The Effects of Probiotic Supplementation on the Incidence of Diarrhea in Cancer Patients Receiving Radiation Therapy: A Systematic Review with Meta-Analysis and Trial Sequential Analysis of Randomized Controlled Trials
Abstract
:1. Introduction
2. Methods
2.1. Design and Data Sources
2.2. Data Extraction and Quality Assessment
2.3. Statistical Analysis
2.4. Operational Definitions
3. Results
3.1. Description of Included Trials
3.2. Population Characteristics
3.3. Intervention Characteristics
3.4. Quality Assessment (ROB)
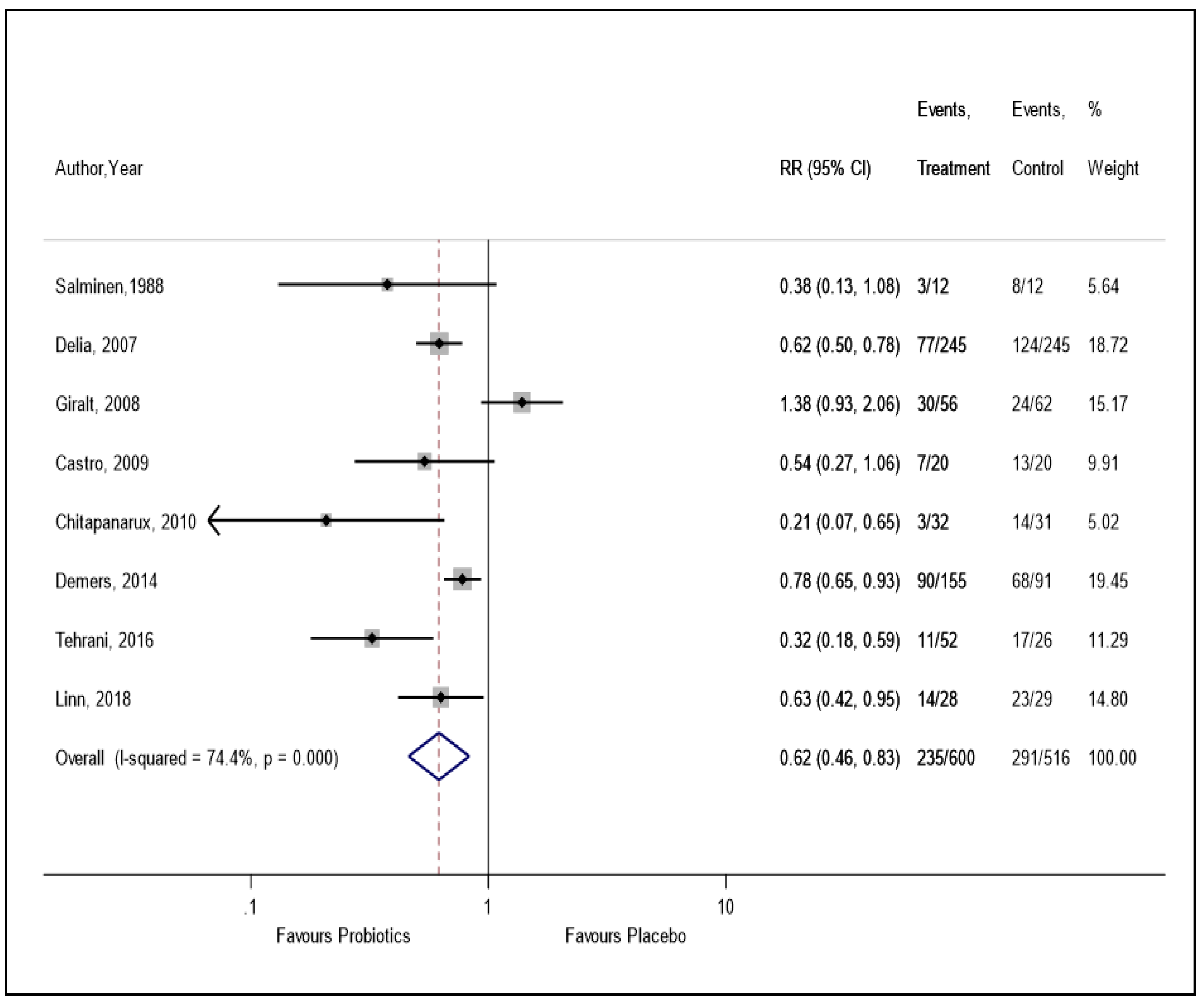
3.5. Primary Outcome: Risk of RID
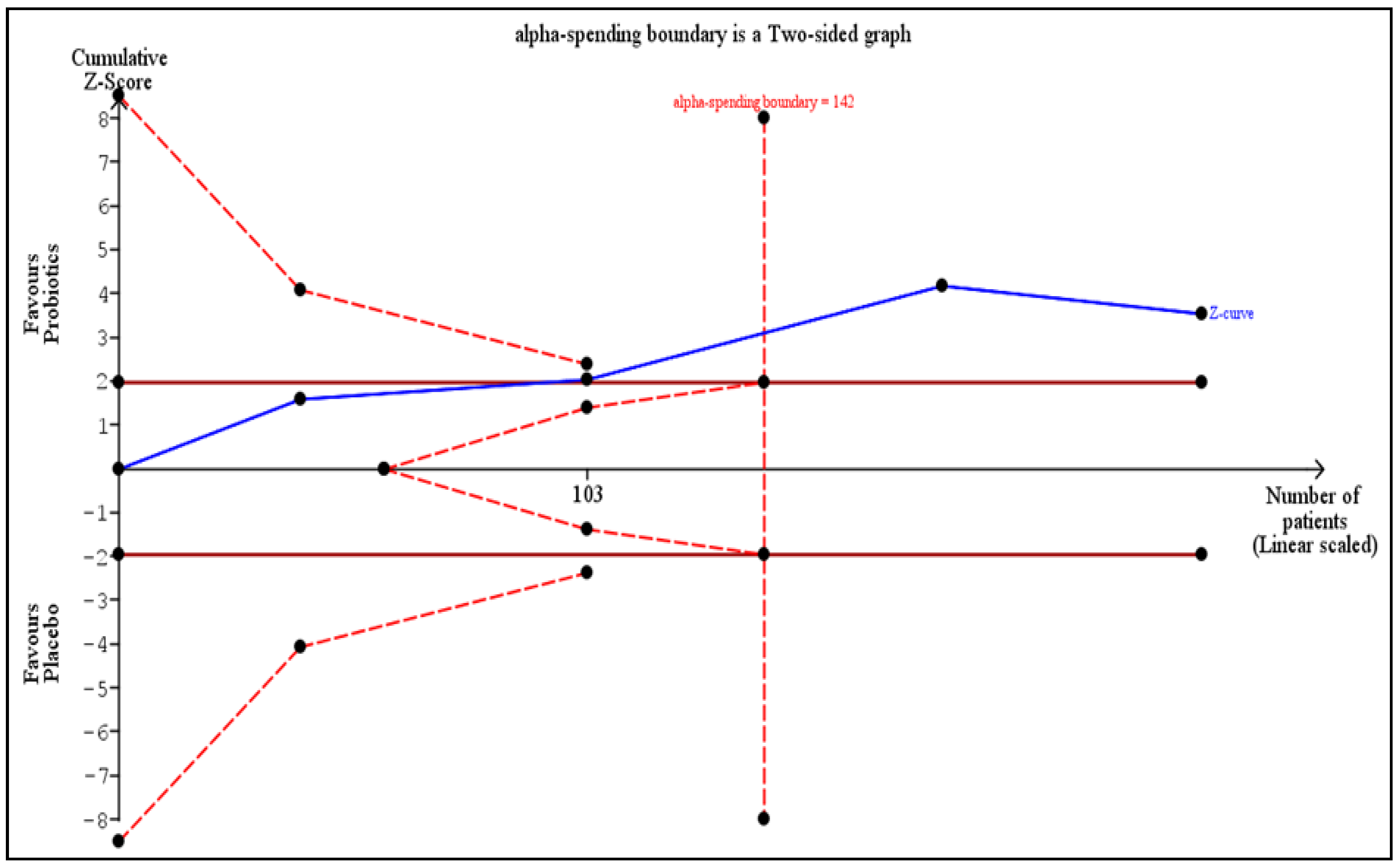
3.6. Trial Sequential Analysis (TSA) for the Primary Outcome
3.7. GRADE Summary of Evidence
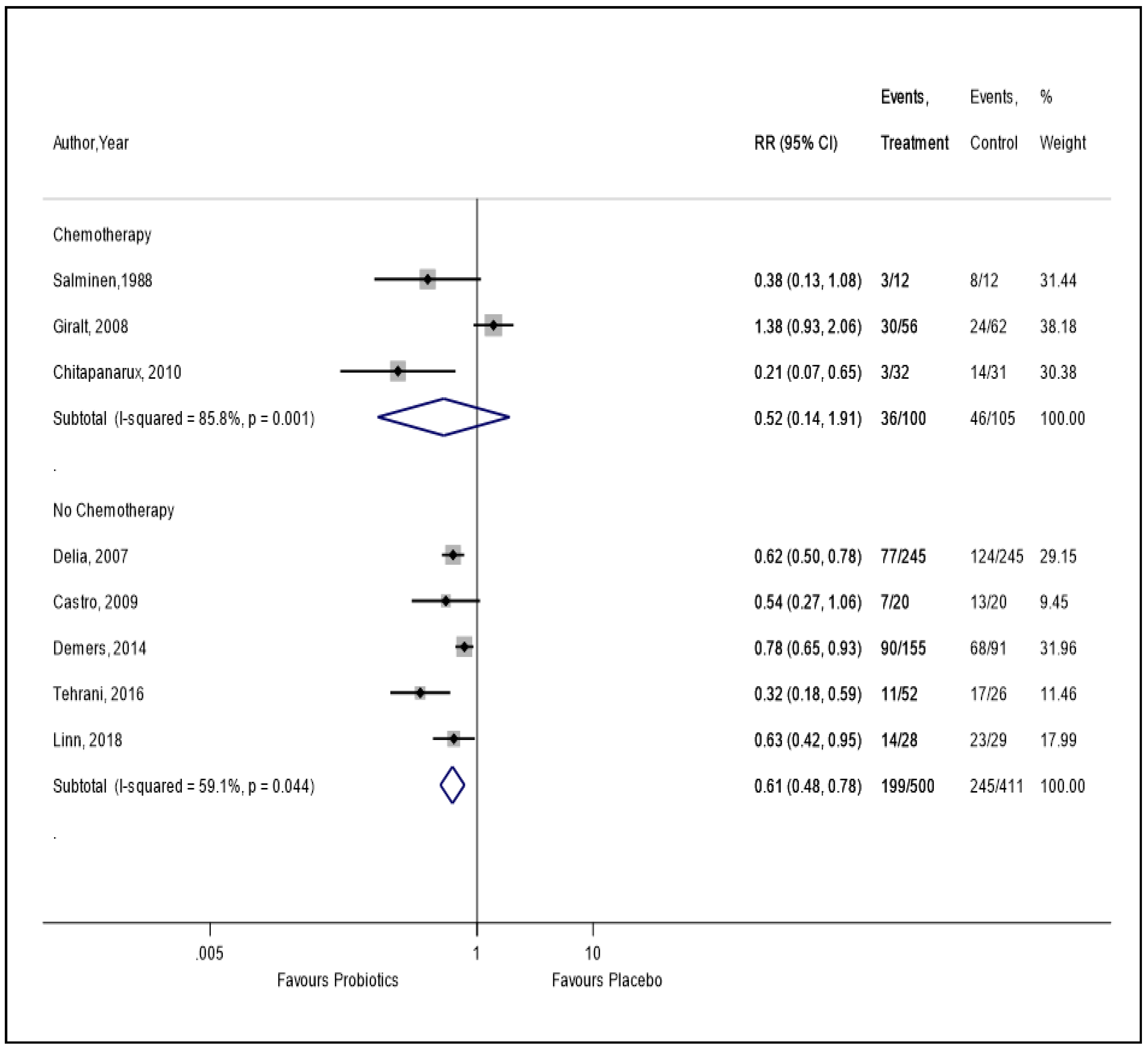
3.8. Secondary Outcomes
4. Discussion
4.1. Main Findings
4.2. Comparison with Previous Meta-Analyses
4.3. Implications for Clinical Practice
4.4. Strengths and Limitations
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| GRADE | Grading of Recommendations, Assessment, Development and Evaluation |
| RCT | Randomized controlled trials |
| RID | Radiation-induced diarrhea |
| RT | Radiation therapy |
| TSA | Trial sequential analysis |
Appendix A
Appendix A.1. Search Strategy in MEDLINE
| Medline | Embase | Cochrane | ||
| 1 | exp DIARRHEA/ | 49,612 | 228,556 | 3029 |
| 2 | exp RADIOTHERAPY/ | 168,386 | 479,346 | 5602 |
| 3 | Radiation-induced Diarrhea.mp. | 53 | 72 | 30 |
| 4 | exp PROBIOTICS/ | 13,609 | 28,098 | 1590 |
| 5 | exp SYNBIOTICS/ | 356 | 1200 | 96 |
| 6 | exp LACTOBACILLUS/ | 25,693 | 39,707 | 1365 |
| 7 | exp BIFIDOBACTERIUM/ | 5163 | 10,399 | 552 |
| 8 | exp SACCHAROMYCES/ | 104,336 | 98,961 | 138 |
| 9 | exp Enterococcus/ | 18,010 | 43,801 | 249 |
| 10 | exp BIFIDOBACTERIUM/ | 5163 | 10,399 | 552 |
| 11 | exp Randomized Controlled Trials as Topic/ | 119,100 | 146,594 | 7449 |
| 12 | exp Clinical Trial/ | 800,652 | 1,329,589 | 173 |
| 13 | random$.ab. | 956,882 | 1,275,640 | 599,733 |
| 14 | 4 or 5 or 6 or 7 or 8 or 9 or 10 | 156,392 | 196,258 | 2516 |
| 15 | 1 or 2 or 3 | 217,601 | 697,441 | 8569 |
| 16 | 11 or 12 or 13 | 1,460,326 | 2,207,279 | 601,980 |
