Deciphering the Nutraceutical Potential of Raphanus sativus—A Comprehensive Overview
Abstract
1. Introduction
2. Antioxidant Effects of Radishes
3. Hepatoprotective Effects of Radishes
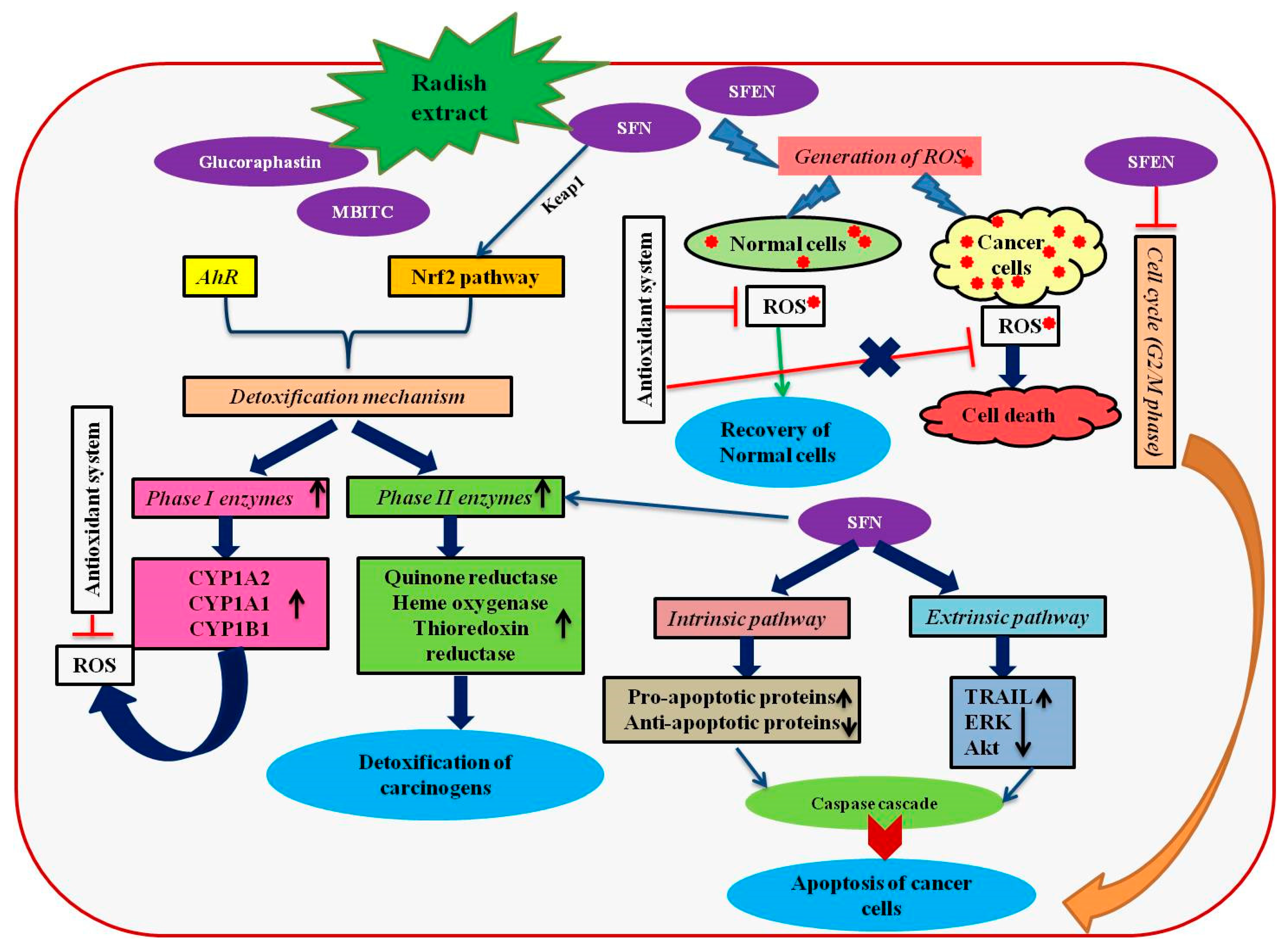
4. Anticancer Effects of R. sativus
4.1. Liver Cancer
4.2. Colon Cancer
4.3. Breast Cancer
4.4. Cervical, Lung and Prostate Cancer
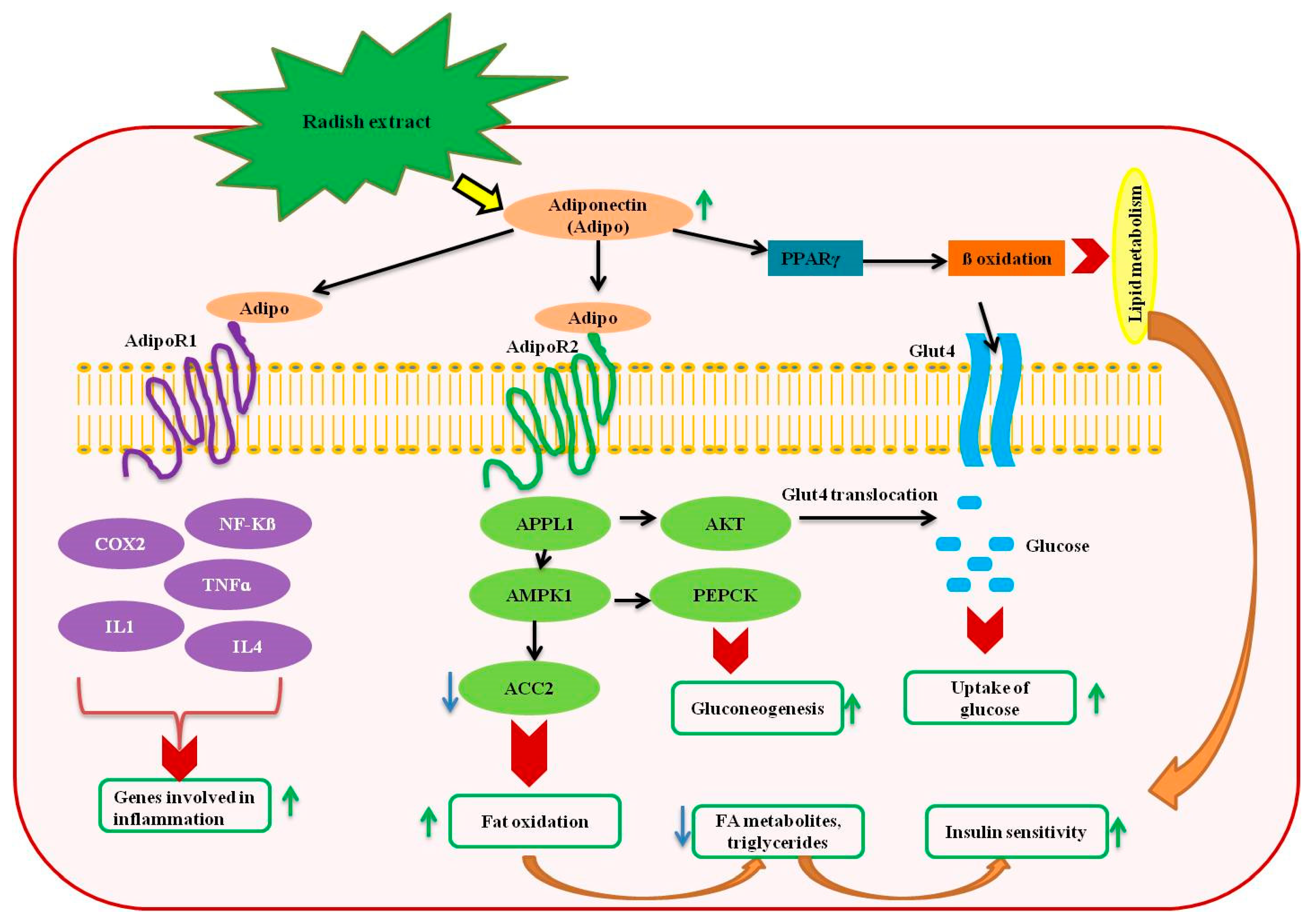
5. Antidiabetic Effects of Radish
6. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Rhodes, M.J.C. Physiological roles for secondary metabolites in plants: Some progress, many outstanding problems. Plant Mol. Biol. 1994, 24, 1–20. [Google Scholar] [CrossRef] [PubMed]
- Luckner, M. Secondary Metabolism in Microorganisms, Plants and Animals; Springer Science & Business Media: Berlin, Germany, 2013. [Google Scholar]
- Seca, A.; Pinto, D. Plant secondary metabolites as anticancer agents: Successes in clinical trials and therapeutic application. Int. J. Mol. Sci. 2018, 19, 263. [Google Scholar] [CrossRef] [PubMed]
- Soundararajan, P.; Kim, J. Anticarcinogenic glucosinolates in cruciferous vegetables and their antagonistic effects on prevention of cancer. Molecules 2018, 23, 2983. [Google Scholar] [CrossRef]
- Beevi, S.S.; Mangamoori, L.N.; Gowda, B.B. Polyphenolics profile and antioxidant properties of Raphanus sativus L. Nat. Prod. Res. 2012, 26, 557–563. [Google Scholar] [CrossRef] [PubMed]
- Castro-Torres, I.G.; De la O-Arciniega, M.; Gallegos-Estudillo, J.; Naranjo-Rodríguez, E.B.; Domínguez-Ortíz, M.Á. Raphanus sativus L. var. niger as a source of phytochemicals for the prevention of cholesterol gallstones. Phytother. Res. 2014, 28, 167–171. [Google Scholar] [CrossRef]
- Curtis, I.S. The noble radish: Past, present and future. Trends Plant Sci. 2003, 8, 305–307. [Google Scholar] [CrossRef]
- Goyeneche, R.; Roura, S.; Ponce, A.; Vega-Galvez, A.; Quispe-Fuentes, I.; Uribe, E.; Di Scala, K. Chemical characterization and antioxidant capacity of red radish (Raphanus sativus L.) leaves and roots. J. Funct. Foods 2015, 16, 256–264. [Google Scholar] [CrossRef]
- Takaya, Y.; Kondo, Y.; Furukawa, T.; Niwa, M. Antioxidant constituents of radish sprout (Kaiware-daikon), Raphanus sativus L. J. Agric. Food Chem. 2003, 51, 8061–8066. [Google Scholar] [CrossRef]
- Tsouvaltzis, P.; Brecht, J.K. Changes in Quality and Antioxidant Enzyme Activities of Bunched and Topped Radish (Raphanus sativus L.) Plants during Storage at 5 or 10C. J. Food Quality 2014, 37, 157–167. [Google Scholar] [CrossRef]
- Beevi, S.S.; Mangamoori, L.N.; Dhand, V.; Ramakrishna, D.S. Isothiocyanate profile and selective antibacterial activity of root, stem and leaf extracts derived from Raphanus sativus L. Foodborne Pathog. Dis. 2009, 6, 129–136. [Google Scholar] [CrossRef]
- Rakhmawati, R.; Anggarwulan, E.; Retnaningtyas, E. Potency of Lobak leaves (Raphanus sativus L. var. Hortensis Back) as anticancer and antimicrobial candidates. Biodivers. J. Biol. Divers. 2009, 10. [Google Scholar] [CrossRef]
- Pocasap, P.; Weerapreeyakul, N.; Barusrux, S. Cancer preventive effect of Thai rat-tailed radish (Raphanus sativus L. var. caudatus Alef). J. Funct. Foods 2013, 5, 1372–1381. [Google Scholar] [CrossRef]
- Kim, S.; Woo, M.; Kim, M.; Noh, J.S.; Song, Y.O. Hot water extracts of pressure-roasted dried radish attenuates hepatic oxidative stress via Nrf2 upregulation in mice fed high-fat diet. Food Sci. Biotechnol. 2017, 26, 1063–1069. [Google Scholar] [CrossRef] [PubMed]
- Baenas, N.; Piegholdt, S.; Schloesser, A.; Moreno, D.A.; Garcia-Viguera, C.; Rimbach, G.; Wagner, A.E. Metabolic activity of radish sprouts derived isothiocyanates in drosophila melanogaster. Int. J. Mol. Sci. 2016, 17, 251. [Google Scholar] [CrossRef]
- Siddiq, A.; Younus, I. The Radish, Raphanus sativus L. Var. caudatus reduces anxiety-like behavior in mice. Metab. Brain Dis. 2018. [Google Scholar] [CrossRef]
- Yuan, G.; Wang, X.; Guo, R.; Wang, Q. Effect of salt stress on phenolic compounds, glucosinolates, myrosinase and antioxidant activity in radish sprouts. Food Chem. 2010, 121, 1014–1019. [Google Scholar] [CrossRef]
