Maternal Choline Supplementation Modulates Placental Markers of Inflammation, Angiogenesis, and Apoptosis in a Mouse Model of Placental Insufficiency
Abstract
:1. Introduction
2. Materials and Methods
2.1. Mice and Diets
2.2. Tissue Collection and Processing
2.3. Quantitative Real-Time RT-PCR
2.4. LC-MS/MS
2.5. Placental Morphometry
2.6. Placental Apoptosis
2.7. Statistical Analysis
3. Results
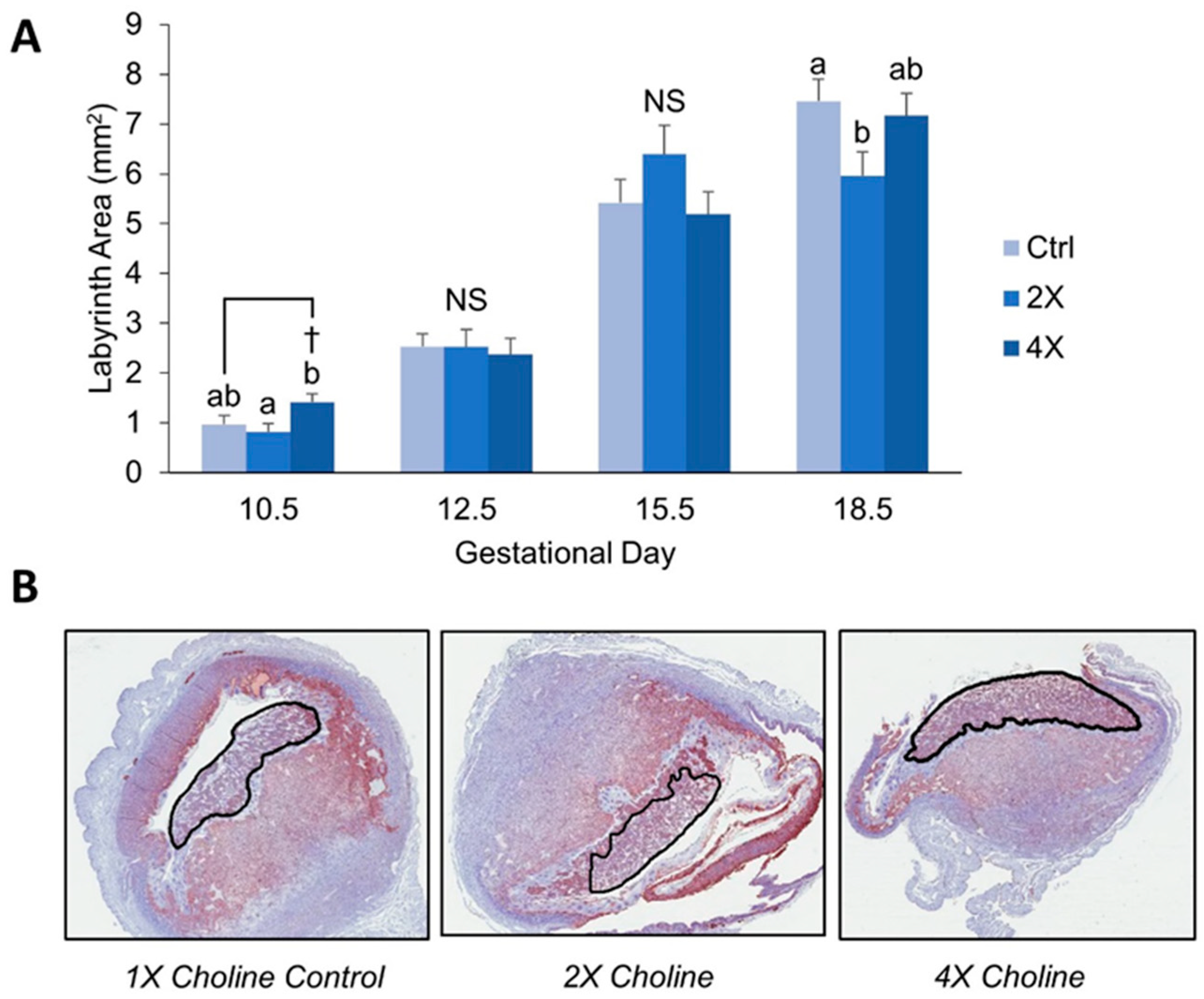
3.1. Placental Morphometry
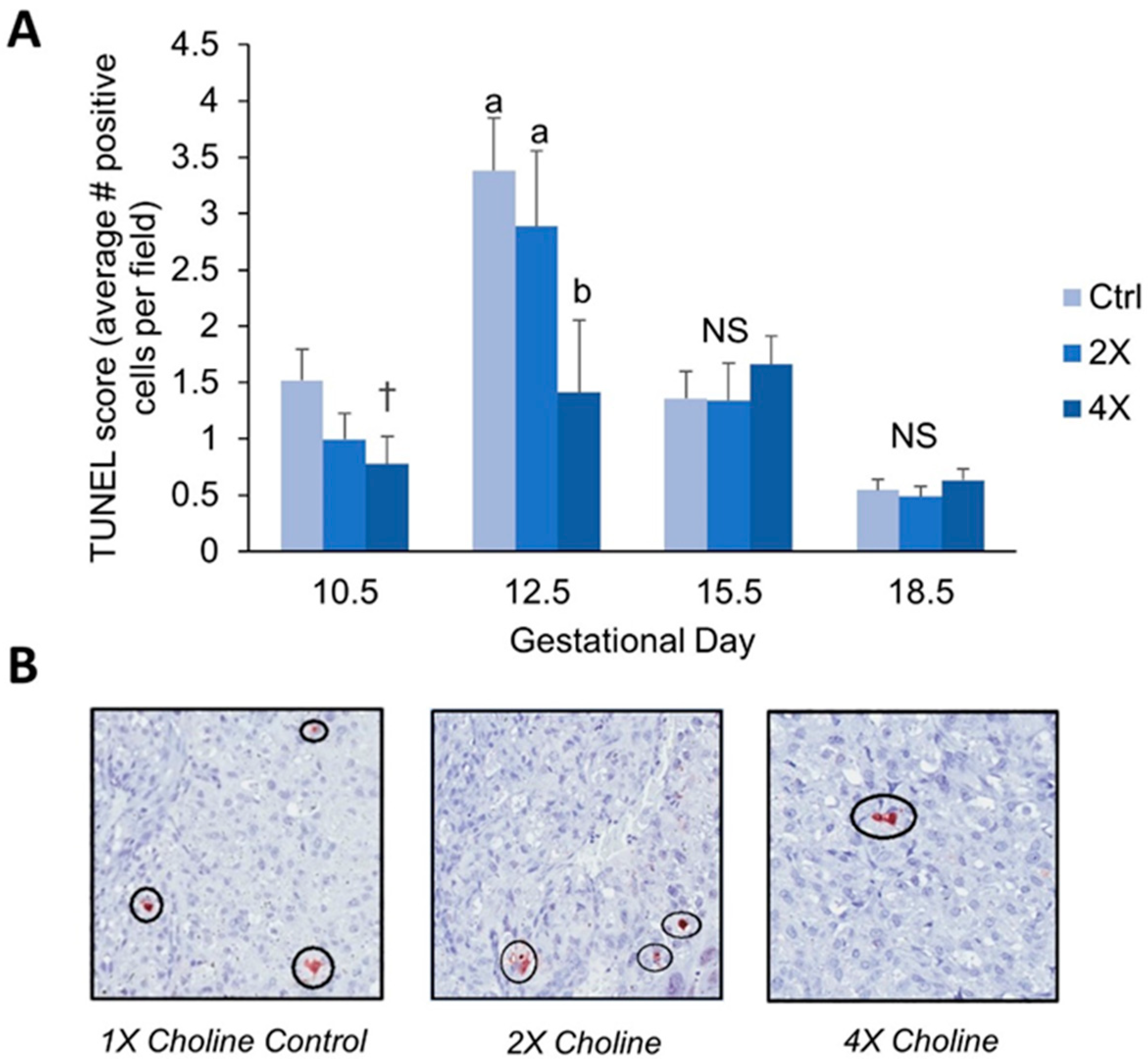
3.2. Placental Apoptosis
3.3. Placental Artery Remodeling
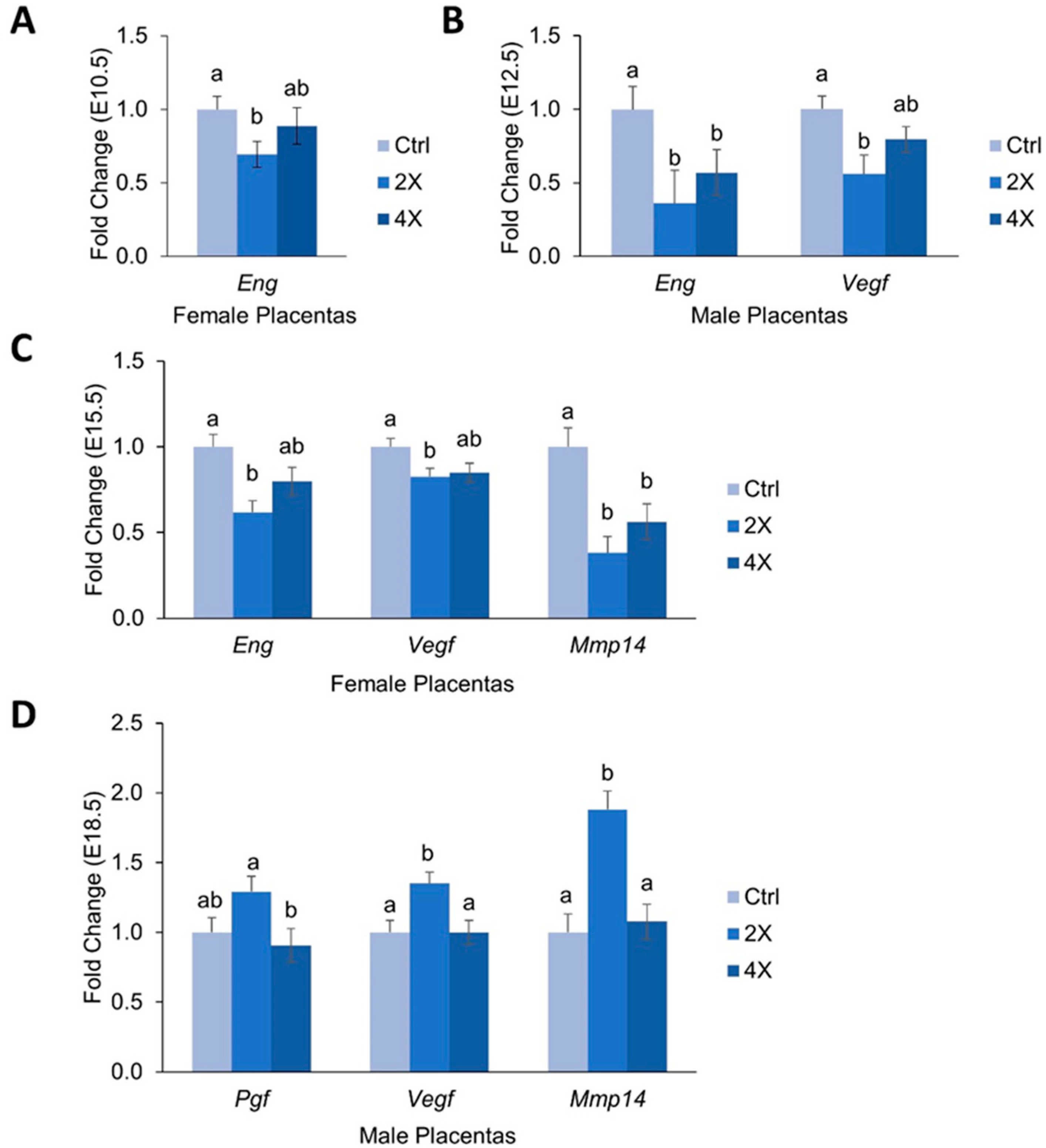
3.4. Placental Expression of Angiogenic Genes
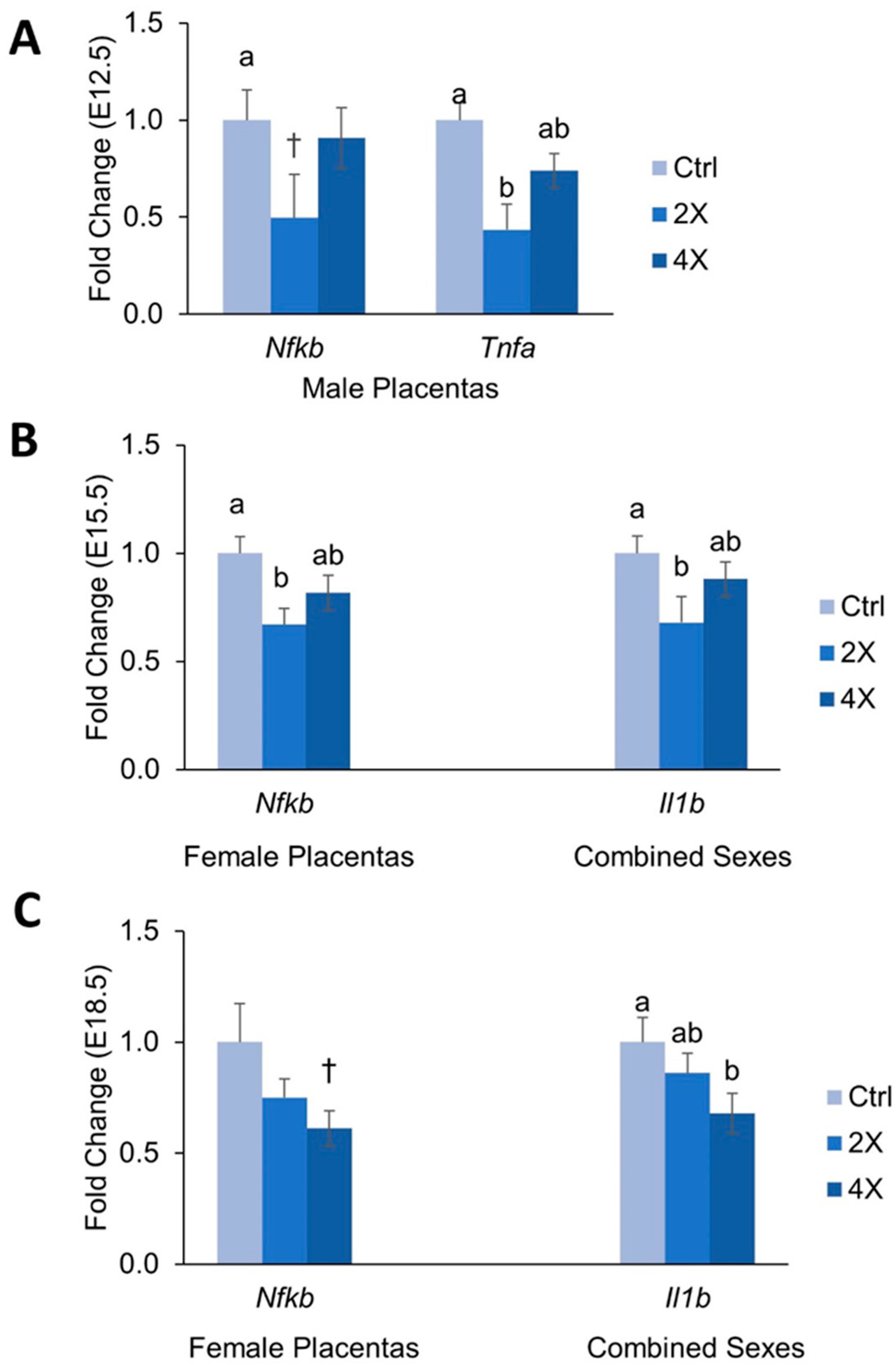
3.5. Placental Expression of Inflammatory Genes
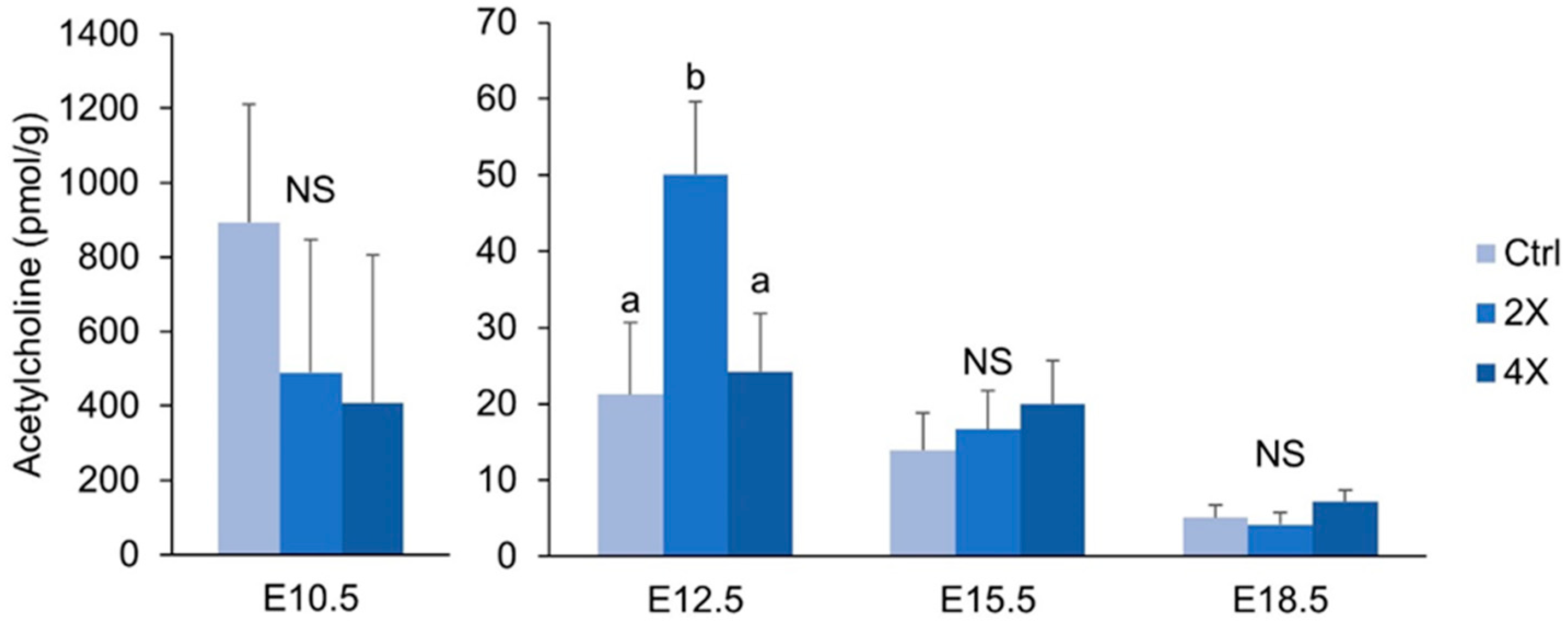
3.6. Placental Acetylcholine
4. Discussion
4.1. Maternal Choline Supplementation in the Dlx3+/− Pregnant Dam Increases Placental Labyrinth Size and Decreases Placental Apoptosis in Mid-Gestation
4.2. Effects of Maternal Choline Supplementation on Placental Inflammatory and Angiogenic Gene Expression Vary by Gestational Time Point and Fetal Sex
4.3. Placental Concentrations of Acetylcholine are Minimally Affected by Maternal Choline Supplementation
4.4. Strengths and Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Chaddha, V.; Viero, S.; Huppertz, B.; Kingdom, J. Developmental biology of the placenta and the origins of placental insufficiency. Semin. Fetal Neonatal Med. 2004, 9, 357–369. [Google Scholar] [CrossRef] [PubMed]
