The Effects of Glucosamine and Chondroitin Sulfate on Gut Microbial Composition: A Systematic Review of Evidence from Animal and Human Studies
Abstract
1. Introduction
2. Materials and Methods
3. Results
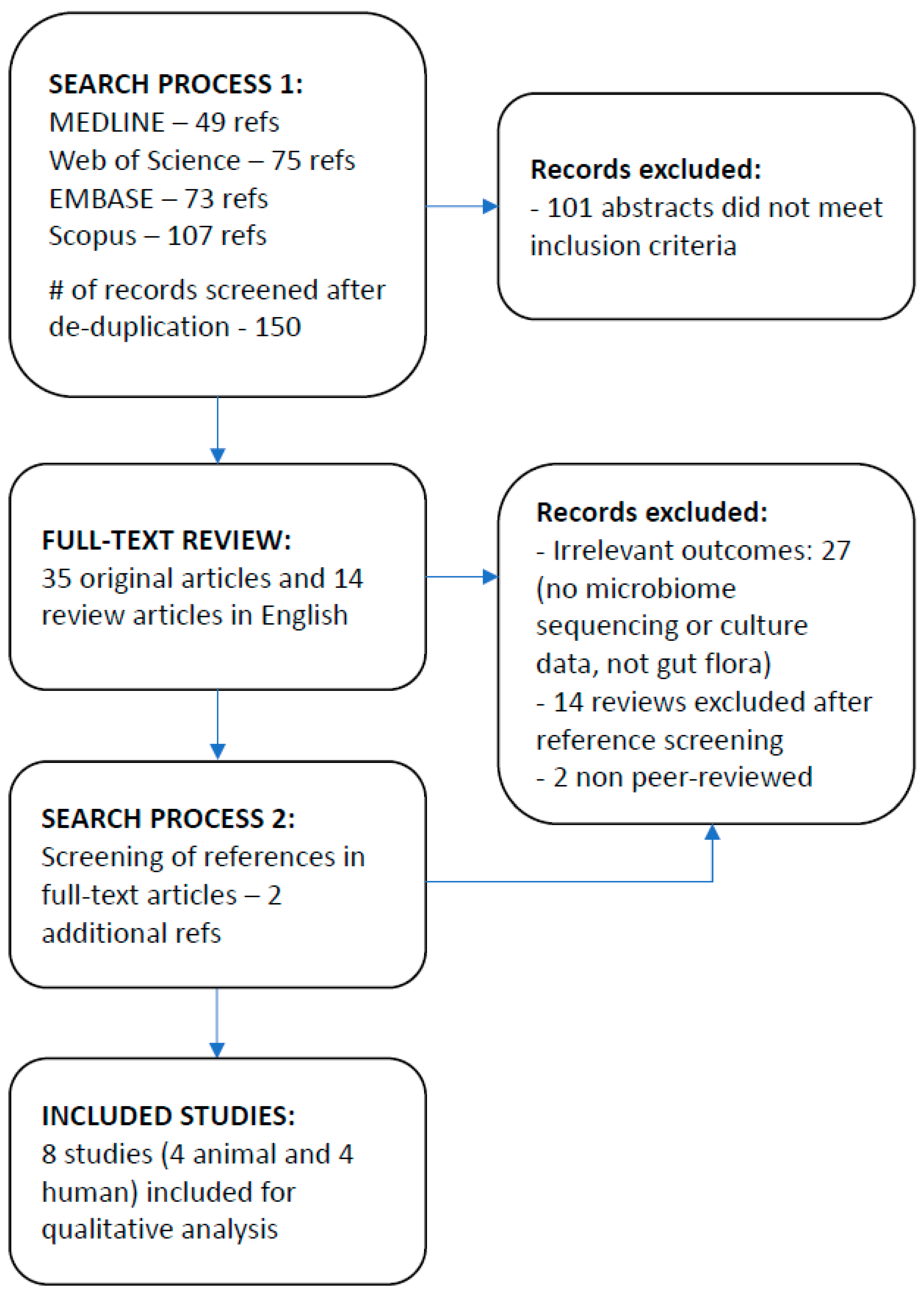
3.1. Study Selection
3.2. Study Characteristics
3.3. Results of Individual Studies
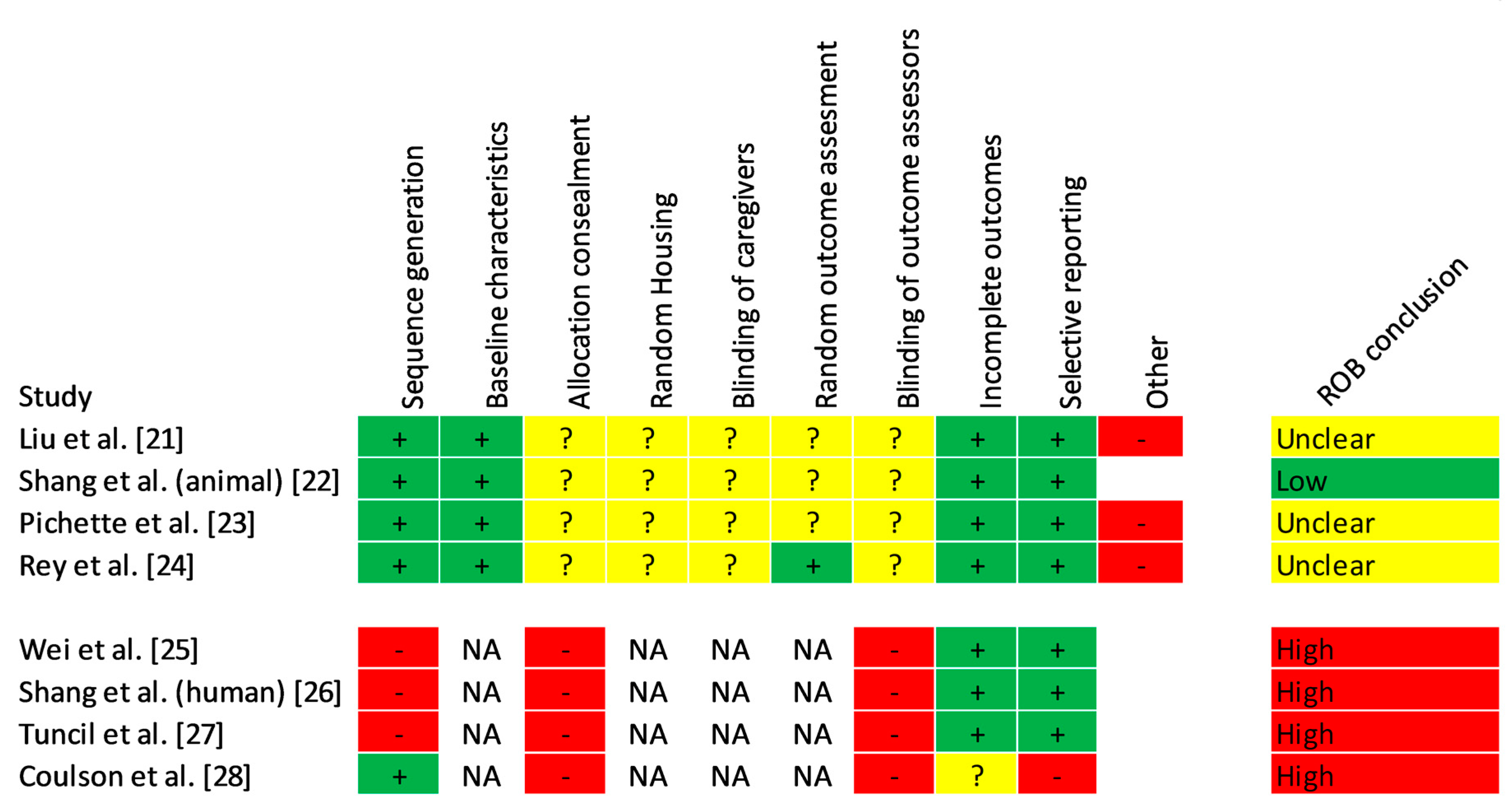
3.4. Risk of Bias
3.5. Synthesis of Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A. Literature Search Algorithms
| #5 | #2 AND #1 Refined by: DOCUMENT TYPES: (ARTICLE) AND LANGUAGES: (ENGLISH) DocType=All document types; Language=All languages; |
| #4 | #2 AND #1 Refined by: DOCUMENT TYPES: (ARTICLE) DocType=All document types; Language=All languages; |
| #3 | #2 AND #1 DocType=All document types; Language=All languages; |
| #2 | TOPIC: (glucosamine or chondroitin) DocType=All document types; Language=All languages; |
| #1 | TOPIC: (microbiom* or microbiot* or microflor* or gut flor* or intestinal flor*) DocType=All document types; Language=All languages; |
| #1. (microbiom* or microbiot* or microflor* or gut flor* or intestinal flor*).mp. (mp=title, abstract, heading word, drug trade name, original title, device manufacturer, drug manufacturer, device trade name, keyword, floating subheading word, candidate term word) |
| #2. (glucosamine or chondroitin).mp. (mp=title, abstract, heading word, drug trade name, original title, device manufacturer, drug manufacturer, device trade name, keyword, floating subheading word, candidate term word) |
| #3. 1 and 2 |
| #4. limit 3 to English language |
| #5. limit 4 to article |
| (TITLE-ABS-KEY (microbiom* OR microbiot* OR microflor* OR (gut AND flor*) OR (intestinal AND flor*))) AND (TITLE-ABS-KEY (glucosamine OR chondroitin)) AND (LIMIT-TO (DOCTYPE, “ar”)) AND (LIMIT-TO (LANGUAGE, “English”)) |
References
- Sawitzke, A.D.; Shi, H.; Finco, M.F.; Dunlop, D.D.; Harris, C.L.; Singer, N.G.; Bradley, J.D.; Silver, D.; Jackson, C.G.; Lane, N.E.; et al. Clinical efficacy and safety of glucosamine, chondroitin sulphate, their combination, celecoxib or placebo taken to treat osteoarthritis of the knee: 2-year results from GAIT. Ann. Rheum. Dis. 2010, 69, 1459–1464. [Google Scholar] [CrossRef] [PubMed]
- Reichenbach, S.; Sterchi, R.; Scherer, M.; Trelle, S.; Bürgi, E.; Bürgi, U.; Dieppe, P.A.; Jüni, P. Meta-analysis: Chondroitin for osteoarthritis of the knee or hip. Ann. Intern. Med. 2007, 146, 580–590. [Google Scholar] [CrossRef]
- Gabay, C.; Medinger-Sadowski, C.; Gascon, D.; Kolo, F.; Finckh, A. Symptomatic effects of chondroitin 4 and chondroitin 6 sulfate on hand osteoarthritis: A randomized, double-blind, placebo-controlled clinical trial at a single center. Arthritis Rheum. 2011, 63, 3383–3391. [Google Scholar] [CrossRef] [PubMed]
- Clegg, D.O.; Reda, D.J.; Harris, C.L.; Klein, M.A.; O’Dell, J.R.; Hooper, M.M.; Bradley, J.D.; Bingham, C.O.; Weisman, M.H.; Jackson, C.G.; et al. Glucosamine, chondroitin sulfate, and the two in combination for painful knee osteoarthritis. N. Engl. J. Med. 2006, 354, 795–808. [Google Scholar] [CrossRef] [PubMed]
- Monfort, J.; Pelletier, J.-P.; Garcia-Giralt, N.; Martel-Pelletier, J. Biochemical basis of the effect of chondroitin sulphate on osteoarthritis articular tissues. Ann. Rheum. Dis. 2008, 67, 735–740. [Google Scholar] [CrossRef]
- Henrotin, Y.; Mobasheri, A.; Marty, M. Is there any scientific evidence for the use of glucosamine in the management of human osteoarthritis? Arthritis Res. Ther. 2012, 14, 201. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, A.; Gilzad-kohan, M.H.; Aghazadeh-Habashi, A.; Jamali, F. Absorption and bioavailability of glucosamine in the rat. J. Pharm. Sci. 2012, 101, 2574–2583. [Google Scholar] [CrossRef]
