Lactose-Free Dairy Products: Market Developments, Production, Nutrition and Health Benefits
Abstract
1. Introduction
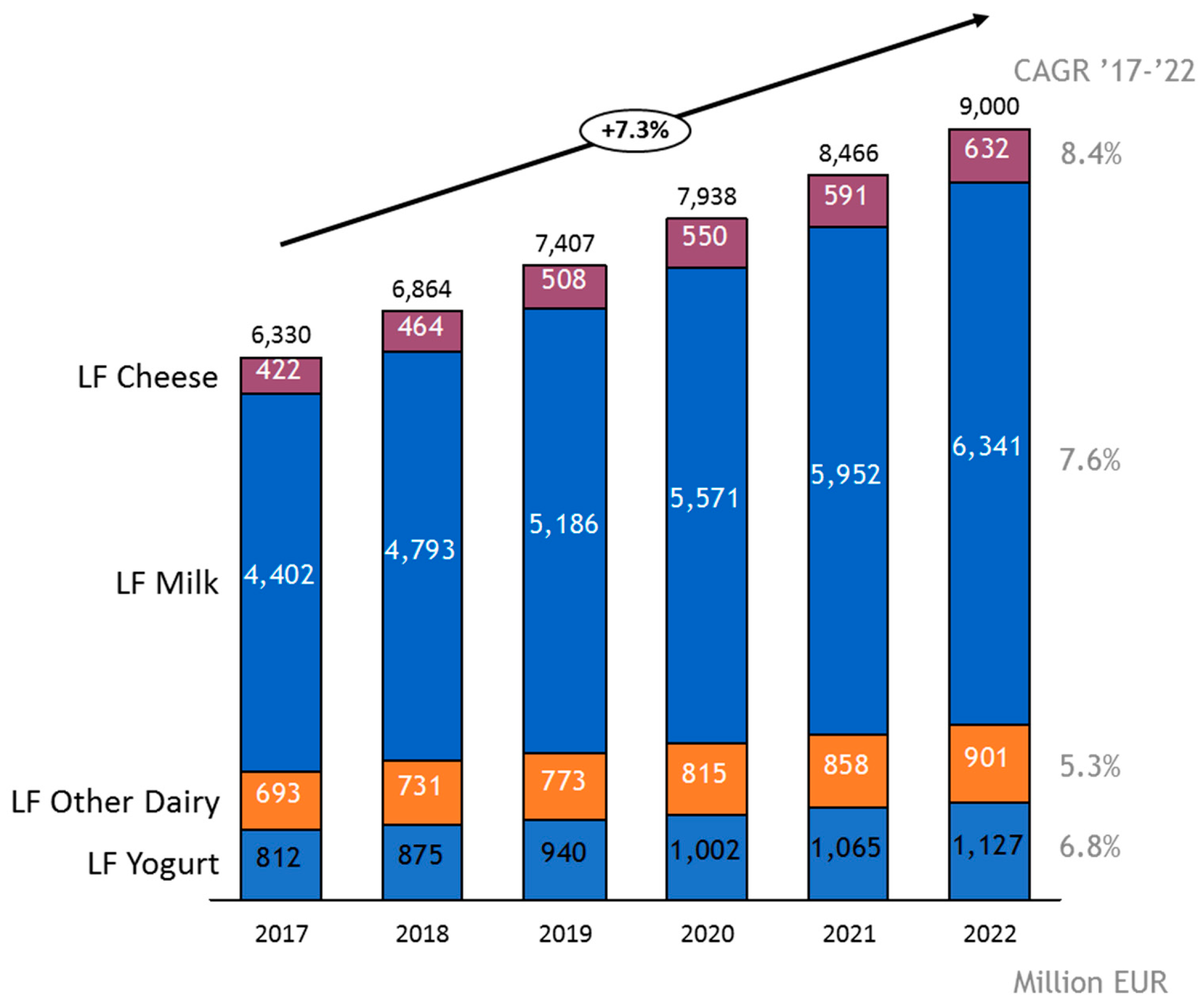
2. Market Developments of Lactose-Free Dairy
3. Production of Lactose-Free Dairy
3.1. Milk
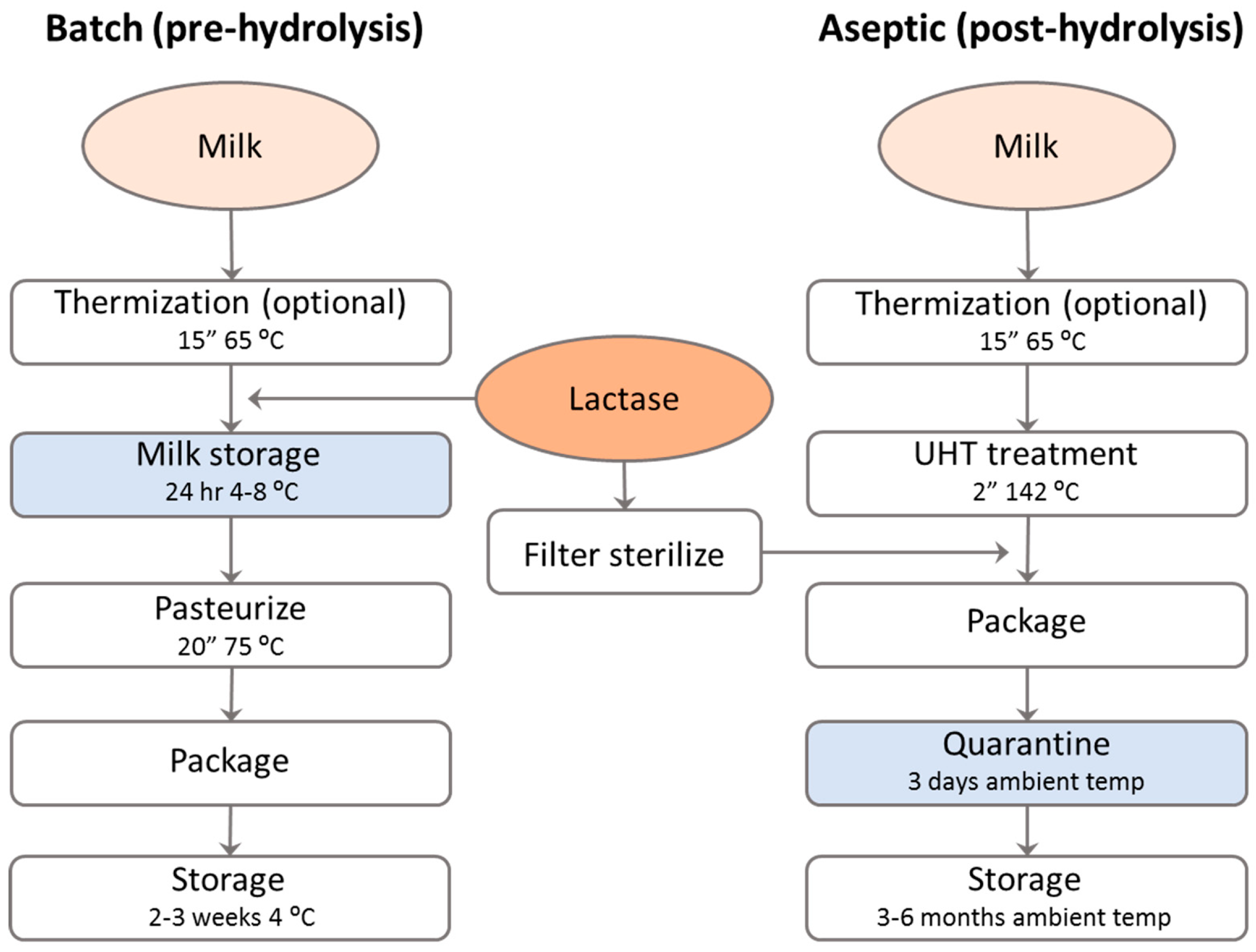
3.1.1. Batch Process (Pre-Hydrolysis)
- The dosage of the enzyme should be sufficient to reach the required boundary for lactose-free during the limited time and low temperature of the incubation. Therefore, the enzyme dosage is relatively high. Enzymes available for this process are selected for their relatively high activity at neutral pH and low temperature. The process control is high since the lactose conversion may be measured in the tank and since the enzyme dosage or incubation time may be adapted during the process.
- The batch incubation requires the occupation of a tank in the factory and the holdup of the milk for a day. The process is, therefore, discontinuous, which may pose a problem for some factories, especially when the productivity is high. A lactase with a higher specific activity under these conditions (like Maxilact® Smart) may help shorten the production time and, hence, may increase the throughput of the factory. Since the pasteurization of the milk is postponed for a day, the milk quality should be impeccable to prevent microbial spoilage.
- The milk product produced with the batch process is relatively insensitive for possible side activities in the enzyme preparation. This is due to the limited time of storage of the milk at refrigerated conditions and to the pasteurization/sterilization after the enzyme incubation, inactivating most enzymatic activities. Although in the past some lactase preparations showed proteolytic side activities [12], these problems seem to be over and very few complaints occur for lactose-free milk produced with the batch process.
- Since lactose hydrolysis leads to a doubling of the sweetness of milk, processes were developed to remove part of the lactose using chromatography or (ultra and nano) filtration techniques combined with the hydrolysis of the remaining lactose so an exact sweetness is regenerated [13,14]. The resulting lactose-free milk produced with this process is of excellent quality, and the taste is almost identical to regular milk. This feature is especially appreciated by the pasteurized milk drinkers in Northern Europe and North America and is, therefore, frequently used in conjunction with the batch process.
3.1.2. Aseptic Process (Post-Hydrolysis)
- The dosage of the enzyme can be much lower compared to the batch process, since both the incubation time and temperature are higher. Process control is, however, absent since the enzyme is only active in the final milk package. E.g., the storage temperature in un-thermostated warehouses may deviate from summer to winter, and the dairy producer should take these aspects into account when dosing the enzyme.