- Salah-Abbès, J.B.; Abbès, S.; Zohra, H.; Oueslati, R. Tunisian radish (Raphanus sativus) extract prevents cadmium-induced immunotoxic and biochemical alterations in rats. J. Immunotoxicol. 2015, 12, 40–47. [Google Scholar] [CrossRef]
- Nakamura, Y.; Nakamura, K.; Asai, Y.; Wada, T.; Tanaka, K.; Matsuo, T.; Okamoto, S.; Meijer, J.; Kitamura, Y.; Nishikawa, A.; et al. Comparison of the glucosinolate? myrosinase systems among daikon (Raphanus sativus, Japanese white radish) varieties. J. Agric. Food Chem. 2008, 56, 2702–2707. [Google Scholar] [CrossRef]
- Kliebenstein, D.J.; Kroymann, J.; Brown, P.; Figuth, A.; Pedersen, D.; Gershenzon, J.; Mitchell-Olds, T. Genetic control of natural variation in Arabidopsis glucosinolate accumulation. Plant Physiol. 2001, 126, 811–825. [Google Scholar] [CrossRef]
- Field, B.; Cardon, G.; Traka, M.; Botterman, J.; Vancanneyt, G.; Mithen, R. Glucosinolate and amino acid biosynthesis in Arabidopsis. Plant Physiol. 2004, 135, 828–839. [Google Scholar] [CrossRef]
- Dawson, G.W.; Hick, A.J.; Bennett, R.N.; Donald, A.; Pickett, J.A.; Wallsgrove, R.M. Synthesis of glucosinolate precursors and investigations into the biosynthesis of phenylalkyl-and methylthioalkylglucosinolates. J. Biol. Chem. 1993, 268, 27154–27159. [Google Scholar] [PubMed]
- Grassi, D.; Desideri, G.; Ferri, C. Flavonoids: Antioxidants against atherosclerosis. Nutrients 2010, 2, 889–902. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.S.; Sun, X.D.; Cao, Y.; Wang, L.; Li, F.J.; Wang, Y.F. Antioxidant and prooxidant properties of acylated pelargonidin derivatives extracted from red radish (Raphanus sativus var. niger, Brassicaceae). Food Chem. Toxicol. 2010, 48, 2712–2718. [Google Scholar] [CrossRef] [PubMed]
- Castaneda-Ovando, A.; de Lourdes Pacheco-Hernández, M.; Páez-Hernández, M.E.; Rodríguez, J.A.; Galán-Vidal, C.A. Chemical studies of anthocyanins: A review. Food Chem. 2009, 113, 859–871. [Google Scholar] [CrossRef]
- Kong, J.M.; Chia, L.S.; Goh, N.K.; Chia, T.F.; Brouillard, R. Analysis and biological activities of anthocyanins. Phytochemistry 2003, 64, 923–933. [Google Scholar] [CrossRef]
- Mazza, G.; Miniati, E. Anthocyanins in Fruits, Vegetables and Grains; CRC Press: Boca Raton, FL, USA, 2018. [Google Scholar]
- Bagchi, D.; Sen, C.K.; Bagchi, M.; Atalay, M. Antiangiogenic, antioxidant and anticarcinogenic properties of a novel anthocyanin-rich berry extract formula. Biochemistry 2004, 69, 75–80. [Google Scholar] [PubMed]
- Renis, M.; Calandra, L.; Scifo, C.; Tomasello, B.; Cardile, V.; Vanella, L.; Bei, R.; Fauci, L.L.; Galvano, F. Response of cell cycle/stress-related protein expression and DNA damage upon treatment of CaCO2 cells with anthocyanins. Br. J. Nutr. 2008, 100, 27–35. [Google Scholar] [CrossRef]
- Shih, P.H.; Yeh, C.T.; Yen, G.C. Anthocyanins induce the activation of phase II enzymes through the antioxidant response element pathway against oxidative stress-induced apoptosis. J. Agric. Food Chem. 2007, 55, 9427–9435. [Google Scholar] [CrossRef]
- Wang, L.S.; Stoner, G.D. Anthocyanins and their role in cancer prevention. Cancer Lett. 2008, 269, 281–290. [Google Scholar] [CrossRef]
- Feng, R.; Ni, H.M.; Wang, S.Y.; Tourkova, I.L.; Shurin, M.R.; Harada, H.; Yin, X.M. Cyanidin-3-rutinoside, a natural polyphenol antioxidant, selectively kills leukemic cells by induction of oxidative stress. J. Biol. Chem. 2007, 282, 13468–13476. [Google Scholar] [CrossRef]
- Koley, T.K.; Khan, Z.; Oulkar, D.; Singh, B.K.; Maurya, A.; Singh, B.; Banerjee, K. High resolution LC-MS characterization of phenolic compounds and the evaluation of antioxidant properties of a tropical purple radish genotype. Arab. J. Chem. 2017. [Google Scholar] [CrossRef]
- Lee, K.B.; Kim, Y.J.; Kim, H.J.; Choi, J.; Kim, J.K. Phytochemical profiles of Brassicaceae vegetables and their multivariate characterization using chemometrics. Appl. Biol. Chem. 2018, 61, 131–144. [Google Scholar] [CrossRef]
- Wei, J.; Miao, H.; Wang, Q. Effect of glucose on glucosinolates, antioxidants and metabolic enzymes in Brassica sprouts. Sci. Hortic. 2011, 129, 535–540. [Google Scholar] [CrossRef]
- Baenas, N.; Ferreres, F.; Garcia-Viguera, C.; Moreno, D.A. Radish sprouts? Characterization and elicitation of novel varieties rich in anthocyanins. Food Res. Int. 2015, 69, 305–312. [Google Scholar] [CrossRef]