- Adebowale, A.; Du Jianpin, L.Z.; Leslie James, L.; Eddington Natalie, D. The bioavailability and pharmacokinetics of glucosamine hydrochloride and low molecular weight chondroitin sulfate after single and multiple doses to beagle dogs. Biopharm. Drug Dispos. 2002, 23, 217–225. [Google Scholar] [CrossRef]
- Ronca, F.; Palmieri, L.; Panicucci, P.; Ronca, G. Anti-inflammatory activity of chondroitin sulfate. Osteoarthr. Cartil. 1998, 6, 14–21. [Google Scholar] [CrossRef]
- Chourasia, M.K.; Jain, S.K. Polysaccharides for colon targeted drug delivery. Drug Deliv. 2004, 11, 129–148. [Google Scholar] [CrossRef]
- Barthe, L.; Woodley, J.; Lavit, M.; Przybylski, C.; Philibert, C.; Houin, G. In vitro intestinal degradation and absorption of chondroitin sulfate, a glycosaminoglycan drug. Arzneimittel-Forschung 2004, 54, 286–292. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Huang, S.-Q.; Li, C.-Q.; Xu, Q.; Zeng, Q.-P. Akkermansia muciniphila May Determine Chondroitin Sulfate Ameliorating or Aggravating Osteoarthritis. Front. Microbiol. 2017. [Google Scholar] [CrossRef] [PubMed]
- Benjdia, A.; Berteau, O. Sulfatases and radical SAM enzymes: Emerging themes in glycosaminoglycan metabolism and the human microbiota. Biochem. Soc. Trans. 2016, 44, 109–115. [Google Scholar] [CrossRef] [PubMed]
- Segarra, S.; Martinez-Subiela, S.; Cerda-Cuellar, M.; Martinez-Puig, D.; Munoz-Prieto, A.; Rodriguez-Franco, F.; Rodríguez-Bertos, A.; Allenspach, K.; Velasco, A.; Cerón, J. Oral chondroitin sulfate and prebiotics for the treatment of canine Inflammatory Bowel Disease: A randomized, controlled clinical trial. BMC Vet. Res. 2016, 12, 49. [Google Scholar] [CrossRef] [PubMed]
- Jin, M.; Iwamoto, T.; Yamada, K.; Satsu, H.; Totsuka, M.; Shimizu, M. Disaccharide derived from chondroitin sulfate A suppressed CpG-induced IL-6 secretion in macrophage-like J774.1 cells. Cytokine 2010, 51, 53–59. [Google Scholar] [CrossRef] [PubMed]
- Corfield, A.P.; Myerscough, N.; Bradfield, N.; Corfield, C.D.A.; Gough, M.; Clamp, J.R.; Durdey, P.; Warren, B.F.; Bartolo, D.C.C.; et al. Colonic mucins in ulcerative colitis: Evidence for loss of sulfation. Glycoconj. J. 1996, 13, 809–822. [Google Scholar] [CrossRef] [PubMed]
- Sicard, J.-F.; Vogeleer, P.; Le Bihan, G.; Olivera, Y.R.; Beaudry, F.; Jacques, M.; Harel, J. N-Acetyl-glucosamine influences the biofilm formation of Escherichia coli. Gut Pathog. 2018, 10, 26. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.-S.; Han, S.-Y.; Ryu, K.-Y.; Kim, D.-H. The degradation of glycosaminoglycans by intestinal microflora deteriorates colitis in mice. Inflammation 2009, 32, 27–36. [Google Scholar] [CrossRef] [PubMed]
- Hooijmans, C.R.; Rovers, M.M.; de Vries, R.B.; Leenaars, M.; Ritskes-Hoitinga, M.; Langendam, M.W. SYRCLE’s risk of bias tool for animal studies. BMC Med. Res. Methodol. 2014, 14, 43. [Google Scholar] [CrossRef]
- Lewin, S.; Bohren, M.; Rashidian, A.; Munthe-Kaas, H.; Glenton, C.; Colvin, C.J.; Garside, R.; Noyes, J.; Booth, A.; Tunçalp, A.; et al. Applying GRADE-CERQual to qualitative evidence synthesis findings—Paper 2: How to make an overall CERQual assessment of confidence and create a Summary of Qualitative Findings table. Implement. Sci. 2018, 13, 10. [Google Scholar] [CrossRef]
- Liu, F.; Zhang, N.; Li, Z.; Wang, X.; Shi, H.; Xue, C.; Li, L.W.; Tang, Q. Chondroitin sulfate disaccharides modified the structure and function of the murine gut microbiome under healthy and stressed conditions. Sci. Rep. 2017, 7, 6783. [Google Scholar] [CrossRef] [PubMed]
