Chili Intake Is Inversely Associated with Chronic Kidney Disease among Adults: A Population-Based Study
Abstract
1. Introduction
2. Materials and Methods
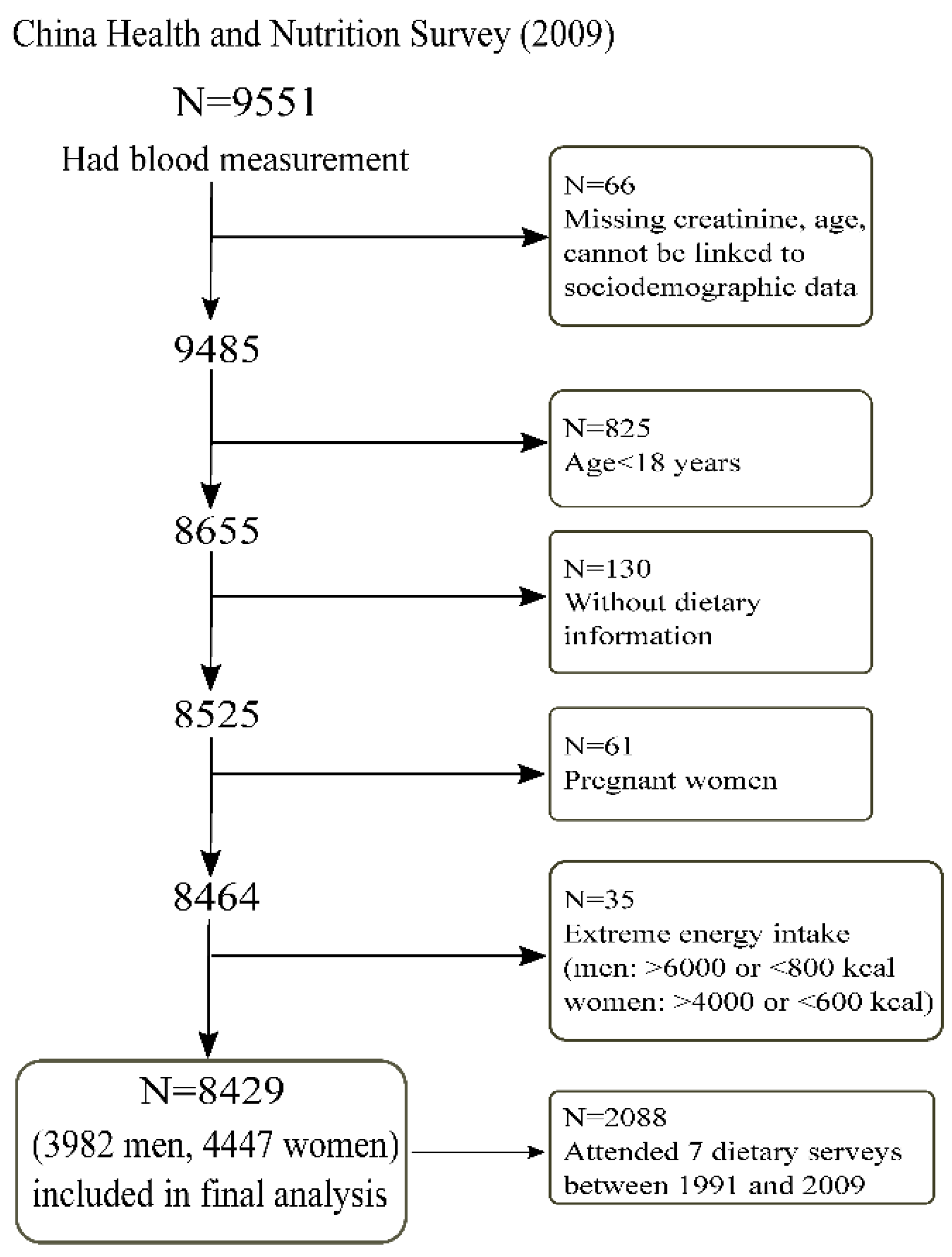
2.1. Study Design and Sample
2.2. Outcome Variable: Estimated GFR and CKD
2.3. Exposure Variable: Cumulative Mean Chili Intake
2.4. Covariates
2.5. Data Analyses
3. Results
3.1. Sample Characteristics
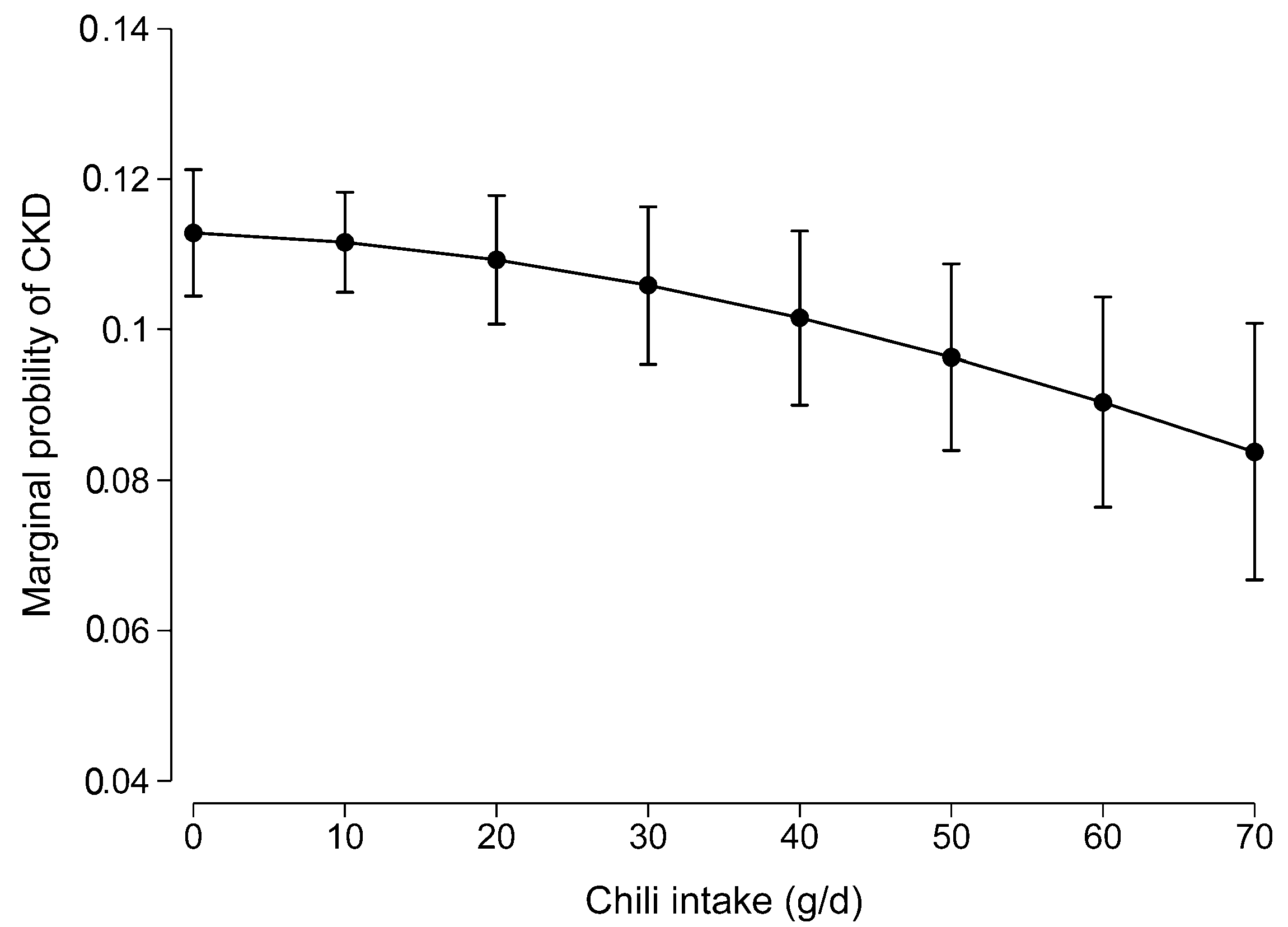
3.2. Association Between Chili Intake and CKD
3.3. Subgroup Analyses by Sociographic Factors and Chronic Conditions
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Hill, N.R.; Fatoba, S.T.; Oke, J.L.; Hirst, J.A.; O’Callaghan, C.A.; Lasserson, D.S.; Hobbs, F.D. Global Prevalence of Chronic Kidney Disease—A Systematic Review and Meta-Analysis. PLoS ONE 2016, 11, e0158765. [Google Scholar] [CrossRef] [PubMed]
- Jha, V.; Garcia-Garcia, G.; Iseki, K.; Li, Z.; Naicker, S.; Plattner, B.; Saran, R.; Wang, A.Y.; Yang, C.W. Chronic kidney disease: Global dimension and perspectives. Lancet 2013, 382, 260–272. [Google Scholar] [CrossRef]
- Banerjee, T.; Liu, Y.; Crews, D.C. Dietary Patterns and CKD Progression. Blood Purif. 2016, 41, 117–122. [Google Scholar] [CrossRef] [PubMed]
- Nadkarni, G.N.; Uribarri, J. Phosphorus and the kidney: What is known and what is needed. Adv. Nutr. 2014, 5, 98–103. [Google Scholar] [CrossRef] [PubMed]
- Karalius, V.P.; Shoham, D.A. Dietary sugar and artificial sweetener intake and chronic kidney disease: A review. Adv. Chronic Kidney Dis. 2013, 20, 157–164. [Google Scholar] [CrossRef] [PubMed]
- Glew, R.H.; Sun, Y.; Horowitz, B.L.; Konstantinov, K.N.; Barry, M.; Fair, J.R.; Massie, L.; Tzamaloukas, A.H. Nephropathy in dietary hyperoxaluria: A potentially preventable acute or chronic kidney disease. World J. Nephrol. 2014, 3, 122–142. [Google Scholar] [CrossRef] [PubMed]
- Lin, J.; Fung, T.T.; Hu, F.B.; Curhan, G.C. Association of dietary patterns with albuminuria and kidney function decline in older white women: A subgroup analysis from the Nurses’ Health Study. Am. J. Kidney Dis. 2011, 57, 245–254. [Google Scholar] [CrossRef]
