Hepatoprotective Mechanisms of Taxifolin on Carbon Tetrachloride-Induced Acute Liver Injury in Mice
Abstract
:1. Introduction
2. Materials and Methods
2.1. Animals
2.2. CCl4-Induced Hepatotoxicity
2.3. Histological Analyses
2.4. MDA Assays
2.5. Measurements of Antioxidant Enzyme Activity
2.6. Statistical Analysis
3. Results
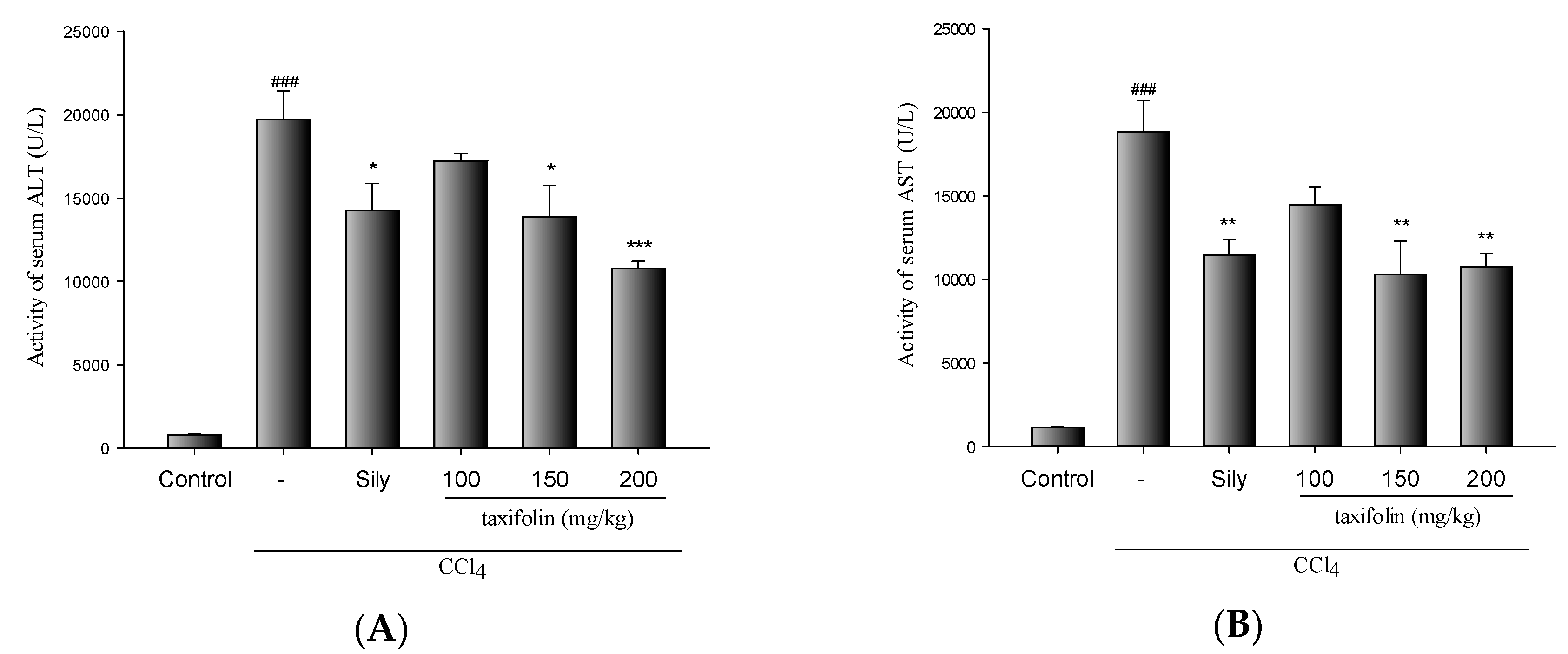
3.1. CCl4-Induced Acute Liver Injury
3.2. Histological Analyses
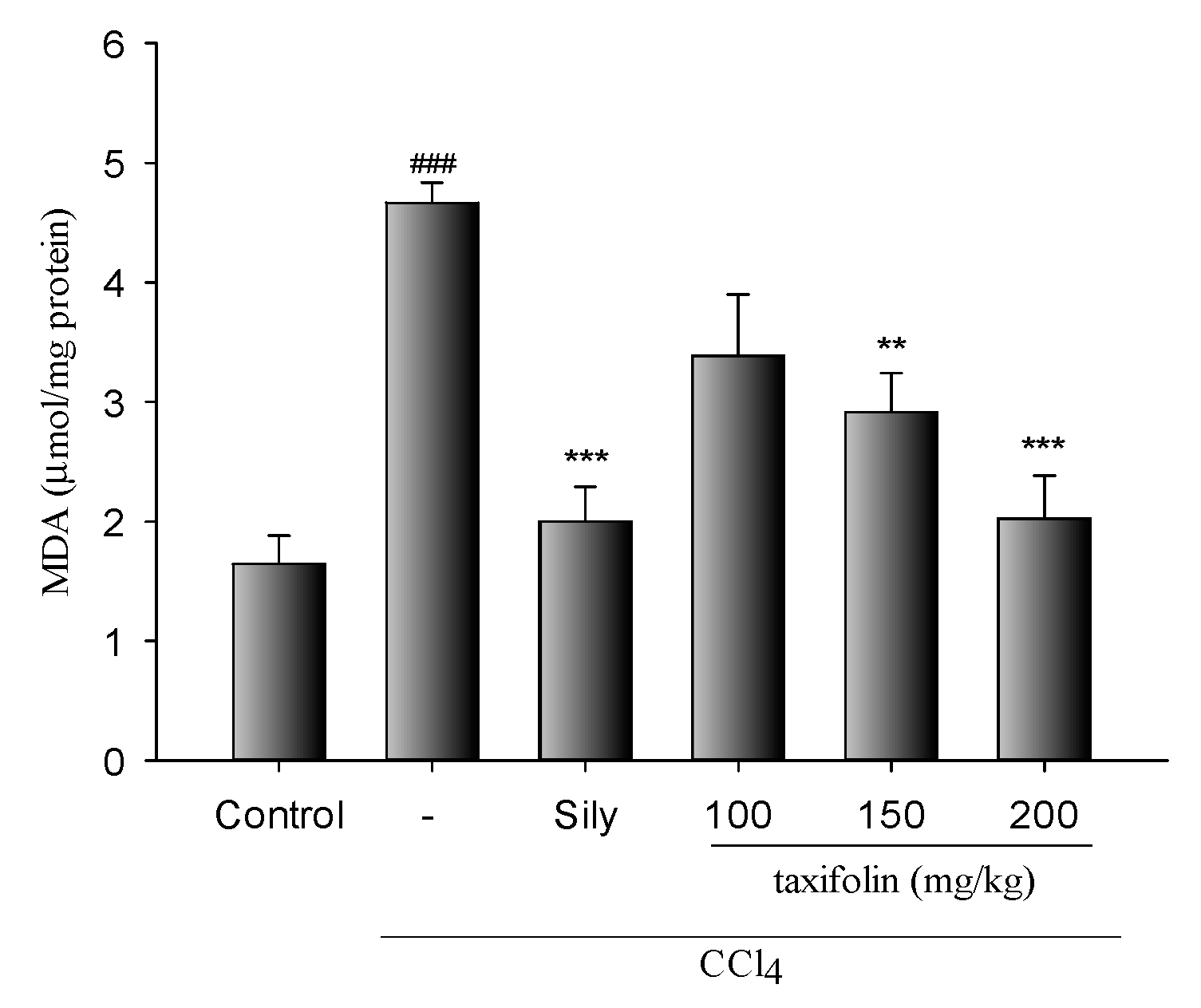
3.3. MDA Levels
3.4. Antioxidant Enzyme Activities
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Nordberg, J.; Arner, E.S. Reactive oxygen species, antioxidants, and the mammalian thioredoxin system. Free Radic. Biol. Med. 2001, 31, 1287–1312. [Google Scholar] [CrossRef]
- Kietzmann, T.; Gorlach, A. Reactive oxygen species in the control of hypoxia-inducible factor-mediated gene expression. Semin. Cell Dev. Biol. 2005, 16, 474–486. [Google Scholar] [CrossRef] [PubMed]
- Seifried, H.E.; Anderson, D.E.; Fisher, E.I.; Milner, J.A. A review of the interaction among dietary antioxidants and reactive oxygen species. J. Nutr. Biochem. 2007, 18, 567–579. [Google Scholar] [CrossRef] [PubMed]
