Bioactivity Evaluation of a Novel Formulated Curcumin
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials and Methods Used in Manufacturing Formulated Curcumin
2.1.1. Preparation of Curcumin Solid Dispersion Loaded Pellet
2.1.2. Measurements of Particle Size and Zeta Potential
2.1.3. Dissolution Test
2.2. Pharmacokinetic Study
2.2.1. Animal Model
2.2.2. Sample Pretreatment
2.2.3. LC/MS/MS Conditions and Data Analysis
2.3. Cell Lines and Cell Culture
2.4. Drug Preparation and Cell Exposure
2.5. Proliferation and Viability Assays
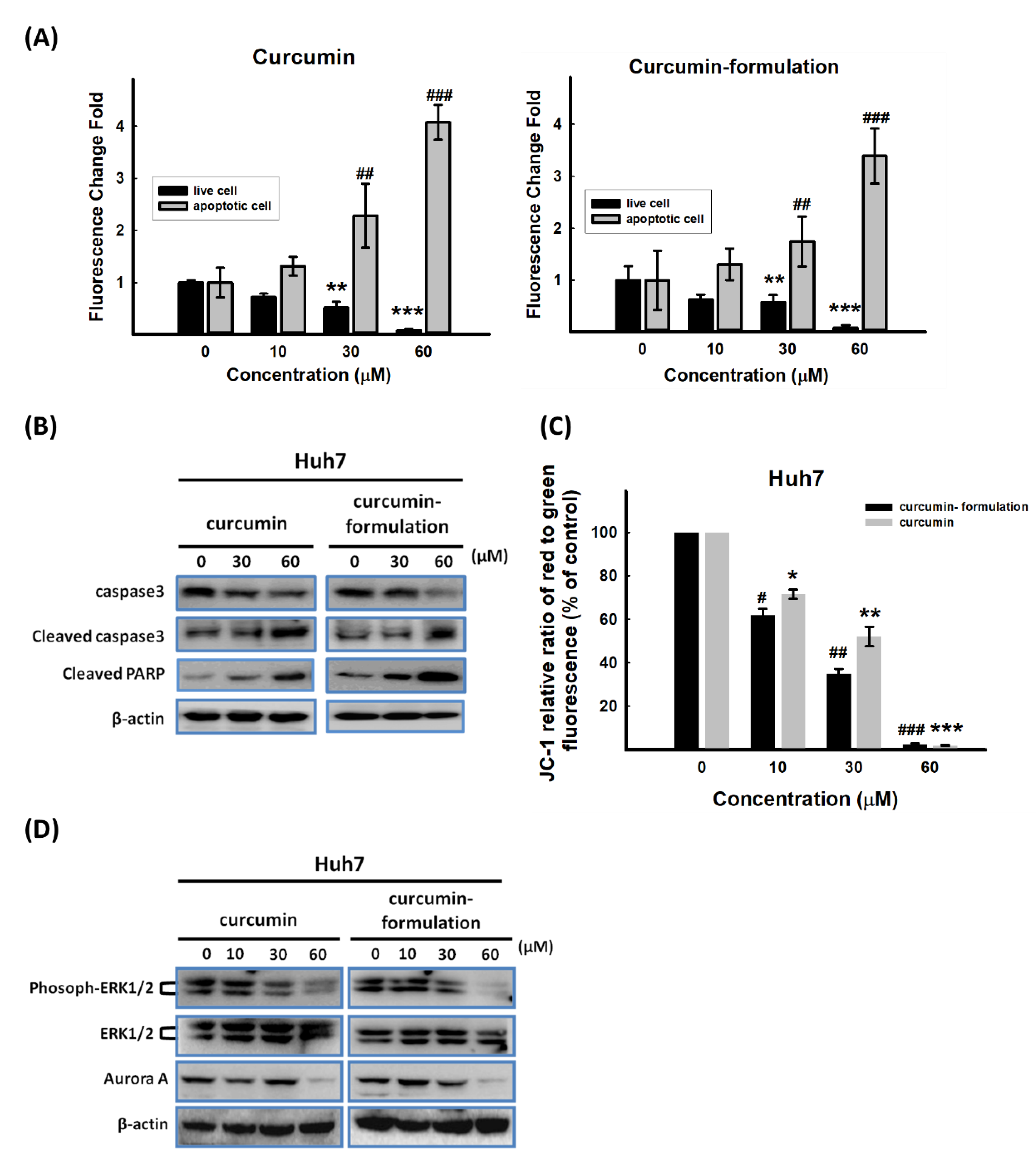
2.6. Mitochondrial Membrane Potential Assay
2.7. Annexin V and Propidium Iodide (PI) Double Staining by Flow Cytometry
2.8. Western Blot
2.9. Cell Cycle Analysis
2.10. Analysis the Similar Mechanism of Gene Expression Profiles of Conventional and Formulated Curcumin Using the L1000 Microarray
2.11. Statistical Analysis
3. Result
3.1. Preparation and Evaluation Formulated Curcumin
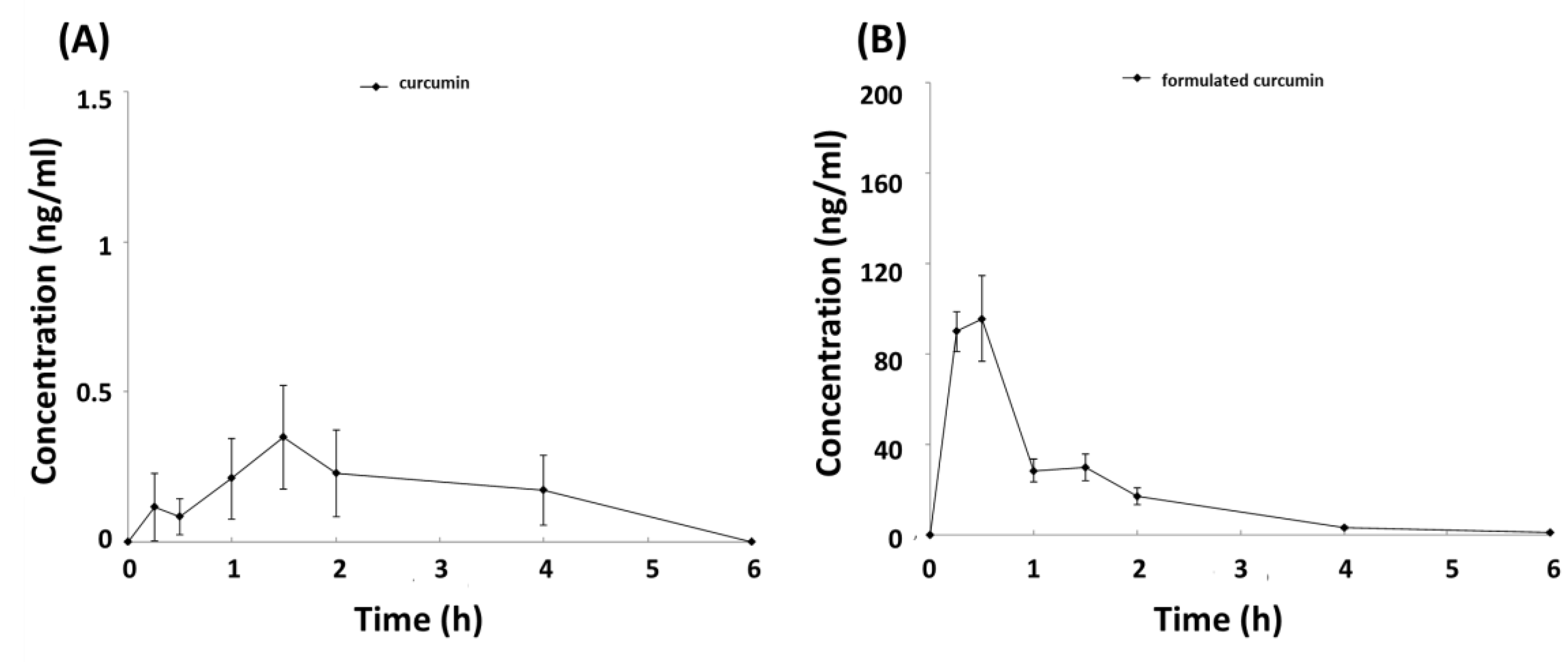
3.2. Oral Administration of Formulated Curcumin Shows an Increase in Bioavailability over Conventional Curcumin via Pharmacokinetic Analysis
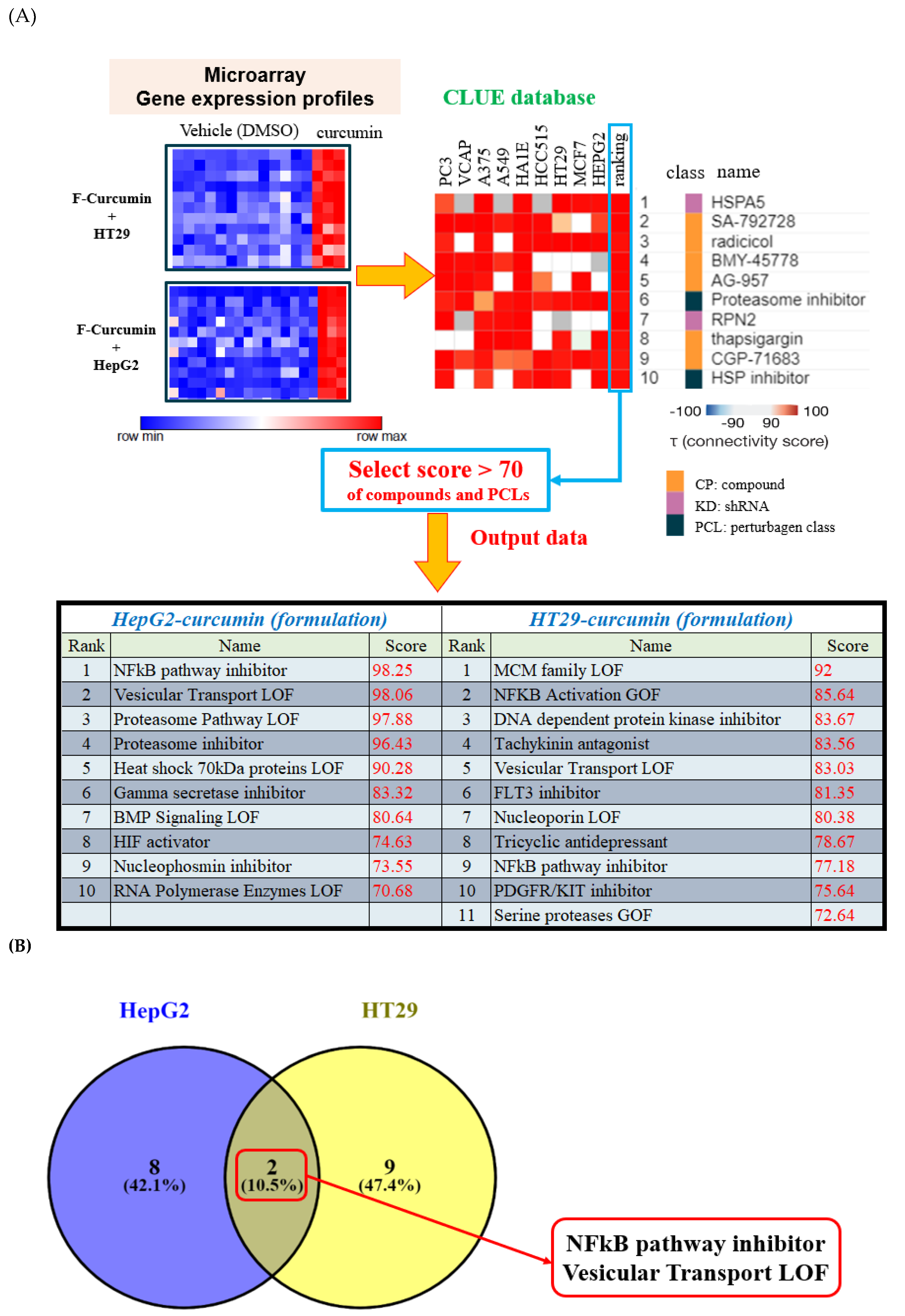
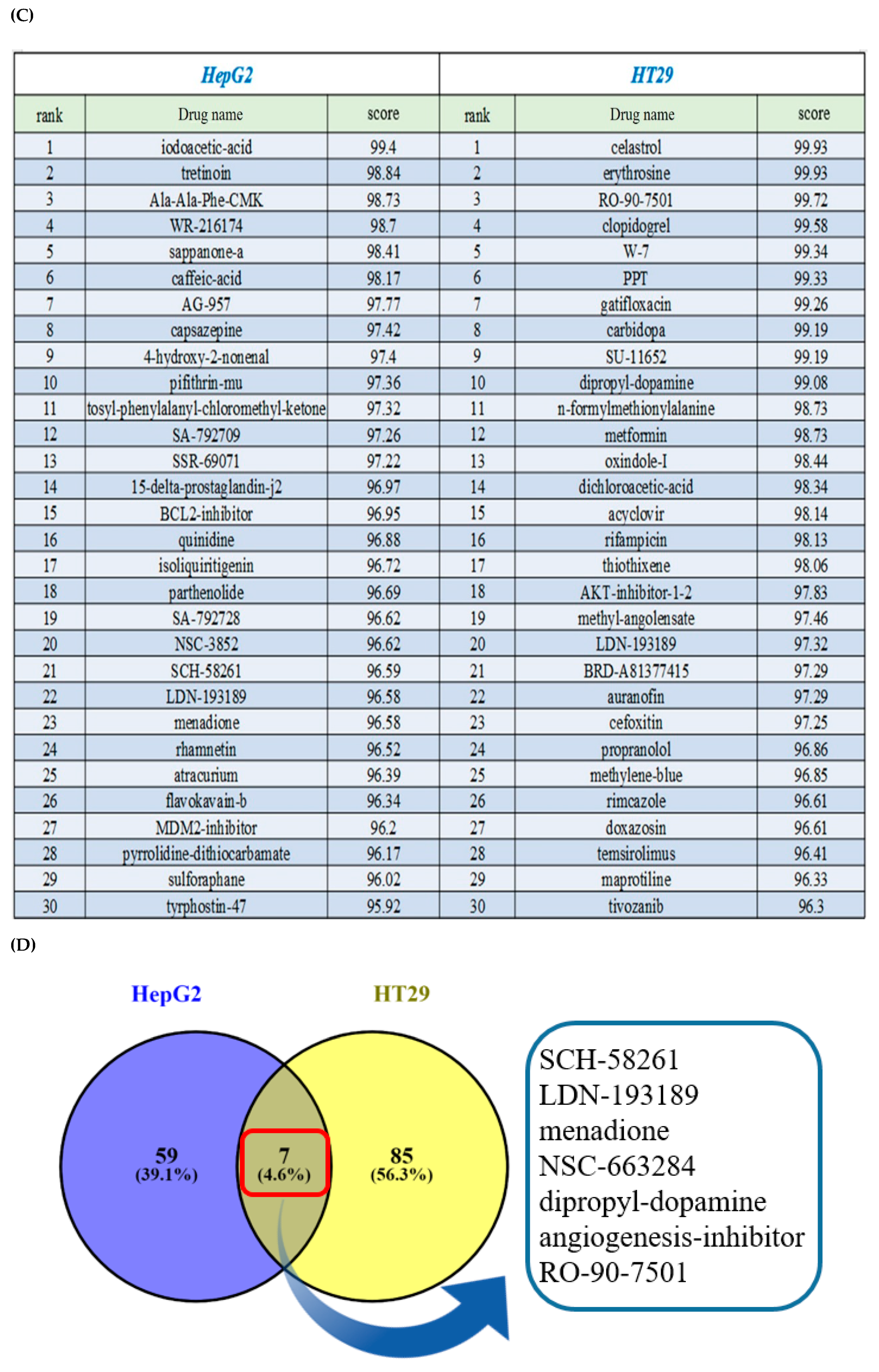
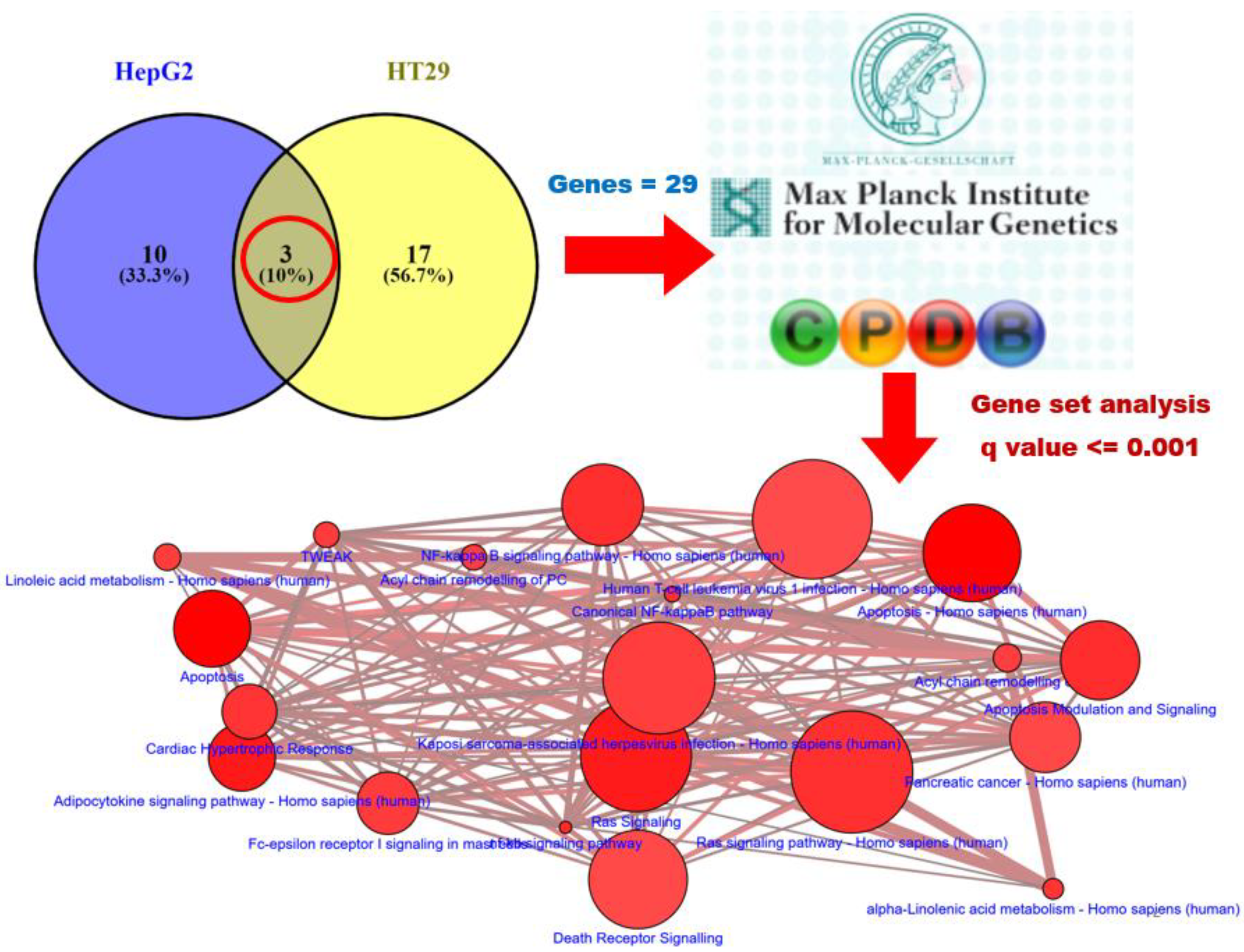
3.3. Gene Expression Analysis of Formulated Curcumin and Prediction of Highly Correlated Pathways
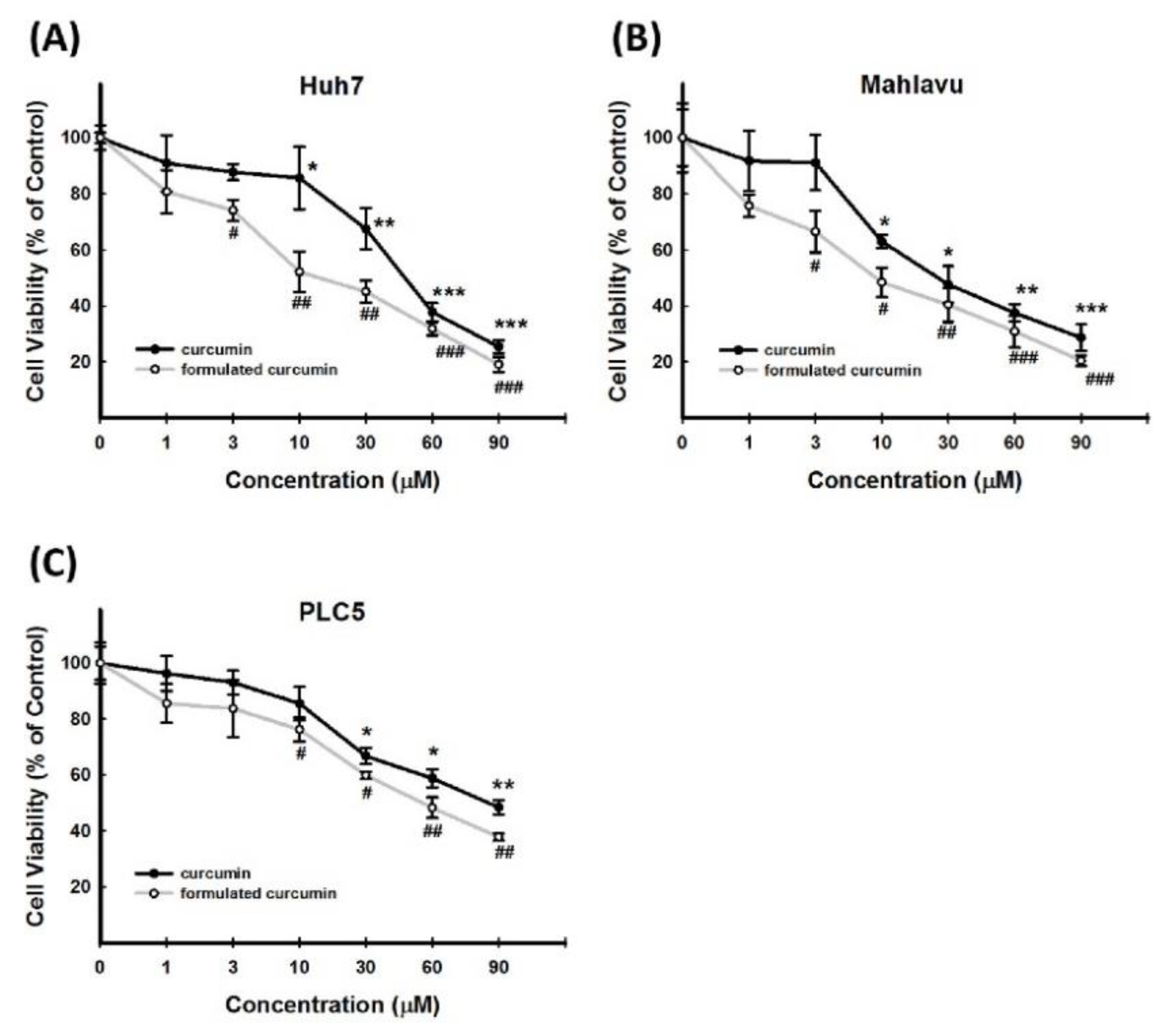
3.4. Formulated Curcumin Displays Stronger Inhibition on Population Growth of Huh7 Cells
3.5. Cytotoxic Effect of Curcumin on Other HCC Cell Lines
3.6. Effects of Curcumin on Apoptosis-Related Protein Expression
3.7. Effects of Combination of Curcumin and Sorafenib
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| Full Name | Abbreviations |
| area under curve | AUC |
| aurora kinase A | AURKA |
| bioavailability | BA |
| dimethyl sulfoxide | DMSO |
| Dulbecco’s modified Eagle’s medium | DMEM |
| enhanced chemiluminescence | ECL |
| extracellular regulated protein kinases | ERK |
| hepatocellular carcinoma | HCC |
| high performance liquid chromatography | HPLC |
| histone deacetylase 1 | HDAC1 |
| janus kinase | JAK |
| limit of quantification | LOQ |
| limits of detection | LOD |
| multidrug resistance protein 1 | MDR1 |
| platelet-derived growth factor receptor beta | PDGFR-β |
| poly (ADP-ribose) polymerase | PARP |
| polydispersity index | PDI |
| polyvinylidene difluoride | PVDF |