| 17 | 14 and 15 and 16 | 485 | 2382 | 229 |
| 18 | limit 17 to (humans and yr = “2016–2018”) | 44 | 308 | 21 |
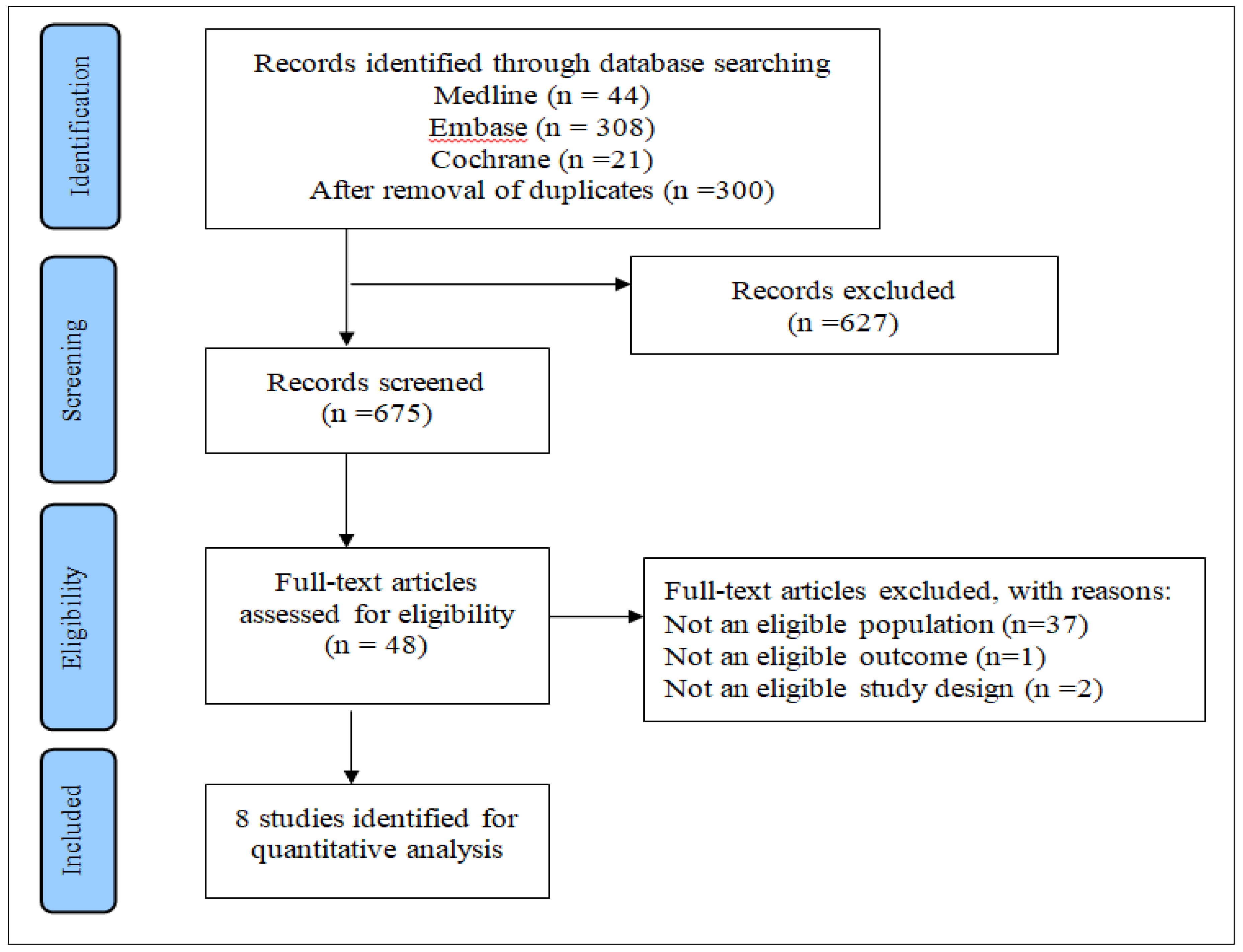
Appendix A.2. PRISMA Flow Diagram
Description of Included Trials
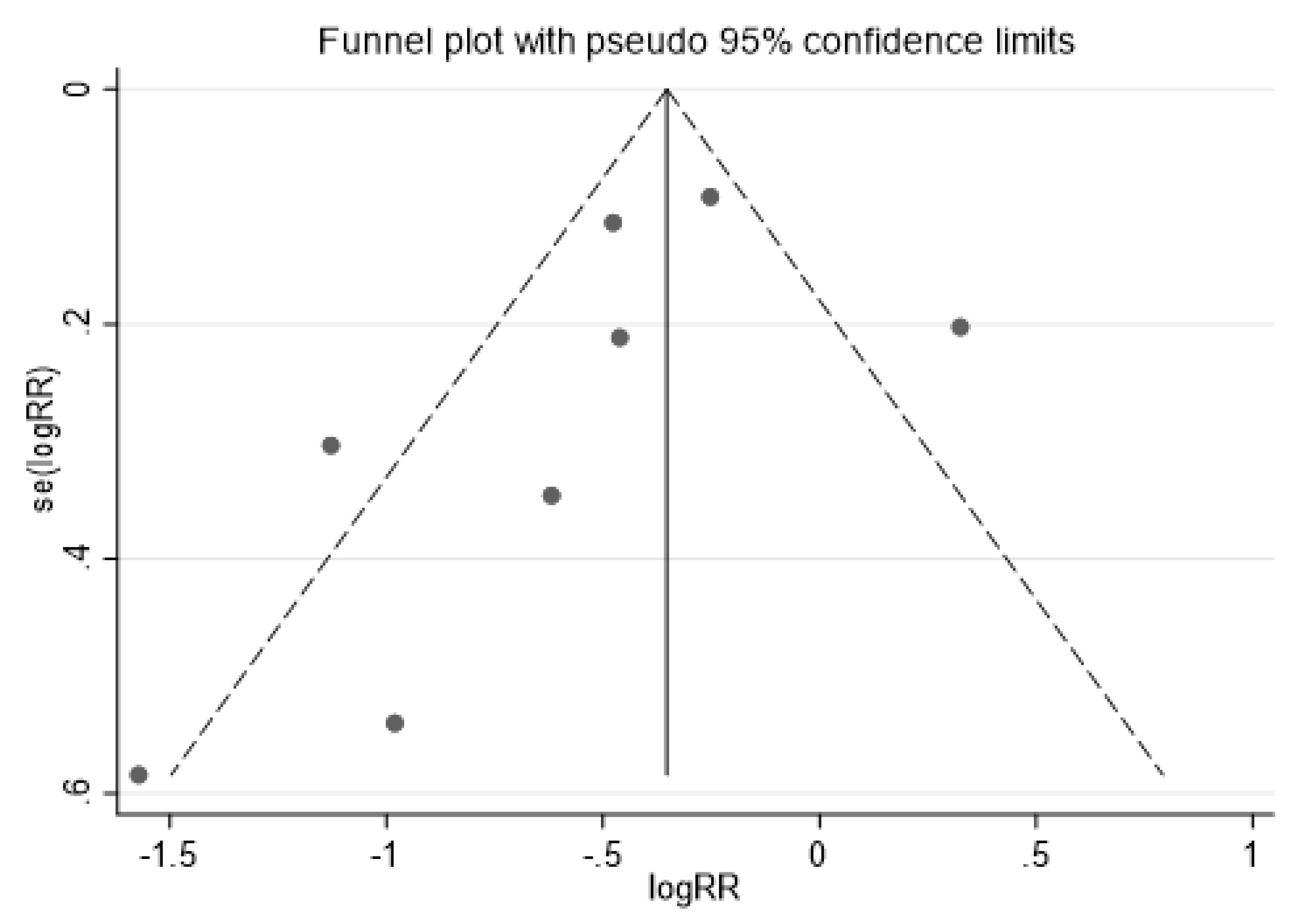
Appendix A.3. Assessment of Primary Outcome (The Funnel Plot Asymmetry Test)
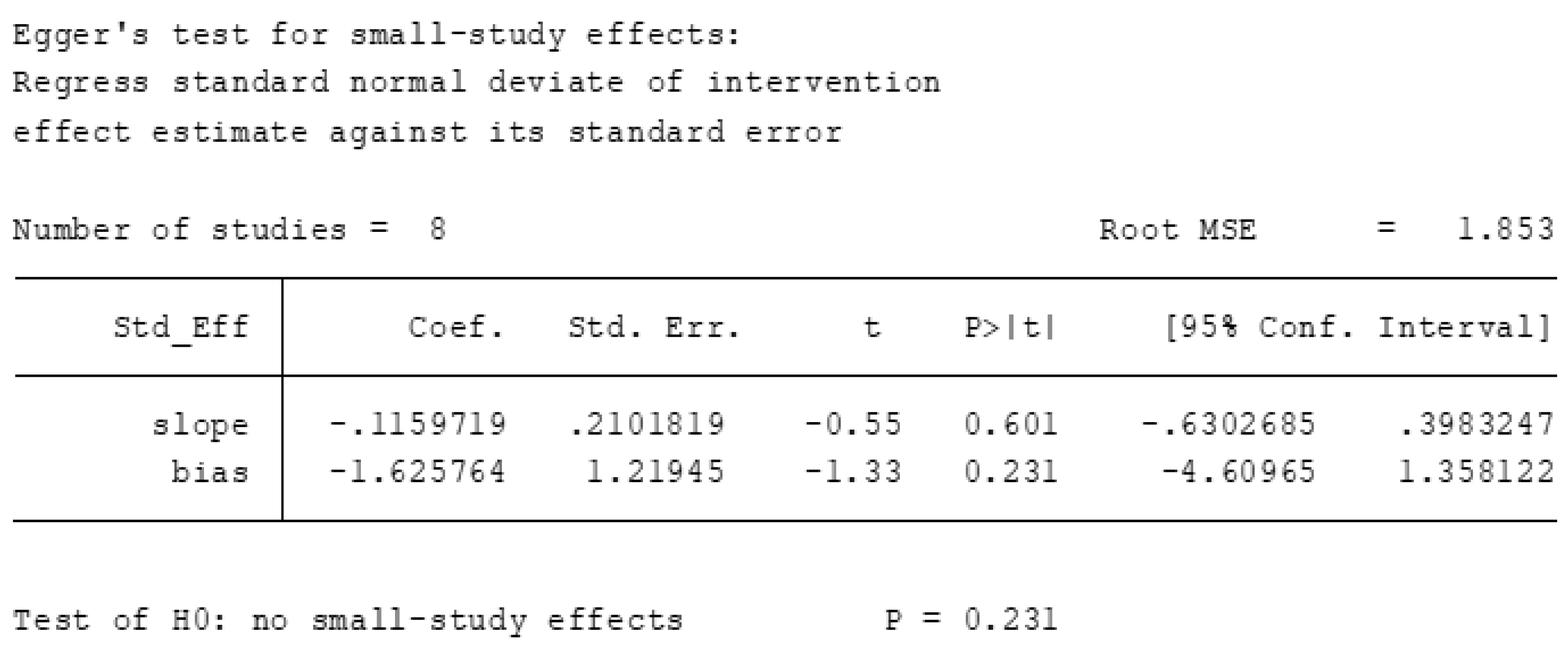
Appendix A.4. Egger’s Regression Test
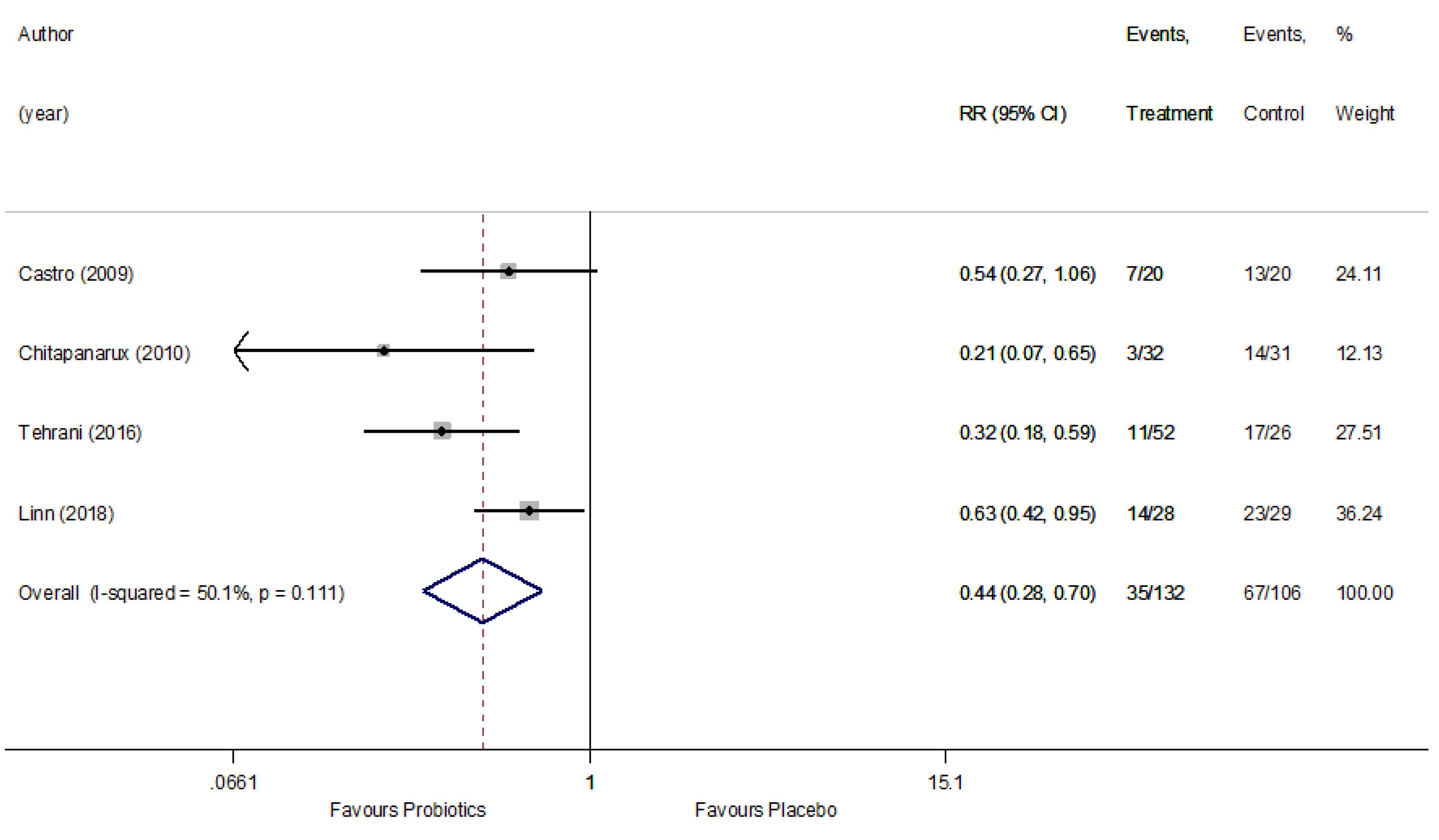
Appendix A.5. Sensitivity Analysis: By Excluding Trials with High ROB
Appendix A.6. Sensitivity Analysis: By Per-Protocol Data
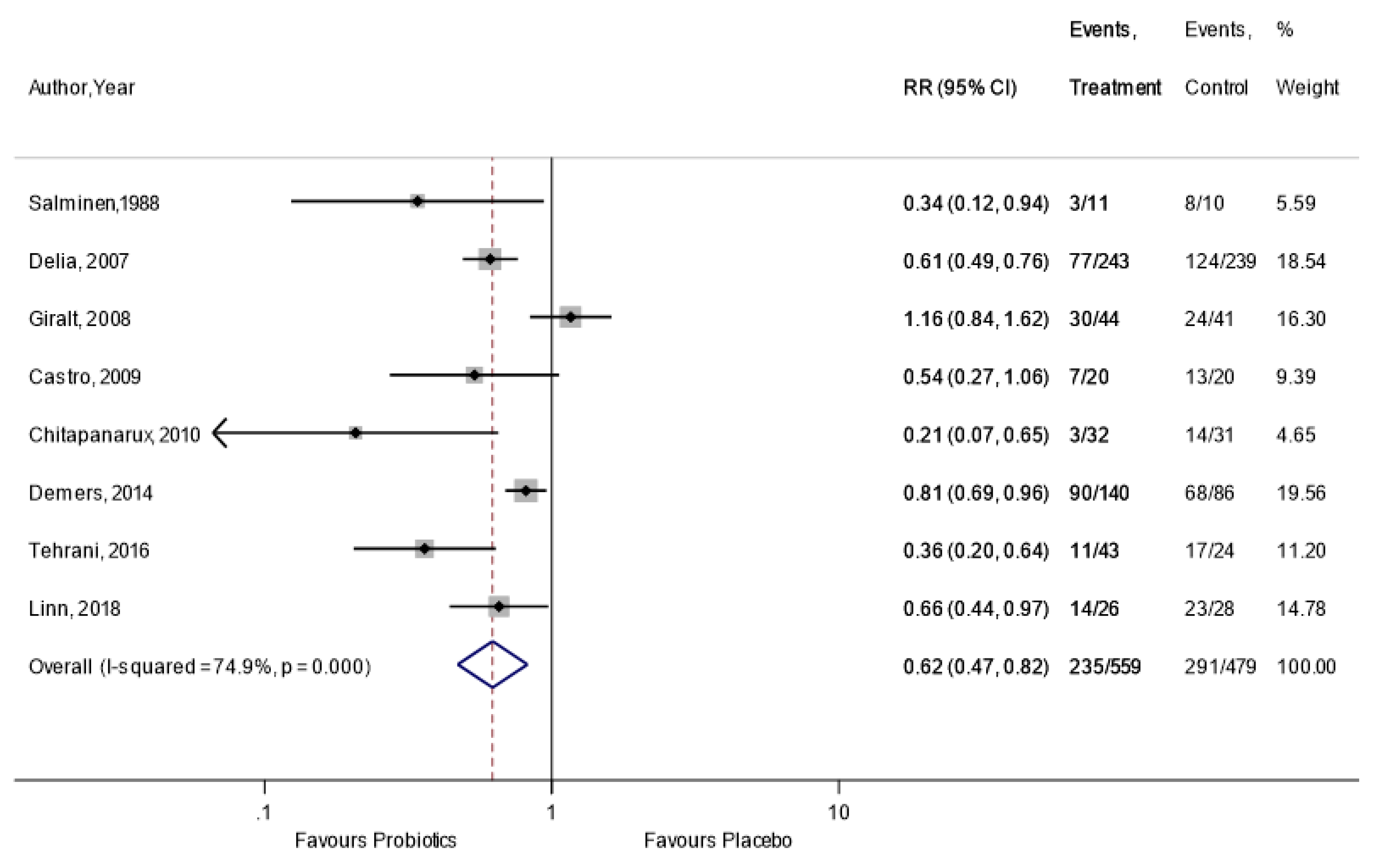
Appendix A.7. GRADE Summary of Evidence
| Certainty Assessment | № of Patients | Effect | Certainty | Importance | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| № of Studies | Study Design | Risk of Bias | Inconsistency | Indirectness | Imprecision | Other Considerations | Probiotics | Placebo | Relative (95% CI) | Absolute (95% CI) | ||
| Outcome: Incidence of Radiation-Induced Diarrhea | ||||||||||||
| Three | Randomized trials | Not serious a | Serious b | Serious c | Not serious d | None e | 107/215 (49.8%) | 105/151 (69.5%) | RR 0.61 (0.39 to 0.96) | 271 fewer per 1000 (from 424 fewer to 28 fewer) | ⨁⨁◯◯ LOW | IMPORTANT |
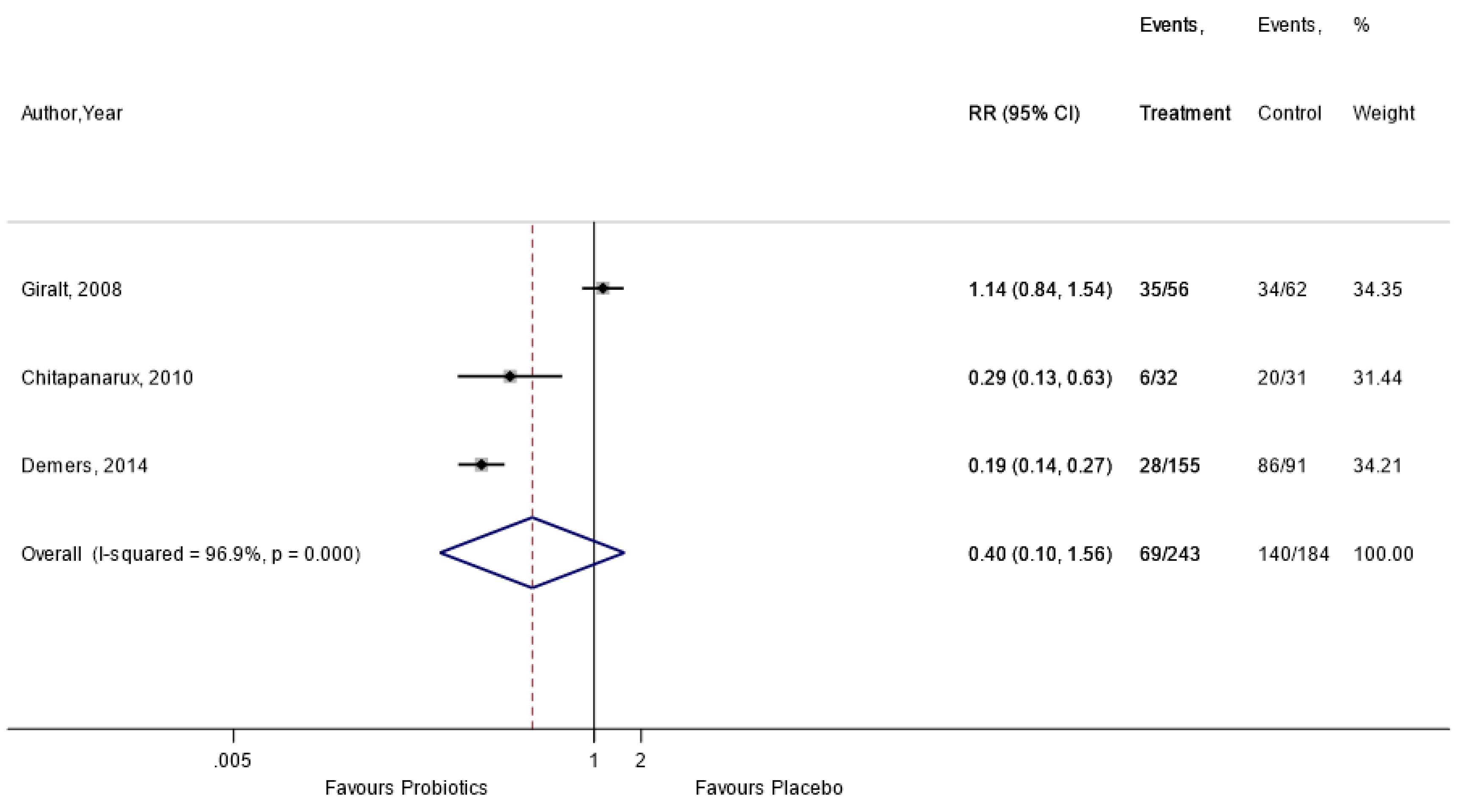
Appendix A.8. Incidence of Antidiarrheal Medication Use
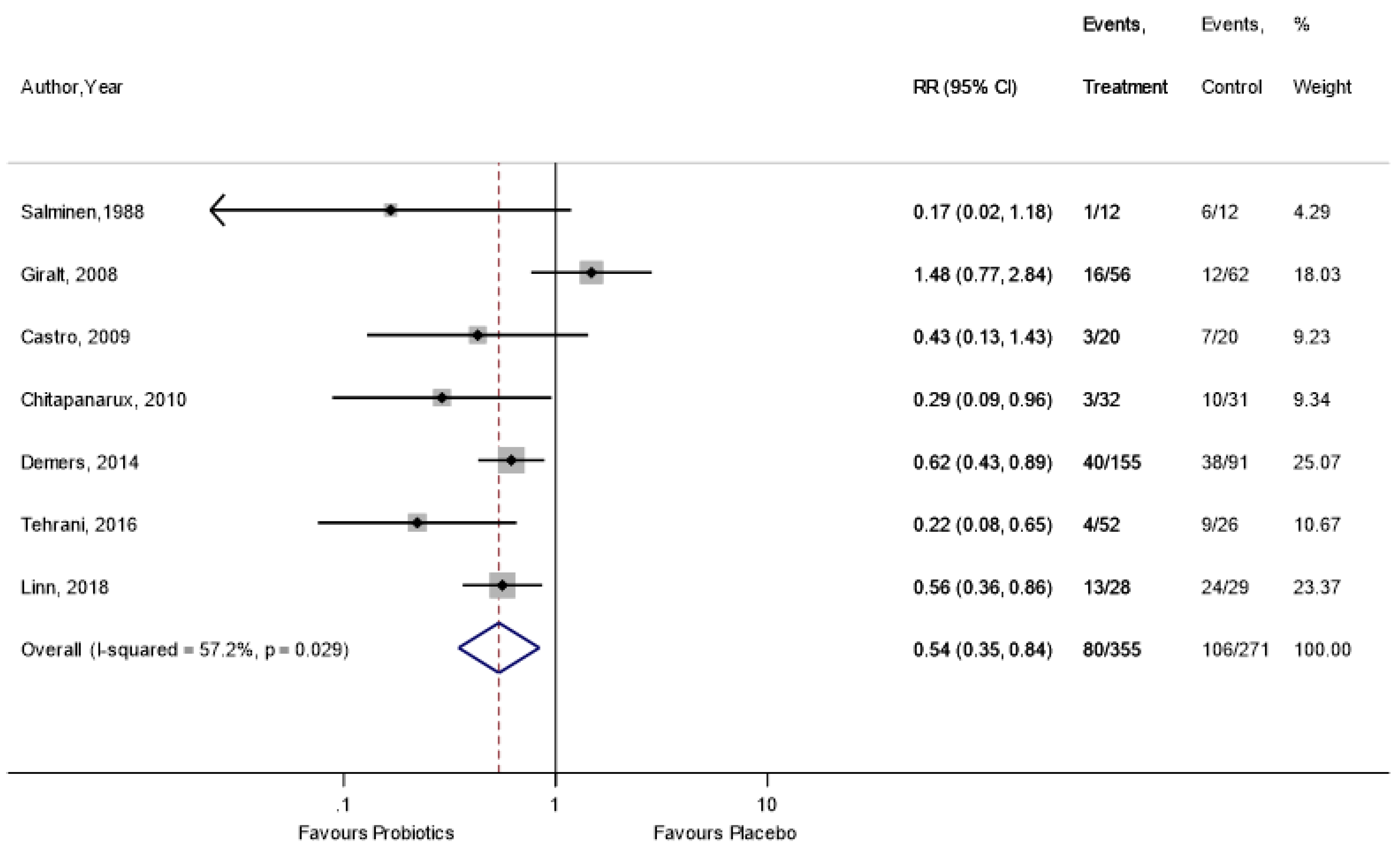
Appendix A.9. Incidence of Soft Stool
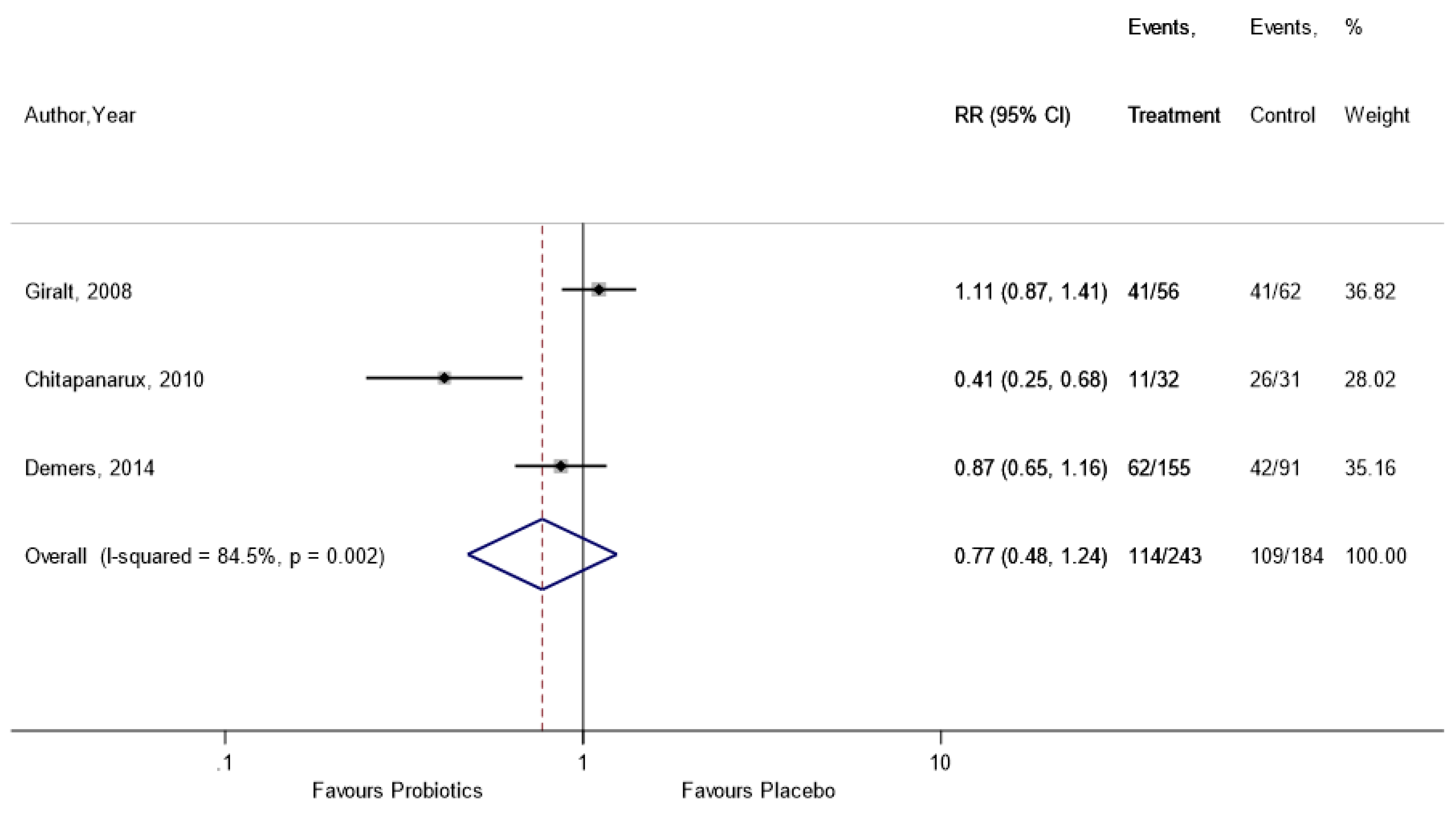
Appendix A.10. Incidence of Watery Stool
Appendix A.11. Post-Hoc Exploratory Analysis on the Effect of Single versus Combined Strains of Probiotics on RID
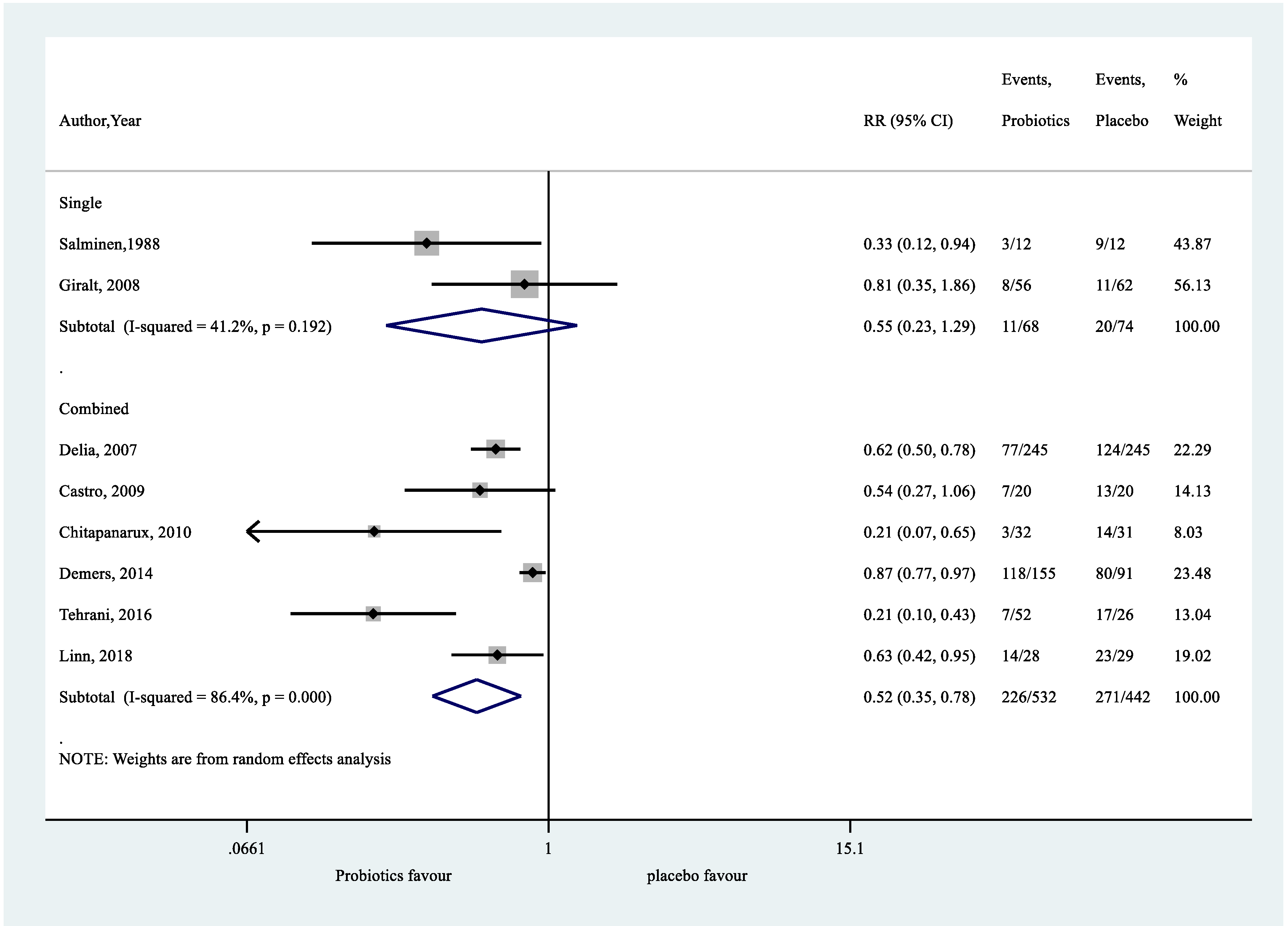
Appendix A.12. Post-Hoc Exploratory Analysis on the Effect of Probiotics, with or without Bifidobacterium, on RID
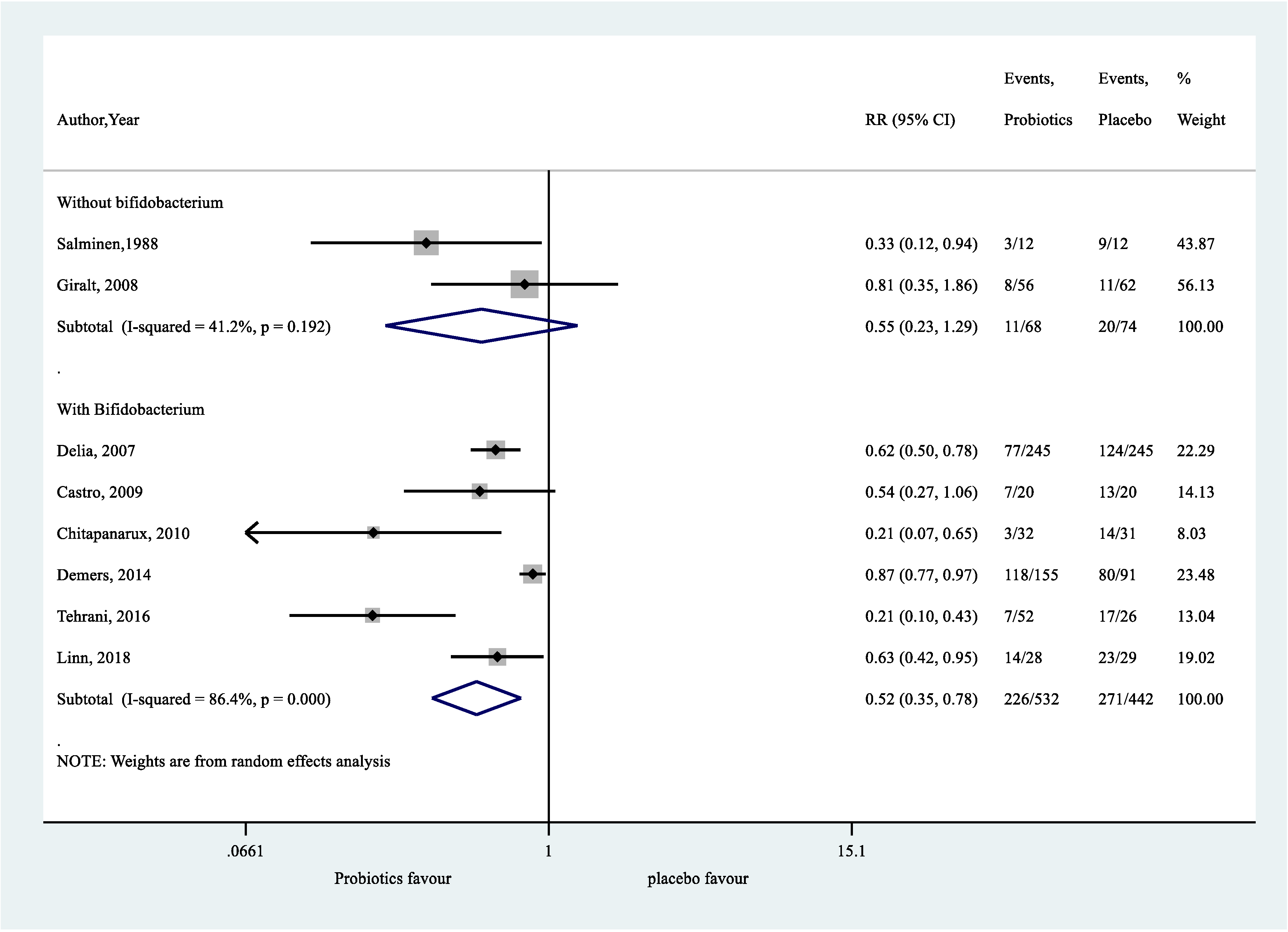
References
- Chen, H.H.; Kuo, M.T. Improving radiotherapy in cancer treatment: Promises and challenges. Oncotarget 2017, 8, 62742. [Google Scholar] [CrossRef] [PubMed]
- Baskar, R.; Itahana, K. Radiation therapy and cancer control in developing countries: Can we save more lives? Int. J. Med. Sci. 2017, 14, 13. [Google Scholar] [CrossRef] [PubMed]