- Tham, N.T.; Hwang, S.R.; Bang, J.H.; Yi, H.; Park, Y.I.; Kang, S.J.; Kang, H.G.; Kim, Y.S.; Ku, H.O. High-content analysis of in vitro hepatocyte injury induced by various hepatotoxicants. J. Vet. Sci. 2019, 20, 34–42. [Google Scholar] [CrossRef] [PubMed]
- Kodavanti, P.R.; Joshi, U.M.; Young, R.A.; Meydrech, E.F.; Mehendale, H.M. Protection of hepatotoxic and lethal effects of CCl4 by partial hepatectomy. Toxicol. Pathol. 1989, 17, 494–505. [Google Scholar] [CrossRef] [PubMed]
- Brattin, W.J.; Glende, E.A., Jr.; Recknagel, R.O. Pathological mechanisms in carbon tetrachloride hepatotoxicity. J. Free Radic. Biol. Med. 1985, 1, 27–38. [Google Scholar] [CrossRef]
- Basu, S. Carbon tetrachloride-induced lipid peroxidation: Eicosanoid formation and their regulation by antioxidant nutrients. Toxicology 2003, 189, 113–127. [Google Scholar] [CrossRef]
- Chu, S.C.; Hsieh, Y.S.; Lin, J.Y. Inhibitory Effects of Flavonoids on Moloney Murine Leukemia Virus Reverse Transcriptase Activity. J. Nat. Prod. 1992, 55, 179–183. [Google Scholar] [CrossRef] [PubMed]
- Kang, J.T.; Kwon, D.K.; Park, S.J.; Kim, S.J.; Moon, J.H.; Koo, O.J.; Jang, G.; Lee, B.C. Quercetin improves the in vitro development of porcine oocytes by decreasing reactive oxygen species levels. J. Vet. Sci. 2013, 14, 15–20. [Google Scholar] [CrossRef] [PubMed]