| propidium iodide | PI |
| signal transducer and activator of transcription | STAT |
| sodium dodecyl sulfate | SDS |
| Tanshinone IIA | TA |
| The Food and Drug Administration | FDA |
| vascular endothelial growth factor receptor | VEGFR |
References
- Sheikh, B.Y.; Sarker, M.M.R.; Kamarudin, M.N.A.; Ismail, A. Prophetic medicine as potential functional food elements in the intervention of cancer: A review. Biomed. Pharm. 2017, 95, 614–648. [Google Scholar] [CrossRef] [PubMed]
- Shahidi, F. Nutraceuticals, Functional Foods and Dietary Supplements in Health and Disease. J. Food Drug Anal. 2012, 20, 226–230. [Google Scholar]
- Sharma, R.A.; Gescher, A.J.; Steward, W.P. Curcumin: The story so far. Eur. J. Cancer. 2005, 41, 1955–1968. [Google Scholar] [CrossRef] [PubMed]
- Oyama, Y.; Masuda, T.; Nakata, M.; Chikahisa, L.; Yamazaki, Y.; Miura, K.; Okagawa, M. Protective actions of 5’-n-alkylated curcumins on living cells suffering from oxidative stress. Eur. J. Pharmacol. 1998, 360, 65–71. [Google Scholar] [CrossRef]
- Maheshwari, R.K.; Singh, A.K.; Gaddipati, J.; Srimal, R.C. Multiple biological activities of curcumin: A short review. Life Sci. 2006, 78, 2081–2087. [Google Scholar] [CrossRef]
- Ono, K.; Hasegawa, K.; Naiki, H.; Yamada, M. Curcumin has potent anti-amyloidogenic effects for Alzheimer’s beta-amyloid fibrils in vitro. J. Neurosci. Res. 2004, 75, 742–750. [Google Scholar] [CrossRef]
- Pal, S.; Choudhuri, T.; Chattopadhyay, S.; Bhattacharya, A.; Datta, G.K.; Das, T.; Sa, G. Mechanisms of curcumin-induced apoptosis of Ehrlich’s ascites carcinoma cells. Biochem. Biophys. Res. Commun. 2001, 288, 658–665. [Google Scholar] [CrossRef]
- Pal, S.; Bhattacharyya, S.; Choudhuri, T.; Datta, G.K.; Das, T.; Sa, G. Amelioration of immune cell number depletion and potentiation of depressed detoxification system of tumor-bearing mice by curcumin. Cancer Detect. Prev. 2005, 29, 470–478. [Google Scholar] [CrossRef]
- Nautiyal, J.; Banerjee, S.; Kanwar, S.S.; Yu, Y.; Patel, B.B.; Sarkar, F.H.; Majumdar, A.P. Curcumin enhances dasatinib-induced inhibition of growth and transformation of colon cancer cells. Int. J. Cancer 2011, 128, 951–961. [Google Scholar] [CrossRef]
- Duvoix, A.; Blasius, R.; Delhalle, S.; Schnekenburger, M.; Morceau, F.; Henry, E.; Dicato, M.; Diederich, M. Chemopreventive and therapeutic effects of curcumin. Cancer Lett. 2005, 223, 181–190. [Google Scholar] [CrossRef]