- Shang, Q.; Shi, J.; Song, G.; Zhang, M.; Cai, C.; Hao, J.; Li, G.; Yu, G. Structural modulation of gut microbiota by chondroitin sulfate and its oligosaccharide. Int. J. Biol. Macromol. 2016, 89, 489–498. [Google Scholar] [CrossRef] [PubMed]
- Pichette, J.; Fynn-Sackey, N.; Gagnon, J. Hydrogen Sulfide and Sulfate Prebiotic Stimulates the Secretion of GLP-1 and Improves Glycemia in Male Mice. Endocrinology 2017, 158, 3416–3425. [Google Scholar] [CrossRef] [PubMed]
- Rey, F.E.; Gonzalez, M.D.; Cheng, J.; Wu, M.; Ahern, P.P.; Gordon, J.I. Metabolic niche of a prominent sulfate-reducing human gut bacterium. Proc. Natl. Acad. Sci. USA 2013, 110, 13582–13587. [Google Scholar] [CrossRef] [PubMed]
- Wei, C.-Y.; Liao, N.-B.; Zhang, Y.; Ye, X.-Q.; Li, S.; Hu, Y.-Q.; Liu, D.-H.; Linhardt, R.J.; Wang, X.; Chen, S.G. In vitro fermentation behaviors of fucosylated chondroitin sulfate from Pearsonothuria graeffei by human gut microflora. Int. J. Biol. Macromol. 2017, 102, 1195–1201. [Google Scholar] [CrossRef] [PubMed]
- Shang, Q.; Yin, Y.; Zhu, L.; Li, G.; Yu, G.; Wang, X. Degradation of chondroitin sulfate by the gut microbiota of Chinese individuals. J. Biol. Macromol. 2016, 1, 112–118. [Google Scholar] [CrossRef] [PubMed]
- Tuncil, Y.E.; Nakatsu, C.H.; Kazem, A.E.; Arioglu-Tuncil, S.; Reuhs, B.; Martens, E.C.; Hamaker, B.R. Delayed utilization of some fast-fermenting soluble dietary fibers by human gut microbiota when presented in a mixture. J. Funct. Food 2017, 32, 347–357. [Google Scholar] [CrossRef]
- Coulson, S.; Butt, H.; Vecchio, P.; Gramotnev, H.; Vitetta, L. Green-lipped mussel extract (Perna canaliculus) and glucosamine sulphate in patients with knee osteoarthritis: Therapeutic efficacy and effects on gastrointestinal microbiota profiles. Inflammopharmacology 2013, 21, 79–90. [Google Scholar] [CrossRef]
- David, L.A.; Maurice, C.F.; Carmody, R.N.; Gootenberg, D.B.; Button, J.E.; Wolfe, B.E.; Ling, A.V.; Devlin, A.S.; Varma, Y.; Fischbach, M.A.; et al. Diet rapidly and reproducibly alters the human gut microbiome. Nature 2014, 505, 559–563. [Google Scholar] [CrossRef]
- Fava, F.; Gitau, R.; Griffin, B.A.; Gibson, G.R.; Tuohy, K.M.; Lovegrove, J.A. The type and quantity of dietary fat and carbohydrate alter faecal microbiome and short-chain fatty acid excretion in a metabolic syndrome ‘at-risk’ population. Int. J. Obes. 2013, 37, 216–223. [Google Scholar] [CrossRef]
- Tuncil, Y.E.; Xiao, Y.; Porter, N.T.; Reuhs, B.L.; Martens, E.C.; Hamaker, B.R. Reciprocal Prioritization to Dietary Glycans by Gut Bacteria in a Competitive Environment Promotes Stable Coexistence. mBio 2017, 8. [Google Scholar] [CrossRef] [PubMed]
- Salyers, A.A.; Vercellotti, J.R.; West, S.E.; Wilkins, T.D. Fermentation of mucin and plant polysaccharides by strains of Bacteroides from the human colon. Appl. Environ. Microbiol. 1977, 33, 319–322. [Google Scholar] [PubMed]
- Martens, E.C.; Chiang, H.C.; Gordon, J.I. Mucosal Glycan Foraging Enhances Fitness and Transmission of a Saccharolytic Human Gut Bacterial Symbiont. Cell Host Microbe 2008, 4, 447–457. [Google Scholar] [CrossRef]
- Roth, C.; Petricevic, M.; John, A.; Goddard-Borger, E.D.; Davies, G.J.; Williams, S.J. Structural and mechanistic insights into a Bacteroides vulgatus retaining N-acetyl-beta-galactosaminidase that uses neighbouring group participation. Chem. Commun. 2016, 52, 11096–11099. [Google Scholar] [CrossRef] [PubMed]
- Desai, M.S.; Seekatz, A.M.; Koropatkin, N.M.; Kamada, N.; Hickey, C.A.; Wolter, M.; Pudlo, N.A.; Kitamoto, S.; Terrapon, N.; Muller, A.; et al. A Dietary Fiber-Deprived Gut Microbiota Degrades the Colonic Mucus Barrier and Enhances Pathogen Susceptibility. Cell 2016, 167, 1339–1353.e21. [Google Scholar] [CrossRef] [PubMed]