- Nettleton, J.A.; Steffen, L.M.; Palmas, W.; Burke, G.L.; Jacobs, D.R., Jr. Associations between microalbuminuria and animal foods, plant foods, and dietary patterns in the Multiethnic Study of Atherosclerosis. Am. J. Clin. Nutr. 2008, 87, 1825–1836. [Google Scholar] [CrossRef]
- Hsu, C.C.; Jhang, H.R.; Chang, W.T.; Lin, C.H.; Shin, S.J.; Hwang, S.J.; Huang, M.C. Associations between dietary patterns and kidney function indicators in type 2 diabetes. Clin. Nutr. 2014, 33, 98–105. [Google Scholar] [CrossRef]
- Shi, Z.; Taylor, A.W.; Riley, M.; Byles, J.; Liu, J.; Noakes, M. Association between dietary patterns, cadmium intake and chronic kidney disease among adults. Clin. Nutr. 2018, 37, 276–284. [Google Scholar] [CrossRef]
- Jhee, J.H.; Kee, Y.K.; Park, J.T.; Chang, T.I.; Kang, E.W.; Yoo, T.H.; Kang, S.W.; Han, S.H. A Diet Rich in Vegetables and Fruit and Incident CKD: A Community-Based Prospective Cohort Study. Am. J. Kidney Dis. 2019, 74, 491–500. [Google Scholar] [CrossRef] [PubMed]
- Rozin, P.; Schiller, D. The nature and acquisition of a preference for chili pepper by humans. Motiv. Emot. 1980, 4, 77–101. [Google Scholar] [CrossRef]
- Astrup, A.; Kristensen, M.; Gregersen, N.T.; Belza, A.; Lorenzen, J.K.; Due, A.; Larsen, T.M. Can bioactive foods affect obesity? Ann. N. Y. Acad. Sci. 2010, 1190, 25–41. [Google Scholar] [CrossRef] [PubMed]
- Lv, J.; Qi, L.; Yu, C.; Yang, L.; Guo, Y.; Chen, Y.; Bian, Z.; Sun, D.; Du, J.; Ge, P.; et al. Consumption of spicy foods and total and cause specific mortality: Population based cohort study. BMJ 2015, 351, h3942. [Google Scholar] [CrossRef] [PubMed]
- Shi, Z.; Riley, M.; Taylor, A.W.; Page, A. Chilli consumption and the incidence of overweight and obesity in a Chinese adult population. Int. J. Obes. 2017, 41, 1074–1079. [Google Scholar] [CrossRef] [PubMed]
- Shi, Z.; Riley, M.; Brown, A.; Page, A. Chilli intake is inversely associated with hypertension among adults. Clin. Nutr. ESPEN 2018, 23, 67–72. [Google Scholar] [CrossRef]
- Shi, Z.; El-Obeid, T.; Riley, M.; Li, M.; Page, A.; Liu, J. High Chili Intake and Cognitive Function among 4582 Adults: An Open Cohort Study over 15 Years. Nutrients 2019, 11, 1183. [Google Scholar] [CrossRef]
- Sun, F.; Xiong, S.; Zhu, Z. Dietary Capsaicin Protects Cardiometabolic Organs from Dysfunction. Nutrients 2016, 8, 174. [Google Scholar] [CrossRef]