- Kunwar, A.; Barik, A.; Mishra, B.; Rathinasamy, K.; Pandey, R.; Priyadarsini, K.I. Quantitative cellular uptake, localization and cytotoxicity of curcumin in normal and tumor cells. Biochim. Biophys. Acta 2008, 1780, 673–679. [Google Scholar] [CrossRef]
- Cao, J.; Liu, Y.; Jia, L.; Zhou, H.M.; Kong, Y.; Yang, G.; Jiang, L.P.; Li, Q.J.; Zhong, L.F. Curcumin induces apoptosis through mitochondrial hyperpolarization and mtDNA damage in human hepatoma G2 cells. Free Radic. Biol. Med. 2007, 43, 968–975. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.Z.; Zhang, B.Y.; Yuan, J.; Mao, J.W.; Mei, W.J. [Effect of curcumin on JAK-STAT signaling pathway in hepatoma cell lines]. Yao Xue Xue Bao Acta Pharm. Sin. 2009, 44, 1434–1439. [Google Scholar]
- Deguchi, A. Curcumin targets in inflammation and cancer. Endocr. Metab. Immune Disord. Drug Targets 2015, 15, 88–96. [Google Scholar] [CrossRef] [PubMed]
- Lin, L.I.; Ke, Y.F.; Ko, Y.C.; Lin, J.K. Curcumin inhibits SK-Hep-1 hepatocellular carcinoma cell invasion in vitro and suppresses matrix metalloproteinase-9 secretion. Oncology 1998, 55, 349–353. [Google Scholar] [CrossRef] [PubMed]
- Magini, A.; Polchi, A.; Di Meo, D.; Mariucci, G.; Sagini, K.; De Marco, F.; Cassano, T.; Giovagnoli, S.; Dolcetta, D.; Emiliani, C. TFEB activation restores migration ability to Tsc1-deficient adult neural stem/progenitor cells. Hum. Mol. Genet. 2017, 26, 3303–3312. [Google Scholar] [CrossRef]
- Pan, M.H.; Huang, T.M.; Lin, J.K. Biotransformation of curcumin through reduction and glucuronidation in mice. Drug Metab. Dispos. Biol. Fate Chem. 1999, 27, 486–494. [Google Scholar]
- Vareed, S.K.; Kakarala, M.; Ruffin, M.T.; Crowell, J.A.; Normolle, D.P.; Djuric, Z.; Brenner, D.E. Pharmacokinetics of curcumin conjugate metabolites in healthy human subjects. Cancer Epidemiol. Biomark. Prev. 2008, 17, 1411–1417. [Google Scholar] [CrossRef]
- Wahlstrom, B.; Blennow, G. A study on the fate of curcumin in the rat. Acta Pharmacol. Toxicol. 1978, 43, 86–92. [Google Scholar] [CrossRef]
- Siviero, A.; Gallo, E.; Maggini, V.; Gori, L.; Mugelli, A.; Firenzuoli, F.; Vannacci, A. Curcumin, a golden spice with a low bioavailability. J. Herb. Med. 2015, 5, 57–70. [Google Scholar] [CrossRef]