- Classen, J.; Belka, C.; Paulsen, F.; Budach, W.; Hoffmann, W.; Bamberg, M. Radiation-induced gastrointestinal toxicity. Pathophysiology, approaches to treatment and prophylaxis. Strahlenther. Onkol. 1998, 174, 82–84. [Google Scholar] [PubMed]
- Guarner, F.; Khan, A.G.; Garisch, J.; Eliakim, R.; Gangl, A.; Thomson, A.; Krabshuis, J.; Lemair, T.; Kaufmann, P.; De Paula, J.A. World gastroenterology organisation global guidelines: Probiotics and prebiotics october 2011. J. Clin. Gastroenterol. 2012, 46, 468–481. [Google Scholar] [CrossRef] [PubMed]
- Liu, M.-M.; Li, S.-T.; Shu, Y.; Zhan, H.-Q. Probiotics for prevention of radiation-induced diarrhea: A meta-analysis of randomized controlled trials. PLoS ONE 2017, 12, e0178870. [Google Scholar] [CrossRef]
- Wei, D.; Heus, P.; van de Wetering, F.T.; van Tienhoven, G.; Verleye, L.; Scholten, R.J. Probiotics for the prevention or treatment of chemotherapy-or radiotherapy-related diarrhoea in people with cancer. Cochrane Database Syst. Rev. 2018. [Google Scholar] [CrossRef]
- Wang, Y.; Yao, N.; Wei, K.; Jiang, L.; Hanif, S.; Wang, Z.; Pei, C. The efficacy and safety of probiotics for prevention of chemoradiotherapy-induced diarrhea in people with abdominal and pelvic cancer: A systematic review and meta-analysis. Eur. J. Clin. Nutr. 2016, 70, 1246. [Google Scholar] [CrossRef]
- Bowen, J.M.; Gibson, R.J.; Coller, J.K.; Blijlevens, N.; Bossi, P.; Al-Dasooqi, N.; Bateman, E.H.; Chiang, K.; de Mooij, C.; Mayo, B. Systematic review of agents for the management of cancer treatment-related gastrointestinal mucositis and clinical practice guidelines. Supportive Care Cancer 2019, 27, 4011–4022. [Google Scholar] [CrossRef]
- Giralt, J.; Regadera, J.P.; Verges, R.; Romero, J.; de la Fuente, I.; Biete, A.; Villoria, J.; Cobo, J.M.; Guarner, F. Effects of probiotic Lactobacillus casei DN-114 001 in prevention of radiation-induced diarrhea: Results from multicenter, randomized, placebo-controlled nutritional trial. Int. J. Radiat. Oncol. Biol. Phys. 2008, 71, 1213–1219. [Google Scholar] [CrossRef]