- Benjdia, A.; Martens, E.C.; Gordon, J.I.; Berteau, O. Sulfatases and a radical S-adenosyl-l-methionine (AdoMet) enzyme are key for mucosal foraging and fitness of the prominent human gut symbiont, Bacteroides thetaiotaomicron. J. Biol. Chem. 2011, 286, 25973–25982. [Google Scholar] [CrossRef] [PubMed]
- Hori, Y.; Hoshino, J.; Yamazaki, C.; Sekiguchi, T.; Miyauchi, S.; Horie, K. Effects of chondroitin sulfate on colitis induced by dextran sulfate sodium in rats. Jpn. J. Pharmacol. 2001, 85, 155–160. [Google Scholar] [CrossRef]
- Raghavan, V.; Groisman, E.A. Species-specific dynamic responses of gut bacteria to a mammalian glycan. J. Bacteriol 2015, 197, 1538–1548. [Google Scholar] [CrossRef]
- Sugahara, K.; Kojima, T. Specificity studies of bacterial sulfatases by means of structurally defined sulfated oligosaccharides isolated from shark cartilage chondroitin sulfate D. Eur. J. Biochem. 1996, 239, 865–870. [Google Scholar] [CrossRef]
- Ulmer, J.E.; Vilen, E.M.; Namburi, R.B.; Benjdia, A.; Beneteau, J.; Malleron, A.; Bonnaffé, D.; Driguez, P.A.; Descroix, K.; Lassalle, G.; et al. Characterization of glycosaminoglycan (GAG) sulfatases from the human gut symbiont Bacteroides thetaiotaomicron reveals the first GAG-specific bacterial endosulfatase. J. Biol. Chem. 2014, 289, 24289–24303. [Google Scholar] [CrossRef]
- Yang, J.-Y.; Lee, Y.-S.; Kim, Y.; Lee, S.-H.; Ryu, S.; Fukuda, S.; Hase, K.; Yang, C.S.; Lim, H.S.; Kim, M.S.; et al. Gut commensal Bacteroides acidifaciens prevents obesity and improves insulin sensitivity in mice. Mucosal. Immunol. 2017, 10, 104–116. [Google Scholar] [CrossRef] [PubMed]
- Loubinoux, J.; Bronowicki, J.-P.; Pereira, I.A.C.; Mougenel, J.-L.; Faou, A.E. Sulfate-reducing bacteria in human feces and their association with inflammatory bowel diseases. FEMS Microbiol. Ecol. 2002, 40, 107–112. [Google Scholar] [CrossRef] [PubMed]
- Salzman, N.H.; Jong H de Paterson, Y.; Harmsen, H.J.M.; Welling, G.W.; Bos, N.A. Analysis of 16S libraries of mouse gastrointestinal microflora reveals a large new group of mouse intestinal bacteria. Microbiology 2002, 148, 3651–3660. [Google Scholar] [CrossRef] [PubMed]
- Rooks, M.G.; Veiga, P.; Wardwell-Scott, L.H.; Tickle, T.; Segata, N.; Michaud, M.; Gallini, C.A.; Beal, C.; van Hylckama-Vlieg, J.E.; Ballal, S.A.; et al. Gut microbiome composition and function in experimental colitis during active disease and treatment-induced remission. ISME J. 2014, 8, 1403–1417. [Google Scholar] [CrossRef] [PubMed]
- Martín, R.; Martín, C.; Escobedo, S.; Suárez, J.E.; Quirós, L.M. Surface glycosaminoglycans mediate adherence between HeLa cells and Lactobacillus salivarius Lv72. BMC Microbiol. 2013, 13, 210. [Google Scholar] [CrossRef] [PubMed]
- Cartmell, A.; Lowe, E.C.; Baslé, A.; Firbank, S.J.; Ndeh, D.A.; Murray, H.; Terrapon, N.; Lombard, V.; Henrissat, B.; Turnbull, J.E.; et al. How members of the human gut microbiota overcome the sulfation problem posed by glycosaminoglycans. Proc. Natl. Acad. Sci. USA 2017, 114, 7037–7042. [Google Scholar] [CrossRef] [PubMed]
- Rogowski, A.; Briggs, J.A.; Mortimer, J.C.; Tryfona, T.; Terrapon, N.; Lowe, E.C.; Baslé, A.; Morland, C.; Day, A.M.; Zheng, H.; et al. Glycan complexity dictates microbial resource allocation in the large intestine. Nat. Commun. 2015, 6, 7481. [Google Scholar] [CrossRef] [PubMed]
- Mahowald, M.A.; Rey, F.E.; Seedorf, H.; Turnbaugh, P.J.; Fulton, R.S.; Wollam, A.; Shah, N.; Wang, C.; Magrini, V.; Wilson, R.K.; et al. Characterizing a model human gut microbiota composed of members of its two dominant bacterial phyla. Proc. Natl. Acad. Sci. USA 2009, 106, 5859–5864. [Google Scholar] [CrossRef]