- Pajak, P.; Socha, R.; Gałkowska, D.; Roznowski, J.; Fortuna, T. Phenolic profile and antioxidant activity in selected seeds and sprouts. Food Chem. 2014, 143, 300–306. [Google Scholar] [CrossRef] [PubMed]
- Luo, X.; Zhang, H.; Duan, Y.; Chen, G. Protective effects of radish (Raphanus sativus L.) leaves extract against hydrogen peroxide-induced oxidative damage in human fetal lung fibroblast (MRC-5) cells. Biomed. Pharmacother. 2018, 103, 406–414. [Google Scholar] [CrossRef] [PubMed]
- Ricardo, L.L.; Bernardi, D.I.; Mantovanelli, G.C.; Moreno, B.P.; Mito, M.S.; Silva, A.A.; de Oliveira, R.S., Jr.; Ishii-Iwamoto, E.L.; Sarragiotto, M.H.; Baldoqui, D.C. Phytochemical investigation and phytotoxic activity of aerial parts of oilseed radish (Raphanus sativus var. oleifer Stokes). Biochem. Syst. Ecol. 2018, 78, 52–58. [Google Scholar] [CrossRef]
- Force, L.E.; O’Hare, T.J.; Wong, L.S.; Irving, D.E. Impact of cold storage on glucosinolate levels in seed-sprouts of broccoli, rocket, white radish and kohl-rabi. Postharvest Biol. Technol. 2007, 44, 175–178. [Google Scholar] [CrossRef]
- Hanlon, P.R.; Webber, D.M.; Barnes, D.M. Aqueous extract from Spanish black radish (Raphanus sativus L. Var. niger) induces detoxification enzymes in the HepG2 human hepatoma cell line. J. Agric. Food Chem. 2007, 55, 6439–6446. [Google Scholar] [CrossRef]
- Li, R.; Song, D.; Vriesekoop, F.; Cheng, L.; Yuan, Q.; Liang, H. Glucoraphenin, sulforaphene and antiproliferative capacity of radish sprouts in germinating and thermal processes. Eur. Food Res. Technol. 2017, 243, 547–554. [Google Scholar] [CrossRef]
- Ahn, M.; Kim, J.; Hong, S.; Kim, J.; Ko, H.; Lee, N.H.; Kim, G.O.; Shin, T. Black Radish (Raphanus sativus L. var. niger) Extract mediates its hepatoprotective effect on carbon tetrachloride-induced hepatic injury by attenuating oxidative stress. J. Med. Food 2018, 21, 866–875. [Google Scholar] [CrossRef] [PubMed]
- Syed, S.N.; Rizvi, W.; Kumar, A.; Khan, A.A.; Moin, S.; Ahsan, A. In Vitro Antioxidant and In Vivo Hepatoprotective Activity of Leave Extract of Raphanus sativus in Rats Using CCL 4 Model. Afr. J. Tradit. Complement. Altern. Med. 2014, 11, 102–106. [Google Scholar] [CrossRef] [PubMed]
- Dash, R.N.; Habibuddin, M.; Baruah, D.B. Anthocyanins fraction of red radish (Raphanus sativus L.) protects hepatic damage induced by carbon tetrachloride in albino rats. J. Exp. Integr. Med. 2013, 3, 43–50. [Google Scholar] [CrossRef]
- Kim, J.; Ahn, M.; Kim, S.E.; Lee, H.S.; Kim, H.K.; Kim, G.O.; Shin, T. Hepatoprotective effect of fermented black radish (Raphanus sativus L. var. niger) in CCl4 induced liver injury in rats. J. Prev. Vet. Med. 2017, 41, 143–149. [Google Scholar] [CrossRef]
- Lee, S.W.; Yang, K.M.; Kim, J.K.; Nam, B.H.; Lee, C.M.; Jeong, M.H.; Seo, S.Y.; Kim, G.Y.; Jo, W.S. Effects of white radish (Raphanus sativus) enzyme extract on hepatotoxicity. Toxicol. Res. 2012, 28, 165. [Google Scholar] [CrossRef] [PubMed]
- You, H.; Hao, R.; Li, R.; Zhang, L.; Zhu, Y.; Luo, Y. The effect of radish sourced 4-(Methylthio)-3-butenyl isothiocyanate on ameliorating the severity of high fat diet inducted nonalcoholic fatty liver disease in rats. Int. J. Clin. Exp. Med. 2015, 8, 15910. [Google Scholar] [PubMed]
- Niture, S.K.; Khatri, R.; Jaiswal, A.K. Regulation of Nrf2? an update. Free Radic. Biol. Med. 2014, 66, 36–44. [Google Scholar] [CrossRef]
- Rafatullah, S.; Al-Sheikh, A.; Alqasoumi, S.; Al-Yahya, M.; ElTahir, K.; Galal, A. Protective effect of fresh radish juice (Raphanus sativus L.) against carbon tetrachloride-induced hepatotoxicity. Int. J. Pharmacol. 2008, 4, 130–134. [Google Scholar] [CrossRef]
- Gutiérrez, R.M.; Solís, R.V. Hepatoprotective and inhibition of oxidative stress in liver of Prostechea michuacana. Rec. Nat. Prod. 2009, 3, 46. [Google Scholar]
- Wolf, P.L. Biochemical diagnosis of liver disease. Indian J. Clin. Biochem. 1999, 14, 59–90. [Google Scholar] [CrossRef]
- Seakins, A.; Robinson, D.S. The effect of the administration of carbon tetrachloride on the formation of plasma lipoproteins in the rat. Biochem. J. 1963, 86, 401. [Google Scholar] [CrossRef] [PubMed]