- Brok, J.; Thorlund, K.; Wetterslev, J.; Gluud, C. Apparently conclusive meta-analyses may be inconclusive—trial sequential analysis adjustment of random error risk due to repetitive testing of accumulating data in apparently conclusive neonatal meta-analyses. Int. J. Epidemiol. 2008, 38, 287–298. [Google Scholar] [CrossRef]
- Wetterslev, J.; Thorlund, K.; Brok, J.; Gluud, C. Trial sequential analysis may establish when firm evidence is reached in cumulative meta-analysis. J. Clin. Epidemiol. 2008, 61, 64–75. [Google Scholar] [CrossRef] [PubMed]
- Mansouri-Tehrani, H.; Khorasgani, M.R.; Roayaei, M. Effects of probiotics with or without honey on radiation-induced diarrhea. Int. J. Radiat. Res. 2016, 14, 205–213. [Google Scholar] [CrossRef]
- Linn, Y.H.; Thu, K.K.; Win, N.H.H. Effect of probiotics for the prevention of acute radiation-induced diarrhoea among cervical cancer patients: A randomized double-blind placebo-controlled study. Probiotics Antimicrob. Proteins 2019, 11, 638–647. [Google Scholar] [CrossRef] [PubMed]
- Higgins, J.P.; Green, S. Cochrane Handbook for Systematic Reviews of Interventions; Version 5.1.0; The Cochrane Collaboration: Oxford, UK, 2011. [Google Scholar]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. Ann. Intern. Med. 2009, 151, 264–269. [Google Scholar] [CrossRef]
- Higgins, J.P.; Altman, D.G.; Gøtzsche, P.C.; Jüni, P.; Moher, D.; Oxman, A.D.; Savović, J.; Schulz, K.F.; Weeks, L.; Sterne, J.A. The Cochrane Collaboration’s tool for assessing risk of bias in randomised trials. BMJ 2011, 343, d5928. [Google Scholar] [CrossRef]
- Egger, M.; Smith, G.D.; Schneider, M.; Minder, C. Bias in meta-analysis detected by a simple, graphical test. BMJ 1997, 315, 629–634. [Google Scholar] [CrossRef]
- Guyatt, G.H.; Oxman, A.D.; Vist, G.E.; Kunz, R.; Falck-Ytter, Y.; Alonso-Coello, P.; Schünemann, H.J. GRADE: An emerging consensus on rating quality of evidence and strength of recommendations. BMJ 2008, 336, 924–926. [Google Scholar] [CrossRef]
- Amarenco, G. Bristol Stool Chart: Étude prospective et monocentrique de «l’introspection fécale» chez des sujets volontaires. Progrès en Urologie 2014, 24, 708–713. [Google Scholar] [CrossRef]
- Lewis, S.; Heaton, K. Stool form scale as a useful guide to intestinal transit time. Scand. J. Gastroenterol. 1997, 32, 920–924. [Google Scholar] [CrossRef]