- The aseptic process requires special equipment and consumable costs, and especially for the in-factory filtration, it requires highly skilled operators to prevent microbial contamination of the milk during lactase injection. However, the process can be operated full-continuous when organized properly, and that is a major advantage for factories that require a high throughput.
- The aseptic process for making lactose-free UHT milk could only be fully developed after major improvements in the quality of the lactase enzymes. Besides the removal of proteolytic side activity, it was also found that arylsulfatase side activity in the lactose preparation may lead to severe medicinal off-flavors during storage due to p-cresol formation from sulphonated-cresol that is naturally present in the milk [16]. A producer of lactose-free UHT milk should consider using only the highest quality lactases for this process to prevent problems during shelf life. Arylsulfatase-free lactases (like all Maxilact® products) are currently commercially available.
- Lactose hydrolysis in milk leads to an increased presence of monosaccharides, and therefore, the Maillard reaction is more efficient. Limited proteolysis by proteases present in the milk or originating from the lactase preparation may enhance the reaction. This results in the increased formation of off-flavors, in the browning of lactose-free milk when compared to regular milk and in a reduced nutritional value when stored at increased temperatures [10,17,18]. The increased Maillard reaction is probably the most important determinant of the reduced shelf life of lactose-free UHT milk compared to regular UHT milk. Although it has been suggested in the past that lactose-free UHT milk production using the batch process may lead to even more browning compared to milk produced via the aseptic process [19], recent data show that the storage conditions (temperature) and choice of the lactase are much more relevant for determining shelf life [10]. Excellent shelf life was found for lactose-free UHT milk produced with the batch process, and milk browning during storage is, therefore, largely independent of the production process that is used.
3.2. Fermented Milk Products
- It has been suggested that the lactic acid bacteria present in yoghurt will survive the stomach, and the lactase enzyme present in these bacteria aids the digestion of (or part of) the lactose in the small intestine. The monosaccharides are both consumed by the bacteria and taken up in the small intestine, so lactose intolerance symptoms are reduced. Some dairy companies claim to produce a yoghurt containing special cultures that have this effect. This hypothesis will only hold if the yoghurt bacteria and their intracellular lactase enzyme will survive the stomach. Indeed, the pasteurization of yoghurt seems to worsen lactose intolerance symptoms.
- A second explanation that was put forward suggests that the lactose in yoghurt is better digested due to the decreased transit time of a viscous yoghurt meal compared to liquid milk. Due to this, any residual lactase in the small intestine will have more time to digest lactose and, thereby, reduces intolerance symptoms. It has indeed been found that having a meal together with a glass of milk will reduce symptoms, suggesting that transit time may play a role in lactose digestion.
3.3. Other Dairy Products
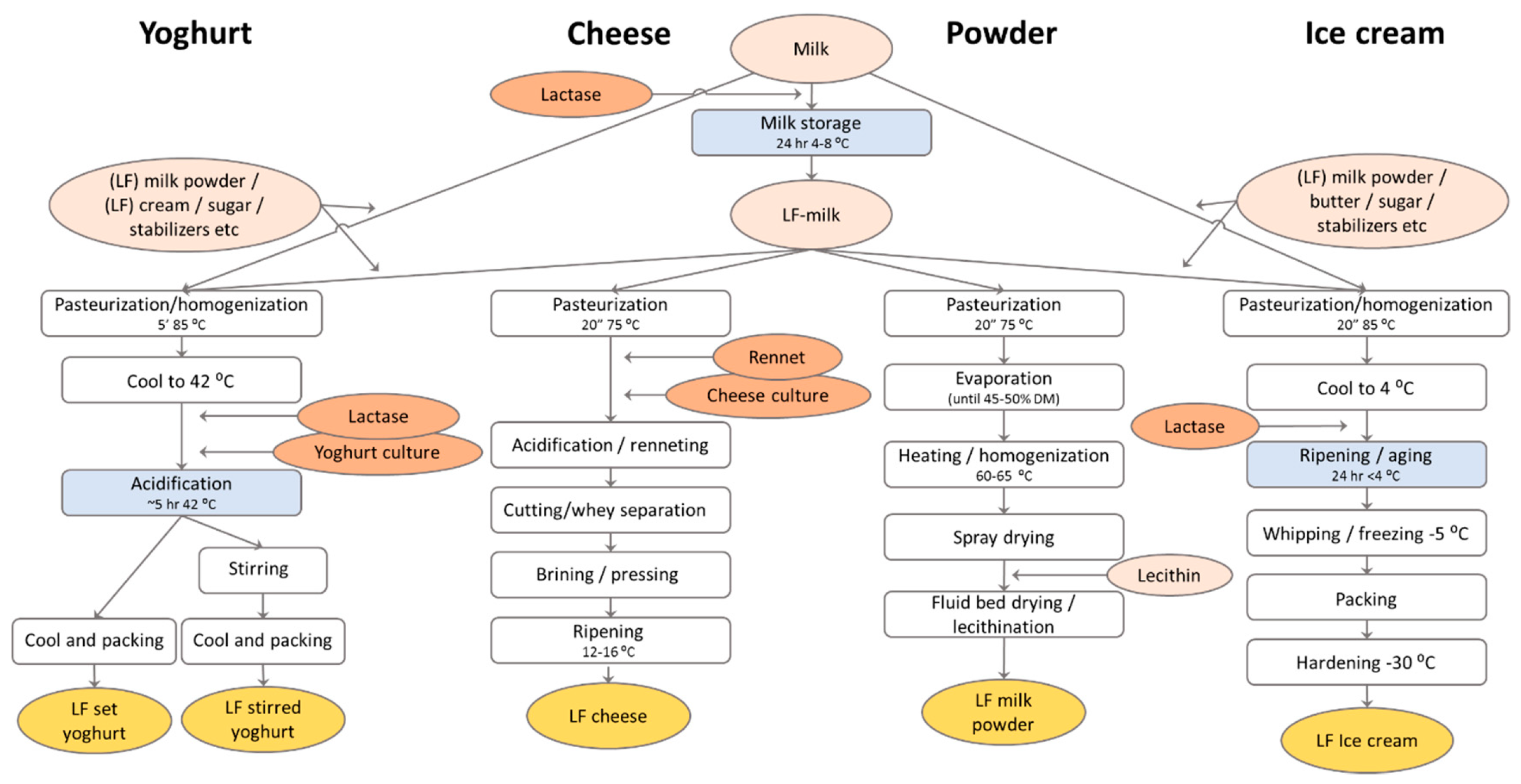
- Flavored milk is made with a process that is familiar to the milk process (Figure 2). However, the generation of the extra sweetness by the lactase treatment is an advantage since it will allow a reduction of sugar addition, similar to the situation in most yoghurts [24]. Flavored lactose-free milk also has much less problems with Maillard-related off-flavors and browning than regular lactose-free UHT milk, since the product often has a strong flavor and color by itself. For some flavored milks, like highly sugared chocolate milk, lactose hydrolysis may be insufficient to fully replace all sugar addition [27], so the addition of additional sweeteners may still be required.
