Multiple Micronutrient Powder Reduces Vitamin E Deficiency in Brazilian Children: A Pragmatic, Controlled Clinical Trial
Abstract
:1. Introduction
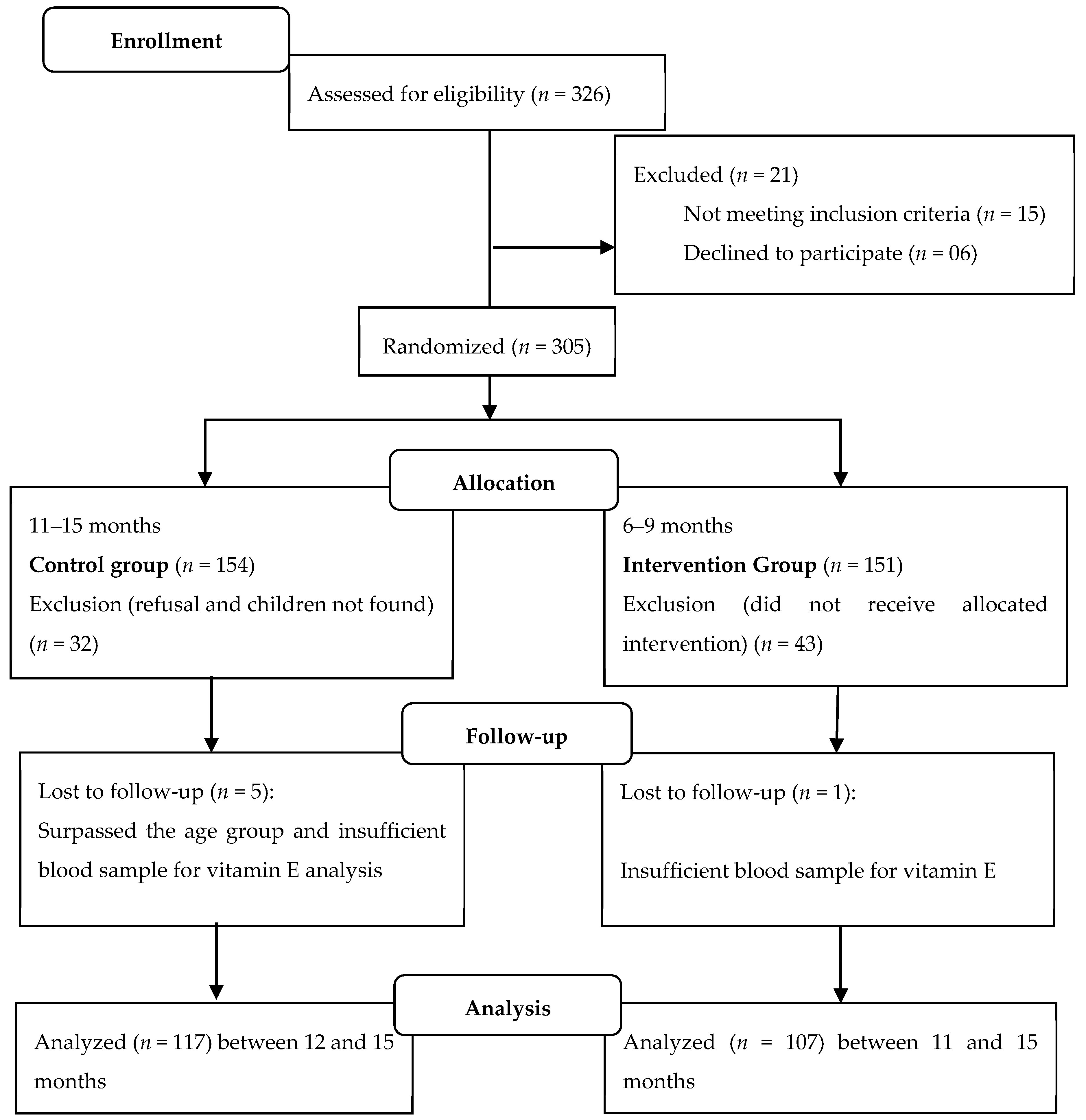
2. Materials and Methods
2.1. Study Design and Population
2.2. Data Collection
2.3. Groups and Interventions
2.4. Anthropometric Assessment
2.5. Biochemical Assessment
2.6. Statistical Analysis
2.7. Ethical Aspects
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| AGP | Alpha-1-glycoprotein |
| CG | Control group |
| CRP | C-reactive protein |
| ENFAC | Estudo Nacional de Fortificação Caseira da Alimentação Complementar |
| ICF | Informed Consent Form |
| HIV | Human Immunodeficiency Virus |
| HPLC | High Performance Liquid Chromatography |
| IG | Intervention group |
| IRIS | International Research on Infant Supplementation |
| IQR | Interquartile range |
| PSE | Health at School Program |
| PR | Prevalence ratio |
| SD | Standard deviation |
| WHO | World Health Organization |
References
- Jiang, Q. Natural forms of vitamin E: Metabolism, antioxidant, and anti-inflammatory activities and their role in disease prevention and therapy. Free Radic. Biol. Med. 2014, 72, 76–90. [Google Scholar] [CrossRef] [PubMed]
- Marquez, M.; Yépez, C.E.; Sútil-Naranjo, R.; Rincón, M. Aspectos básicos y determinación de las vitaminas antioxidantes E y A. Investig. Clin. 2002, 43, 191–204. [Google Scholar]
- Raizman, J.E.; Cohen, A.H.; Teodoro-Morrison, T.; Wan, B.; Khun-Chen, M.; Wilkenson, C.; Bevilaqua, V.; Adeli, K. Pediatric reference value distributions for vitamins A and E in the CALIPER cohort and establishment of age stratified reference intervals. Clin. Biochem. 2014, 47, 812–815. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Vitamin and Mineral Requirements in Human Nutrition: Report of a Joint FAO/WHO Expert Consultation; World Health Organization: Bangkok, Thailand, 1998. [Google Scholar]
- Nordic Nutrition Recommendations. Part 3: Vitamin A, D, E, K, Thiamin, Riboflavin, Niacin, Vitamin B6, Folate, Vitamin B12, Biotin, Pantothenic Acid and Vitamin C; Nordic Council of Ministers: Copenhagen, Denmark, 2014. [Google Scholar]