- Tremblay, A.; Arguin, H.; Panahi, S. Capsaicinoids: A spicy solution to the management of obesity? Int. J. Obes. 2016, 40, 1198–1204. [Google Scholar] [CrossRef]
- Janssens, P.L.; Hursel, R.; Martens, E.A.; Westerterp-Plantenga, M.S. Acute effects of capsaicin on energy expenditure and fat oxidation in negative energy balance. PLoS ONE 2013, 8, e67786. [Google Scholar] [CrossRef]
- Whiting, S.; Derbyshire, E.J.; Tiwari, B. Could capsaicinoids help to support weight management? A systematic review and meta-analysis of energy intake data. Appetite 2014, 73, 183–188. [Google Scholar] [CrossRef] [PubMed]
- Ludy, M.J.; Moore, G.E.; Mattes, R.D. The effects of capsaicin and capsiate on energy balance: Critical review and meta-analyses of studies in humans. Chem. Senses 2012, 37, 103–121. [Google Scholar] [CrossRef] [PubMed]
- Yoshioka, M.; St-Pierre, S.; Drapeau, V.; Dionne, I.; Doucet, E.; Suzuki, M.; Tremblay, A. Effects of red pepper on appetite and energy intake. Br. J. Nutr. 1999, 82, 115–123. [Google Scholar] [CrossRef] [PubMed]
- Rios-Silva, M.; Santos-Alvarez, R.; Trujillo, X.; Cardenas-Maria, R.Y.; Lopez-Zamudio, M.; Bricio-Barrios, J.A.; Leal, C.; Saavedra-Molina, A.; Huerta-Trujillo, M.; Espinoza-Mejia, K.; et al. Effects of Chronic Administration of Capsaicin on Biomarkers of Kidney Injury in Male Wistar Rats with Experimental Diabetes. Molecules 2018, 24, 36. [Google Scholar] [CrossRef]
- Yu, S.Q.; Ma, S.; Wang, D.H. Activation of TRPV1 Prevents Salt-Induced Kidney Damage and Hypertension After Renal Ischemia-Reperfusion Injury in Rats. Kidney Blood Press. Res. 2018, 43, 1285–1296. [Google Scholar] [CrossRef]
- Li, J.; Wang, D.H. Increased GFR and renal excretory function by activation of TRPV1 in the isolated perfused kidney. Pharmacol. Res. 2008, 57, 239–246. [Google Scholar] [CrossRef]
- Zhu, Y.; Wang, Y.; Wang, D.H. Diuresis and natriuresis caused by activation of VR1-positive sensory nerves in renal pelvis of rats. Hypertension 2005, 46, 992–997. [Google Scholar] [CrossRef]