- Delia, P.; Sansotta, G.; Donato, V.; Frosina, P.; Messina, G.; De Renzis, C.; Famularo, G. Use of probiotics for prevention of radiation-induced diarrhea. World J. Gastroenterol. WJG 2007, 13, 912. [Google Scholar] [CrossRef]
- Salminen, E.; Elomaa, I.; Minkkinen, J.; Vapaatalo, H.; Salminen, S. Preservation of intestinal integrity during radiotherapy using live Lactobacillus acidophilus cultures. Clin. Radiol. 1988, 39, 435–437. [Google Scholar] [CrossRef]
- Demers, M.; Dagnault, A.; Desjardins, J. A randomized double-blind controlled trial: Impact of probiotics on diarrhea in patients treated with pelvic radiation. Clin. Nutr. 2014, 33, 761–767. [Google Scholar] [CrossRef] [PubMed]
- Castro, M.; Sanchez, P.; Glasberg, J.; Horie, L.; Waitzberg, D. P112 effects of probiotic in prevention of radiation-induced diarrhea. Clin. Nutr. Suppl. 2009, 2, 72–73. [Google Scholar] [CrossRef]
- Chitapanarux, I.; Chitapanarux, T.; Traisathit, P.; Kudumpee, S.; Tharavichitkul, E.; Lorvidhaya, V. Randomized controlled trial of live lactobacillus acidophilus plus bifidobacterium bifidum in prophylaxis of diarrhea during radiotherapy in cervical cancer patients. Radiat. Oncol. 2010, 5, 31. [Google Scholar] [CrossRef] [PubMed]
- Touchefeu, Y.; Montassier, E.; Nieman, K.; Gastinne, T.; Potel, G.; Bruley des Varannes, S.; Le Vacon, F.; de La Cochetière, M. Systematic review: The role of the gut microbiota in chemotherapy-or radiation-induced gastrointestinal mucositis–current evidence and potential clinical applications. Aliment. Pharmacol. Ther. 2014, 40, 409–421. [Google Scholar] [CrossRef]
- Johansson, M.E.; Gustafsson, J.K.; Sjöberg, K.E.; Petersson, J.; Holm, L.; Sjövall, H.; Hansson, G.C. Bacteria penetrate the inner mucus layer before inflammation in the dextran sulfate colitis model. PLoS ONE 2010, 5, e12238. [Google Scholar] [CrossRef]
- Bosset, J.; Calais, G.; Daban, A.; Berger, C.; Radosevic-Jelic, L.; Maingon, P.; Bardet, E.; Pierart, M.; Briffaux, A. Preoperative chemoradiotherapy versus preoperative radiotherapy in rectal cancer patients: Assessment of acute toxicity and treatment compliance: Report of the 22921 randomised trial conducted by the EORTC Radiotherapy Group. Eur. J. Cancer 2004, 40, 219–224. [Google Scholar] [CrossRef]