- Giraud, D.W.; Kim, Y.N.; Cho, Y.O.; Driskell, J.A. Vitamin E inadequacy observed in a group of 2 to 6-years-old children living in Kwangju, Republic of Korea. Int. J. Vitam. Nutr. Res. 2008, 78, 148–155. [Google Scholar] [CrossRef] [PubMed]
- Luna, R.C.P.; Nascimento, C.C.C.; Bandeira, G.T.P.; Oliveira, A.F.; Monteiro, M.G.C.A.; Barbosa, F.P.T.; Albuquerque, T.M.; Silva, A.H.A.; Moraes, R.M.; Lima, R.T.; et al. Baixo consumo habitual de alimentos fonte de vitamina E em população infantil. Rev. Inst. Adolfo Lutz 2011, 70, 213–219. [Google Scholar]
- Paiva-Bandeira, G.T.; Rios-Asciutti, L.S.; Riviera, A.A.; Rodrigues-Gonçalves, M.C.; Pordeus-De-Lima, R.C.; Albuquerque, T.M.; Marcos-De-Morais, R.; Toledo-Vianna, R.P.; Farias-De-Oliveira, A.; Teixeira-Lima, R.; et al. Relação entre renda e consumo alimentar habitual de β-caroteno, vitamina C e vitamina E de crianças. Rev. Salud Publica 2011, 13, 386–397. [Google Scholar] [PubMed]
- Fares, S.; Sethom, M.M.; Khouaja-Mokrani, C.; Jabnoun, S.; Feki, M.; Kaabachi, N. Vitamina A, E, and D deficiencies in Tunisian very low birth weight neonates: Prevalence and risk factors. Pediatrics Neonatol. 2014, 55, 196–201. [Google Scholar] [CrossRef] [PubMed]
- Van Biervliet, S.; Van Biervliet, J.; Bernard, D.; Matthys, M.; Vercaemst, R.; Blaton, A.V. Serum α-tocopherol and selenium in Belgian infants and children. Biol. Trace Elem. Res. 2001, 79, 115–120. [Google Scholar] [CrossRef]
- Augusto, R.A.; Cobayashi, F.; Cardoso, M.A. Associations between low consumption of fruits and vegetables and nutritional deficiencies in Brazilian schoolchildren. Public Health Nutr. 2014, 18, 927–935. [Google Scholar] [CrossRef] [PubMed]
- Morais, G.C. Fatores Associados ao Estado Nutricional de Vitamina E em Lactentes Atendidos da Atenção Básica de Saúde de Goiânia-GO. Dissertação. Master’s Thesis (Mestrado em Nutrição e Saúde), Faculdade de Nutrição, Universidade Federal de Goiás, Goiânia, Brazil, 2016. [Google Scholar]
- Traber, M.G. Vitamin E inadequacy in humans: Causes and consequences. Adv. Nutr. 2014, 5, 503–514. [Google Scholar] [CrossRef] [PubMed]
- National Institutes of Health. Vitamin E: Fact Sheet for Health Professionals. Rockville. 2013. Available online: https://ods.od.nih.gov/factsheets/VitaminE-HealthProfessional/ (accessed on 19 January 2016).