- Azizieh, F.; Raghupathy, R.; Makhseed, M. Maternal Cytokine Production Patterns in Women with Pre-eclampsia. Am. J. Reprod. Immunol. 2005, 54, 30–37. [Google Scholar] [CrossRef] [PubMed]
- Cartwright, J.E.; Whitley, G.S.J. Trophoblast-mediated spiral artery remodelling: A role for apoptosis. J. Anat. 2009, 215, 21–26. [Google Scholar]
- Cheng, M.-H.; Wang, P.-H. Placentation abnormalities in the pathophysiology of preeclampsia. Expert Rev. Mol. Diagn. 2009, 9, 37–49. [Google Scholar] [CrossRef] [PubMed]
- Venkatesha, S.; Toporsian, M.; Lam, C.; Hanai, J.-I.; Mammoto, T.; Kim, Y.M.; Bdolah, Y.; Lim, K.-H.; Yuan, H.-T.; Libermann, T.A.; et al. Soluble endoglin contributes to the pathogenesis of preeclampsia. Nat. Med. 2006, 12, 642–649. [Google Scholar] [CrossRef] [PubMed]
- Boij, R.; Svensson-Arvelund, J.; Nilsson-Ekdahl, K.; Sandholm, K.; Lindahl, T.L.; Pálonek, E.; Garle, M.; Berg, G.; Ernerudh, J.; Jenmalm, M.; et al. Biomarkers of Coagulation, Inflammation, and Angiogenesis are Independently Associated with Preeclampsia. Am. J. Reprod. Immunol. 2012, 68, 258–270. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jiang, X.; West, A.A.; Caudill, M.A. Maternal choline supplementation: A nutritional approach for improving offspring health? Trends Endocrinol. Metab. 2014, 25, 263–273. [Google Scholar] [CrossRef] [PubMed]
- Caudill, M.A. Pre- and Postnatal Health: Evidence of Increased Choline Needs. J. Am. Diet. Assoc. 2010, 110, 1198–1206. [Google Scholar] [CrossRef]