- Song, J.X.; Sun, Y.R.; Peluso, I.; Zeng, Y.; Yu, X.; Lu, J.H.; Xu, Z.; Wang, M.Z.; Liu, L.F.; Huang, Y.Y.; et al. A novel curcumin analog binds to and activates TFEB in vitro and in vivo independent of MTOR inhibition. Autophagy 2016, 12, 1372–1389. [Google Scholar] [CrossRef] [PubMed]
- Magini, A.; Polchi, A.; Di Meo, D.; Buratta, S.; Chiaradia, E.; Germani, R.; Emiliani, C.; Tancini, B. Curcumin analogue C1 promotes hex and gal recruitment to the plasma membrane via mTORC1-independent TFEB activation. Int. J. Mol. Sci. 2019, 20, 1363. [Google Scholar] [CrossRef] [PubMed]
- Vikash, K.; Kumar, M.S.; Amit, L.; Singh, R. Multiple unit dosage form-pellet and pelletization techniques: An overview. Int. J. Res. Ayurveda Pharm. 2011, 2, 121–125. [Google Scholar]
- Dhirendra, K.; Lewis, S.; Udupa, N.; Atin, K. Solid dispersions: A review. Pak. J. Pharm. Sci. 2009, 22, 234–246. [Google Scholar]
- Lamb, J.; Crawford, E.D.; Peck, D.; Modell, J.W.; Blat, I.C.; Wrobel, M.J.; Lerner, J.; Brunet, J.P.; Subramanian, A.; Ross, K.N.; et al. The Connectivity Map: Using gene-expression signatures to connect small molecules, genes, and disease. Science 2006, 313, 1929–1935. [Google Scholar] [CrossRef]
- Subramanian, A.; Narayan, R.; Corsello, S.M.; Peck, D.D.; Natoli, T.E.; Lu, X.; Gould, J.; Davis, J.F.; Tubelli, A.A.; Asiedu, J.K.; et al. A Next Generation Connectivity Map: L1000 Platform and the First 1,000,000 Profiles. Cell 2017, 171, 1437–1452. [Google Scholar] [CrossRef]
- Guo, J.; Li, L.; Guo, B.; Liu, D.; Shi, J.; Wu, C.; Chen, J.; Zhang, X.; Wu, J. Mechanisms of resistance to chemotherapy and radiotherapy in hepatocellular carcinoma. Transl. Cancer Res. 2018, 7, 765–781. [Google Scholar] [CrossRef]
- Lohitesh, K.; Chowdhury, R.; Mukherjee, S. Resistance a major hindrance to chemotherapy in hepatocellular carcinoma: An insight. Cancer Cell Int. 2018, 18, 44. [Google Scholar] [CrossRef]
- Llovet, J.M.; Ricci, S.; Mazzaferro, V.; Hilgard, P.; Gane, E.; Blanc, J.F.; de Oliveira, A.C.; Santoro, A.; Raoul, J.L.; Forner, A.; et al. Sorafenib in advanced hepatocellular carcinoma. N. Eng. J. Med. 2008, 359, 378–390. [Google Scholar] [CrossRef]
- Reig, M.; Bruix, J. Sorafenib for hepatocellular carcinoma: Global validation. Gastroenterology 2009, 137, 1171–1173. [Google Scholar] [CrossRef]
- Welker, M.W.; Lubomierski, N.; Gog, C.; Herrmann, E.; Engels, K.; Vogl, T.J.; Bechstein, W.O.; Zeuzem, S.; Trojan, J. Efficacy and safety of sorafenib in advanced hepatocellular carcinoma under daily practice conditions. J. Chemother. 2010, 22, 205–211. [Google Scholar] [CrossRef] [PubMed]
