A Review of the Role of Green Tea (Camellia sinensis) in Antiphotoaging, Stress Resistance, Neuroprotection, and Autophagy
Abstract
1. Introduction
2. Reactive Oxygen Species, Oxidative Stress, and Antioxidants
3. Photoaging
4. Camellia Sinensis (Green Tea)
5. Protective Effects of Green Tea
5.1. Antiphotoaging Properties of Green Tea
Green Tea as an AntiphotoagingAgent: Expected Mode of Action
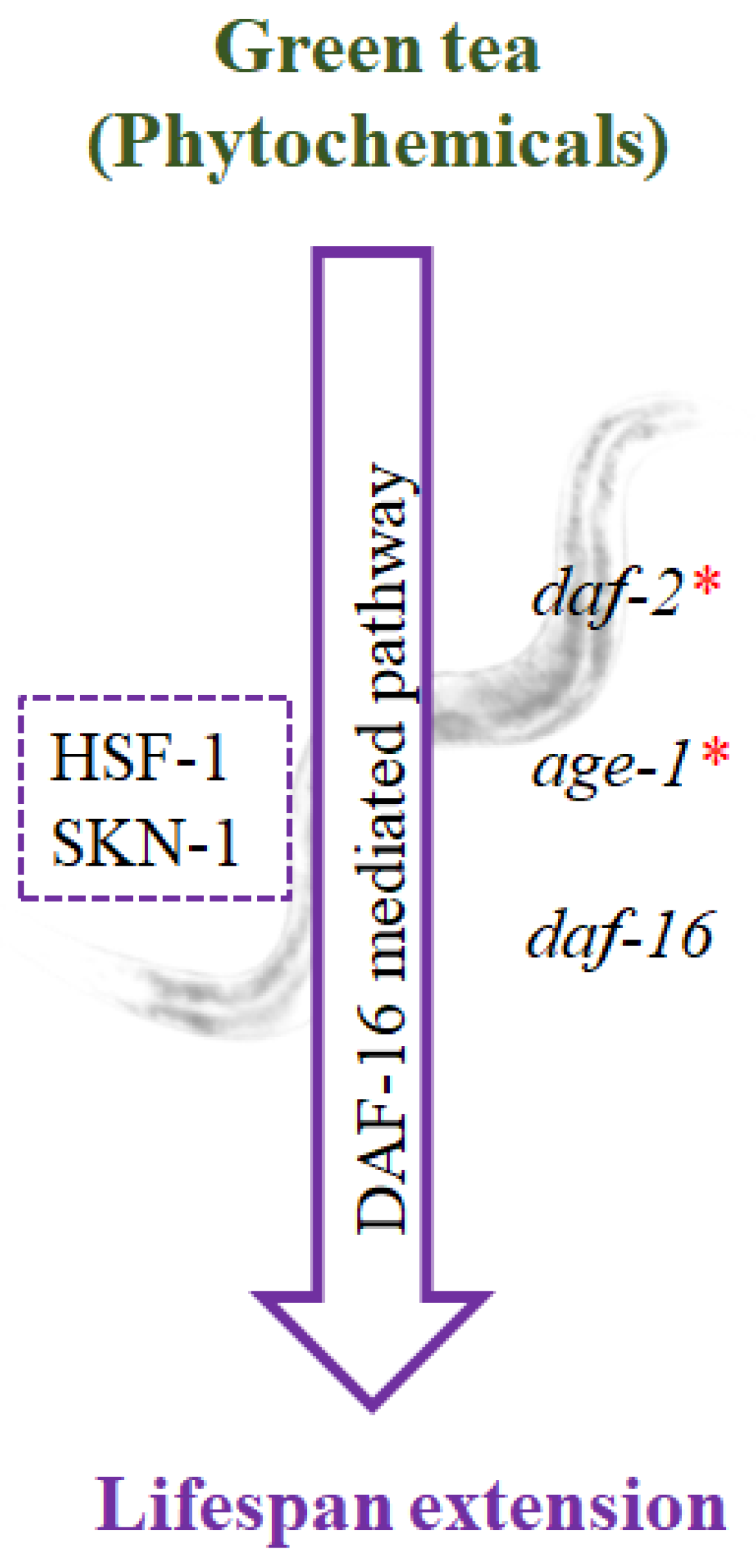
5.2. Stress Resistance Properties of Green Tea
5.3. Neuroprotective Properties of Green Tea
5.4. Autophagy Properties of Green Tea
6. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Graham, H.N. Green tea composition, consumption, and polyphenol chemistry. Prev. Med. 1992, 21, 334–350. [Google Scholar] [CrossRef]
- Gardner, E.J.; Ruxton, C.H.; Leeds, A.R. Black tea—Helpful or harmful? A review of the evidence. Eur. J. Clin. Nutr. 2007, 61, 3–18. [Google Scholar] [CrossRef] [PubMed]
- Wierzejska, R. Tea and health—A review of the current state of knowledge. Przegl. Epidemiol. 2014, 68, 595–599. [Google Scholar]
- Yamamoto, T.; Juneja, L.R.; Chu, D.; Kim, M. Chemistry and Applications of Green Tea; CRC Press: Boca Raton, FL, USA, 1997; pp. 6–34. [Google Scholar]
- Vuong, Q.V. Epidemiological evidence linking tea consumption to human health: A review. Crit. Rev. Food Sci. Nutr. 2014, 54, 523–536. [Google Scholar] [CrossRef] [PubMed]
- Scharbert, S.; Jezussek, M.; Hofmann, T. Evaluation of the taste contribution of theaflavins in black tea infusions using the taste activity concept. Eur. Food Res. Technol. 2004, 218, 442–447. [Google Scholar] [CrossRef]
- Vuong, Q.V.; Stathopoulos, C.E.; Nguyen, M.; Golding, J.B.; Roach, P.D. Isolation of green tea catechins and their utilisation in the food industry. Food Rev. Int. 2011, 27, 227–247. [Google Scholar] [CrossRef]
- Hayat, K.; Iqbal, H.; Malik, U.; Bilal, U.; Mushtaq, S. Tea and its consumption: Benefits and risks. Crit. Rev. Food Sci. Nutr. 2015, 55, 939–954. [Google Scholar] [CrossRef] [PubMed]
- Cabrera, C.; Artacho, R.; Gimenez, R. Beneficial effects of green tea—A review. J. Am. Coll. Nutr. 2006, 25, 79–99. [Google Scholar] [CrossRef] [PubMed]
- Dufresne, C.J.; Farnworth, E.R. A review of latest research findings on the health promotion properties of tea. J. Nutr. Biochem. 2001, 12, 404–421. [Google Scholar] [CrossRef]
- Rietveld, A.; Wiseman, S. Antioxidant effects of tea: Evidence from human clinical trials. J. Nutr. 2003, 133, 3285–3292. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.S.; Hong, J. Prevention of chronic diseases by tea: Possible mechanisms and human relevance. Annu. Rev. Nutr. 2013, 17, 161–181. [Google Scholar] [CrossRef] [PubMed]
- Childs, B.G.; Durik, M.; Baker, D.J.; van Deursen, J.M. Cellular senescence in aging and age-related disease: From mechanisms to therapy. Nat Med. 2015, 21, 1424–1435. [Google Scholar] [CrossRef] [PubMed]
- Santilli, V.; Bernetti, A.; Mangone, M.; Paoloni, M. Clinical definition of sarcopenia. Clin. Cases Miner. Bone Metab. 2014, 11, 177–180. [Google Scholar] [CrossRef] [PubMed]
- Pandel, R.; Poljsak, B.; Godic, A.; Dahmane, R. Skin photoaging and the role of antioxidants in its prevention. ISRN Dermatol. 2013, 2013, 930164. [Google Scholar] [CrossRef] [PubMed]
- Puglia, C.; Offerta, A.; Saija, A.; Trombetta, D.; Venera, C. Protective effect of red orange extract supplementation against UV-induced skin damages: Photoaging and solar lentigines. J. Cosmet. Dermatol. 2014, 13, 151–157. [Google Scholar] [CrossRef] [PubMed]
- Elmets, C.A.; Anderson, C.Y. Sunscreens and photocarcinogenesis: An objective assessment. Photochem. Photobiol. 1996, 63, 435–440. [Google Scholar] [CrossRef] [PubMed]
- Bosch, R.; Philips, N.; Suárez-Pérez, J.A.; Juarranz, A.; Devmurari, A.; Chalensouk-Khaosaat, J.; González, S. Mechanisms of Photoaging and Cutaneous Photocarcinogenesis, and Photoprotective Strategies with Phytochemicals. Antioxidants 2015, 4, 248–268. [Google Scholar] [CrossRef] [PubMed]
- Holick, M.F. Vitamin D deficiency. N. Engl. J. Med. 2007, 357, 266–281. [Google Scholar] [CrossRef] [PubMed]
- Gulcin, I. Antioxidant activity of food constituents: An overview. Arch. Toxicol. 2012, 86, 345–391. [Google Scholar] [CrossRef] [PubMed]
- Sohanaki, H.; Baluchnejadmojarad, T.; Nikbakht, F.; Roghani, M. Pelargonidin improves memory deficit in amyloid β25-35 rat model of Alzheimer’s disease by inhibition of glial activation, cholinesterase, and oxidative stress. Biomed. Pharmacother. 2016, 83, 85–91. [Google Scholar] [CrossRef] [PubMed]
- Gulcin, I. Antioxidant and antiradical activities of L-carnitine. Life Sci. 2006, 78, 803–811. [Google Scholar] [CrossRef] [PubMed]
- de Barboza, G.D.; Guizzardi, S.; Moine, L.; de Talamoni, N.T. Oxidative stress, antioxidants and intestinal calcium absorption. World J. Gastroenterol. 2017, 23, 2841–2853. [Google Scholar] [CrossRef] [PubMed]
- Delaunay-Moisan, A.; Appenzeller-Herzog, C. The antioxidant machinery of the endoplasmic reticulum: Protection and signaling. Free Radic. Biol. Med. 2015, 83, 341–351. [Google Scholar] [CrossRef] [PubMed]
- Peng, S.; Hou, Y.; Yao, J.; Fang, J. Activation of Nrf2-driven antioxidant enzymes by cardamonin confers neuroprotection of PC12 cells against oxidative damage. Food Funct. 2017, 8, 997–1007. [Google Scholar] [CrossRef] [PubMed]
- Evans, M.D.; Cooke, M.S. Factors contributing to the outcome of oxidative damage to nucleic acids. Bioessays 2004, 26, 533–542. [Google Scholar] [CrossRef] [PubMed]
- Radman, M. Protein damage, radiation sensitivity and aging. DNA Repair 2016, 44, 186–192. [Google Scholar] [CrossRef] [PubMed]
- Zhang, P.Y.; Xu, X.; Li, X.C. Cardiovascular diseases: Oxidative damage and antioxidant protection. Eur. Rev. Med. Pharmacol Sci. 2014, 18, 3091–3096. [Google Scholar] [PubMed]
- Aruoma, O.I. Nutrition and health aspects of free radicals and antioxidants. Food. Chem. Toxicol. 1994, 62, 671–683. [Google Scholar] [CrossRef]
- Nieradko-Iwanicka, B.; Borzęcki, A. Subacute poisoning of mice with deltamethrin produces memory impairment, reduced locomotor activity, liver damage and changes in blood morphology in the mechanism of oxidative stress. Pharmacol. Rep. 2015, 67, 535–541. [Google Scholar] [CrossRef] [PubMed]