- Inker, L.A.; Schmid, C.H.; Tighiouart, H.; Eckfeldt, J.H.; Feldman, H.I.; Greene, T.; Kusek, J.W.; Manzi, J.; Van Lente, F.; Zhang, Y.L.; et al. Estimating glomerular filtration rate from serum creatinine and cystatin C. N. Engl. J. Med. 2012, 367, 20–29. [Google Scholar] [CrossRef]
- Webster, A.C.; Nagler, E.V.; Morton, R.L.; Masson, P. Chronic Kidney Disease. Lancet 2017, 389, 1238–1252. [Google Scholar] [CrossRef]
- Hu, F.B.; Stampfer, M.J.; Rimm, E.; Ascherio, A.; Rosner, B.A.; Spiegelman, D.; Willett, W.C. Dietary fat and coronary heart disease: A comparison of approaches for adjusting for total energy intake and modeling repeated dietary measurements. Am. J. Epidemiol. 1999, 149, 531–540. [Google Scholar] [CrossRef]
- Zhai, F.Y.; Du, S.F.; Wang, Z.H.; Zhang, J.G.; Du, W.W.; Popkin, B.M. Dynamics of the Chinese diet and the role of urbanicity, 1991–2011. Obes. Rev. 2014, 15, 16–26. [Google Scholar] [CrossRef] [PubMed]
- Yao, M.; McCrory, M.A.; Ma, G.; Tucker, K.L.; Gao, S.; Fuss, P.; Roberts, S.B. Relative influence of diet and physical activity on body composition in urban Chinese adults. Am. J. Clin. Nutr. 2003, 77, 1409–1416. [Google Scholar] [CrossRef] [PubMed]
- Yan, S.; Li, J.; Li, S.; Zhang, B.; Du, S.; Gordon-Larsen, P.; Adair, L.; Popkin, B. The expanding burden of cardiometabolic risk in China: The China Health and Nutrition Survey. Obes. Rev. 2012, 13, 810–821. [Google Scholar] [CrossRef] [PubMed]
- American Diabetes, A. Diagnosis and classification of diabetes mellitus. Diabetes Care 2014, 37, S81–S90. [Google Scholar] [CrossRef] [PubMed]
- Chobanian, A.V.; Bakris, G.L.; Black, H.R.; Cushman, W.C.; Green, L.A.; Izzo, J.L., Jr.; Jones, D.W.; Materson, B.J.; Oparil, S.; Wright, J.T., Jr.; et al. Seventh report of the Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure. Hypertension 2003, 42, 1206–1252. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Cui, Y.; Jin, R.; Lang, H.; Yu, H.; Sun, F.; He, C.; Ma, T.; Li, Y.; Zhou, X.; et al. Enjoyment of Spicy Flavor Enhances Central Salty-Taste Perception and Reduces Salt Intake and Blood Pressure. Hypertension 2017, 70, 1291–1299. [Google Scholar] [CrossRef]
- Janssens, P.L.; Hursel, R.; Westerterp-Plantenga, M.S. Capsaicin increases sensation of fullness in energy balance, and decreases desire to eat after dinner in negative energy balance. Appetite 2014, 77, 44–49. [Google Scholar] [CrossRef]