- Morimoto, M.; Numata, K.; Kondo, M.; Hidaka, H.; Takada, J.; Shibuya, A.; Kobayashi, S.; Ohkawa, S.; Okuse, C.; Morita, S.; et al. Higher discontinuation and lower survival rates are likely in elderly Japanese patients with advanced hepatocellular carcinoma receiving sorafenib. Hepatol. Res. 2011, 41, 296–302. [Google Scholar] [CrossRef] [PubMed]
- Liao, S.C.; Hsu, H.W.; Chuang, K.L.; Huang, Z.Y.; Lin, K.T.; Hsu, W.H.; Chang, K.H.; Huang, C.F.; Su, C.L. Using the Pleiotropic Characteristics of Curcumin to Validate the Potential Application of a Novel Gene Expression Screening Platform. Nutrients 2019, 11, 1397. [Google Scholar] [CrossRef]
- Jendrossek, V.; Handrick, R.; Belka, C. Celecoxib activates a novel mitochondrial apoptosis signaling pathway. FASEB J. 2003, 17, 1547–1549. [Google Scholar] [CrossRef] [PubMed]
- Wieder, T.; Essmann, F.; Prokop, A.; Schmelz, K.; Schulze-Osthoff, K.; Beyaert, R.; Dorken, B.; Daniel, P.T. Activation of caspase-8 in drug-induced apoptosis of B-lymphoid cells is independent of CD95/Fas receptor-ligand interaction and occurs downstream of caspase-3. Blood 2001, 97, 1378–1387. [Google Scholar] [CrossRef]
- Engels, I.H.; Stepczynska, A.; Stroh, C.; Lauber, K.; Berg, C.; Schwenzer, R.; Wajant, H.; Janicke, R.U.; Porter, A.G.; Belka, C.; et al. Caspase-8/FLICE functions as an executioner caspase in anticancer drug-induced apoptosis. Oncogene 2000, 19, 4563–4573. [Google Scholar] [CrossRef]
- Shin, H.K.; Kim, J.; Lee, E.J.; Kim, S.H. Inhibitory effect of curcumin on motility of human oral squamous carcinoma YD-10B cells via suppression of ERK and NF-kappaB activations. Phytother. Res. 2010, 24, 577–582. [Google Scholar] [CrossRef]
- Xie, Y.Q.; Wu, X.B.; Tang, S.Q. Curcumin treatment alters ERK-1/2 signaling in vitro and inhibits nasopharyngeal carcinoma proliferation in mouse xenografts. Int. J. Clin. Exp. Med. 2014, 7, 108–114. [Google Scholar]
- Guo, Y.; Shan, Q.; Gong, Y.; Lin, J.; Shi, F.; Shi, R.; Yang, X. Curcumin induces apoptosis via simultaneously targeting AKT/mTOR and RAF/MEK/ERK survival signaling pathways in human leukemia THP-1 cells. Pharmazie 2014, 69, 229–233. [Google Scholar]
- Wolanin, K.; Magalska, A.; Mosieniak, G.; Klinger, R.; McKenna, S.; Vejda, S.; Sikora, E.; Piwocka, K. Curcumin affects components of the chromosomal passenger complex and induces mitotic catastrophe in apoptosis-resistant Bcr-Abl-expressing cells. Mol. Cancer Res. 2006, 4, 457–469. [Google Scholar] [CrossRef]