- World Health Organization. Guideline: Use of Multiple Micronutrient Powders for Home Fortification of Foods Consumed by Infants and Children 6–23 Months of Age; World Health Organization: Geneva, Switzerland, 2011. [Google Scholar]
- Ministério da Saúde; Secretaria de Atenção à Saúde; Departamento de Atenção Básica. Estratégia de fortificação da alimentação infantil com micronutrientes (vitaminas e minerais) em pó; Manual de operacional/Ministério da Saúde, Secretaria de Atenção à Saúde, Departamento de Atenção Básica; Ministério da Saúde: Brasília, Brazil, 2014. [Google Scholar]
- Brazil. Resolução RDC nº344, de 13 de dezembro de 2002. Aprova o regulamento técnico para a fortificação das farinhas de trigo e das farinhas de milho com ferro e ácido fólico. Agência Nacional de Vigilância Sanitária. Available online: http://189.28.128.100/dab/docs/portaldab/documentos/resolucao_rdc344_2002.pdf (accessed on 10 March 2019).
- Ministério da Saúde. Cadernos de Atenção Básica: Carências de Micronutrients; Ministério da Saúde: Brasília, Brazil, 2007. [Google Scholar]
- Cardoso, M.A.; Augusto, R.A.; Bortolini, G.A.; Oliveira, C.S.M.; Tietzmann, D.C.; Sequeira, L.A.S.; Hadler, M.C.C.M.; Peixoto, M.R.G.; Muniz, P.T.; Vitolo, M.R.; et al. Effect of providing multiple micronutrients in powder through primary healthcare on anemia in young Brazilian children: A multicentre pragmatic controlled trial. PLoS ONE 2016, 11, e0151097. [Google Scholar] [CrossRef] [PubMed]
- Untoro, J.; Karyadi, E.; Wibowo, L.; Erhardt, M.W.; Gross, R. Multiple micronutrient supplements improve micronutrient status and anemia but not growth and morbidity of Indonesian infants: A randomized, double-blind, placebo-controlled trial. J. Nutr. 2005, 135, S639–S645. [Google Scholar] [CrossRef] [PubMed]
- Smuts, C.M.; Lombard, C.J.; Benadé, A.J.S.; Dhansay, M.A.; Berger, J.; Hop, L.T.; De Romaña, G.L.; Untoro, J.; Karyadi, E.; Erhardt, J.; et al. Efficacy of a foodlet-based multiple micronutrient supplement for preventing growth faltering, anemia, and micronutrient deficiency of infants: The four country IRIS trial pooled data analysis. J. Nutr. 2005, 135, 631S–638S. [Google Scholar] [CrossRef] [PubMed]
- De Romaña, G.L.; Cusirramos, S.; De Romaña, D.; Gross, R. Efficacy of multiple micronutrient supplementation for improving anemia, micronutrient status, growth and morbidity of Peruvian infants. J. Nutr. 2005, 135, S646–S652. [Google Scholar] [CrossRef] [PubMed]
- Hop, L.T.; Berger, J. Multiple micronutrient supplementation improves anemia, micronutrient nutrient status, and growth of Vietnamese infants: Double-blind, randomized, placebo-controlled trial. J. Nutr. 2005, 135, 660S–665S. [Google Scholar] [CrossRef] [PubMed]
- Santos, N.C.C.B.; Vaz, E.M.C.; Nogueira, J.A.; Toso, B.R.G.O.; Collet, N.; Reichert, A.P.S. Presença e extensão dos atributos de atenção primária à saúde da criança em distintos modelos de cuidado. Cad. Saude Publica 2018, 34, e00014216. [Google Scholar] [CrossRef] [PubMed]
- Organização Pan-Americana da Saúde. Inovando o Papel da Atenção Primária nas Redes de Atenção à Saúde: Resultados do Laboratório de Inovação em Quatro Capitais Brazileiras; Organização Pan-Americana da Saúde: Brasília, Brazil, 2011. [Google Scholar]