| First Author | Year/Area | Mean Age | Probiotics/Placebo | Probiotics with diarrhea/Placebo with diarrhea | Primary Tumor Site | Type of therapy | Total Radiation Dose | Chemotherapy |
|---|---|---|---|---|---|---|---|---|
| Linn | 2018/Myanmar | 52.5-57.38 | 26/28 | 14/23 | Cervical Carcinoma | external beam pelvic radiotherapy | 50Gy | Not specified |
| Tehrani | 2016/Iran | 62 | 22/24 | 7/17 | Pelvic cancers (colorectal, prostate, endometrial, bladder, ovary, cervix, bone sarcoma) | conventional radiotherapy | 4000 to 5000 cGy (1.8Gy/day) with 18 MV | Not specified |
| Salminen | 1988/Finland | 40–75 | 11/10 | 3/9 | Cervix or uterus carcinoma | Internal and external pelvic RT and intracavitary caesium | 50Gy for pelvic, 80Gy for the tumour | Intracavitary caesium |
| Delia | 2007/Italy | No | 243/239 | 77/124 | Sigmoid, rectal or cervical cancers | Postoperative RT | 60–70 Gy | Not specified |
| Giralt | 2008/Spain | ≤18 | 44/41 | 8/11 | Endometrial adenocarcinoma or advanced cervical squamous cell carcinoma | Postoperative RT concomitant weekly cisplatin (only for patients with cervical cancer) | 45–50.4 Gy | Weekly Cisplatin 40 mg/m2 |
| Castro | 2009/Brazil | NR | 20/20 | 7/13 | Cervical or endometrial cancer | RT treatments | NR | Not specified |
| Chitapanarux | 2010/Thailand | 18–65 | 32/31 | 3/14 | Cervical cancer | Pelvic RT and weekly cisplatin | 200 cGy per fraction, five fractions per week | Weekly cisplatin 40 mg/m2 for 6 weeks |
| Demers | 2014/Canada | >18 | 140/86 | 118/80 | Gynecologic, rectal, or prostate cancer | RT for gynecologic cancers without chemotherapy, gynecologic or rectal cancers with chemotherapy | 40 Gy for the pelvic level | Not specified |
| First Author | Daily Dosage | Medication usage | Route | Timing | Probiotics Source | Diarrhea grade | Numbers of patients going into randomization | Numbers of patients assigned to treatment | Numbers of patients assigned to placebo | Genus | Species | Strain | Single or combinations | With or without Bifidobacterium |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Linn | 1.75 × 109 CFU | t.i.d | Oral | from the first day of radiotherapy until the end of radiotherapy | Fame Pharmaceuticals, Myanmar | Common Terminology Criteria for Adverse Events | 57 | 28 | 29 | Lactobacillus, Bifidobacterium | L. acidophilus, B. animalis | L. acidophilus subsp LA-5, B. animalis subsp. Lactis BB-12 | Combined | With Bifidobacterium |