- Liu, H.S.; Ke, C.S.; Cheng, H.C.; Huang, C.Y.; Su, C.L. Curcumin-induced mitotic spindle defect and cell cycle arrest in human bladder cancer cells occurs partly through inhibition of aurora A. Mol. Pharmacol. 2011, 80, 638–646. [Google Scholar] [CrossRef] [PubMed]
- Ke, C.S.; Liu, H.S.; Yen, C.H.; Huang, G.C.; Cheng, H.C.; Huang, C.Y.; Su, C.L. Curcumin-induced Aurora-A suppression not only causes mitotic defect and cell cycle arrest but also alters chemosensitivity to anticancer drugs. J. Nutr. Biochem. 2014, 25, 526–539. [Google Scholar] [CrossRef] [PubMed]
- Li, B.; Takeda, T.; Tsuiji, K.; Wong, T.F.; Tadakawa, M.; Kondo, A.; Nagase, S.; Yaegashi, N. Curcumin induces cross-regulation between autophagy and apoptosis in uterine leiomyosarcoma cells. Int. J. Gynecol. Cancer 2013, 23, 803–808. [Google Scholar] [CrossRef] [PubMed]
- Zhou, T.; Ye, L.; Bai, Y.; Sun, A.; Cox, B.; Liu, D.; Li, Y.; Liotta, D.; Snyder, J.P.; Fu, H.; et al. Autophagy and apoptosis in hepatocellular carcinoma induced by EF25-(GSH)2: A novel curcumin analog. PLoS ONE 2014, 9. [Google Scholar] [CrossRef] [PubMed]
- Lao, C.D.; Ruffin, M.T.T.; Normolle, D.; Heath, D.D.; Murray, S.I.; Bailey, J.M.; Boggs, M.E.; Crowell, J.; Rock, C.L.; Brenner, D.E. Dose escalation of a curcuminoid formulation. BMC Complementary Altern. Med. 2006, 6, 10. [Google Scholar] [CrossRef]
- Sharma, R.A.; Euden, S.A.; Platton, S.L.; Cooke, D.N.; Shafayat, A.; Hewitt, H.R.; Marczylo, T.H.; Morgan, B.; Hemingway, D.; Plummer, S.M.; et al. Phase I clinical trial of oral curcumin: Biomarkers of systemic activity and compliance. Clin. Cancer Res. 2004, 10, 6847–6854. [Google Scholar] [CrossRef]
- Vasconcelos, T.; Sarmento, B.; Costa, P. Solid dispersions as strategy to improve oral bioavailability of poor water soluble drugs. Drug Discov. Today 2007, 12, 1068–1075. [Google Scholar] [CrossRef]
- Ye, G.; Wang, S.; Heng, P.W.; Chen, L.; Wang, C. Development and optimization of solid dispersion containing pellets of itraconazole prepared by high shear pelletization. Int. J. Pharm. 2007, 337, 80–87. [Google Scholar] [CrossRef]
- Li, J.; Liu, P.; Liu, J.P.; Zhang, W.L.; Yang, J.K.; Fan, Y.Q. Novel Tanshinone II A ternary solid dispersion pellets prepared by a single-step technique: In vitro and in vivo evaluation. Eur. J. Pharm. Biopharm. 2012, 80, 426–432. [Google Scholar] [CrossRef]