- Dairy powders can be produced from milk or whey that is made lactose-free via the batch process (Figure 3). A major problem is the presence of a high concentration of monosaccharides in the treated milk, leading to a drop in the glass-transition temperature. Hence, this product will lead to the fouling of the spray dryer when the drying conditions are not adapted [28]. The much milder spray drying conditions lead to dramatic decreases in the productivity of the drying process and to increased costs. Additionally, the lactose-free milk (or whey) powder is highly hygroscopic, leading to caking during storage when not packaged with extra caution. Due to these challenging technical problems, lactose-free dairy powders is still a small market in contrast to regular milk powders.
- Cheese can be made lactose-free by incubating the cheese milk with lactase before renneting (Figure 3). This is mostly useful for young, fresh cheeses that are known to contain a significant amount of lactose, and lactases are currently used for this purpose. In ripened cheeses, all lactose will have been consumed by the lactic acid bacteria, so no lactase incubation is required. In contrast to the situation in yoghurt, the treatment of cheese milk with lactase is in the older literature often mentioned to stimulate the acidification during cheese making. Also, the lactase addition is mentioned to increase cheese flavor formation during ripening. However, it is not entirely clear if these effects are due to the stimulation of the cheese microbial flora due to the hydrolysis of the lactose or to residual proteolytic activity present in the lactase preparations that were commercially available in the past. The release of amino acids from milk proteins may have stimulated the cheese culture activity in these experiments. The latter explanation seems to be the most likely (see, for example, the discussion on this subject in Reference [29]).
- Ice cream can also be made lactose-free by either using lactose-free milk and powders in the ice cream mix [30] or by adding the lactase enzyme after pasteurization and incubation during the aging period before freezing (Figure 3). Because of the increase in monosaccharide content after lactose hydrolysis, the freezing point of the ice cream mix will decrease. This will lead to a softer ice cream at the same temperature. Although for some frozen desserts this may be an advantage because of the “soft scoop”, it will also lead to faster melting. Since the sweetness increases due to lactose hydrolysis, the ice cream maker can, however, decide to decrease sugar addition and, thereby, increase the melting temperature again. Since such changes in the recipe will decrease total solids in the mix, additional measures may be required to comply to the local regulation for ice cream. Lactase treatment is also used in ice cream to prevent lactose crystallization. Especially when whey powder or WPC is used in the ice cream mix, the amount of lactose may be high enough to form crystals during freezing. This leads to a sensory defect that is called “sandiness”. Lactase treatment can prevent the formation of sandiness in ice cream by splitting lactose into glucose and galactose, which are more soluble at low temperature. It was found that lactose hydrolysis increases the apparent viscosity of the ice cream mix, decreases the freezing point, increases the sweetness to allow a 25% reduction in sugar addition, decreases sandiness, and improves the overall acceptability of ice cream [30].