- Zhang, L.; Long, J.; Jiang, W.; Shi, Y.; He, X.; Zhou, Z.; Li, Y.; Yeung, R.O.; Wang, J.; Matsushita, K.; et al. Trends in Chronic Kidney Disease in China. N. Engl. J. Med. 2016, 375, 905–906. [Google Scholar] [CrossRef]
- Hawkins, M.S.; Sevick, M.A.; Richardson, C.R.; Fried, L.F.; Arena, V.C.; Kriska, A.M. Association between physical activity and kidney function: National Health and Nutrition Examination Survey. Med. Sci. Sports Exerc. 2011, 43, 1457–1464. [Google Scholar] [CrossRef]
- Beddhu, S.; Baird, B.C.; Zitterkoph, J.; Neilson, J.; Greene, T. Physical activity and mortality in chronic kidney disease (NHANES III). Clin. J. Am. Soc. Nephrol. 2009, 4, 1901–1906. [Google Scholar] [CrossRef]
- Jung, S.H.; Kim, H.J.; Oh, G.S.; Shen, A.; Lee, S.; Choe, S.K.; Park, R.; So, H.S. Capsaicin ameliorates cisplatin-induced renal injury through induction of heme oxygenase-1. Mol. Cells 2014, 37, 234–240. [Google Scholar] [CrossRef] [PubMed]

| None | 1–20 g/day | 20.1–50 g/day | ≥50.1 g/day | p-Value | |
|---|---|---|---|---|---|
| N | 3390 | 2617 | 1733 | 689 | |
| Chili intake (g/day), mean (SD) | 0.0 (0.0) | 9.7 (5.6) | 32.8 (8.4) | 74.5 (25.8) | <0.001 |
| Traditional dietary pattern, mean (SD) | −0.1 (0.9) | −0.0 (0.8) | 0.2 (0.7) | 0.4 (0.8) | <0.001 |
| Modern dietary pattern, mean (SD) | 0.4 (1.0) | 0.2 (0.8) | 0.0 (0.8) | −0.1 (0.7) | <0.001 |
| Energy intake (kcal/day), mean (SD) | 2074.1 (610.7) | 2117.2 (648.7) | 2196.0 (630.5) | 2310.8 (699.8) | <0.001 |
| Fat intake (g/day), mean (SD) | 71.8 (33.1) | 74.1 (35.9) | 77.2 (37.9) | 79.7 (40.5) | <0.001 |
| Protein intake (g/day), mean (SD) | 65.7 (22.2) | 65.0 (23.2) | 66.6 (22.3) | 69.6 (25.6) | <0.001 |
| Carbohydrate intake (g/day), mean (SD) | 286.9 (100.9) | 291.5 (101.1) | 303.9 (98.3) | 324.0 (110.2) | <0.001 |
| Age (years), mean (SD) | 50.3 (16.0) | 52.2 (14.1) | 51.3 (14.3) | 48.9 (14.7) | <0.001 |
| BMI (kg/m2), mean (SD) | 23.4 (3.5) | 23.5 (3.5) | 23.3 (3.4) | 22.9 (3.3) | <0.001 |
| BMI status, n (%) | 0.004 | ||||
| Underweight | 222 (6.7%) | 157 (6.1%) | 98 (5.8%) | 42 (6.3%) | |
| Normal | 2102 (63.0%) | 1612 (62.7%) | 1091 (64.3%) | 469 (70.2%) | |