- Elderman, M.; Hugenholtz, F.; Belzer, C.; Boekschoten, M.; van Beek, A.; de Haan, B.; Savelkoul, H.; de Vos, P.; Faas, M. Sex and strain dependent differences in mucosal immunology and microbiota composition in mice. Biol. Sex. Differ. 2018, 9, 26. [Google Scholar] [CrossRef]
- Dominianni, C.; Sinha, R.; Goedert, J.J.; Pei, Z.; Yang, L.; Hayes, R.B.; Ahn, J. Sex, body mass index, and dietary fiber intake influence the human gut microbiome. PLoS ONE 2015, 10, e0124599. [Google Scholar] [CrossRef]
- Nakayama, Y.; Narita, T.; Mori, A.; Uesaka, S.; Miyazaki, K.; Ito, H. The effects of age and sex on chondroitin sulfates in normal synovial fluid. Arthritis Rheum. 2002, 46, 2105–2108. [Google Scholar] [CrossRef] [PubMed]
- Bak, Y.-K.; Lampe, J.W.; Sung, M.-K. Effects of dietary supplementation of glucosamine sulfate on intestinal inflammation in a mouse model of experimental colitis. J. Gastroenterol. Hepatol. 2014, 29, 957–963. [Google Scholar] [CrossRef] [PubMed]
- Berry, D.; Mader, E.; Lee, T.K.; Woebken, D.; Wang, Y.; Zhu, D.; Palatinszky, M.; Schintlmeister, A.; Schmid, M.C.; Hanson, B.T.; et al. Tracking heavy water (D2O) incorporation for identifying and sorting active microbial cells. Proc. Natl. Acad. Sci. USA 2015, 112, E194–E203. [Google Scholar] [CrossRef] [PubMed]
| Title | Compound Studied | Dose | Comparator | Duration of Exposure | Participants (N) | Microbiome Assessment Method | Microorganism Identification Reference | ||
|---|---|---|---|---|---|---|---|---|---|
| Animal Studies | |||||||||
| 1 | Liu, F. et al., 2017 [21] | Chondroitin sulfate disaccharides modified the structure and function of the murine gut microbiome under healthy and stressed conditions | CS disaccharides CS-4s and CS-6s | 150 mg/kg | PBS + Both groups fed ad libitum Maintenance Purified Diet | 16 days | Balb/c male mouse (N = 30, randomly assigned to 4 groups) | 16S sequencing | QIIME pipeline, GreenGene database |
| 2 | Shang, Q et al., 2016 [22] | Structural modulation of gut microbiota by chondroitin sulfate and its oligosaccharide | CS isomers CSA, CSC, CSO | 150 mg/kg | Normal saline + Both groups fed standard lab diet | 6 weeks | Kunming male and female mouse (N = 48, randomly assigned to 8 groups, 6 mice each) | 16S sequencing | UPARSE pipeline, database not reported |
| 3 | Pichette, J. et al., 2017 [23] | Hydrogen sulfide and sulfate prebiotic stimulates the secretion of GLP-1 and improves glycemia in male mice | CS | 3% wt/wt | No supplement + Both groups fed diet low in fermentable carbohydrate | 4 weeks | Male wild-type C57BL/6 mouse (N = 26, randomly assigned to 2 groups) | Targeted PCR | NA |
| 4 | Rey, F. et al., 2013 [24] | Metabolic niche of a prominent sulfate-reducing human gut bacterium | CS | 3% wt/wt | No supplement + Both groups fed High fat/high sugar diet | 1 week | NMRI gnobiotic male germ-free mouse, artificial humanized microflora (N = 20 per group) | COPRO-seq | NA |
| Human Studies | |||||||||
| 5 | Wei, C. et al., 2017 [25] | In vitro fermentation behaviors of fucosylated chondroitin sulfate from Pearsonothuria graeffei by human gut microflora | Fucosylated CS from sea cucumber | growth media | none | 72 h | n = 6 healthy human fecal samples/in vitro | 16S-based Real-time quantitative PCR | BLAST |
| 6 | Shang, Q. et al., 2016 [26] | Degradation of chondroitin sulfate by the gut microbiota of Chinese individuals | CS as a sole carbon source in growth medium | growth media | none | 72 h | n = 6 healthy human fecal samples/in vitro | 16S sequencing | BLAST |