- Dulce de Leche is produced by heating sugared concentrated milk until caramelization occurs due to Maillard reactions. Lactase treatment of the milk will not only make the final product lactose-free but also stimulate the Maillard reaction due to the release of galactose. Hence, treatment with lactase may enhance the flavor and color formation in the production process for Dulce de Leche. Additionally, like in ice cream, lactose hydrolysis prevents the occurrence of sandiness when the final product is stored refrigerated.
- For many dairies, the cheese whey is a valuable by-product that is fractionated into proteins, lactose and milk salts and sold as WPC, WPI and different grades of lactose. However, not all dairies have the capabilities or facilities to produce these products. The sweet dairy flavor of liquid whey or whey permeate may also be upgraded by digesting the lactose with a neutral lactase, either in free form, immobilized or as cell suspension, before concentrating it into a syrup or powder [31]. Such lactose-free products may be used as sweetener in, for example, ice cream, confectionary or bakery applications. An additional advantage of hydrolyzing the lactose in whey or whey permeates is that a microbially stable syrup may be formed without crystallization problems, thereby saving on drying costs. Depending on the pH of the substrate, either neutral or acid lactase may be used for the conversion [31].
4. Health Aspects of Lactose-Free Dairy
4.1. Nutritional Difference between Lactose-Free Dairy and Normal Dairy
4.2. Potential Health and Economic Impact of Lactose-Free Dairy vs. Dairy Avoidance
5. Conclusions and Future Scope
Author Contributions
Funding
Conflicts of Interest
References
- Silanikove, N.; Leitner, G.; Merin, U. The Interrelationships between Lactose Intolerance and the Modern Dairy Industry: Global Perspectives in Evolutional and Historical Backgrounds. Nutrients 2015, 7, 7312–7331. [Google Scholar] [CrossRef] [PubMed]
- Walstra, P.; Geurts, T.J.; Noomen, A.; Jellema, A.; van Boekel, M.A.J.S. Dairy Technology: Principles of Milk Properties and Processes; Marcel Dekker Inc.: New York, NY, USA, 1999; Chapter 22; pp. 555–600. [Google Scholar]
- The Really BIG List of Lactose Percentages. Available online: http://www.stevecarper.com/li/list_of_lactose_percentages.htm (accessed on 3 January 2019).
- Suchy, F.J.; Brannon, P.M.; Carpenter, T.O.; Fernandez, J.R.; Gilsanz, V.; Gould, J.B.; Hall, K.; Hui, S.L.; Lupton, J.; Mennella, J.; et al. National Institutes of Health Consensus Development Conference: Lactose intolerance and health. Ann. Intern. Med. 2010, 152, 792–796. [Google Scholar] [CrossRef] [PubMed]
- Dekker, P.J.T. Enzymes Exogenous to Milk in Dairy Technology: β-D-Galactosidase. In Reference Module in Food Sciences, 1st ed.; Elsevier: Amsterdam, The Netherlands, 2016; pp. 1–8. [Google Scholar]
- Euromonitor Database. Available online: https://www.euromonitor.com/ (accessed on 1 March 2018).
- Mintel Global New Product Database. Available online: http://www.mintel.com/global-new-products-database (accessed on 1 March 2018).
- Van Scheppingen, W.B.; van Hilten, P.H.; Vijverberg, M.P.; Duchateau, A.L.L. Selective and sensitive determination of lactose in low-lactose dairy products with HPAEC-PAD. J. Chromatogr. B. 2017, 1060, 395–399. [Google Scholar] [CrossRef] [PubMed]
- Harju, M. Chromatographic and enzymatic removal of lactose from milk. Bull. Int. Dairy Fed. 2004, 389, 4–8. [Google Scholar]
- Troise, A.D.; Bandini, E.; De Donno, R.; Meijer, G.; Trezzi, M.; Fogliano, V. The quality of low lactose milk is affected by the side proteolytic activity of the lactase used in the production process. Food Res. Int. 2016, 89, 514–525. [Google Scholar] [CrossRef] [PubMed]
- Finocchiaro, T.; Olson, N.F.; Richardson, T. Use of immobilized lactase in milk systems. Adv. Biochem. Eng. 1980, 15, 71–88. [Google Scholar]
- Mittal, S.B.; Newell, G.; Hourigan, J.A.; Zadow, J.G. The effect of protease contamination in lactase on the flavour of lactose-hydrolysed milks. Aust. J. Dairy Technol. 1991, 46, 46–47. [Google Scholar]
- Jelen, P.; Tossavainen, O. Low lactose and lactose-free dairy products–prospects, technologies and applications. Aust. J. Dairy Technol. 2003, 58, 161–165. [Google Scholar]
- Harju, M.; Kallioinen, H.; Tossavainen, O. Lactose hydrolysis and other conversions in dairy products: Technological aspects. Int. Dairy J. 2012, 22, 104–109. [Google Scholar] [CrossRef]
- Dahlqvist, A.; Asp, N.-G.; Burvall, A.; Rausing, H. Hydrolysis of lactose in milk and whey with minute amounts of lactase. J. Dairy Res. 1977, 44, 541–548. [Google Scholar] [CrossRef]