- Torres-Durán, P.V.; Miranda-Zamora, R.; Paredes-Carbajal, M.C.; Mascher, D.; Díaz-Zagoya, J.C.; Juárez-Oropeza, M.A. Spirulina maxima prevents induction of fatty liver by carbon tetrachloride in the rat. IUBMB Life 1998, 44, 787–793. [Google Scholar] [CrossRef]
- Gupta, P.; Kim, B.; Kim, S.H.; Srivastava, S.K. Molecular targets of isothiocyanates in cancer: Recent advances. Mol. Nutr. Food Res. 2014, 58, 1685–1707. [Google Scholar] [CrossRef]
- Rampal, G.; Khanna, N.; Thind, T.S.; Arora, S.; Vig, A.P. Role of isothiocyanates as anticancer agents and their contributing molecular and cellular mechanisms. Med. Chem. Drug Discov. 2012, 3, 79–93. [Google Scholar]
- Schmidt, J.V.; Bradfield, C.A. Ah receptor signaling pathways. Annu. Rev. Cell Dev. Biol. 1996, 12, 55–89. [Google Scholar] [CrossRef]
- Lee, J.M.; Johnson, J.A. An important role of Nrf2-ARE pathway in the cellular defense mechanism. BMB Rep. 2004, 37, 139–143. [Google Scholar] [CrossRef]
- Zhang, L.; Savas, Ü.; Alexander, D.L.; Jefcoate, C.R. Characterization of the mouse CYP1B1 gene identification of an enhancer region that directs aryl hydrocarbon receptor-mediated constitutive and induced expression. J. Biol. Chem. 1998, 273, 5174–5183. [Google Scholar] [CrossRef]
- Dong, L.; Ma, Q.; Whitlock, J.P. DNA Binding by the Heterodimeric Ah Receptor Relationship to dioxin-induced CYP1A1 transcription in vivo. J. Biol. Chem. 1996, 271, 7942–7948. [Google Scholar] [CrossRef]
- Li, W.; Harper, P.A.; Tang, B.K.; Okey, A.B. Regulation of cytochrome P450 enzymes by aryl hydrocarbon receptor in human cells: CYP1A2 expression in the LS180 colon carcinoma cell line after treatment with 2, 3, 7, 8-tetrachlorodibenzo-p-dioxin or 3-methylcholanthrene. Biochem. Pharmacol. 1998, 56, 599–612. [Google Scholar] [CrossRef]
- Favreau, L.V.; Pickett, C.B. Transcriptional regulation of the rat NAD (P) H: Quinone reductase gene. Identification of regulatory elements controlling basal level expression and inducible expression by planar aromatic compounds and phenolic antioxidants. J. Biol. Chem. 1991, 266, 4556–4561. [Google Scholar]
- Clarke, J.D.; Dashwood, R.H.; Ho, E. Multi-targeted prevention of cancer by sulforaphane. Cancer Lett. 2008, 269, 291–304. [Google Scholar] [CrossRef] [PubMed]
- Juge, N.; Mithen, R.F.; Traka, M. Molecular basis for chemoprevention by sulforaphane: A comprehensive review. Cell. Mol. Life Sci. 2007, 64, 1105. [Google Scholar] [CrossRef] [PubMed]
- Yeh, C.T.; Yen, G.C. Chemopreventive functions of sulforaphane: A potent inducer of antioxidant enzymes and apoptosis. J. Funct. Foods 2009, 1, 23–32. [Google Scholar] [CrossRef]
- Ahn, Y.H.; Hwang, Y.; Liu, H.; Wang, X.J.; Zhang, Y.; Stephenson, K.K.; Boronina, T.N.; Cole, R.N.; Dinkova-Kostova, A.T.; Talalay, P.; et al. Electrophilic tuning of the chemoprotective natural product sulforaphane. Proc. Natl. Acad. Sci. USA 2010, 2010. [Google Scholar] [CrossRef] [PubMed]
- Jin, C.Y.; Moon, D.O.; Lee, J.D.; Heo, M.S.; Choi, Y.H.; Lee, C.M.; Park, Y.M.; Kim, G.Y. Sulforaphane sensitizes tumor necrosis factor-related apoptosis-inducing ligand-mediated apoptosis through downregulation of ERK and Akt in lung adenocarcinoma A549 cells. Carcinogenesis 2006, 28, 1058–1066. [Google Scholar] [CrossRef] [PubMed]
- Kaur, I.P. Inhibition of cooked food-induced mutagenesis by dietary constituents: Comparison of two natural isothiocyanates. Food Chem. 2009, 112, 977–981. [Google Scholar]
- Papi, A.; Orlandi, M.; Bartolini, G.; Barillari, J.; Iori, R.; Paolini, M.; Ferroni, F.; Fumo, M.G.; Pedulli, G.F.; Valgimigli, L. Cytotoxic and antioxidant activity of 4-methylthio-3-butenyl isothiocyanate from Raphanus sativus L. (Kaiware Daikon) sprouts. J. Agric. Food Chem. 2008, 56, 875–883. [Google Scholar] [CrossRef] [PubMed]
- Kim, W.K.; Kim, J.H.; Jeong, D.H.; Chun, Y.H.; Kim, S.H.; Cho, K.J.; Chang, M.J. Radish (Raphanus sativus L. leaf) ethanol extract inhibits protein and mRNA expression of ErbB2 and ErbB3 in MDA-MB-231 human breast cancer cells. Nutr. Res. Pract. 2011, 5, 288–293. [Google Scholar] [CrossRef] [PubMed]
- Slamon, D.J.; Clark, G.M. Amplification of c-erbB-2 and Aggressive Human Breast Tumors?: Response. Science 1988, 240, 1796–1798. [Google Scholar] [CrossRef]