- Halliwell, B.; Gutteridge, J.M.C. Free Radicals in Biology and Medicine, 2nd ed.; Clarendon Press: Oxford, UK, 1989. [Google Scholar]
- He, L.; He, T.; Farrar, S.; Ji, L.; Liu, T.; Ma, X. Antioxidants Maintain Cellular Redox Homeostasis by Elimination of Reactive Oxygen Species. Cell. Physiol. Biochem. 2017, 44, 532–553. [Google Scholar] [CrossRef] [PubMed]
- Sies, H. Oxidative stress: Oxidants and antioxidants. Exp. Physiol. 1997, 82, 291–295. [Google Scholar] [CrossRef] [PubMed]
- Somogyi, A.; Rosta, K.; Pusztai, P.; Tulassay, Z.; Nagy, G. Antioxidant measurements. Physiol. Meas. 2007, 28, R41–R55. [Google Scholar] [CrossRef] [PubMed]
- Riederer, P.; Sofic, E.; Rausch, W.D.; Schmidt, B.; Reynolds, G.P.; Jellinger, K.; Youdim, M.B. Transition metals, ferritin, glutathione, and ascorbic acid in parkinsonian brains. J. Neurochem. 1989, 52, 515–520. [Google Scholar] [CrossRef] [PubMed]
- Li, F.; Wang, Y.; Li, D.; Chen, Y.; Qiao, X.; Fardous, R.; Lewandowski, A.; Liu, J.; Chan, T.H.; Dou, Q.P. Perspectives on the recent developments with green tea polyphenols in drug discovery. Expert Opin. Drug Discov. 2018, 24, 1–18. [Google Scholar]
- Bocco, A.; Cuvelier, M.E.; Richard, H.; Berset, C. Antioxidant activity and phenolic composition of citrus peel and seed extracts. J. Agric. Food. Chem. 1998, 46, 2123–2129. [Google Scholar] [CrossRef]
- Eberhardt, M.V.; Lee, C.Y.; Liu, R.H. Antioxidant activity of fresh apples. Nature 2000, 405, 903–904. [Google Scholar] [CrossRef] [PubMed]
- Ganesan, K.; Kumar, K.S.; Rao, P.V.S. Comparative assessment of antioxidant activity in three edible species of green seaweed, Enteromorpha from Okha, Northwest coast of India. Innov. Food Sci. Emerg. 2011, 12, 73–78. [Google Scholar] [CrossRef]
- Nichols, J.A.; Katiyar, S.K. Skin photoprotection by natural polyphenols: Anti-inflammatory, antioxidant and DNA repair mechanisms. Arch. Dermatol. Res. 2010, 302, 71–83. [Google Scholar] [CrossRef] [PubMed]
- Weber, P.; Bendich, A.; Schalch, W. Vitamin C and human health-a review of recent data relevant to human requirements. Int. J. Vit. Nut. Res. 1996, 66, 19–30. [Google Scholar]
- Gulcin, I.; Bursal, E.; Şehitoglu, H.M.; Bilsel, M.; Goren, A.C. Polyphenol contents and antioxidant activity of lyophilized aqueous extract of propolis from Erzurum, Turkey. Food Chem. Toxicol. 2010, 48, 2227–2238. [Google Scholar] [CrossRef] [PubMed]
- Maiani, G.; Caston, M.J.; Catasta, G.; Toti, E.; Cambrodon, I.G.; Bysted, A.; Granado-Lorencio, F.; Olmedilla-Alonso, B.; Knuthsen, P.; Valoti, M.; et al. Carotenoids: Actual knowledge on food sources, intakes, stability and bioavailability and their protective role in humans. Mol. Nut. Food Res. 2009, 53, S194–S218. [Google Scholar] [CrossRef] [PubMed]
- Stahl, W.; Sies, H. Antioxidant activity of carotenoids. Mol. Asp. Med. 2003, 24, 345–351. [Google Scholar] [CrossRef]
- Ding, L.; Gao, X.; Hu, J.; Yu, S. (−)Epigallocatechin-3-gallate attenuates anesthesia-induced memory deficit in young mice via modulation of nitric oxide expression. Mol. Med. Rep. 2018, 18, 4813–4820. [Google Scholar] [CrossRef] [PubMed]
- Rutkowski, M.; Grzegorczyk, K. Adverse effects of antioxidative vitamins. Int. J. Occup. Med. Environ. Health 2012, 25, 105–121. [Google Scholar] [CrossRef] [PubMed]
- Meyers, D.G.; Maloley, P.A.; Weeks, D. Safety of antioxidant vitamins. Arch Intern. Med. 1996, 156, 925–935. [Google Scholar] [CrossRef] [PubMed]
- Petruk, G.; Del Giudice, R.; Rigano, M.M.; Monti, D.M. Antioxidants from Plants Protect against Skin Photoaging. Oxid. Med. Cell. Longev. 2018, 2018, 1454936. [Google Scholar] [CrossRef] [PubMed]
- Slominski, A.T.; Zmijewski, M.A.; Skobowiat, C.; Zbytek, B.; Slominski, R.M.; Steketee, J.D. Sensing the environment: Regulation of local and global homeostasis by the skin neuroendocrine system. Adv. Anat. Embryol. Cell Biol. 2012, 212, 1–115. [Google Scholar]
- Lorencini, M.; Brohem, C.A.; Dieamant, G.C.; Zanchin, N.I.; Maibach, H.I. Active ingredients against human epidermal aging. Ageing Res. Rev. 2014, 15, 100–115. [Google Scholar] [CrossRef] [PubMed]
- Roh, E.; Kim, J.E.; Kwon, J.Y.; Park, J.S.; Bode, A.M.; Dong, Z.; Lee, K.W. Molecular mechanisms of green tea polyphenols with protective effects against skin photoaging. Crit. Rev. Food Sci. Nutr. 2017, 57, 1631–1637. [Google Scholar] [CrossRef] [PubMed]
- Afaq, F.; Mukhtar, H. Effects of solar radiation on cutaneous detoxification pathways. J. Photochem. Photobiol. B 2001, 63, 61–69. [Google Scholar] [CrossRef]
- Dunaway, S.; Odin, R.; Zhou, L.; Ji, L.; Zhang, Y.; Kadekaro, A.L. Natural Antioxidants: Multiple Mechanisms to Protect Skin from Solar Radiation. Front. Pharmacol. 2018, 9, 392. [Google Scholar] [CrossRef] [PubMed]
- Cichorek, M.; Wachulska, M.; Stasiewicz, A.; Tymińska, A. Skin melanocytes: Biology and development. Postep. Dermatol. Alergol. 2013, 30, 30–41. [Google Scholar] [CrossRef] [PubMed]
- Fisher, G.J.; Wang, Z.Q.; Datta, S.C.; Varani, J.; Kang, S.; Voorhees, J.J. Pathophysiology of premature skin aging induced by ultraviolet light. N. Engl. J. Med. 1997, 337, 1419–1428. [Google Scholar] [CrossRef] [PubMed]
- Brenneisen, P.; Sies, H.; Scharffetter-Kochanek, K. Ultraviolet-B irradiation and matrix metalloproteinases: From induction via signaling to initial events. Ann. N. Y. Acad. Sci. 2002, 973, 31–43. [Google Scholar] [CrossRef] [PubMed]
- Smijs, T.G.; Pavel, S. Titanium dioxide and zinc oxide nanoparticles in sunscreens: Focus on their safety and effectiveness. Nanotechnol. Sci. Appl. 2011, 4, 95–112. [Google Scholar] [CrossRef] [PubMed]
- Afzal, M.; Safer, A.M.; Menon, M. Green tea polyphenols and their potential role in health and disease. Inflammopharmacology 2015, 23, 151–161. [Google Scholar] [CrossRef] [PubMed]
- Khalatbary, A.R.; Khademi, E. The green tea polyphenolic catechin epigallocatechin gallate and neuroprotection. Nutr. Neurosci. 2018, 25, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Mukhtar, H.; Ahmad, N. Tea polyphenols: Prevention of cancer and optimizing health. Am. J. Clin. Nutr. 2000, 71 (Suppl. 6), 1698S–1702S. [Google Scholar] [CrossRef]