- De Swaaf, M.P.M.; van Dijk, A.A.; Edens, L.; Dekker, P.J.T. Enzyme Preparation Yielding A Clean Taste. EP1954808 B1, 28 November 2006. [Google Scholar]
- Jansson, T.; Jensen, H.B.; Sundekilde, U.K.; Clausen, M.R.; Eggers, N.; Larsen, L.B.; Ray, C.; Andersen, H.J.; Bertram, H.C. Chemical and proteolysis-derived changes during long-term storage of lactose-hydrolyzed ultrahigh-temperature (UHT) milk. J. Agric. Food Chem. 2014, 62, 11270–11278. [Google Scholar] [CrossRef] [PubMed]
- Evangelisti, F.; Calcagno, C.; Nardi, S.; Zunin, P. Deterioration of protein fraction by Maillard reaction in dietetic milks. J. Dairy Res. 1999, 66, 237–243. [Google Scholar] [CrossRef] [PubMed]
- Mendoza, M.R.; Olano, A.; Villamiel, M. Chemical indicators of heat treatment in fortified and special milks. J. Agric. Food Chem. 2005, 53, 2995–2999. [Google Scholar] [CrossRef] [PubMed]
- Kies, A.K. Authorised EU health claims related to the management of lactose intolerance: Reduced lactose content, dietary lactase supplements and live yoghurt cultures. In Foods, Nutrients and Food Ingredients with Authorised EU Health Claims; Sadler, M.J., Ed.; Woodhead Publishing: Cambridge, UK, 2014; Chapter 9; pp. 177–211. [Google Scholar]
- Savaiano, D.A. Lactose digestion from yogurt: Mechanism and relevance. Am. J. Clin. Nutr. 2014, 99, 1251S–1255S. [Google Scholar] [CrossRef] [PubMed]
- Kárnyáczki, Z.; Csanádi, J. Texture profile properties, sensory evaluation, and susceptibility to syneresis of yoghurt prepared from lactose-free milk. Acta Aliment. 2017, 46, 403–410. [Google Scholar]
- Adhikari, K.; Dooley, L.M.; Chambers, E., IV; Bhumiratana, N. Sensory characteristics of commercial lactose-free milks manufactured in the United States. Lebensm. Wiss. Technol. 2010, 43, 113–118. [Google Scholar] [CrossRef]
- McCain, H.R.; Kaliappan, S.; Drake, M.A. Sugar reduction in dairy products. J. Dairy Sci. 2018, 101, 8619–8640. [Google Scholar] [CrossRef] [PubMed]
- Garrigues, C.; Gilleladen, C.; Curic-Bawden, M.; Janzen, T.; Birkelund, M.; Buchhorn, G.L.; Soerensen, K.I.; Christensen, N.; Svane, C.; Riis, S.; et al. Method of Producing A Fermented Milk Product with Improved Control of Post-Acidification. WO2015/193459 Al, 18 June 2015. [Google Scholar]
- Sahin, A.; Hamamci, H.; Garayev, S. Rheological properties of lactose-free dairy desserts. Food Sci. Technol. Int. 2016, 22, 609–620. [Google Scholar] [CrossRef] [PubMed]
- Li, X.E.; Lopetcharat, K.; Qiu, Y.; Drake, M.A. Sugar reduction of skim chocolate milk and viability of alternative sweetening through lactose hydrolysis. J. Dairy Sci. 2015, 98, 1455–1466. [Google Scholar] [CrossRef] [PubMed]
- Ferreira Torres, J.K.; Stephani, R.; Miranda-Tavares, G.; de Carvalho, A.F.; Golin Bueno Costa, R.; Rocha de Almeida, C.E.; Ramos Almeida, M.; Cappa de Oliveira, L.F.; Schuck, P.; Tuler Perrone, I. Technological aspects of lactose-hydrolyzed milk powder. Food Res. Int. 2017, 101, 45–53. [Google Scholar] [CrossRef] [PubMed]
- Marschke, R.J.; Nickerson, D.E.J.; Jarrett, W.D.; Dulley, J.R. A cause of increased proteolysis in cheddar cheese manufactured from milk containing added Maxilact. Aust. J. Dairy Technol. 1980, 35, 84–88. [Google Scholar]
- Abbasi, S.; Saeedabadian, A. Influences of lactose hydrolysis of milk and sugar reduction on some physical properties of ice cream. J. Food Sci. 2015, 52, 367–374. [Google Scholar] [CrossRef]
- Panesar, P.S.; Kennedy, J.F. Biotechnological approaches for the value addition of whey. Crit. Rev. Biotechnol. 2012, 32, 327–348. [Google Scholar] [CrossRef] [PubMed]
- da-Costa-Pinto, E.A.; Collares, E.F. Chronic lactose intake modifies the gastric emptying of monosaccharides but not of disaccharides in weanling rats. Braz. J. Med. Biol. Res. 1997, 30, 723–726. [Google Scholar] [CrossRef] [PubMed]
- Gutzwiller, A. Glucose and galactose absorption after ingestion of milk containing hydrolysed lactose in calves with diarrhoea. J. Vet. Med. A Physiol. Pathol. Clin. Med. 2000, 47, 495–500. [Google Scholar] [CrossRef] [PubMed]
- Ercan, N.; Nuttall, F.Q.; Gannon, M.C.; Redmon, J.B.; Sheridan, K.J. Effects of glucose, galactose, and lactose ingestion on the plasma glucose and insulin response in persons with non-insulin-dependent diabetes mellitus. Metabolism 1993, 42, 1560–1567. [Google Scholar] [CrossRef]
- Bailey, R.K.; Fileti, C.P.; Keith, J.; Tropez-Sims, S.; Price, W.; Allison-Ottey, S.D. Lactose intolerance and health disparities among African Americans and Hispanic Americans: An updated consensus statement. J. Natl. Med. Assoc. 2013, 105, 112–127. [Google Scholar] [CrossRef]
- United States Department of Agriculture Food (USDA). USDA Food Composition Databases. Available online: https://ndb.nal.usda.gov/ (accessed on 9 September 2018).