| 7 | Tuncil, Y. et al., 2017 [27] | Delayed utilization of some fast-fermenting soluble dietary fibers by human gut microbiota when presented in a mixture | CS as a sole carbon source in growth medium | growth media | none | 12 h | n = 3 healthy human fecal samples/in vitro | 16S sequencing | QIIME pipeline, GreenGene database |
| 8 | Coulson, S. et al., 2013 [28] | Green-lipped mussel extract (Perna canaliculus) and glucosamine sulfate in patients with knee osteoarthritis: Therapeutic efficacy and effects on gastrointestinal microbiota profiles | 12% CS from green-lipped mussel extract or GS | 350 mg of CS/day | 3000 mg of GS/day | 12 weeks | n = 11 men and 29 women (38 total, randomized to green-lipped mussel extract or GS) | MALDI-TOF Mass spectrometry | MALDI Byotyper |
| Total Gut Microbial Diversity | Microorganism | Abundance Change Attributed to Intervention (If Reported) and Direction of Change | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Phylum | Class | Order | Family | Genus | Species | ||||||
| Animal Studies | |||||||||||
| 1 | Liu et al., 2017 [21] | No change in total number of OTUs, Chao1, Shannon, inverse Simpson indices | Bacteroidetes | Bacteroidia | Bacteroidales | Bacteroidaceae | Bacteroides | acidifaciens | 0.12% | 0.22% | Increased * |
| Bacteroidetes | Bacteroidia | Bacteroidales | Bacteroidaceae | Bacteroides | 0.0007% | 0.20% | Increased * | ||||
| Bacteroidetes | Bacteroidia | Bacteroidales | Bacteroidaceae | Bacteroides | 0.0004% | 0.06% | Increased * | ||||
| Firmicutes | Bacilli | Bacillales | Bacillaceae | Lysinibacillus | boronitolerans | 0.0000% | 0.0031% | Increased * | |||
| Proteobacteria | Alphaproteobacteria | Rhizobiales | Brucellaceae | Pseudochrobactrum | 0.0014% | 0.0041% | Increased * | ||||
| Firmicutes | Clostridia | Clostridiales | 0.01% | 0.15% | Increased * | ||||||
| Bacteroidetes | Bacteroidia | Bacteroidales | S24-7 | 0.02% | 0.13% | Increased * | |||||
| 2 | Shang et al., 2016 (animal) a [22] | No consistent difference in the number of OTUs, Chao1, Shannon, Simpson indices | Bacteroidetes | Bacteroidia | Bacteroidales | Rikenellaceae | Alistipes | increased | |||
| Bacteroidetes | Bacteroidia | Bacteroidales | S24-7 | increased in M, decreased in F | |||||||
| Bacteroidetes | Bacteroidia | Bacteroidales | Bacteroidaceae | Bacteroides | decreased in M, increased in F | ||||||
| Proteobacteria | Epsilonproteobacteria | Campylobacterales | Helicobacteraceae | Helicobacter | decreased in M, no change in F | ||||||
| Firmicutes | Clostridia | Clostridiales | Lachnospiraceae | NK4A136 | decreased | ||||||
| 3 | Pichette et al., 2017 b [23] | Not studied | Proteobacteria | Deltaproteobacteria | Desulfovibrionales | Desulfovibrionaceae | Desulfovibrio | piger | 0.10% | 0.13% | Increased * |
| 4 | Rey et al., 2013 b [24] | Not studied | Proteobacteria | Deltaproteobacteria | Desulfovibrionales | Desulfovibrionaceae | Desulfovibrio | piger | 2.30% | 3.50% | Increased * |
| Human Studies | |||||||||||
| 5 | Wei et al., 2017 [25] | Not studied | Firmicutes | Clostridia | Clostridiales | Clostridiaceae | Clostridium | increased | |||
| Actinobacteria | Actinobacteria | Bifidobacteriales | Bifidobacteriaceae | Bifidobacterium | increased | ||||||
| Bacteroidetes | Bacteroidia | Bacteroidales | Prevotellaceae | Prevotella | increased | ||||||
| Firmicutes | Bacilli | Lactobacillales | Lactobacillaceae | Lactobacillus | decreased | ||||||
| Proteobacteria | Gammaproteobacteria | Enterobacterales | Enterobacteriaceae | decreased | |||||||
| 6 | Shang et al., 2016 (human) c [26] | Not studied | Bacteroidetes | Bacteroidia | Bacteroidales | Bacteroidaceae | Bacteroides | thetaiotaomicron | increased | ||
| Bacteroidetes | Bacteroidia | Bacteroidales | Bacteroidaceae | Bacteroides | thetaiotaomicron 82 | increased | |||||
