 ± 28.8 | 0.44 |
| Cholesterol (mg/dL) | 178.0 ± 8.4 | 207.1 ± 18.4 | 0.28 |
| Calories | 2381.7 ± 263.0 | 2261.7 ± 263.0 | 0.70 |
| CHO (g) | 291.4 ± 29.4 | 244.1 ± 21.2 | 0.21 |
| Sugar (g) | 107.1 ± 17.2 | 93.7 ± 8.7 | 0.45 |
| Total Fiber (g) | 23.6 ± 2.8 | 19.0 ± 1.3 | 0.10 |
| Fat (g) | 97.7 ± 14.6 | 91.4 ± 26.5 | 0.66 |
| Protein (g) | 86.4 ± 10.9 | 86.5 ± 7.2 | 0.99 |
| NGT (n = 8) | Prediabetes (n = 17) | P-Value | |
|---|---|---|---|
| Fasting PG (mg/dL) | 95.2 ± 1.3 | 104.9 ± 2.1 * | 0.006 |
| 120 min PG (mg/dL) | 105.7 ± 9.2 | 136.8 ± 9.2 | 0.63 |
| PG tAUC120 (mg/dL·min) | 15,046.6 ± 550.0 | 17,593.8 ± 1089.3 * | 0.05 |
| Fasting Insulin (μU/mL) | 13.8 ± 2.2 | 12.8 ± 2.4 * | 0.03 |
| 120 min Insulin (μU/mL) | 55.3 ± 11.3 | 107.0 ± 11.8 * | 0.01 |
| Insulin tAUC120 (μU/mL·min) | 53.9 ± 1.9 | 53.7 ± 2.9 | 0.20 |
| Fasting FFA (mEq/L) | 0.52 ± 0.07 | 0.53 ± 0.03 | 0.92 |
| 120 min FFA (mEq/L) | 0.12 ± 0.05 | 0.22 ± 0.05 | 0.20 |
| FFA tAUC120 (mEq/L·min) | 31.3 ± 6.4 | 45.7 ± 6.7 | 0.20 |
| HOMA-IR | 2.4 ± 0.4 | 4.5 ± 0.7 * | 0.02 |
| Adipose insulin resistance | 19.2 ± 3.9 | 35.9 ± 6.0 * | 0.03 |
| Whole-Body Insulin Sensitivity | 4.0 ± 1.0 | 2.1 ± 0.3 * | 0.02 |
| HbA1c (%) | 5.4 ± 0.09 | 5.6 ± 0.07 | 0.11 |
| NGT (n = 8) | Prediabetes (n = 17) | ANOVA (P-Value) | ||||
|---|---|---|---|---|---|---|
| 0 min | 120 min | 0 min | 120 min | Time | GxT | |
| Systolic BP (mmHg) | 125.0 ± 3.7 | 134.1 ± 6.7 * | 129.2 ± 4.8 | 137.6 ± 4.4 * | 0.02 | 0.88 |
| Diastolic BP (mmHg) | 71.9 ± 4.1 | 77.1 ± 4.6 * | 71.3 ± 3.3 | 81.7 ± 2.7 * | 0.009 | 0.35 |
| MAP (mmHg) | 87.2 ± 3.5 | 99.4 ± 5.8 * | 91.4 ± 3.7 | 99.4 ± 3.3 * | 0.001 | 0.42 |
| Pulse Pressure (mmHg) | 51.6 ± 2.8 | 41.9 ± 3.2 * | 57.9 ± 2.3 | 42.3 ± 2.0 * | <0.001 | 0.22 |
| RHR (bpm) | 66.8 ± 2.0 | 66.2 ± 2.0 * | 66.6 ± 2.9 | 63.4 ± 1.8 * | 0.02 | 0.44 |
| AIx (%) | 32.1 ± 7.8 | 17.3 ± 9.5 | 23.4 ± 2.1 | 20.3 ± 2.7 | 0.06 | 0.21 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Eichner, N.Z.M.; Gilbertson, N.M.; Musante, L.; La Salvia, S.; Weltman, A.; Erdbrügger, U.; Malin, S.K. An Oral Glucose Load Decreases Postprandial Extracellular Vesicles in Obese Adults with and without Prediabetes. Nutrients 2019, 11, 580. https://doi.org/10.3390/nu11030580
Eichner NZM, Gilbertson NM, Musante L, La Salvia S, Weltman A, Erdbrügger U, Malin SK. An Oral Glucose Load Decreases Postprandial Extracellular Vesicles in Obese Adults with and without Prediabetes. Nutrients. 2019; 11(3):580. https://doi.org/10.3390/nu11030580
Chicago/Turabian StyleEichner, Natalie Z. M., Nicole M. Gilbertson, Luca Musante, Sabrina La Salvia, Arthur Weltman, Uta Erdbrügger, and Steven K. Malin. 2019. "An Oral Glucose Load Decreases Postprandial Extracellular Vesicles in Obese Adults with and without Prediabetes" Nutrients 11, no. 3: 580. https://doi.org/10.3390/nu11030580
APA StyleEichner, N. Z. M., Gilbertson, N. M., Musante, L., La Salvia, S., Weltman, A., Erdbrügger, U., & Malin, S. K. (2019). An Oral Glucose Load Decreases Postprandial Extracellular Vesicles in Obese Adults with and without Prediabetes. Nutrients, 11(3), 580. https://doi.org/10.3390/nu11030580