- Jiang, X.; Bar, H.Y.; Yan, J.; Jones, S.; Brannon, P.M.; West, A.A.; Perry, C.A.; Ganti, A.; Pressman, E.; Devapatla, S.; et al. A higher maternal choline intake among third-trimester pregnant women lowers placental and circulating concentrations of the antiangiogenic factor fms-like tyrosine kinase-1 (sFLT1). FASEB J. 2013, 27, 1245–1253. [Google Scholar] [CrossRef]
- Jiang, X.; Jones, S.; Andrew, B.Y.; Ganti, A.; Malysheva, O.V.; Giallourou, N.; Brannon, P.M.; Roberson, M.S.; Caudill, M.A. Choline Inadequacy Impairs Trophoblast Function and Vascularization in Cultured Human Placental Trophoblasts. J. Cell. Physiol. 2014, 229, 1016–1027. [Google Scholar] [CrossRef]
- Kwan, S.T. (Cecilia); King, J.H.; Yan, J.; Jiang, X.; Wei, E.; Fomin, V.G.; Roberson, M.S.; Caudill, M.A. Maternal choline supplementation during murine pregnancy modulates placental markers of inflammation, apoptosis and vascularization in a fetal sex-dependent manner. Placenta 2017, 53, 57–65. [Google Scholar] [PubMed]
- Clark, P.; Brown, J.; Li, S.; Woods, A.; Han, L.; Sones, J.; Preston, R.; Southard, T.; Davisson, R.; Roberson, M.; et al. Distal-less 3 haploinsufficiency results in elevated placental oxidative stress and altered fetal growth kinetics in the mouse. Placenta 2012, 33, 830–838. [Google Scholar] [CrossRef] [Green Version]