| Bacteroidetes | Bacteroidia | Bacteroidales | Bacteroidaceae | Bacteroides | ovatus | increased | |||||
| Firmicutes | Clostridia | Clostridiales | Clostridiaceae | Clostridium | hathewayi | increased | |||||
| 7 | Tuncil, Y. et al., 2017 [27] | Decreased Shannon index in two of three donor samples; no change in 3rd | Bacteroidetes | Bacteroidia | Bacteroidales | Bacteroidaceae | Bacteroides | increased | |||
| Bacteroidetes | Bacteroidia | Bacteroidales | Tannerellaceae | Parabacteroides | Increased 3 to 20-fold * | ||||||
| 8 | Coulson et al., 2013 d [28] | No difference in number of species before and after treatment in both groups | GLM (Chondroitin Sulfate source) group | ||||||||
| Proteobacteria | Gammaproteobacteria | Enterobacterales | Enterobacteriaceae | 1.93 × 10 7 | 6.70 × 10 7 | increased | |||||
| Firmicutes | Clostridia | Clostridiales | Eubacteriaceae | Eubacteria | 8.14 × 10 9 | 16.10 × 10 9 | increased | ||||
| Firmicutes | Bacilli | Lactobacillales | Streptococcaceae | Streptococcus | 1.09 × 10 7 | 3.65 × 10 7 | increased | ||||
| Bacteroidetes | Bacteroidia | Bacteroidales | Bacteroidaceae | Bacteroides | 1.26 × 10 10 | 2.05 × 10 10 | increased | ||||
| Firmicutes | Clostridia | Clostridiales | Clostridiaceae | Clostridium | 2.04 × 10 9 | 0.95 × 10 9 | decreased | ||||
| Firmicutes | Bacilli | Bacillales | Staphylococcaceae | Staphylococcus | 4.51 × 10 6 | 0.02 × 10 6 | decreased | ||||
| Firmicutes | Bacilli | Lactobacillales | Enterococcaceae | Enterococcus | 1.80 × 10 7 | 0.66 × 10 7 | decreased | ||||
| Yeast | 8.52 × 10 3 | 4.19 × 10 3 | decreased | ||||||||
| Firmicutes | Bacilli | Lactobacillales | Lactobacillaceae | Lactobacillus | 1.08 × 10 9 | 0.45 × 10 9 | decreased | ||||
| Glucosamine Sulfate group | |||||||||||
| Firmicutes | Bacilli | Bacillales | Staphylococcaceae | Staphylococcus | 7.03 × 10 5 | 0.48 × 10 5 | decreased | ||||
| Firmicutes | Bacilli | Lactobacillales | Enterococcaceae | Enterococcus | 6.18 × 10 6 | 0.63 × 10 6 | decreased | ||||
| Firmicutes | Clostridia | Clostridiales | Clostridiaceae | Clostridium | 8.22 × 10 9 | 6.13 × 10 9 | decreased | ||||
| Summary of Review Finding | Studies | Methodological Limitations | Coherence | Adequacy | Relevance | CERQual Assessment | Explanation of CERQual Assessment |
|---|---|---|---|---|---|---|---|
| 1. Chondroitin sulfate supplementation has no effect on the overall gut bacterial diversity in mice and humans | 1, 2, 7, 8 | Moderate concerns: Studies varied in sampling technique, bacteria identification methods, and in reporting of results. | Moderate concerns: three studies reported concordant results, and one reported a decrease in overall diversity in 2 out of 3 subjects | Moderate concerns: evidence comes from two good quality mouse studies, and two low-quality human studies. | No concerns: The presence of this finding in both mouse and human studies conveys higher relevance. | Low | There was moderate coherence among studies, however confidence was downgraded due to moderate concerns about methodology, coherence, and adequacy. |
| 2. Exposure to Chondroitin sulfate increases the relative abundance of genus Bacteroides in mice and humans | 1, 2, 5, 6, 7, 8 | Serious concerns: Studies varied widely in sampling technique, chondroitin exposure methodology (in vivo vs in vitro) and in reporting of results. | No concerns: Both mouse studies and three of the four human studies showed coherence in this finding. Fourth human study did not report | Moderate concerns: There were two good quality mouse studies, and four low-quality human studies. | No concerns: The presence of this finding in both mouse and human studies, and in both sexes conveys higher relevance than if it was observed in one species/one sex only. | Moderate | There was high coherence among studies, and high relevance, however confidence was downgraded due to serious concerns about methodology and adequacy. |