- Lohman, T.G.; Roche, A.F.; Martorel, R. Anthropometric Standardization Reference Manual; Human Kinetics Books: Champaign, IL, USA, 1988. [Google Scholar]
- Gomes, L.F.; Alves, A.F.; Sevanian, A.; Peres, C.D.E.A.; Cendoroglo, M.S.; De Mello-Almada, C.; Quirino, L.M.; Ramos, L.R.; Junqueira, V.B. Role of beta2-glycoprotein I, LDL-, and antioxidant concentrations in hypercholesterolemic elderly subjects. Antioxid Redox Signal 2004, 6, 237–244. [Google Scholar] [CrossRef] [PubMed]
- Thurnham, D.I.; Davies, J.A.; Crump, B.J.; Situnayake, R.D. The use of different lipids to express serum tocopherol: Lipid ratios for the measurement of vitamin E status. Ann. Clin. Biochem. 1986, 23, 514–520. [Google Scholar] [CrossRef] [PubMed]
- Thurnham, D.I.; Mccabe, G.P.; Northrop-Clewes, C.A.; Nestel, P. Effects of subclinical infection on plasma retinol concentrations and assessment of prevalence of vitamin A deficiency: Meta-analysis. Lancet 2003, 362, 2052–2058. [Google Scholar] [CrossRef]
- Abizari, A.R.; Azupogo, F.; Brouwer, I.D. Subclinical inflammation influences the association between vitamin A and iron status among schoolchildren in Ghana. PLoS ONE 2017, 12, e0170747. [Google Scholar] [CrossRef] [PubMed]
- Kumar, M.V.; Rajagopalan, S. Impact of a multiple-micronutrient food supplement on the nutritional status of schoolchildren. Food Nutr. Bull. 2006, 27, 203–210. [Google Scholar] [CrossRef] [PubMed]
- Lodge, J.K.; Hall, W.L.; Jeanes, Y.M.; Proteggente, A.R. Physiological factors influencing vitamina E biokinetics. Ann. N. Y. Acad. Sci. 2004, 1031, 60–73. [Google Scholar] [CrossRef] [PubMed]
- Ministério da Saúde. Orientações Para a Coleta e Análise de Dados Antropométricos em Serviços de Saúde; Ministério da Saúde: Brasília, Brazil, 2011. [Google Scholar]
- Institute of Medicine. Dietary Reference Intakes for Vitamin C, Vitamin E, Selenium, and Carotenoids; National Academy Press: Washington, DC, USA, 2000. [Google Scholar]
- Rivera, J.A.; González-Cossío, T.; Flores, M.; Romero, M.; Rivera, M.; Téllez-Rojo, M.M.; Rosado, J.L.; Brown, K.H. Multiple micronutrient supplementation increases the growth of Mexican infants. Am. J. Clin. Nutr. 2001, 74, 657–663. [Google Scholar] [CrossRef] [PubMed]
- Dror, D.K.; Allen, L.H. Vitamin E deficiency in developing countries. Food Nutr. Bull. 2011, 32, 124–143. [Google Scholar] [CrossRef] [PubMed]
- Millward, D.J. Nutrition, infection and stunting: The roles of deficiencies of individual nutrients and foods, and of inflammation, as determinants of reduced linear growth of children. Nutr. Res. Rev. 2017, 30, 50–72. [Google Scholar] [CrossRef] [PubMed]
- Suchdev, O.S.; Williams, A.M.; Mei, Z.; Flores-Ayala, R.; Pasricha, S.; Rogers, L.M.; Namaste, S.M.L. Assessment of iron status in settings of inflammation: Challenges and potential approaches. Am. J. Clin. Nutr. 2017, 106, 1626S–1633S. [Google Scholar] [CrossRef] [PubMed]
- Wit, J.M.; Himes, J.H.; Van Buuren, S.; Denno, D.M.; Suchdev, P.S. Practical Application of Linear Growth Measurements in Clinical Research in Low- and Middle-Income Countries. Horm. Res. Paediatr. 2017, 88, 79–90. [Google Scholar] [CrossRef] [PubMed]
| Variable | CG (n = 117) | IG (n = 107) | p Value |
|---|---|---|---|