- Morasso, M.I.; Grinberg, A.; Robinson, G.; Sargent, T.D.; Mahon, K.A. Placental failure in mice lacking the homeobox gene Dlx3. Proc. Natl. Acad. Sci. USA 1999, 96, 162–167. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- King, J.H.; Kwan, S.T.C.; Yan, J.; Klatt, K.C.; Roberson, M.S.; Caudill, M.A. Maternal Choline Supplementation Alters Fetal Growth Patterns in a Mouse Model of Placental Insufficiency. Nutrients 2017, 9, 765. [Google Scholar] [CrossRef] [PubMed]
- Murthi, P.; Kalionis, B.; Rajaraman, G.; Keogh, R.J.; Costa, F.D.S. The Role of Homeobox Genes in the Development of Placental Insufficiency. Fetal Diagn. Ther. 2012, 32, 225–230. [Google Scholar] [CrossRef] [PubMed]
- Schmittgen, T.D.; Livak, K.J. Analyzing real-time PCR data by the comparative CT method. Nat. Protoc. 2008, 3, 1101–1108. [Google Scholar] [CrossRef] [PubMed]
- Meller, M.; Vadachkoria, S.; Luthy, D.; Williams, M. Evaluation of housekeeping genes in placental comparative expression studies. Placenta 2005, 26, 601–607. [Google Scholar] [CrossRef] [PubMed]
- Mehedint, M.G.; Craciunescu, C.N.; Zeisel, S.H. Maternal dietary choline deficiency alters angiogenesis in fetal mouse hippocampus. Proc. Natl. Acad. Sci. USA 2010, 107, 12834–12839. [Google Scholar] [CrossRef] [Green Version]
- Holm, P.I.; Ueland, P.M.; Kvalheim, G.; Lien, E.A. Determination of Choline, Betaine, and Dimethylglycine in Plasma by a High-Throughput Method Based on Normal-Phase Chromatography-Tandem Mass Spectrometry. Clin. Chem. 2003, 49, 286–294. [Google Scholar] [CrossRef] [Green Version]