- Yang, K.Y.; Lin, L.C.; Tseng, T.Y.; Wang, S.C.; Tsai, T.H. Oral bioavailability of curcumin in rat and the herbal analysis from Curcuma longa by LC-MS/MS. J. Chromatogr. B 2007, 853, 183–189. [Google Scholar] [CrossRef]
- Khalil, N.M.; do Nascimento, T.C.; Casa, D.M.; Dalmolin, L.F.; de Mattos, A.C.; Hoss, I.; Romano, M.A.; Mainardes, R.M. Pharmacokinetics of curcumin-loaded PLGA and PLGA-PEG blend nanoparticles after oral administration in rats. Colloids Surf. B Biointerfaces 2013, 101, 353–360. [Google Scholar] [CrossRef] [PubMed]
- Marczylo, T.H.; Verschoyle, R.D.; Cooke, D.N.; Morazzoni, P.; Steward, W.P.; Gescher, A.J. Comparison of systemic availability of curcumin with that of curcumin formulated with phosphatidylcholine. Cancer Chemother. Pharmacol. 2007, 60, 171–177. [Google Scholar] [CrossRef] [PubMed]
- Darvesh, A.S.; Aggarwal, B.B.; Bishayee, A. Curcumin and liver cancer: A review. Curr. Pharm. Biotechnol. 2012, 13, 218–228. [Google Scholar] [CrossRef] [PubMed]
- Lin, Z.Z.; Jeng, Y.M.; Hu, F.C.; Pan, H.W.; Tsao, H.W.; Lai, P.L.; Lee, P.H.; Cheng, A.L.; Hsu, H.C. Significance of Aurora B overexpression in hepatocellular carcinoma. Aurora B Overexpression in HCC. BMC Cancer 2010, 10, 461. [Google Scholar] [CrossRef]
- Carmena, M.; Earnshaw, W.C. The cellular geography of aurora kinases. Nat. Rev. Mol. Cell Biol. 2003, 4, 842–854. [Google Scholar] [CrossRef]
- Jeng, Y.M.; Peng, S.Y.; Lin, C.Y.; Hsu, H.C. Overexpression and amplification of Aurora-A in hepatocellular carcinoma. Clin. Cancer Res. 2004, 10, 2065–2071. [Google Scholar] [CrossRef]
- Woo, J.H.; Kim, Y.H.; Choi, Y.J.; Kim, D.G.; Lee, K.S.; Bae, J.H.; Min, D.S.; Chang, J.S.; Jeong, Y.J.; Lee, Y.H.; et al. Molecular mechanisms of curcumin-induced cytotoxicity: Induction of apoptosis through generation of reactive oxygen species, down-regulation of Bcl-XL and IAP, the release of cytochrome c and inhibition of Akt. Carcinogenesis 2003, 24, 1199–1208. [Google Scholar] [CrossRef]
- Du, J.; Hannon, G.J. Suppression of p160ROCK bypasses cell cycle arrest after Aurora-A/STK15 depletion. Proc. Natl. Acad. Sci. USA 2004, 101, 8975–8980. [Google Scholar] [CrossRef]
- Ning, L.; Wentworth, L.; Chen, H.; Weber, S.M. Down-regulation of Notch1 signaling inhibits tumor growth in human hepatocellular carcinoma. Am. J. Transl. Res. 2009, 1, 358–366. [Google Scholar]