- Takeuchi, K.; Ito, F. EGF receptor in relation to tumor development: Molecular basis of responsiveness of cancer cells to EGFR-targeting tyrosine kinase inhibitors. FEBS J. 2010, 277, 316–326. [Google Scholar] [CrossRef]
- Burgess, A.W.; Cho, H.S.; Eigenbrot, C.; Ferguson, K.M.; Garrett, T.P.; Leahy, D.J.; Lemmon, M.A.; Sliwkowski, M.X.; Ward, C.W.; Yokoyama, S. An open-and-shut case? Recent insights into the activation of EGF/ErbB receptors. Mol. Cell 2003, 12, 541–552. [Google Scholar] [CrossRef]
- Citri, A.; Yarden, Y. EGF–ERBB signalling: Towards the systems level. Nat. Rev. Mol. Cell Biol. 2006, 7, 505. [Google Scholar] [CrossRef] [PubMed]
- Janmaat, M.L.; Kruyt, F.A.; Rodriguez, J.A.; Giaccone, G. Response to epidermal growth factor receptor inhibitors in non-small cell lung cancer cells: Limited antiproliferative effects and absence of apoptosis associated with persistent activity of extracellular signal-regulated kinase or Akt kinase pathways. Clin. Cancer Res. 2003, 9, 2316–2326. [Google Scholar] [PubMed]
- Yuan, Z.Q.; Sun, M.; Feldman, R.I.; Wang, G.; Ma, X.L.; Jiang, C.; Coppola, D.; Nicosia, S.V.; Cheng, J.Q. Frequent activation of AKT2 and induction of apoptosis by inhibition of phosphoinositide-3-OH kinase/Akt pathway in human ovarian cancer. Oncogene 2000, 19, 2324. [Google Scholar] [CrossRef] [PubMed]
- Bellacosa, A.; De Feo, D.; Godwin, A.K.; Bell, D.W.; Cheng, J.Q.; Altomare, D.A.; Wan, M.; Dubeau, L.; Scambia, G.; Masciullo, V.; et al. Molecular alterations of the AKT2 oncogene in ovarian and breast carcinomas. Int. J. Cancer 1995, 64, 280–285. [Google Scholar] [CrossRef] [PubMed]
- Sen, P.; Mukherjee, S.; Ray, D.; Raha, S. Involvement of the Akt/PKB signaling pathway with disease processes. Mol. Cell. Biochem. 2003, 253, 241–246. [Google Scholar] [CrossRef] [PubMed]
- Cardone, M.H.; Roy, N.; Stennicke, H.R.; Salvesen, G.S.; Franke, T.F.; Stanbridge, E.; Frisch, S.; Reed, J.C. Regulation of cell death protease caspase-9 by phosphorylation. Science 1998, 282, 1318–1321. [Google Scholar] [CrossRef]
- Brunet, A.; Bonni, A.; Zigmond, M.J.; Lin, M.Z.; Juo, P.; Hu, L.S.; Anderson, M.J.; Arden, K.C.; Blenis, J.; Greenberg, M.E. Akt promotes cell survival by phosphorylating and inhibiting a Forkhead transcription factor. Cell 1999, 96, 857–868. [Google Scholar] [CrossRef]
- Tang, E.D.; Nuñez, G.; Barr, F.G.; Guan, K.L. Negative regulation of the forkhead transcription factor FKHR by Akt. J. Biol. Chem. 1999, 274, 16741–16746. [Google Scholar] [CrossRef]
- Pawlik, A.; Wała, M.; Hać, A.; Felczykowska, A.; Herman-Antosiewicz, A. Sulforaphene, an isothiocyanate present in radish plants, inhibits proliferation of human breast cancer cells. Phytomedicine 2017, 29, 1–10. [Google Scholar] [CrossRef]
- Mondal, A.; Biswas, R.; Rhee, Y.H.; Kim, J.; Ahn, J.C. Sulforaphene promotes Bax/Bcl2, MAPK-dependent human gastric cancer AGS cells apoptosis and inhibits migration via EGFR, p-ERK1/2 downregulation. Gen. Physiol. Biophys. 2016, 35, 25–34. [Google Scholar] [PubMed]
- Lee, S.J.; Zhang, J.; Choi, A.M.; Kim, H.P. Mitochondrial dysfunction induces formation of lipid droplets as a generalized response to stress. Oxid. Med. Cell. Longev. 2013, 2013, 327167. [Google Scholar] [CrossRef] [PubMed]
- Xiao, D.; Powolny, A.A.; Antosiewicz, J.; Hahm, E.R.; Bommareddy, A.; Zeng, Y.; Desai, D.; Amin, S.; Herman-Antosiewicz, A.; Singh, S.V. Cellular responses to cancer chemopreventive agent D, L-sulforaphane in human prostate cancer cells are initiated by mitochondrial reactive oxygen species. Pharm. Res. 2009, 26, 1729–1738. [Google Scholar] [CrossRef] [PubMed]
- Gobeil, S.; Boucher, C.C.; Nadeau, D.; Poirier, G.G. Characterization of the necrotic cleavage of poly (ADP-ribose) polymerase (PARP-1): Implication of lysosomal proteases. Cell Death Differ. 2001, 8, 588. [Google Scholar] [CrossRef] [PubMed]
- Chaitanya, G.V.; Alexander, J.S.; Babu, P.P. PARP-1 cleavage fragments: Signatures of cell-death proteases in neurodegeneration. Cell Commun. Signal. 2010, 8, 31. [Google Scholar] [CrossRef] [PubMed]
- Salah-Abbes, J.B.; Abbes, S.; Houas, Z.; Abdel-Wahhab, M.A.; Oueslati, R. Zearalenone induces immunotoxicity in mice: Possible protective effects of radish extract (Raphanus sativus). J. Pharm. Pharmacol. 2008, 60, 761–770. [Google Scholar] [CrossRef] [PubMed]