- Yan, J.; Wang, W.; Gregory, J.F., III; Malysheva, O.; Brenna, J.T.; Stabler, S.P.; Allen, R.H.; Caudill, M.A. MTHFR C677T genotype influences the isotopic enrichment of one-carbon metabolites in folate-compromised men consuming d9-choline. Am. J. Clin. Nutr. 2011, 93, 348–355. [Google Scholar] [CrossRef]
- Berghorn, K.A.; Clark, P.A.; Encarnacion, B.; DeRegis, C.J.; Folger, J.K.; I Morasso, M.; Soares, M.J.; Wolfe, M.W.; Roberson, M.S. Developmental expression of the homeobox protein Distal-less 3 and its relationship to progesterone production in mouse placenta. J. Endocrinol. 2005, 186, 315–323. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jonge, W.J.; Ulloa, L. The alpha7 nicotinic acetylcholine receptor as a pharmacological target for inflammation. Br. J. Pharmacol. 2009, 151, 915–929. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Arias, H.R.; Richards, V.E.; Ng, D.; Ghafoori, M.E.; Le, V.; Mousa, S.A. Role of non-neuronal nicotinic acetylcholine receptors in angiogenesis. Int. J. Biochem. Cell Biol. 2009, 41, 1441–1451. [Google Scholar] [CrossRef] [PubMed]
- DiFederico, E.; Genbacev, O.; Fisher, S.J. Preeclampsia Is Associated with Widespread Apoptosis of Placental Cytotrophoblasts within the Uterine Wall. Am. J. Pathol. 1999, 155, 293–301. [Google Scholar] [CrossRef] [Green Version]
- Allaire, A. Placental apoptosis in preeclampsia. Obstet. Gynecol. 2000, 96, 271–276. [Google Scholar] [PubMed]
- Ishihara, N.; Matsuo, H.; Murakoshi, H.; Laoag-Fernandez, J.B.; Samoto, T.; Maruo, T. Increased apoptosis in the syncytiotrophoblast in human term placentas complicated by either preeclampsia or intrauterine growth retardation. Am. J. Obstet. Gynecol. 2002, 186, 158–166. [Google Scholar] [CrossRef] [PubMed]