- Nagao, T.; Hase, T.; Tokimitsu, I. A green tea extract high in catechins reduces body fat and cardiovascular risks in humans. Obesity 2007, 15, 1473–1483. [Google Scholar] [CrossRef] [PubMed]
- Moore, R.J.; Jackson, K.G.; Minihane, A.M. Green tea (Camellia sinensis) catechins and vascular function. Br. J. Nutr. 2009, 102, 1790–1802. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.; Wang, Y.; Xie, Z.; Zhou, Y.; Zhang, Y.; Wan, X. The anti-obesity effects of green tea in human intervention and basic molecular studies. Eur. J. Clin. Nutr. 2014, 68, 1075–1087. [Google Scholar] [CrossRef] [PubMed]
- Lee, L.S.; Kim, S.H.; Kim, Y.B.; Kim, Y.C. Quantitative analysis of major constituents in green tea with different plucking periods and their antioxidant activity. Molecules 2014, 19, 9173–9186. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.L.; Juan, I.M.; Chen, Y.L.; Liang, Y.C.; Lin, J.K. Composition of polyphenols in fresh tea leaves and associations of their oxygen-radical-absorbing capacity with antiproliferative actions in fibroblast cells. J. Agric. Food Chem. 1996, 44, 1387–1394. [Google Scholar] [CrossRef]
- Chen, C.N.; Liang, C.M.; Lai, J.R.; Tsai, Y.J.; Tsay, J.S.; Lin, J.K. Capillary electrophoretic determination of theanine, caffeine, and catechins in fresh tea leaves and oolong tea and their effects on rat neurosphere adhesion and migration. J. Agric. Food. Chem. 2003, 51, 7495–7503. [Google Scholar] [CrossRef] [PubMed]
- Chu, C.; Deng, J.; Man, Y.; Qu, Y. Green Tea Extracts Epigallocatechin-3-gallate for Different Treatments. Biomed. Res. Int. 2017, 2017, 5615647. [Google Scholar] [CrossRef] [PubMed]
- Zheng, X.X.; Xu, Y.L.; Li, S.H.; Liu, X.X.; Hui, R.; Huang, X.H. Green tea intake lowers fasting serum total and LDL cholesterol in adults: A meta-analysis of 14 randomized controlled trials. Am. J. Clin. Nutr. 2011, 94, 601–610. [Google Scholar] [CrossRef] [PubMed]
- Venables, M.C.; Hulston, C.J.; Cox, H.R.; Jeukendrup, A.E. Green tea extract ingestion, fat oxidation, and glucose tolerance in healthy humans. Am. J. Clin. Nutr. 2008, 87, 778–784. [Google Scholar] [CrossRef] [PubMed]
- Roberts, J.D.; Roberts, M.G.; Tarpey, M.D.; Weekes, J.C.; Thomas, C.H. The effect of a decaffeinated green tea extract formula on fat oxidation, body composition and exercise performance. J. Int. Soc. Sports Nutr. 2015, 12, 1. [Google Scholar] [CrossRef] [PubMed]
- Hursel, R.; Viechtbauer, W.; Westerterp-Plantenga, M.S. The effects of green tea on weight loss and weight maintenance: A meta-analysis. Int. J. Obes. (Lond.) 2009, 33, 956–961. [Google Scholar] [CrossRef] [PubMed]
- Mao, X.; Gu, C.; Chen, D.; Yu, B.; He, J. Oxidative stress-induced diseases and tea polyphenols. Oncotarget 2017, 8, 81649–81661. [Google Scholar] [CrossRef] [PubMed]
- Coentrao-Pde, A.; Teixeira, V.L.; Netto, A.D. Antioxidant activity of polyphenols from green and toasted mate tea. Nat. Prod. Commun. 2011, 6, 651–656. [Google Scholar] [PubMed]
- Forester, S.C.; Lambert, J.D. The role of antioxidant versus pro-oxidant effects of green tea polyphenols in cancer prevention. Mol. Nutr. Food Res. 2011, 55, 844–854. [Google Scholar] [CrossRef] [PubMed]
- Lambert, J.D.; Elias, R.J. The antioxidant and pro-oxidant activities of green tea polyphenols: A role in cancer prevention. Arch. Biochem. Biophys. 2010, 501, 65–72. [Google Scholar] [CrossRef] [PubMed]
- Yu, Y.; Deng, Y.; Lu, B.M.; Liu, Y.X.; Li, J.; Bao, J.K. Green tea catechins: A fresh flavor to anticancer therapy. Apoptosis 2013, 19, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Chikara, S.; Nagaprashantha, L.D.; Singhal, J.; Horne, D.; Awasthi, S.; Singhal, S.S. Oxidative stress and dietary phytochemicals: Role in cancer chemoprevention and treatment. Cancer Lett. 2018, 413, 122–134. [Google Scholar] [CrossRef] [PubMed]
- Fung, S.T.; Ho, C.K.; Choi, S.W.; Chung, W.Y.; Benzie, I.F. Comparison of catechin profiles in human plasma and urine after single dosing and regular intake of green tea (Camellia sinensis). Br. J. Nutr. 2013, 109, 2199–2207. [Google Scholar] [CrossRef] [PubMed]
- Iso, H.; Date, C.; Wakai, K.; Fukui, M.; Tamakoshi, A.; JACC Study Group. The relationship between green tea intake and type 2 diabetes in Japanese adults. Ann. Intern. Med. 2006, 144, 554–562. [Google Scholar] [CrossRef]
- Wu, C.H.; Lu, F.H.; Chang, C.S.; Chang, T.C.; Wang, R.H.; Chang, C.J. Relationship among habitual tea consumption, percent body fat, and body fat distribution. Obes. Res. 2003, 11, 1088–1095. [Google Scholar] [CrossRef] [PubMed]
- Martin, M.A.; Goya, L.; Ramos, S. Protective effects of tea, red wine and cocoa in diabetes. Evidences from human studies. Food Chem. Toxicol. 2017, 109, 302–314. [Google Scholar] [CrossRef] [PubMed]
- Pastoriza, S.; Mesías, M.; Cabrera, C.; Rufián-Henares, J.A. Healthy properties of green and white teas: An update. Food Funct. 2017, 8, 2650–2662. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Zheng, Y.; Cheng, X.; Meng, M.; Luo, Y.; Li, B. The anti-photoaging effect of antioxidant collagen peptides from silver carp (Hypophthalmichthys molitrix) skin is preferable to tea polyphenols and casein peptides. Food Funct. 2017, 8, 1698–1707. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.O.; Kim, S.N.; Kim, Y.C. Anti-wrinkle Effects of Water Extracts of Teas in Hairless Mouse. Toxicol. Res. 2014, 30, 283–289. [Google Scholar] [CrossRef] [PubMed]
- Meeran, S.M.; Mantena, S.K.; Elmets, C.A.; Katiyar, S.K. (−)-Epigallocatechin-3-gallate prevents photocarcinogenesis in mice through interleukin-12-dependent DNA repair. Cancer Res. 2006, 66, 5512–5520. [Google Scholar] [CrossRef] [PubMed]
- Conney, A.H.; Wang, Z.Y.; Huang, M.T.; Ho, C.T.; Yang, C.S. Inhibitory effect of green tea on tumorigenesis by chemicals and ultraviolet light. Prev. Med. 1992, 21, 361–369. [Google Scholar] [CrossRef]
- Sevin, A.; Oztaş, P.; Senen, D.; Han, U.; Karaman, C.; Tarimci, N.; Kartal, M.; Erdoğan, B. Effects of polyphenols on skin damage due to ultraviolet A rays: An experimental study on rats. J. Eur. Acad. Dermatol. Venereol. 2007, 21, 650–656. [Google Scholar] [CrossRef] [PubMed]