- Beevi, S.S.; Mangamoori, L.N.; Subathra, M.; Edula, J.R. Hexane extract of Raphanus sativus L. roots inhibits cell proliferation and induces apoptosis in human cancer cells by modulating genes related to apoptotic pathway. Plant Food Hum. Nutr. 2010, 65, 200–209. [Google Scholar] [CrossRef] [PubMed]
- Yang, M.; Wang, H.; Zhou, M.; Liu, W.; Kuang, P.; Liang, H.; Yuan, Q. The natural compound sulforaphene, as a novel anticancer reagent, targeting PI3K-AKT signaling pathway in lung cancer. Oncotarget 2016, 7, 76656. [Google Scholar] [CrossRef]
- Dal-Ré, R. Worldwide clinical interventional studies on leading causes of death: A descriptive analysis. Ann. Epidemiol. 2011, 21, 727–731. [Google Scholar] [CrossRef]
- Banihani, S. Radish (Raphanus sativus) and diabetes. Nutrients 2017, 9, 1014. [Google Scholar] [CrossRef]
- Vigersky, R.A.; Filmore-Nassar, A.; Glass, A.R. Thyrotropin suppression by metformin. J. Clin. Endocrinol. Metab. 2006, 91, 225–227. [Google Scholar] [CrossRef] [PubMed]
- Vidon, N.; Chaussade, S.; Noel, M.; Franchisseur, C.; Huchet, B.; Bernier, J.J. Metformin in the digestive tract. Diabetes Res. Clin. Pract. 1988, 4, 223–229. [Google Scholar] [CrossRef]
- Taniguchi, H.; Muroi, R.; Kobayashi-Hattori, K.; Uda, Y.; Oishi, Y.; Takita, T. Differing effects of water-soluble and fat-soluble extracts from Japanese radish (Raphanus sativus) sprouts on carbohydrate and lipid metabolism in normal and streptozotocin-induced diabetic rats. J. Nutr. Sci. Vitaminol. 2007, 53, 261–266. [Google Scholar] [CrossRef] [PubMed]
- Broadhurst, C.L.; Polansky, M.M.; Anderson, R.A. Insulin-like biological activity of culinary and medicinal plant aqueous extracts in vitro. J. Agric. Food Chem. 2000, 48, 849–852. [Google Scholar] [CrossRef]
- Taniguchi, H.; Kobayashi-Hattori, K.; Tenmyo, C.; Kamei, T.; Uda, Y.; Sugita-Konishi, Y.; Oishi, Y.; Takita, T. Effect of Japanese radish (Raphanus sativus) sprout (Kaiware-daikon) on carbohydrate and lipid metabolisms in normal and streptozotocin-induced diabetic rats. Phytother. Res. 2006, 20, 274–278. [Google Scholar] [CrossRef]
- Aly, T.A.; Fayed, S.A.; Ahmed, A.M.; Rahim, E.A.E. Effect of Egyptian radish and clover sprouts on blood sugar and lipid metabolisms in diabetic rats. Glob. J. Biotechnol. Biochem. 2015, 10, 16–21. [Google Scholar]
- Antonopoulos, A.S.; Margaritis, M.; Coutinho, P.; Shirodaria, C.; Psarros, C.; Herdman, L.; Krasopoulos, G. Adiponectin as a link between type 2 diabetes and vascular NADPH oxidase activity in the human arterial wall: The regulatory role of perivascular adipose tissue. Diabetes 2015, 64, 2207–2219. [Google Scholar] [CrossRef]
- Okada, Y.; Okada, M.; Sagesaka, Y. Screening of dried plant seed extracts for adiponectin production activity and tumor necrosis factor-alpha inhibitory activity on 3T3-L1 adipocytes. Plant Food. Hum. Nutr. 2010, 65, 225–232. [Google Scholar] [CrossRef]
- Ghoshal, K.; Bhattacharyya, M. Adiponectin: Probe of the molecular paradigm associating diabetes and obesity. World J. Diabetes 2015, 6, 151. [Google Scholar] [CrossRef]
- Fruebis, J.; Tsao, T.S.; Javorschi, S.; Ebbets-Reed, D.; Erickson, M.R.S.; Yen, F.T.; Bihain, B.E.; Lodish, H.F. Proteolytic cleavage product of 30-kDa adipocyte complement-related protein increases fatty acid oxidation in muscle and causes weight loss in mice. Proc. Natl. Acad. Sci. USA 2001, 98, 2005–2010. [Google Scholar] [CrossRef]
- Deepa, S.S.; Dong, L.Q. APPL1: Role in adiponectin signaling and beyond. Am. J. Physiol.-Endoc. Metab. 2009, 296, E22–E36. [Google Scholar] [CrossRef]
- Lihn, A.S.; Pedersen, S.B.; Richelsen, B. Adiponectin: Action, regulation and association to insulin sensitivity. Obes. Rev. 2005, 6, 13–21. [Google Scholar] [CrossRef]
- Zhu, W.; Cheng, K.K.; Vanhoutte, P.M.; Lam, K.S.; Xu, A. Vascular effects of adiponectin: Molecular mechanisms and potential therapeutic intervention. Clin. Sci. 2008, 114, 361–374. [Google Scholar] [CrossRef] [PubMed]
- Yamauchi, T.; Kamon, J.; Waki, H.; Terauchi, Y.; Kubota, N.; Hara, K.; Mori, Y.; Ide, T.; Murakami, K.; Tsuboyama-Kasaoka, N.; et al. The fat-derived hormone adiponectin reverses insulin resistance associated with both lipoatrophy and obesity. Nat. Med. 2001, 7, 941. [Google Scholar] [CrossRef] [PubMed]