- Heaney, R.P.; Dowell, M.S.; Rafferty, K.; Bierman, J. Bioavailability of the calcium in fortified soy imitation milk, with some observations on method. Am. J. Clin. Nutr. 2000, 71, 1166–1169. [Google Scholar] [CrossRef] [PubMed]
- Kwak, H.-S.; Lee, W.-J.; Lee, M.-R. Revisiting lactose as an enhancer of calcium absorption. Int. Dairy J. 2012, 22, 147–151. [Google Scholar] [CrossRef]
- Survey: 75% of People With Lactose Intolerance Avoid Dairy Foods. Available online: http://www.marketwired.com/press-release/survey-75-of-people-with-lactose-intolerance-avoid-dairy-foods-1751282.htm (accessed on 3 January 2019).
- Half of Dairy Consumers in the U.S. Also Use Dairy Alternatives, New Research Out of Cargill Shows. Available online: http://www.nutritionaloutlook.com/food-beverage/half-dairy-consumers-us-also-use-dairy-alternatives-new-research-out-cargill-shows (accessed on 3 January 2019).
- Barr, S.I. Perceived lactose intolerance in adult Canadians: A national survey. Appl. Physiol. Nutr. Metab. 2013, 38, 830–835. [Google Scholar] [CrossRef] [PubMed]
- USDA Food Composition Databases. Available online: https://ndb.nal.usda.gov/ndb/search/list?home=true (accessed on 3 January 2019).
- Nicklas, T.A.; Qu, H.; Hughes, S.O.; He, M.; Wagner, S.E.; Foushee, H.R.; Shewchuk, R.M. Self-perceived lactose intolerance results in lower intakes of calcium and dairy foods and is associated with hypertension and diabetes in adults. Am. J. Clin. Nutr. 2011, 94, 191–198. [Google Scholar] [CrossRef] [PubMed]
- Segal, E.; Dvorkin, L.; Lavy, A.; Rozen, G.S.; Yaniv, I.; Raz, B.; Tamir, A.; Ish-Shalom, S. Bone density in axial and appendicular skeleton in patients with lactose intolerance: Influence of calcium intake and vitamin D status. J. Am. Coll. Nutr. 2003, 22, 201–207. [Google Scholar] [CrossRef] [PubMed]
- Buchowski, M.S.; Semenya, J.; Johnson, A.O. Dietary calcium intake in lactose maldigesting intolerant and tolerant African-American women. J. Am. Coll. Nutr. 2002, 21, 47–54. [Google Scholar] [CrossRef] [PubMed]
- Corazza, G.R.; Benati, G.; Di Sario, A.; Tarozzi, C.; Strocchi, A.; Passeri, M.; Gasbarrini, G. Lactose intolerance and bone mass in postmenopausal Italian women. Br. J. Nutr. 1995, 73, 479–487. [Google Scholar] [CrossRef] [PubMed]
- Heaney, R.P. Calcium, dairy products and osteoporosis. J. Am. Coll. Nutr. 2000, 19, 83S–99S. [Google Scholar] [CrossRef] [PubMed]
- Huncharek, M.; Muscat, J.; Kupelnick, B. Impact of dairy products and dietary calcium on bone-mineral content in children: Results of a meta-analysis. Bone 2008, 43, 312–321. [Google Scholar] [CrossRef] [PubMed]
- Miller, G.D.; DiRienzo, D.D.; Reusser, M.E.; McCarron, D.A. Benefits of dairy product consumption on blood pressure in humans: A summary of the biomedical literature. J. Am. Coll. Nutr. 2000, 19, 147S–164S. [Google Scholar] [CrossRef] [PubMed]
- Pasin, G.; Comerford, K.B. Dairy foods and dairy proteins in the management of type 2 diabetes: A systematic review of the clinical evidence. Adv. Nutr. 2015, 6, 245–259. [Google Scholar] [CrossRef] [PubMed]
- Appleby, P.; Roddam, A.; Allen, N.; Key, T. Comparative fracture risk in vegetarians and nonvegetarians in EPIC-Oxford. Eur. J. Clin. Nutr. 2007, 61, 1400–1406. [Google Scholar] [CrossRef] [PubMed]