- Lim, J.Y.; Kim, O.K.; Lee, J.; Lee, M.J.; Kang, N.; Hwang, J.K. Protective effect of the standardized green tea seed extract on UVB-induced skin photoaging in hairless mice. Nutr. Res. Pract. 2014, 8, 398–403. [Google Scholar] [CrossRef] [PubMed]
- Clarke, K.A.; Dew, T.P.; Watson, R.E.; Farrar, M.D.; Osman, J.E.; Nicolaou, A.; Rhodes, L.E.; Williamson, G. Green tea catechins and their metabolites in human skin before and after exposure to ultraviolet radiation. J. Nutr. Biochem. 2016, 27, 203–210. [Google Scholar] [CrossRef] [PubMed]
- Mnich, C.D.; Hoek, K.S.; Virkki, L.V.; Farkas, A.; Dudli, C.; Laine, E.; Urosevic, M.; Dummer, R. Green tea extract reduces induction of p53 and apoptosis in UVB-irradiated human skin independent of transcriptional controls. Exp. Dermatol. 2009, 18, 69–77. [Google Scholar] [CrossRef] [PubMed]
- Elmets, C.A.; Singh, D.; Tubesing, K.; Matsui, M.; Katiyar, S.; Mukhtar, H. Cutaneous photoprotection from ultraviolet injury by green tea polyphenols. J. Am. Acad. Dermatol. 2001, 44, 425–432. [Google Scholar] [CrossRef] [PubMed]
- Camouse, M.M.; Domingo, D.S.; Swain, F.R.; Conrad, E.P.; Matsui, M.S.; Maes, D.; Declercq, L.; Cooper, K.D.; Stevens, S.R.; Baron, E.D. Topical application of green and white tea extracts provides protection from solar-simulated ultraviolet light in human skin. Exp. Dermatol. 2009, 18, 522–526. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.H.; Wu, Y.; Wei, H.C.; Xu, Y.Y.; Jia, L.L.; Chen, J.; Yang, X.S.; Dong, G.H.; Gao, X.H.; Chen, H.D. Protective effects of green tea extracts on photoaging and photommunosuppression. Skin Res. Technol. 2009, 15, 338–345. [Google Scholar] [CrossRef] [PubMed]
- Hong, Y.H.; Jung, E.Y.; Shin, K.S.; Yu, K.W.; Chang, U.J.; Suh, H.J. Tannase-converted green tea catechins and their anti-wrinkle activity in humans. J. Cosmet. Dermatol. 2013, 12, 137–143. [Google Scholar] [CrossRef] [PubMed]
- Bowe, W.P.; Pugliese, S. Cosmetic benefits of natural ingredients. J. Drugs. Dermatol. 2014, 13, 1021–1025. [Google Scholar] [PubMed]
- Janjua, R.; Munoz, C.; Gorell, E.; Rehmus, W.; Egbert, B.; Kern, D.; Chang, A.L. A two-year, double-blind, randomized placebo-controlled trial of oral green tea polyphenols on the long-term clinical and histologic appearance of photoaging skin. Dermatol. Surg. 2009, 35, 1057–1065. [Google Scholar] [CrossRef] [PubMed]
- Zheng, S.Q.; Huang, X.B.; Xing, T.K.; Ding, A.J.; Wu, G.S.; Luo, H.R. Chlorogenic Acid Extends the Lifespan of Caenorhabditis elegans via Insulin/IGF-1 Signaling Pathway. J. Gerontol. A Biol. Sci. Med. Sci. 2017, 72, 464–472. [Google Scholar] [PubMed]
- Kenyon, C.J. The genetics of aging. Nature 2010, 464, 504–512. [Google Scholar] [CrossRef] [PubMed]
- Garigan, D.; Hsu, A.L.; Fraser, A.G.; Kamath, R.S.; Ahringer, J.; Kenyon, C. Genetic analysis of tissue aging in Caenorhabditis elegans: A role for heat-shock factor and bacterial proliferation. Genetics 2002, 161, 1101–1112. [Google Scholar] [PubMed]
- Herndon, L.A.; Schmeissner, P.J.; Dudaronek, J.M.; Brown, P.A.; Listner, K.M.; Sakano, Y.; Paupard, M.C.; Hall, D.H.; Driscoll, M. Stochastic and genetic factors influence tissue-specific decline in ageing C. elegans. Nature 2002, 419, 808–814. [Google Scholar] [CrossRef] [PubMed]
- Tullet, J.M.; Hertweck, M.; An, J.H.; Baker, J.; Hwang, J.Y.; Liu, S.; Oliveira, R.P.; Baumeister, R.; Blackwell, T.K. Direct inhibition of the longevity-promoting factor SKN-1 by insulin-like signaling in C. elegans. Cell 2008, 132, 1025–1038. [Google Scholar] [CrossRef] [PubMed]
- Prasanth, M.I.; Santoshram, G.S.; Bhaskar, J.P.; Balamurugan, K. Ultraviolet-A triggers photoaging in model nematode Caenorhabditis elegans in a DAF-16 dependent pathway. Age 2016, 38, 27. [Google Scholar] [CrossRef] [PubMed]
- Yiannakopoulou, E.C. Targeting oxidative stress response by green tea polyphenols: Clinical implications. Free Radic. Res. 2013, 47, 667–671. [Google Scholar] [CrossRef] [PubMed]
- Babu, P.V.; Liu, D. Green tea catechins and cardiovascular health: An update. Curr. Med. Chem. 2008, 15, 1840–1850. [Google Scholar] [CrossRef] [PubMed]
- Zarse, K.; Jabin, S.; Ristow, M. L-Theanine extends lifespan of adult Caenorhabditis elegans. Eur. J. Nutr. 2012, 51, 765–768. [Google Scholar] [CrossRef] [PubMed]
- Xiao, R.Y.; Hao, J.; Ding, Y.H.; Che, Y.Y.; Zou, X.J.; Liang, B. Transcriptome Profile Reveals that Pu-Erh Tea Represses the Expression of Vitellogenin Family to Reduce Fat Accumulation in Caenorhabditis elegans. Molecules 2016, 21, 1379. [Google Scholar] [CrossRef] [PubMed]
- Xiong, L.G.; Chen, Y.J.; Tong, J.W.; Gong, Y.S.; Huang, J.A.; Liu, Z.H. Epigallocatechin-3-gallate promotes healthy lifespan through mitohormesis during early-to-mid adulthood in Caenorhabditis elegans. Redox Biol. 2018, 14, 305–315. [Google Scholar] [CrossRef] [PubMed]
- Cantó, C.; Gerhart-Hines, Z.; Feige, J.N.; Lagouge, M.; Noriega, L.; Milne, J.C.; Elliott, P.J.; Puigserver, P.; Auwerx, J. AMPK regulates energy expenditure by modulating NAD+ metabolism and SIRT1 activity. Nature 2009, 458, 1056–1060. [Google Scholar] [CrossRef] [PubMed]
- Massie, H.R.; Aiello, V.R.; Williams, T.R. Inhibition of iron absorption prolongs the life span of Drosophila. Mech. Ageing Dev. 1993, 67, 227–237. [Google Scholar] [CrossRef]
- Lopez, T.; Schriner, S.E.; Okoro, M.; Lu, D.; Chiang, B.T.; Huey, J.; Jafari, M. Green tea polyphenols extend the lifespan of male Drosophila melanogaster while impairing reproductive fitness. J. Med. Food. 2014, 17, 1314–1321. [Google Scholar] [CrossRef] [PubMed]
- Lopez, T.E.; Pham, H.M.; Nguyen, B.V.; Tahmasian, Y.; Ramsden, S.; Coskun, V.; Schriner, S.E.; Jafari, M. Green tea polyphenols require the mitochondrial iron transporter, mitoferrin, for lifespan extension in Drosophila melanogaster. Arch. Insect. Biochem. Physiol. 2016, 93, 210–221. [Google Scholar] [CrossRef] [PubMed]
- Wagner, A.E.; Piegholdt, S.; Rabe, D.; Baenas, N.; Schloesser, A.; Eggersdorfer, M.; Stocker, A.; Rimbach, G. Epigallocatechin gallate affects glucose metabolism and increases fitness and lifespan in Drosophila melanogaster. Oncotarget 2015, 6, 30568–30578. [Google Scholar] [CrossRef] [PubMed]