| Tehrani | 1.5 × 109 CFU, 1.5 × 1010 CFU, 3.5 × 109 CFU 2.5 × 108 CFU, 1 × 1010 CFU 5 × 108, CFU, 1.5 × 108 CFU | b.d | Oral | one week before starting pelvic radiotherapy until the end of radiotherapy | Zist Takhmir Company, Tehran, Iran | Common Toxicity Criteria of the National Cancer Institute | 52 | 26 | 26 | lactobacillus, Bifidobacterium, Streptococcus | L. casei, L. acidophilus, L. rhamnosus, L bulgaricus, B. breve, B. longum, S. thermophilus, | Nil | Combined | With Bifidobacterium |
| Salminen | 2 × 109 CFU | q.d. | Oral | 5 days prior to radiotherapy,10 days after finishing radiotherapy | NA | NR | 24 | 12 | 12 | Lactobacillus | L. acidophilus | L. acidophilus (NCDO 1748) | Single | Without bifidobacterium |
| Delia | 1.35 × 1012 CFU | t.i.d. | Oral | The first day of RT until the end of therapy | VSL Pharmaceuticals, Fort Lauderdale, MD, USA | WHO grading | 490 | 245 | 245 | Lactobacillus & Bifidobacterium & Streptococcus | L. casei, L. plantarum, L. acidophilus, and L. delbruekii subsp. Bulgaricus & B. longum, B. breve, B. infantis, S. salivarius | S. salivrius subsp. Thermophilus | Combined | With Bifidobacterium |
| Giralt | 3 × 108 CFU | t.i.d. | Oral | One week | NR | Common Toxicity Criteria of the NCI | 118 | 44 out of 56 | 41 out of 62 | Lactobacillus | L. casei | Lactobacillus casei DN-114 001 | Single | Without bifidobacterium |
| Castro | NR | NR | Oral | NR | NR | Common Toxicity Criteria of the NCI | 40 | 20 | 20 | Lactobacillus & Bifidobacterium | L. casei & B. breve | L. casei subsp shirota & B. breve spp. | Combined | With Bifidobacterium |
| Chitapanarux | 4 × 109 CFU | b.i.d | Oral | 7 days before RT and continuing everyday during RT | Laboratio, Farmaceutico SIT, Mede, Italy | Common Toxicity Criteria of the NCI | 63 | 32 | 31 | Lactobacillus & Bifidobacterium | L. acidophilus & B. bifidum | L. acidophilus viv Lyophisat & B. bifidum viv Lyophisat | Combined | With Bifidobacterium |