- Yoon, M.J.; Lee, G.Y.; Chung, J.J.; Ahn, Y.H.; Hong, S.H.; Kim, J.B. Adiponectin increases fatty acid oxidation in skeletal muscle cells by sequential activation of AMP-activated protein kinase, p38 mitogen-activated protein kinase and peroxisome proliferator–activated receptor α. Diabetes 2006, 55, 2562–2570. [Google Scholar] [CrossRef] [PubMed]
- Maassen, J.A.; Janssen, G.M.C.; Lemkes, H.H.J.P. Mitochondrial diabetes mellitus. J. Endocrinol. Investig. 2002, 25, 477–484. [Google Scholar] [CrossRef] [PubMed]
- Tiwari, A.K. Revisiting “Vegetables” to combat modern epidemic of imbalanced glucose homeostasis. Pharmacog. Mag. 2014, 10, S207. [Google Scholar] [CrossRef]
- Huang, C.F.; Chen, Y.W.; Yang, C.Y.; Lin, H.Y.; Way, T.D.; Chiang, W.; Liu, S.H. Extract of lotus leaf (Nelumbo nucifera) and its active constituent catechin with insulin secretagogue activity. J. Agric. Food Chem. 2011, 59, 1087–1094. [Google Scholar] [CrossRef]
- Chaturvedi, P. Inhibitory response of Raphanus sativus on lipid peroxidation in albino rats. Evid.-Based Complment. Altern. Med. 2008, 5, 55–59. [Google Scholar] [CrossRef]
- Lugasi, A.; Dworschák, E.; Blázovics, A.; Kery, A. Antioxidant and free radical scavenging properties of squeezed juice from black radish (Raphanus sativus L. var niger) root. Phytother. Res. 1998, 12, 502–506. [Google Scholar] [CrossRef]
- Forbes-Hernández, T.Y.; Gasparrini, M.; Afrin, S.; Cianciosi, D.; González-Paramás, A.M.; Santos-Buelga, C.; Bompadre, S. Strawberry (cv. Romina) methanolic extract and anthocyanin-enriched fraction improve lipid profile and antioxidant status in HepG2 cells. Int. J. Mol. Sci. 2017, 18, 1149. [Google Scholar] [CrossRef] [PubMed]
- Fahey, J.W.; Talalay, P. Antioxidant functions of sulforaphane: A potent inducer of phase II detoxication enzymes. Food Chem. Toxicol. 1999, 37, 973–979. [Google Scholar] [CrossRef]
- Kumakura, K.; Kato, R.; Kobayashi, T.; Sekiguchi, A.; Kimura, N.; Takahashi, H.; Takahashi, A.; Matsuoka, H. Nutritional content and health benefits of sun-dried and salt-aged radish (takuan-zuke). Food Chem. 2017, 231, 33–41. [Google Scholar] [CrossRef] [PubMed]
| Name | Class | Tissue | Reference |
|---|---|---|---|
| 1,2-dihydroxyferuloyl-gentiobiose | Phenolic acid | Leaves | [33] |
| 13Z-ß-Carotene | Carotenoids | Sprouts | [34] |
| 3-Butenyl isothiocyanate | Isothiocyanates | Pod & flower | [13] |
| 4-methoxyglucobrassicin | Glucosinolate | Sprouts | [35] |
| 4-OH-glucobrassicin | Glucosinolate | Sprouts | [35] |
| 6-Prenyl-naringenin | Flavonone | Root | [33] |
| 6,7,30,40-Tetrahydroxyisoflavone | Isoflavonoids | Leaves | [33] |
| 9Z-ß–carotene | Carotenoids | Sprouts | [34] |
| α-Carotene | Carotenoids | Sprouts | [34] |
| Antheraxanthin | Carotenoids | Sprouts | [34] |
| Anthocyanin-3-O-(cinnamoyl) sophoroside-5-O-glucoside derivatives | Anthocyanin | Sprouts | [36] |
| Anthocyanin 3-O-sophoroside-5-O-(malonyl) glucoside derivatives | Anthocyanin | Sprouts | [36] |
| Apigenin | Flavonoids | Sprouts & seeds | [37] |
| Apigenin-7-O-neohesperidoside | Flavone | Leaves | [33] |
| Apigenin-7-O-rutinoside | Flavone | Leaves | [33] |
| Caffeic acid | Phenolic acid | Sprouts & seeds | [37] |
| Caffeoylmalic acid | Polyphenols | Leaves | [38] |
| Chrysoeriol-7-O-apiosyl-glucoside | Flavone | Leaves | [33] |
| Cyanidin-3-O-caffeoyl-p-coumaroyl-sophoroside- 5-O-glucoside | Anthocyanin | Root | [33] |
| Cyanidin-3-O-di-p-coumaroyl-sophoroside-5-Omalonylglucoside | Anthocyanin | Root | [33] |
| Cyanidin-3-O-glucoside | Anthocyanin | Leaves | [33] |
| Cyanidin-3-O-p-coumaroyl-feruloyl-sophoroside- 5-O-glucoside | Anthocyanin | Root | [33] |
| Cyanidin-3-O-rhamnoside | Anthocyanin | Leaves | [33] |
| Cyanidin-3-O-sophoroside-5-O-glucoside | Anthocyanin | Leaves | [33] |
| Cyanidin-3-O-sophoroside-5-O-malonylglucoside | Anthocyanin | Leaves, root | [33] |
| Cyanidin-3-O-xylosyl-p-coumaroyl-glucosylgalactoside | Anthocyanin | Leaves | [33] |
| Delphinidin-3-O-rutinoside | Anthocyanin | Root | [33] |
| Dihydro-caffeoyl-3-O-glucuronide | Phenolic acid | Root | [33] |
| Dihydro-kaempherol-3-O-rutinoside | Dihydroflavonol | Leaves | [33] |