- Winiarska-Mieczan, A. Protective effect of tea against lead and cadmium-induced oxidative stress-a review. Biometals 2018, 31, 909–926. [Google Scholar] [CrossRef] [PubMed]
- Vayalil, P.K.; Mittal, A.; Hara, Y.; Elmets, C.A.; Katiyar, S.K. Green tea polyphenols prevent ultraviolet light-induced oxidative damage and matrix metalloproteinases expression in mouse skin. J. Investig. Dermatol. 2004, 122, 1480–1487. [Google Scholar] [CrossRef] [PubMed]
- Mi, Y.; Zhang, W.; Tian, H.; Li, R.; Huang, S.; Li, X.; Qi, G.; Liu, X. EGCG evokes Nrf2 nuclear translocation and dampens PTP1B expression to ameliorate metabolic misalignment under insulin resistance condition. Food Funct. 2018, 9, 1510–1523. [Google Scholar] [CrossRef] [PubMed]
- Smith, R.E.; Tran, K.; Smith, C.C.; McDonald, M.; Shejwalkar, P.; Hara, K. The Role of the Nrf2/ARE Antioxidant System in Preventing Cardiovascular Diseases. Diseases 2016, 4, 34. [Google Scholar] [CrossRef] [PubMed]
- Onishi, S.; Ishino, M.; Kitazawa, H.; Yoto, A.; Shimba, Y.; Mochizuki, Y.; Unno, K.; Meguro, S.; Tokimitsu, I.; Miura, S. Green tea extracts ameliorate high-fat diet-induced muscle atrophy in senescence-accelerated mouse prone-8 mice. PLoS ONE 2018, 13, e0195753. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.J.; Rhee, S.J. Green tea catechins protect rats from microwave-induced oxidative damage to heart tissue. J. Med. Food. 2004, 7, 299–304. [Google Scholar] [CrossRef] [PubMed]
- Takabayashi, F.; Harada, N.; Tahara, S.; Kaneko, T.; Hara, Y. Effect of green tea catechins on the amount of 8-hydroxydeoxyguanosine (8-OHdG) in pancreatic and hepatic DNA after a single administration of N-nitrosobis(2-oxopropyl)amine (BOP). Pancreas 1997, 15, 109–112. [Google Scholar] [CrossRef] [PubMed]
- Roychoudhury, S.; Agarwal, A.; Virk, G.; Cho, C.L. Potential role of green tea catechins in the management of oxidative stress-associated infertility. Reprod. Biomed. Online 2017, 34, 487–498. [Google Scholar] [CrossRef] [PubMed]
- Lombardo, F.; Fiducia, M.; Lunghi, R.; Marchetti, L.; Palumbo, A.; Rizzo, F.; Koverech, A.; Lenzi, A.; Gandini, L. Effects of a dietary supplement on chronic pelvic pain syndrome (Category IIIA), leucocytospermia and semen parameters. Andrologia 2012, 44 (Suppl. 1), 672–678. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.G.; Yu, S.D.; Xu, Z.R. Improvement in bovine embryo production in vitro by treatment with green tea polyphenols during in vitro maturation of oocytes. Anim. Reprod. Sci. 2007, 100, 22–31. [Google Scholar] [CrossRef] [PubMed]
- O’Sullivan, J.; Sheridan, J.; Mulcahy, H.; Tenniswood, M.; Morrissey, C. The effect of green tea on oxidative damage and tumour formation in Lobund-Wistar rats. Eur. J. Cancer Prev. 2008, 17, 489–501. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.H.; Liu, T.Z.; Chen, C.H.; Wong, C.H.; Chen, C.H.; Lu, F.J.; Chen, S.C. The efficacy of protective effects of tannic acid, gallic acid, ellagic acid, and propyl gallate against hydrogen peroxide-induced oxidative stress and DNA damages in IMR-90 cells. Mol. Nutr. Food Res. 2007, 51, 962–968. [Google Scholar] [CrossRef] [PubMed]
- Hakim, I.A.; Harris, R.B.; Brown, S.; Chow, H.H.; Wiseman, S.; Agarwal, S.; Talbot, W. Effect of increased tea consumption on oxidative DNA damage among smokers: A randomized controlled study. J. Nutr. 2003, 133, 3303S–3309S. [Google Scholar] [CrossRef] [PubMed]
- Bettuzzi, S.; Brausi, M.; Rizzi, F.; Castagnetti, G.; Peracchia, G.; Corti, A. Chemoprevention of human prostate cancer by oral administration of green tea catechins in volunteers with high-grade prostate intraepithelial neoplasia: A preliminary report from a one-year proof-of-principle study. Cancer Res. 2006, 66, 1234–1240. [Google Scholar] [CrossRef] [PubMed]
- Holzer, N.; Braun, K.F.; Ehnert, S.; Egaña, J.T.; Schenck, T.L.; Buchholz, A.; Schyschka, L.; Neumaier, M.; Benzing, S.; Stöckle, U.; et al. Green tea protects human osteoblasts from cigarette smoke-induced injury: Possible clinical implication. Langenbecks Arch. Surg. 2012, 397, 467–474. [Google Scholar] [CrossRef] [PubMed]
- Sinha, D.; Roy, M. Antagonistic role of tea against sodium arsenite-induced oxidative DNA damage and inhibition of DNA repair in Swiss albino mice. J. Environ. Pathol. Toxicol. Oncol. 2011, 30, 311–322. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Ramiro, I.; Martín, M.A.; Ramos, S.; Bravo, L.; Goya, L. Comparative effects of dietary flavanols on antioxidant defences and their response to oxidant-induced stress on Caco2 cells. Eur. J. Nutr. 2011, 50, 313–322. [Google Scholar] [CrossRef] [PubMed]
- Xue, K.X.; Wang, S.; Ma, G.J.; Zhou, P.; Wu, P.Q.; Zhang, R.F.; Xu, Z.; Chen, W.S.; Wang, Y.Q. Micronucleus formation in peripheral-blood lymphocytes from smokers and the influence of alcohol- and tea-drinking habits. Int. J. Cancer 1992, 50, 702–705. [Google Scholar] [CrossRef] [PubMed]
- Váli, L.; Hahn, O.; Kupcsulik, P.; Drahos, A.; Sárváry, E.; Szentmihályi, K.; Pallai, Z.; Kurucz, T.; Sípos, P.; Blázovics, A. Oxidative stress with altered element content and decreased ATP level of erythrocytes in hepatocellular carcinoma and colorectal liver metastases. Eur. J. Gastroenterol. Hepatol. 2008, 20, 393–398. [Google Scholar] [CrossRef] [PubMed]
- Uttara, B.; Singh, A.V.; Zamboni, P.; Mahajan, R.T. Oxidative stress and neurodegenerative diseases: A review of upstream and downstream antioxidant therapeutic options. Curr. Neuropharmacol. 2009, 7, 65–74. [Google Scholar] [CrossRef] [PubMed]
- Caruana, M.; Vassallo, N. Tea Polyphenols in Parkinson’s Disease. Adv. Exp. Med. Biol. 2015, 863, 117–137. [Google Scholar] [PubMed]
- Bieschke, J.; Russ, J.; Friedrich, R.P.; Ehrnhoefer, D.E.; Wobst, H.; Neugebauer, K.; Wanker, E.E. EGCG remodels mature alpha-synuclein and amyloid-beta fibrils and reduces cellular toxicity. Proc. Natl. Acad. Sci. USA 2010, 107, 7710–7715. [Google Scholar] [CrossRef] [PubMed]
- Chesser, A.S.; Ganeshan, V.; Yang, J.; Johnson, G.V.W. Epigallocatechin-3-gallate enhances clearance of phosphorylated tau in primary neurons. Nutr. Neurosci. 2016, 19, 21–31. [Google Scholar] [CrossRef] [PubMed]