- Mestan, K.; Yu, Y.; Matoba, N.; Cerda, S.; Demmin, B.; Pearson, C.; Ortiz, K.; Wang, X. Placental Inflammatory Response Is Associated with Poor Neonatal Growth: Preterm Birth Cohort Study. Pediatrics 2010, 125, 891–898. [Google Scholar] [CrossRef] [PubMed]
- Christiansen, O.B.; Nielsen, H.S.; Kolte, A.M. Inflammation and miscarriage. Semin. Fetal Neonatal Med. 2006, 11, 302–308. [Google Scholar] [CrossRef]
- Zhang, M.; Han, X.; Bao, J.; Yang, J.; Shi, S.-Q.; Garfield, R.E.; Liu, H. Choline Supplementation During Pregnancy Protects Against Gestational Lipopolysaccharide-Induced Inflammatory Responses. Reprod. Sci. 2017, 25, 74–85. [Google Scholar] [CrossRef]
- Wallace, T.C.; Fulgoni, V.L. Usual Choline Intakes Are Associated with Egg and Protein Food Consumption in the United States. Nutrients 2017, 9, 839. [Google Scholar] [CrossRef]
- Wada, Y.; Ozaki, H.; Abe, N.; Mori, A.; Sakamoto, K.; Nagamitsu, T.; Nakahara, T.; Ishii, K. Role of Vascular Endothelial Growth Factor in Maintenance of Pregnancy in Mice. Endocrinology 2013, 154, 900–910. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lappas, M. Nuclear factor- B mediates placental growth factor induced pro-labour mediators in human placenta. Mol. Hum. Reprod. 2012, 18, 354–361. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Scott, N.M.; Hodyl, N.A.; Murphy, V.E.; Osei-Kumah, A.; Wyper, H.; Hodgson, D.M.; Smith, R.; Clifton, V.L. Placental Cytokine Expression Covaries with Maternal Asthma Severity and Fetal Sex. J. Immunol. 2009, 182, 1411–1420. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kaitu’U-Lino, T.J.; Palmer, K.R.; Whitehead, C.L.; Williams, E.; Lappas, M.; Tong, S. MMP-14 Is Expressed in Preeclamptic Placentas and Mediates Release of Soluble Endoglin. Am. J. Pathol. 2012, 180, 888–894. [Google Scholar] [CrossRef] [PubMed]
- Muralimanoharan, S.; Maloyan, A.; Myatt, L. Evidence of sexual dimorphism in the placental function with severe preeclampsia. Placenta 2013, 34, 1183–1189. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Martin, E.; Smeester, L.; Bommarito, P.A.; Grace, M.R.; Boggess, K.; Kuban, K.; Karagas, M.R.; Marsit, C.J.; O’Shea, T.M.; Fry, R.C.; et al. Sexual epigenetic dimorphism in the human placenta: Implications for susceptibility during the prenatal period. Epigenomics 2017, 9, 267–278. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Schadt, E.E.; Wang, S.; Wang, H.; Arnold, A.P.; Ingram-Drake, L.; Drake, T.A.; Lusis, A.J. Tissue-specific expression and regulation of sexually dimorphic genes in mice. Genome Res. 2006, 16, 995–1004. [Google Scholar] [CrossRef] [Green Version]
- Kwan, S.T. (Cecilia); King, J.H.; Yan, J.; Wang, Z.; Jiang, X.; Hutzler, J.S.; Klein, H.R.; Brenna, J.T.; Roberson, M.S.; Caudill, M.A.; et al. Maternal Choline Supplementation Modulates Placental Nutrient Transport and Metabolism in Late Gestation of Mouse Pregnancy. J. Nutr. 2017, 147, 2083–2092. [Google Scholar]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