- Wang, W.; Zhang, B.; Chen, H.; Zhang, L. Anticancer activities of curcumin on human hepatocarcinoma cell line Sk-hep-1. Zhongguo Zhong Yao Za Zhi 2010, 35, 485–488. [Google Scholar]
- Singh, S.; Singh, P.P.; Roberts, L.R.; Sanchez, W. Chemopreventive strategies in hepatocellular carcinoma. Nat. Rev. Gastroenterol. Hepatol. 2014, 11, 45–54. [Google Scholar] [CrossRef] [PubMed]
- Abou-Alfa, G.K.; Johnson, P.; Knox, J.J.; Capanu, M.; Davidenko, I.; Lacava, J.; Leung, T.; Gansukh, B.; Saltz, L.B. Doxorubicin plus sorafenib vs doxorubicin alone in patients with advanced hepatocellular carcinoma: A randomized trial. JAMA 2010, 304, 2154–2160. [Google Scholar] [CrossRef] [PubMed]


| Properties of Formulated Curcumin | Value |
|---|---|
| Pellet size (µm) | 830–1000 |
| Curcumin level (%) | 3.3 |
| Z-average (nm) | 141.9 ± 5.1 |
| Polydispersity index (PDI) | 0.308 ± 0.029 |
| Zeta potential (mV) | –2.49 ± 0.39 |
| Parameters | Oral | |
|---|---|---|
| Conventional Curcumin* (n = 5) | Formulated Curcumin (n = 6) | |
| 500 mg/kg | 60 mg/kg | |
| Cmax (ng/mL) | 0.704 ± 0.272 | 109.200 ± 41.651 |
| AUC0-t (h × ng/mL) | 1.1 ± 1.2 | 111.8 ± 16.4 |
| AUC0-t/Dose | 0.0022 ± 0.0024 | 1.863 ± 0.273 |
| Tmax(h) | 1.25 ± 0.83 | 0.38 ± 0.14 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liao, S.-C.; Hsu, W.-H.; Huang, Z.-Y.; Chuang, K.-L.; Lin, K.-T.; Tseng, C.-L.; Tsai, T.-H.; Dao, A.-H.; Su, C.-L.; Huang, C.-Y.F. Bioactivity Evaluation of a Novel Formulated Curcumin. Nutrients 2019, 11, 2982. https://doi.org/10.3390/nu11122982
Liao S-C, Hsu W-H, Huang Z-Y, Chuang K-L, Lin K-T, Tseng C-L, Tsai T-H, Dao A-H, Su C-L, Huang C-YF. Bioactivity Evaluation of a Novel Formulated Curcumin. Nutrients. 2019; 11(12):2982. https://doi.org/10.3390/nu11122982
Chicago/Turabian StyleLiao, Se-Chun, Wei-Hsiang Hsu, Zi-Yi Huang, Kun-Lin Chuang, Kuan-Ting Lin, Chia-Ling Tseng, Tung-Hu Tsai, Anh-Hoang Dao, Chun-Li Su, and Chi-Ying F. Huang. 2019. "Bioactivity Evaluation of a Novel Formulated Curcumin" Nutrients 11, no. 12: 2982. https://doi.org/10.3390/nu11122982
APA StyleLiao, S.-C., Hsu, W.-H., Huang, Z.-Y., Chuang, K.-L., Lin, K.-T., Tseng, C.-L., Tsai, T.-H., Dao, A.-H., Su, C.-L., & Huang, C.-Y. F. (2019). Bioactivity Evaluation of a Novel Formulated Curcumin. Nutrients, 11(12), 2982. https://doi.org/10.3390/nu11122982