- Vogel, K.A.; Martin, B.R.; McCabe, L.D.; Peacock, M.; Warden, S.J.; McCabe, G.P.; Weaver, C.M. The effect of dairy intake on bone mass and body composition in early pubertal girls and boys: A randomized controlled trial. Am. J. Clin. Nutr. 2017, 105, 1214–1229. [Google Scholar] [CrossRef] [PubMed]
- Merrilees, M.J.; Smart, E.J.; Gilchrist, N.L.; Frampton, C.; Turner, J.G.; Hooke, E.; March, R.L.; Maguire, P. Effects of diary food supplements on bone mineral density in teenage girls. Eur. J. Nutr. 2000, 39, 256–262. [Google Scholar] [CrossRef] [PubMed]
- Avenell, A.; Mak, J.C.; O’Connell, D. Vitamin D and vitamin D analogues for preventing fractures in post-menopausal women and older men. Cochrane Database Syst. Rev. 2014, 4, CD000227. [Google Scholar] [CrossRef] [PubMed]
- McGrane, M.M.; Essery, E.; Obbagy, J.; Lyon, J.; MacNeil, P.; Spahn, J.; Van Horn, L. Dairy Consumption, Blood Pressure, and Risk of Hypertension: An Evidence-Based Review of Recent Literature. Curr. Cardiovasc. Risk Rep. 2011, 5, 287–298. [Google Scholar] [CrossRef] [PubMed]
- Casellas, F.; Aparici, A.; Pérez, M.J.; Rodríguez, P. Perception of lactose intolerance impairs health-related quality of life. Eur. J. Clin. Nutr. 2016, 70, 1068–1072. [Google Scholar] [CrossRef] [PubMed]
- Erminia, R.; Tiziana, M.; Antonio, N.; Loris, B.; Silvia, P.; Pierpaolo, D.; Ilaria, B. HRQoL questionnaire evaluation in lactose intolerant patients with adverse reactions to foods. Intern. Emerg. Med. 2013, 8, 493–496. [Google Scholar] [CrossRef] [PubMed]
- Lötters, F.J.; Lenoir-Wijnkoop, I.; Fardellone, P.; Rizzoli, R.; Rocher, E.; Poley, M.J. Dairy foods and osteoporosis: An example of assessing the health-economic impact of food products. Osteoporos. Int. 2013, 24, 139–150. [Google Scholar] [CrossRef] [PubMed]
| per 100 g | % of DV per 244 g Cup | ||
|---|---|---|---|
| Macronutrients | |||
| Energy | kcal | 61 | |
| Protein | g | 3.2 | 15% |
| Total lipid (fat) | g | 3.3 | 12% |
| Carbohydrate | g | 4.8 | 4% |
| Choline | mg | 34.9 | 15% 1 |
| Minerals | |||
| Calcium, Ca | mg | 113 | 28% |
| Iron, Fe | mg | 0.03 | 0% |
| Magnesium, Mg | mg | 10 | 6% |
| Phosphorus, P | mg | 84 | 20% |
| Potassium, K | mg | 132 | 7% |
| Sodium, Na | mg | 43 | 13% |
| Zinc, Zn | mg | 0.37 | 6% |
| Vitamins | |||
| Vitamin C | mg | 0 | 0% |
| Thiamin | mg | 0.046 | 7% |
| Riboflavin | mg | 0.169 | 24% |
| Niacin | mg | 0.089 | 1% |
| Vitamin B-6 | mg | 0.036 | 4% |
| Folate, DFE | µg | 5 | 3% |
| Vitamin B12 | µg | 0.45 | 18% |
| Vitamin A, RAE | µg | 46 | 8% |
| Vitamin E | mg | 0.07 | 1% |
| Vitamin D | µg | 0.1 | 1% |
| Vitamin K | µg | 0.3 | 1% |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dekker, P.J.T.; Koenders, D.; Bruins, M.J. Lactose-Free Dairy Products: Market Developments, Production, Nutrition and Health Benefits. Nutrients 2019, 11, 551. https://doi.org/10.3390/nu11030551
Dekker PJT, Koenders D, Bruins MJ. Lactose-Free Dairy Products: Market Developments, Production, Nutrition and Health Benefits. Nutrients. 2019; 11(3):551. https://doi.org/10.3390/nu11030551
Chicago/Turabian StyleDekker, Peter J. T., Damiet Koenders, and Maaike J. Bruins. 2019. "Lactose-Free Dairy Products: Market Developments, Production, Nutrition and Health Benefits" Nutrients 11, no. 3: 551. https://doi.org/10.3390/nu11030551
APA StyleDekker, P. J. T., Koenders, D., & Bruins, M. J. (2019). Lactose-Free Dairy Products: Market Developments, Production, Nutrition and Health Benefits. Nutrients, 11(3), 551. https://doi.org/10.3390/nu11030551