- Fei, T.; Fei, J.; Huang, F.; Xie, T.; Xu, J.; Zhou, Y.; Yang, P. The anti-aging and anti-oxidation effects of tea water extract in Caenorhabditis elegans. Exp. Gerontol. 2017, 97, 89–96. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, A.; Watanabe, T.; Fujita, T.; Hasegawa, T.; Saito, M.; Suganuma, M. Green tea aroma fraction reduces β-amyloid peptide-induced toxicity in Caenorhabditis elegans transfected with human β-amyloid minigene. Biosci. Biotechnol. Biochem. 2014, 78, 1206–1211. [Google Scholar] [CrossRef] [PubMed]
- He, M.; Liu, M.Y.; Wang, S.; Tang, Q.S.; Yao, W.F.; Zhao, H.S.; Wei, M.J. Research on EGCG improving the degenerative changes of the brain in AD model mice induced with chemical drugs. Zhong Yao Cai 2012, 35, 1641–1644. [Google Scholar] [PubMed]
- Walker, J.M.; Klakotskaia, D.; Ajit, D.; Weisman, G.A.; Wood, W.G.; Sun, G.Y.; Serfozo, P.; Simonyi, A.; Schachtman, T.R. Beneficial effects of dietary EGCG and voluntary exercise on behavior in an Alzheimer’s disease mouse model. J. Alzheimers Dis. 2015, 44, 561–572. [Google Scholar] [CrossRef] [PubMed]
- Rezai-Zadeh, K.; Arendash, G.W.; Hou, H.; Fernandez, F.; Jensen, M.; Runfeldt, M.; Shytle, R.D.; Tan, J. Green tea epigallocatechin-3-gallate (EGCG) reduces beta-amyloid mediated cognitive impairment and modulates tau pathology in Alzheimer transgenic mice. Brain Res. 2008, 1214, 177–187. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.J.; Choi, D.Y.; Yun, Y.P.; Han, S.B.; Oh, K.W.; Hong, J.T. Epigallocatechin-3-gallate prevents systemic inflammation-induced memory deficiency and amyloidogenesis via its anti-neuroinflammatory properties. J. Nutr. Biochem. 2013, 24, 298–310. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.L.; Chen, T.F.; Chiu, M.J.; Way, T.D.; Lin, J.K. Epigallocatechin gallate (EGCG) suppresses beta-amyloid-induced neurotoxicity through inhibiting c-Abl/FE65 nuclear translocation and GSK3 beta activation. Neurobiol. Aging 2009, 30, 81–92. [Google Scholar] [CrossRef] [PubMed]
- Wei, J.C.C.; Huang, H.C.; Chen, W.J.; Huang, C.N.; Peng, C.H.; Lin, C.L. Epigallocatechin gallate attenuates amyloid β-induced inflammation and neurotoxicity in EOC 13.31 microglia. Eur. J. Pharmacol. 2016, 770, 16–24. [Google Scholar]
- Cascella, M.; Bimonte, S.; Muzio, M.R.; Schiavone, V.; Cuomo, A. The efficacy of Epigallocatechin-3-gallate (green tea) in the treatment of Alzheimer’s disease: An overview of pre-clinical studies and translational perspectives in clinical practice. Infect. Agent Cancer 2017, 12, 36. [Google Scholar] [CrossRef] [PubMed]
- Schimidt, H.L.; Garcia, A.; Martins, A.; Mello-Carpes, P.B.; Carpes, F.P. Green tea supplementation produces better neuroprotective effects than red and black tea in Alzheimer-like rat model. Food Res. Int. 2017, 100 Pt 1, 442–448. [Google Scholar] [CrossRef]
- Chen, M.; Wang, T.; Yue, F.; Li, X.; Wang, P.; Li, Y.; Chan, P.; Yu, S. Tea polyphenols alleviate motor impairments, dopaminergic neuronal injury, and cerebral α-synuclein aggregation in MPTP-intoxicated parkinsonian monkeys. Neuroscience 2015, 286, 383–392. [Google Scholar] [CrossRef] [PubMed]
- Jimenez-Del-Rio, M.; Guzman-Martinez, C.; Velez-Pardo, C. The effects of polyphenols on survival and locomotor activity in Drosophila melanogaster exposed to iron and paraquat. Neurochem. Res. 2010, 35, 227–238. [Google Scholar] [CrossRef] [PubMed]
- Chan, D.K.; Woo, J.; Ho, S.C.; Pang, C.P.; Law, L.K.; Ng, P.W.; Hung, W.T.; Kwok, T.; Hui, E.; Orr, K.; et al. Genetic and environmental risk factors for Parkinson’s disease in a Chinese population. J. Neurol. Neurosurg. Psychiatry 1998, 65, 781–784. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Zhang, W.B.; Zhu, J.H.; Fu, G.S.; Zhou, B.Q. Breviscapine ameliorates hypertrophy of cardiomyocytes induced by high glucose in diabetic rats via the PKC signaling pathway. Acta Pharmacol. Sin. 2009, 30, 1081–1091. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.S.; Quon, M.J.; Kim, J.A. New insights into the mechanisms of polyphenols beyond antioxidant properties; lessons from the green tea polyphenol, epigallocatechin 3-gallate. Redox Biol. 2014, 2, 187–195. [Google Scholar] [CrossRef] [PubMed]
- Stefani, M.; Rigacci, S. Beneficial properties of natural phenols: Highlight on protection against pathological conditions associated with amyloid aggregation. Biofactors 2014, 40, 482–493. [Google Scholar] [CrossRef] [PubMed]
- Calgarotto, A.K.; Maso, V.; Junior, G.C.F.; Nowill, A.E.; Filho, P.L.; Vassallo, J.; Saad, S.T.O. Antitumor activities of Quercetin and Green Tea in xenografts of human leukemia HL60 cells. Sci. Rep. 2018, 8, 3459. [Google Scholar] [CrossRef] [PubMed]
- Fahie, K.; Zachara, N.E. Molecular Functions of Glycoconjugates in Autophagy. J. Mol. Biol. 2016, 428, 3305–3324. [Google Scholar] [CrossRef] [PubMed]
- Enkhbat, T.; Nishi, M.; Yoshikawa, K.; Jun, H.; Tokunaga, T.; Takasu, C.; Kashihara, H.; Ishikawa, D.; Tominaga, M.; Shimada, M. Epigallocatechin-3-gallate Enhances Radiation Sensitivity in Colorectal Cancer Cells Through Nrf2 Activation and Autophagy. Anticancer Res. 2018, 38, 6247–6252. [Google Scholar] [CrossRef] [PubMed]
- Sun, X.H.; Yin, M.S.; Mu, Y.L. Alterations of mTOR pathway and autophagy in early type 2 diabetic cardiomyopathy in rats. Zhonghua Bing Li Xue Za Zhi. 2016, 45, 707–710. [Google Scholar] [PubMed]
- Wang, Q.; He, W.Y.; Zeng, Y.Z.; Hossain, A.; Gou, X. Inhibiting autophagy overcomes docetaxel resistance in castration-resistant prostate cancer cells. Int. Urol. Nephrol. 2018, 50, 675–686. [Google Scholar] [CrossRef] [PubMed]
- Prasansuklab, A.; Tencomnao, T. Amyloidosis in Alzheimer’s Disease: The Toxicity of Amyloid Beta (Aβ), Mechanisms of Its Accumulation and Implications of Medicinal Plants for Therapy. Evid. Based Complement. Alternat. Med. 2013, 2013, 413808. [Google Scholar] [CrossRef] [PubMed]
- Thongrakard, V.; Titone, R.; Follo, C.; Morani, F.; Suksamrarn, A.; Tencomnao, T.; Isidoro, C. Turmeric toxicity in A431 epidermoid cancer cells associates with autophagy degradation of anti-apoptotic and anti-autophagic p53 mutant. Phytother. Res. 2014, 28, 1761–1769. [Google Scholar] [CrossRef] [PubMed]