| Overweight | 859 (25.7%) | 682 (26.5%) | 454 (26.8%) | 135 (20.2%) | |
| Obese | 153 (4.6%) | 120 (4.7%) | 54 (3.2%) | 22 (3.3%) | |
| Sex, n (%) | 0.006 | ||||
| Men | 1546 (45.6%) | 1226 (46.8%) | 852 (49.2%) | 358 (52.0%) | |
| Women | 1844 (54.4%) | 1391 (53.2%) | 881 (50.8%) | 331 (48.0%) | |
| Income, n (%) | <0.001 | ||||
| Low | 901 (26.9%) | 753 (29.0%) | 484 (28.4%) | 226 (33.3%) | |
| Medium | 1161 (34.7%) | 757 (29.2%) | 583 (34.2%) | 242 (35.6%) | |
| High | 1286 (38.4%) | 1086 (41.8%) | 638 (37.4%) | 211 (31.1%) | |
| Education, n (%) | 0.31 | ||||
| Low | 1373 (40.5%) | 1092 (41.8%) | 730 (42.2%) | 305 (44.4%) | |
| Medium | 1183 (34.9%) | 872 (33.4%) | 586 (33.9%) | 238 (34.6%) | |
| High | 830 (24.5%) | 647 (24.8%) | 412 (23.8%) | 144 (21.0%) | |
| Hypertension, n (%) | 964 (28.6%) | 765 (29.5%) | 430 (25.1%) | 135 (19.9%) | <0.001 |
| Diabetes, n (%) | 388 (11.4%) | 305 (11.7%) | 150 (8.7%) | 61 (8.9%) | 0.002 |
| Urbanization, n (%) | <0.001 | ||||
| Low | 514 (15.2%) | 390 (14.9%) | 236 (13.6%) | 85 (12.3%) | |
| Medium | 1014 (29.9%) | 1015 (38.8%) | 677 (39.1%) | 322 (46.7%) | |
| High | 1862 (54.9%) | 1212 (46.3%) | 820 (47.3%) | 282 (40.9%) | |
| Smoking, n (%) | 0.006 | ||||
| Non-smoker | 2389 (70.5%) | 1776 (67.9%) | 1174 (67.8%) | 456 (66.3%) | |
| Ex-smoker | 126 (3.7%) | 90 (3.4%) | 51 (2.9%) | 15 (2.2%) | |
| Current smoker | 874 (25.8%) | 749 (28.6%) | 507 (29.3%) | 217 (31.5%) | |
| High sensitivity CRP (mg/dL), mean (SD) | 1.0 (0.0–2.0) | 1.0 (0.0–2.0) | 1.0 (0.0–2.0) | 1.0 (0.0–2.0) | 0.71 |
| CKD, n (%) | 445 (13.1%) | 305 (11.7%) | 207 (11.9%) | 51 (7.4%) | <0.001 |
| Physical activity (MET hour/week), mean (SD) | 120.4 (105.1) | 130.0 (112.6) | 121.3 (104.5) | 120.8 (106.2) | 0.006 |
| None | 1–20 g/day | 20.1–50 g/day | ≥50.1 g/day | p Value | |
|---|---|---|---|---|---|
| N = 3390 | N = 2617 | N = 1733 | N = 689 | ||
| Model 1 | 1.00 | 0.82 (0.68–0.98) | 0.97 (0.79–1.19) | 0.63 (0.45–0.89) | 0.057 |
| Model 2 | 1.00 | 0.81 (0.66–0.99) | 0.83 (0.66–1.05) | 0.48 (0.33–0.71) | 0.001 |
| Model 3 | 1.00 | 0.82 (0.67–1.01) | 0.83 (0.65–1.05) | 0.51 (0.35–0.75) | 0.001 |
| Sensitivity analysis | 1.00 | 0.78 (0.54–1.11) | 0.73 (0.49–1.08) | 0.47 (0.26–0.85) | 0.012 |