| Age of child (months), mean, and SD | 13.65 ± 0.87 | 12.94 ± 0.94 | <0.001 1 |
| Female gender, n (%) | 55 (47.01) | 53 (49.53) | 0.71 2 |
| Birth weight, mean and SD | 3247.5 ± 469.8 | 3288.5 ± 426.1 | 0.49 3 |
| Maternal education <9 years, n (%) | 33 (28.45) | 21 (19.81) | 0.13 2 |
| Mother’s age (years), median (IQR) | 28 (24; 32) | 27 (22; 31) | 0.18 1 |
| Current breastfeeding, n (%) | 67 (57.26) | 59 (55.14) | 0.75 2 |
| Number of prenatal consultations, median (IQR) | 8 (6;10) | 8 (6;9) | 0.80 1 |
| Race, n (%) | 0.95 4 | ||
| White | 9 (7.76) | 10 (9.35) | |
| Brown | 102 (87.93) | 93 (86.92) | |
| Black | 5 (4.31) | 4 (3.74) |
| Variable | CG (n = 117) | IG (n = 107) | p Value |
|---|---|---|---|
| Anthropometric data | |||
| Weight (kg), mean and SD | 10.02 ± 1.20 | 9.85 ± 1.19 | 0.29 1 |
| Length (cm), median (IQR) | 77 (75; 79) | 77.8 (75; 79.5) | 0.68 2 |
| LAZ (Z-score), mean and SD | 0.07 ± 1.13 | 0.41 ± 1.09 | 0.02 1 |
| Clinical data | |||
| Presence of fever, n (%) | 39 (33.33) | 24 (22.43) | 0.07 3 |
| Presence of cough, n (%) | 53 (45.30) | 21 (19.63) | <0.001 3 |
| Presence of wheezing, n (%) | 35 (29.91) | 16 (14.95) | 0.008 3 |
| Presence of diarrhea, n (%) | 27 (23.08) | 22 (20.56) | 0.65 3 |
| Biochemical data | |||
| Serum α-tocopherol (µmol/L), median (IQR) | 3.6 (0.09; 9.7) | 17.2 (12.9; 21.2) | <0.001 2 |
| CRP (mg/L), median (IQR) | 0.5 (0.2; 1.5) | 0.3 (0.2; 1.0) | 0.19 2 |
| AGP (mg/dL), median (IQR) | 74.5 (61.8; 109.5) | 72.4 (61.4; 92.3) | 0.08 2 |
| Variable | CG (n = 117) | IG (n = 107) | p Value |
|---|---|---|---|
| Biochemical data | |||
| Vitamin E deficiency (<11.6 µmol/L), n (%) | 95 (85.6) | 16 (14.4) | <0.001 1 |
| Vitamin E deficiency, adjusted PR (95% CI) | 1 | 0.18 (0.11; 0.30) | <0.001 2 |
| CRP > 5 mg/L, n (%) | 14 (12.39) | 6 (5.83) | 0.096 1 |
| CRP > 5 mg/L, adjusted PR (95% CI) | 1 | 0.40 (0.13; 1.29) | 0.13 2 |
| AGP > 100 mg/dL, n (%) | 37 (33.04) | 20 (19.42) | 0.024 1 |
| AGP, adjusted PR (95% CI) | 1 | 0.52 (0.31; 0.85) | 0.009 3 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
M. C. Lobo, L.; M. Schincaglia, R.; G. Peixoto, M.d.R.; C. M. Hadler, M.C., ENFAC Working Group. Multiple Micronutrient Powder Reduces Vitamin E Deficiency in Brazilian Children: A Pragmatic, Controlled Clinical Trial. Nutrients 2019, 11, 2730. https://doi.org/10.3390/nu11112730
M. C. Lobo L, M. Schincaglia R, G. Peixoto MdR, C. M. Hadler MC ENFAC Working Group. Multiple Micronutrient Powder Reduces Vitamin E Deficiency in Brazilian Children: A Pragmatic, Controlled Clinical Trial. Nutrients. 2019; 11(11):2730. https://doi.org/10.3390/nu11112730
Chicago/Turabian StyleM. C. Lobo, Lina, Raquel M. Schincaglia, Maria do Rosário G. Peixoto, and Maria Claret C. M. Hadler ENFAC Working Group. 2019. "Multiple Micronutrient Powder Reduces Vitamin E Deficiency in Brazilian Children: A Pragmatic, Controlled Clinical Trial" Nutrients 11, no. 11: 2730. https://doi.org/10.3390/nu11112730
APA StyleM. C. Lobo, L., M. Schincaglia, R., G. Peixoto, M. d. R., & C. M. Hadler, M. C., ENFAC Working Group. (2019). Multiple Micronutrient Powder Reduces Vitamin E Deficiency in Brazilian Children: A Pragmatic, Controlled Clinical Trial. Nutrients, 11(11), 2730. https://doi.org/10.3390/nu11112730