King, J.H.; Kwan, S.T.; Yan, J.; Jiang, X.; Fomin, V.G.; Levine, S.P.; Wei, E.; Roberson, M.S.; Caudill, M.A. Maternal Choline Supplementation Modulates Placental Markers of Inflammation, Angiogenesis, and Apoptosis in a Mouse Model of Placental Insufficiency. Nutrients 2019, 11, 374. https://doi.org/10.3390/nu11020374
King JH, Kwan ST, Yan J, Jiang X, Fomin VG, Levine SP, Wei E, Roberson MS, Caudill MA. Maternal Choline Supplementation Modulates Placental Markers of Inflammation, Angiogenesis, and Apoptosis in a Mouse Model of Placental Insufficiency. Nutrients. 2019; 11(2):374. https://doi.org/10.3390/nu11020374
Chicago/Turabian StyleKing, Julia H., Sze Ting (Cecilia) Kwan, Jian Yan, Xinyin Jiang, Vladislav G. Fomin, Samantha P. Levine, Emily Wei, Mark S. Roberson, and Marie A. Caudill. 2019. "Maternal Choline Supplementation Modulates Placental Markers of Inflammation, Angiogenesis, and Apoptosis in a Mouse Model of Placental Insufficiency" Nutrients 11, no. 2: 374. https://doi.org/10.3390/nu11020374
APA StyleKing, J. H., Kwan, S. T., Yan, J., Jiang, X., Fomin, V. G., Levine, S. P., Wei, E., Roberson, M. S., & Caudill, M. A. (2019). Maternal Choline Supplementation Modulates Placental Markers of Inflammation, Angiogenesis, and Apoptosis in a Mouse Model of Placental Insufficiency. Nutrients, 11(2), 374. https://doi.org/10.3390/nu11020374