| Demers | 2.6 × 109 CFU or 3 × 1010 CFU | b.i.d or t.i.d. | Oral | From the first day and ended on the last day of RT | Bifilact, Virage Santé Québec city, Canada | WHO grading | 246 | 91 | 91 | Lactobacillus & Bifidobacterium | L. acidophilus and B. longum | L. acidophilus LAC-361 and B. longum BB-536 | Combined | With Bifidobacterium |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Devaraj, N.K.; Suppiah, S.; Veettil, S.K.; Ching, S.M.; Lee, K.W.; Menon, R.K.; Soo, M.J.; Deuraseh, I.; Hoo, F.K.; Sivaratnam, D. The Effects of Probiotic Supplementation on the Incidence of Diarrhea in Cancer Patients Receiving Radiation Therapy: A Systematic Review with Meta-Analysis and Trial Sequential Analysis of Randomized Controlled Trials. Nutrients 2019, 11, 2886. https://doi.org/10.3390/nu11122886
Devaraj NK, Suppiah S, Veettil SK, Ching SM, Lee KW, Menon RK, Soo MJ, Deuraseh I, Hoo FK, Sivaratnam D. The Effects of Probiotic Supplementation on the Incidence of Diarrhea in Cancer Patients Receiving Radiation Therapy: A Systematic Review with Meta-Analysis and Trial Sequential Analysis of Randomized Controlled Trials. Nutrients. 2019; 11(12):2886. https://doi.org/10.3390/nu11122886
Chicago/Turabian StyleDevaraj, Navin Kumar, Subapriya Suppiah, Sajesh K. Veettil, Siew Mooi Ching, Kai Wei Lee, Rohit Kunnath Menon, Man Jun Soo, Inas Deuraseh, Fan Kee Hoo, and Dhashani Sivaratnam. 2019. "The Effects of Probiotic Supplementation on the Incidence of Diarrhea in Cancer Patients Receiving Radiation Therapy: A Systematic Review with Meta-Analysis and Trial Sequential Analysis of Randomized Controlled Trials" Nutrients 11, no. 12: 2886. https://doi.org/10.3390/nu11122886
APA StyleDevaraj, N. K., Suppiah, S., Veettil, S. K., Ching, S. M., Lee, K. W., Menon, R. K., Soo, M. J., Deuraseh, I., Hoo, F. K., & Sivaratnam, D. (2019). The Effects of Probiotic Supplementation on the Incidence of Diarrhea in Cancer Patients Receiving Radiation Therapy: A Systematic Review with Meta-Analysis and Trial Sequential Analysis of Randomized Controlled Trials. Nutrients, 11(12), 2886. https://doi.org/10.3390/nu11122886