- Li, P.; Ma, K.; Wu, H.Y.; Wu, Y.P.; Li, B.X. Isoflavones Induce BEX2-Dependent Autophagy to Prevent ATR-Induced Neurotoxicity in SH-SY5Y Cells. Cell. Physiol. Biochem. 2017, 43, 1866–1879. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Zhu, S.; Li, J.; Assa, A.; Jundoria, A.; Xu, J.; Fan, S.; Eissa, N.T.; Tracey, K.J.; Sama, A.E.; et al. EGCG stimulates autophagy and reduces cytoplasmic HMGB1 levels in endotoxin-stimulated macrophages. Biochem. Pharmacol. 2011, 81, 1152–1163. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.S.; Montana, V.; Jang, H.J.; Parpura, V.; Kim, J.A. Epigallocatechin gallate (EGCG) stimulates autophagy in vascular endothelial cells: A potential role for reducing lipid accumulation. J. Biol. Chem. 2013, 288, 22693–22705. [Google Scholar] [CrossRef] [PubMed]
- Renaud, J.; Nabavi, S.F.; Daglia, M.; Nabavi, S.M.; Martinoli, M.G. Epigallocatechin-3-Gallate, a Promising Molecule for Parkinson’s Disease? Rejuv. Res. 2015, 18, 257–269. [Google Scholar] [CrossRef] [PubMed]
- Trivedi, P.C.; Bartlett, J.J.; Perez, L.J.; Brunt, K.R.; Legare, J.F.; Hassan, A.; Kienesberger, P.C.; Pulinilkunnil, T. Glucolipotoxicity diminishes cardiomyocyte TFEB and inhibits lysosomal autophagy during obesity and diabetes. Biochim. Biophys. Acta 2016, 1861, 1893–1910. [Google Scholar] [CrossRef] [PubMed]
- Holczer, M.; Besze, B.; Zámbó, V.; Csala, M.; Bánhegyi, G.; Kapuy, O. Epigallocatechin-3-Gallate (EGCG) Promotes Autophagy-Dependent Survival via Influencing the Balance of mTOR-AMPK Pathways upon Endoplasmic Reticulum Stress. Oxid. Med. Cell. Longev. 2018, 2018, 6721530. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.H.; Moon, J.H.; Kim, S.W.; Jeong, J.K.; Nazim, U.M.; Lee, Y.J.; Seol, J.W.; Park, S.Y. EGCG-mediated autophagy flux has a neuroprotection effect via a class III histone deacetylase in primary neuron cells. Oncotarget 2015, 6, 9701–9717. [Google Scholar] [CrossRef] [PubMed]
- Wei, R.; Mao, L.; Xu, P.; Zheng, X.; Hackman, R.M.; Mackenzie, G.G.; Wang, Y. Suppressing glucose metabolism with epigallocatechin-3-gallate (EGCG) reduces breast cancer cell growth in preclinical models. Food Funct. 2018, 9, 5682–5696. [Google Scholar] [CrossRef] [PubMed]
- Grube, S.; Ewald, C.; Kögler, C.; Lawson McLean, A.; Kalff, R.; Walter, J. Achievable Central Nervous System Concentrations of the Green Tea Catechin EGCG Induce Stress in Glioblastoma Cells in Vitro. Nutr. Cancer 2018, 10, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.M.; Wu, C.T.; Yang, T.H.; Chang, Y.A.; Sheu, M.L.; Liu, S.H. Green Tea Catechin Prevents Hypoxia/Reperfusion-Evoked Oxidative Stress-Regulated Autophagy-Activated Apoptosis and Cell Death in Microglial Cells. J. Agric. Food Chem. 2016, 64, 4078–4085. [Google Scholar] [CrossRef] [PubMed]
- Hsieh, C.H.; Lu, C.H.; Kuo, Y.Y.; Chen, W.T.; Chao, C.Y. Studies on the non-invasive anticancer remedy of the triple combination of epigallocatechin gallate, pulsed electric field, and ultrasound. PLoS ONE 2018, 13, e0201920. [Google Scholar] [CrossRef] [PubMed]
- Hashimoto, K.; Sakagami, H. Induction of apoptosis by epigallocatechin gallate and autophagy inhibitors in a mouse macrophage-like cell line. Anticancer Res. 2008, 28, 1713–1718. [Google Scholar] [PubMed]
- Yang, L.; Jin, X.; Yan, J.; Jin, Y.; Yu, W.; Wu, H.; Xu, S. Prevalence of dementia, cognitive status and associated risk factors among elderly of Zhejiang province, China in 2014. Age Ageing 2016, 45, 708–712. [Google Scholar] [CrossRef] [PubMed]
- Xiong, L.G.; Huang, J.A.; Li, J.; Yu, P.H.; Xiong, Z.; Zhang, J.W.; Gong, Y.S.; Liu, Z.H.; Chen, J.H. Black tea increased survival of Caenorhabditis elegans under stress. J. Agric. Food Chem. 2014, 62, 11163–11169. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Farah, B.L.; Sinha, R.A.; Wu, Y.; Singh, B.K.; Bay, B.H.; Yang, C.S.; Yen, P.M. Epigallocatechin-3-gallate (EGCG), a green tea polyphenol, stimulates hepatic autophagy and lipid clearance. PLoS ONE 2014, 9, e87161. [Google Scholar] [CrossRef] [PubMed]
- Zhou, H.; Chen, Y.; Huang, S.W.; Hu, P.F.; Tang, L.J. Regulation of autophagy by tea polyphenols in diabetic cardiomyopathy. J. Zhejiang Univ. Sci. B 2018, 19, 333–341. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Chen, D.; Zhu, K. SOX2OT variant 7 contributes to the synergistic interaction between EGCG and Doxorubicin to kill osteosarcoma via autophagy and stemness inhibition. J. Exp. Clin. Cancer Res. 2018, 37, 37. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; Liu, S.; Xu, J.; Li, W.; Duan, G.; Wang, H.; Yang, H.; Yang, Z.; Zhou, R. A new molecular mechanism underlying the EGCG-mediated autophagic modulation of AFP in HepG2 cells. Cell Death Dis. 2017, 8, e3160. [Google Scholar] [CrossRef] [PubMed]
- Thornfeldt, C.R.; Rizer, R.L. Superior Efficacy of an Herbal-based Cosmeceutical Compared with Common Prescription and Cosmetic Antiaging Therapies. J. Drugs Dermatol. 2016, 15, 218–223. [Google Scholar] [PubMed]
- Giacomotto, J.; Ségalat, L. High-throughput screening and small animal models, where are we? Br. J. Pharmacol. 2010, 160, 204–216. [Google Scholar] [CrossRef] [PubMed]
- Halliwell, B. Are polyphenols antioxidants or pro-oxidants? What do we learn from cell culture and in vivo studies? Arch. Biochem. Biophys. 2008, 476, 107–112. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Yang, N.D.; Zhou, F.; Shen, T.; Duan, T.; Zhou, J.; Shi, Y.; Zhu, X.Q.; Shen, H.M. (−)-Epigallocatechin-3-gallate induces non-apoptotic cell death in human cancer cells via ROS-mediated lysosomal membrane permeabilization. PLoS ONE 2012, 7, e46749. [Google Scholar] [CrossRef] [PubMed]
- Li, G.X.; Chen, Y.K.; Hou, Z.; Xiao, H.; Jin, H.; Lu, G.; Lee, M.J.; Liu, B.; Guan, F.; Yang, Z.; et al. Pro-oxidative activities and dose-response relationship of (−)-epigallocatechin-3-gallate in the inhibition of lung cancer cell growth: A comparative study in vivo and in vitro. Carcinogenesis 2010, 31, 902–910. [Google Scholar] [CrossRef] [PubMed]