| E- ß –carotene | Carotenoids | Sprouts | [34] |
| Ferulic acid | Phenolic acid | Aerial parts | [39] |
| Ferulic acid | Phenolic acid | Sprouts & seeds | [37] |
| Feruloylmalic acid | Phenolic acid | Leaves | [38] |
| Gallic acid | Phenolic acid | Sprouts & seeds | [37] |
| Genistin | Isoflavonoids | Leaves | [33] |
| Glucobrassicin | Glucosinolate | Sprouts | [40] |
| Glucodehydroerucin | Glucosinolate | Sprouts | [40] |
| Glucoraphasatin | Glucosinolate | Whole plant, sprouts | [40,41] |
| Glucoraphenin | Glucosinolate | Sprouts | [42] |
| Indole-3-carbinol | Isothiocyanates | Sprouts | [15] |
| Isorhamnetin-3-O-p-coumaroyl-caffeoylsophorotrioside- 7-O-malonyl-glucoside | Flavanol | Root | [33] |
| Isorhamnetin-3-O-p-coumaroyl-sophorotrioside- 7-O-glucoside | Flavanol | Leaves | [33] |
| Kaemferol | Flavonoids | Sprouts & seeds | [37] |
| Kaempferitrin | Polyphenols | Leaves | [38] |
| Kaempferol-3-O-caffeoyl-sophoroside-7-Oglucoside | Flavanol | Root | [33] |
| Kaempferol-3-O-feruloyl-sophoroside-7-Oglucoside | Flavanol | Root | [33] |
| Kaempferol-3-O-glucoside | Flavanol | Leaves | [33] |
| Kaempferol-3-O-glucosyl-rhamnosyl-glucoside | Flavanol | Leaves | [33] |
| Kaempferol-3-O-p-coumaroyl-sinapoylsophorotrioside-7-O-malonyl-glucoside | Flavanol | Leaves, root | [33] |
| Kaempferol-3-O-p-coumaroyl-sophorotrioside- 7-O-glucoside | Flavanol | Leaves | [33] |
| Kaempferol-3-O-rhamnoside(I) | Flavanol | Leaves | [33] |
| Kaempferol-3-O-rutinoside | Flavanol | Leaves | [33] |
| Kaempferol-3-O-xylosyl-rutinoside | Flavanol | Leaves | [33] |
| Lutein | Carotenoids | Sprouts | [33] |
| Luteolin-7-O-glucoside | Flavone | Leaves | [33] |
| m-Coumaric acid | Phenolic acid | Leaves | [33] |
| Methylgalangin | Flavanol | Leaves | [33] |
| Naringenin-40-O-glucuronide | Flavonone | Leaves | [33] |
| Naringenin-7-O-glucuronide | Flavonone | Leaves | [33] |
| p-Coumaric acid | Phenolic acid | Sprouts & seeds | [37] |
| p-Coumarylmalic acid | Polyphenols | Leaves | [38] |
| Pelargonidin-3-O-caffeoyl-caffeoyl-diglucoside-5-O-malonyl-glucoside | Anthocyanin | Root | [33] |
| Pelargonidin-3-O-feruloyl-diglucoside-5-Oglucoside | Anthocyanin | Leaves | [33] |
| Pelargonidin-3-O-p-coumaroyl-diglucoside-5-Oglucoside | Anthocyanin | Leaves | [33] |
| Pelargonidin-3-O-sambubioside | Anthocyanin | Leaves | [33] |
| Protocatechuic acid | Phenolic acid | Sprouts & seeds | [37] |
| Quercetin | Flavonoids | Sprouts & seeds | [37] |
| Quercetin-3-O-p-coumaroyl-sophoroside-7-Oglucoside | Flavanol | Leaves | [33] |
| Quercetin-3-O-rhamnoside | Flavanol | Leaves | [33] |
| Quercetin-3-O-rhamnosyl-galactoside | Flavanol | Leaves | [33] |
| Sinapic acid | Phenolic acid | Sprouts & seeds | [37] |
| Spinacetin-3-O-(200-p-coumaroyl-glucosyl)(1-6)-(apiosyl(1_2))-glucoside | Flavanol | Root | [33] |
| ß-Cryptoxanthin | Carotenoids | Sprouts | [34] |
| Stigmasterol | Phytosterol | Aerial parts | [39] |
| Sulforaphane | Isothiocyanates | Pod & flower | [13] |
| Sulforaphene | Isothiocyanates | Pod & flower | [13] |
| Violaxanthin | Carotenoids | Sprouts | [34] |
| Zeaxanthin | Carotenoids | Sprouts | [34] |
| β-sitosterol | Phytosterol | Aerial parts | [39] |
| β-sitosterol-3-β-O-D-glucopyranoside | Phytosterol | Aerial parts | [39] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Manivannan, A.; Kim, J.-H.; Kim, D.-S.; Lee, E.-S.; Lee, H.-E. Deciphering the Nutraceutical Potential of Raphanus sativus—A Comprehensive Overview. Nutrients 2019, 11, 402. https://doi.org/10.3390/nu11020402
Manivannan A, Kim J-H, Kim D-S, Lee E-S, Lee H-E. Deciphering the Nutraceutical Potential of Raphanus sativus—A Comprehensive Overview. Nutrients. 2019; 11(2):402. https://doi.org/10.3390/nu11020402
Chicago/Turabian StyleManivannan, Abinaya, Jin-Hee Kim, Do-Sun Kim, Eun-Su Lee, and Hye-Eun Lee. 2019. "Deciphering the Nutraceutical Potential of Raphanus sativus—A Comprehensive Overview" Nutrients 11, no. 2: 402. https://doi.org/10.3390/nu11020402
APA StyleManivannan, A., Kim, J.-H., Kim, D.-S., Lee, E.-S., & Lee, H.-E. (2019). Deciphering the Nutraceutical Potential of Raphanus sativus—A Comprehensive Overview. Nutrients, 11(2), 402. https://doi.org/10.3390/nu11020402