| 3. Chondroitin sulfate supplementation may increase the relative abundance of Desulfovibrio piger in mice | 1, 2, 3, 4 | Moderate concerns: All studies used adequate sampling and in-vivo methodology, but one used artificial gut flora. | Serious concerns: Two studies showed an increase in abundance of D. piger, one showed no increase, and one showed a decrease. | Serious concerns: The two studies that showed an increase in the abundance of D. piger were specifically designed to evaluate D. piger. | Moderate concerns: Evidence for this finding comes from mouse studies only, hence relevance to humans is unclear | Low | There were minor concerns about methods, coherence between studies was low, and relevance unclear. |
| 4. Gut microbial response to chondroitin sulfate exposure may vary depending on chondroitin sulfate source and isoform in mice and humans | 1, 2, 3, 5, 6, 8 | Serious concerns: The wide range of reported changes in the gut microbiome between studies can be explained by several significant limitations in sampling and microbial identification techniques. | No concerns: All studies reported different groups of bacteria in response to CS exposure | Serious concerns: 1–2 studies per isoform or source of CS | Moderate concerns: finding was observed in both animal and human studies, but human studies had very small sample sizes | Low | Given multiple serious limitations in methodology and very low adequacy |
| 5. Gut microbial response to chondroitin sulfate exposure may vary among sexes and individual subjects in mice and humans | 2, 5 | Serious concerns: one of the two studies used a convenience sample of six subjects. | Serious concerns: methods and results were very heterogeneous between the two contributing studies. | Serious concerns: Only one animal and one small in-vitro human study | Moderate concerns: Unclear whether sex differences in mouse microbiome are directly relevant to humans; unclear whether the Chinese human study is relevant to the general population. | Very low | Only two studies, possible methodologic explanations for heterogeneity |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shmagel, A.; Demmer, R.; Knights, D.; Butler, M.; Langsetmo, L.; Lane, N.E.; Ensrud, K. The Effects of Glucosamine and Chondroitin Sulfate on Gut Microbial Composition: A Systematic Review of Evidence from Animal and Human Studies. Nutrients 2019, 11, 294. https://doi.org/10.3390/nu11020294
Shmagel A, Demmer R, Knights D, Butler M, Langsetmo L, Lane NE, Ensrud K. The Effects of Glucosamine and Chondroitin Sulfate on Gut Microbial Composition: A Systematic Review of Evidence from Animal and Human Studies. Nutrients. 2019; 11(2):294. https://doi.org/10.3390/nu11020294
Chicago/Turabian StyleShmagel, Anna, Ryan Demmer, Daniel Knights, Mary Butler, Lisa Langsetmo, Nancy E. Lane, and Kristine Ensrud. 2019. "The Effects of Glucosamine and Chondroitin Sulfate on Gut Microbial Composition: A Systematic Review of Evidence from Animal and Human Studies" Nutrients 11, no. 2: 294. https://doi.org/10.3390/nu11020294
APA StyleShmagel, A., Demmer, R., Knights, D., Butler, M., Langsetmo, L., Lane, N. E., & Ensrud, K. (2019). The Effects of Glucosamine and Chondroitin Sulfate on Gut Microbial Composition: A Systematic Review of Evidence from Animal and Human Studies. Nutrients, 11(2), 294. https://doi.org/10.3390/nu11020294