- Heidler, T.; Hartwig, K.; Daniel, H.; Wenzel, U. Caenorhabditis elegans lifespan extension caused by treatment with an orally active ROS-generator is dependent on DAF-16 and SIR-2.1. Biogerontology 2010, 11, 183–195. [Google Scholar] [CrossRef] [PubMed]
- Bonkovsky, H.L. Hepatotoxicity associated with supplements containing Chinese green tea (Camellia sinensis). Ann. Intern. Med. 2006, 144, 68–71. [Google Scholar] [CrossRef] [PubMed]
- Lambert, J.D.; Sang, S.; Yang, C.S. Possible controversy over dietary polyphenols: Benefits vs. risks. Chem. Res. Toxicol. 2007, 20, 583–585. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.S.; Wang, X.; Lu, G.; Picinich, S.C. Cancer prevention by tea: Animal studies, molecular mechanisms and human relevance. Nat. Rev. Cancer 2009, 9, 429–439. [Google Scholar] [CrossRef] [PubMed]
- Sterelny, K. Humans as model organisms. Proc. Biol. Sci. 2017, 284, 20172115. [Google Scholar] [CrossRef] [PubMed]
- Peluso, I.; Serafini, M. Antioxidants from black and green tea: From dietary modulation of oxidative stress to pharmacological mechanisms. Br. J. Pharmacol. 2017, 174, 1195–1208. [Google Scholar] [CrossRef] [PubMed]
- Pallauf, K.; Rimbach, G.; Rupp, P.M.; Chin, D.; Wolf, I.M. Resveratrol and Lifespan in Model Organisms. Curr. Med. Chem. 2016, 23, 4639–4680. [Google Scholar] [CrossRef] [PubMed]

| S. No | Compound(s)/Extract (s) Used | Dosage Given | Model System | Results | Ref. |
|---|---|---|---|---|---|
| 1 | Polyphenols of green tea | 200 mg/kg | Mice | Increase in hydroxyproline content and catalase activity. Decrease in protein carbonyl content. | [83] |
| 2 | Aqueous extracts of green tea | 2% | Mice | Increase the level of collagen and elastin fibers. Reduced expression of MMP-3 enzymes. | [84] |
| 3 | Seed extracts of green tea | 10 mg/kg, 100 mg/kg and 200 mg/kg | Mice | Increase in collagensynthesis. Reduced wrinkle formation. | [88] |
| 4 | Green tea extract | 0.5% of diet | Mice | Reduce muscle atrophy and mediate insulin resistance. | [117] |
| 5 | Green tea polyphenols | 0.2% wt./vol | Mice | Inhibit protein oxidation induced by UV radiations. | [114] |
| 6 | Epigallocatechin gallate (EGCG) | 1.0 mg/cm2 skin area | Mice | Prevent skin tumor incidence and multiplicity. Reduce the number of cells affected by sunburn. | [85] [87] |
| 7 | EGCG | 2 g/L of drinking water | Mice | Maintain equilibrium during redox reaction. | [115] |
| 8 | EGCG | 2 mg/kg or 6 mg/kg | Mice model for AD | Reduction in the accumulation of Aβ. | [138] |
| 9 | Crude green tea extract | 10 mg/mL in food | Fruit flies | Extension of lifespan. Reduction in total body iron. | [110] |
| 10 | EGCG | 10 mg/mL in food | Drosophila | Extend lifespan. | [112] |
| 11 | EGCG | 200 μM | C. elegans | Extend lifespan. | [107] |
| 12 | Cholinergic acid | 50 μM | C. elegans | Extend lifespan. Delay age-related decline in body movements. | [97] |
| 13 | Theanine | 1 μM | C. elegans | Stress resistance and lifespan extension. | [105] |
| 14 | Green tea extract | 0.025 g/mL and 0.05 g/mL of media | C. elegans | Reduce fat accumulation and lipid droplets. | [106] |
| 15 | Green tea and fractions of green tea aroma | 0.125 and 0.25 mg/mL of green tea and 10 and 100 μg/mL of green tea aroma fraction | Transgenic strains of C. elegans | Delay ofAβ-induced paralysis. | [136,137] |
| No. | Compound(s)/Extract(s) Used | Subjects Used | Duration | Treatment Method | Results | Ref. |
|---|---|---|---|---|---|---|
| 1 | Green tea polyphenols | 12 human volunteers between 18–65 years | 3 months | Consume capsules of green tea polyphenols | Conjugate metabolites in plasma, blister fluid, and skin biopsy samples | [89] |
| 2 | Green tea extract | 18 human volunteers between 21 and 71 | 34 days | Topical application | Reduction in the level of cells with sunburn | [90] |
| 3 | Green tea extract | Human volunteers aged between 18 and 50 | - | Topical application ranging from 0.25 to 10% Topical applicationof 5% EGCG, EC, and EGC | Decreased sunburn cells by 66% 0.5 to 2.5% concentration showed optimum activity Lesser activity when compared to the crude extracts | [91] |
| 4 | Green tea extract | 10 human volunteers | 15 min prior to UV irradiation and immediately after exposure | Topical application | Lesser DNA damage when compared to vehicle control | [92] |
| 5 | Green tea extract | 20 Chinese women | 30 min prior to UV irradiation and 6, 24, and 48 h after exposure | Topical application (2–5%) | 3% of topical application showed less erythema, mild pigmentation, controlled level of thickening of stratum corneum and epidermis, and reduction of matrix metalloproteases | [93] |
| 6 | Green tea extract | 42 Korean females | 8 weeks | Topical application at crow’s feet | Free radical scavenging and antiwrinkle effects | [94,95] |
| 7 | Green tea polyphenols | 56 women aged 25 to 75 | 2 years | Oral supplements of 250 mg green tea polyphenols | Improvement in facial skin and in controlling erythema | [96] |
| 8 | Green tea extract | 2015 subjects aged over 65 years | 6 months | Oral consumption | Reduced the prevalence of Alzheimer’s disease and cognitive impairment | [171] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Prasanth, M.I.; Sivamaruthi, B.S.; Chaiyasut, C.; Tencomnao, T. A Review of the Role of Green Tea (Camellia sinensis) in Antiphotoaging, Stress Resistance, Neuroprotection, and Autophagy. Nutrients 2019, 11, 474. https://doi.org/10.3390/nu11020474
Prasanth MI, Sivamaruthi BS, Chaiyasut C, Tencomnao T. A Review of the Role of Green Tea (Camellia sinensis) in Antiphotoaging, Stress Resistance, Neuroprotection, and Autophagy. Nutrients. 2019; 11(2):474. https://doi.org/10.3390/nu11020474
Chicago/Turabian StylePrasanth, Mani Iyer, Bhagavathi Sundaram Sivamaruthi, Chaiyavat Chaiyasut, and Tewin Tencomnao. 2019. "A Review of the Role of Green Tea (Camellia sinensis) in Antiphotoaging, Stress Resistance, Neuroprotection, and Autophagy" Nutrients 11, no. 2: 474. https://doi.org/10.3390/nu11020474
APA StylePrasanth, M. I., Sivamaruthi, B. S., Chaiyasut, C., & Tencomnao, T. (2019). A Review of the Role of Green Tea (Camellia sinensis) in Antiphotoaging, Stress Resistance, Neuroprotection, and Autophagy. Nutrients, 11(2), 474. https://doi.org/10.3390/nu11020474