| None | 1–20 g/day | 20.1–50 g/day | ≥50.1 g/day | p Value | |
|---|---|---|---|---|---|
| Gender | |||||
| Men | 1.00 | 0.74 (0.52–1.06) | 0.74 (0.50–1.10) | 0.47 (0.25–0.89) | 0.920 |
| Women | 1.00 | 0.87 (0.67–1.13) | 0.90 (0.66–1.21) | 0.53 (0.32–0.87) | |
| Income | |||||
| Low | 1.00 | 0.77 (0.53–1.11) | 0.59 (0.38–0.90) | 0.45 (0.24–0.85) | 0.310 |
| Medium | 1.00 | 0.68 (0.44–1.03) | 0.93 (0.61–1.42) | 0.53 (0.29–0.99) | |
| High | 1.00 | 1.05 (0.76–1.45) | 1.14 (0.77–1.70) | 0.51 (0.23–1.16) | |
| Urbanization | |||||
| Low | 1.00 | 0.98 (0.50–1.90) | 0.36 (0.14–0.94) | 0.30 (0.09–1.05) | 0.769 |
| Medium | 1.00 | 0.83 (0.57–1.22) | 0.84 (0.56–1.26) | 0.47 (0.26–0.85) | |
| High | 1.00 | 0.82 (0.63–1.08) | 0.93 (0.68–1.28) | 0.62 (0.35–1.12) | |
| Overweight/obesity | |||||
| No | 1.00 | 0.88 (0.68–1.12) | 0.75 (0.56–1.00) | 0.50 (0.32–0.78) | 0.350 |
| Yes | 1.00 | 0.71 (0.49–1.04) | 1.06 (0.70–1.61) | 0.55 (0.25–1.23) | |
| Hypertension | |||||
| No | 1.00 | 0.85 (0.64–1.13) | 0.78 (0.57–1.06) | 0.57 (0.36–0.91) | 0.616 |
| Yes | 1.00 | 0.78 (0.57–1.06) | 0.91 (0.63–1.32) | 0.40 (0.20–0.80) | |
| Diabetes | |||||
| No | 1.00 | 0.87 (0.69–1.09) | 0.79 (0.61–1.02) | 0.51 (0.33–0.77) | 0.303 |
| Yes | 1.00 | 0.62 (0.37–1.03) | 1.21 (0.64–2.26) | 0.50 (0.18–1.35) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shi, Z.; Zhang, M.; Liu, J. Chili Intake Is Inversely Associated with Chronic Kidney Disease among Adults: A Population-Based Study. Nutrients 2019, 11, 2949. https://doi.org/10.3390/nu11122949
Shi Z, Zhang M, Liu J. Chili Intake Is Inversely Associated with Chronic Kidney Disease among Adults: A Population-Based Study. Nutrients. 2019; 11(12):2949. https://doi.org/10.3390/nu11122949
Chicago/Turabian StyleShi, Zumin, Ming Zhang, and Jianghong Liu. 2019. "Chili Intake Is Inversely Associated with Chronic Kidney Disease among Adults: A Population-Based Study" Nutrients 11, no. 12: 2949. https://doi.org/10.3390/nu11122949
APA StyleShi, Z., Zhang, M., & Liu, J. (2019). Chili Intake Is Inversely Associated with Chronic Kidney Disease among Adults: A Population-Based Study. Nutrients, 11(12), 2949. https://doi.org/10.3390/nu11122949