- Kang, C.B.; Kim, C.K.; Song, S.H.; Ha, W.S. Study on mechanism of multistep hepatotumorigenesis in rat: Development of hepatotumorigenesis. J. Vet. Sci. 2001, 41, 583–589. [Google Scholar]
- Manna, S.; Bhattacharyya, D.; Mandal, T.K.; Das, S. Repeated dose toxicity of alfa-cypermethrin in rats. J. Vet. Sci. 2004, 5, 241–245. [Google Scholar] [CrossRef] [PubMed]
- Tsai, J.C.; Chiu, C.S.; Chen, Y.C.; Lee, M.S.; Hao, X.Y.; Hsieh, M.T.; Kao, C.P.; Peng, W.H. Hepatoprotective effect of Coreopsis tinctoria flowers against carbon tetrachloride-induced liver damage in mice. BMC Complement Altern. Med. 2017, 17, 139. [Google Scholar] [CrossRef] [PubMed]
- Misra, H.P.; Fridovich, I. The role of superoxide anion in the autoxidation of epinephrine and a simple assay for superoxide dismutase. J. Chem. Biol. 1972, 247, 3170–3175. [Google Scholar]
- Flohe, L.; Gunzler, W.A. Assays of glutathione peroxidase. Methods Enzymol 1984, 105, 114–121. [Google Scholar] [PubMed]
- Carlberg, I.; Mannervik, B. Glutathione reductase. Methods Enzymol 1985, 113, 484–490. [Google Scholar] [PubMed]
- Kwon, Y.S.; Jang, K.H.; Jang, I.H. The effects of Korean red ginseng (ginseng radix rubra) on liver regeneration after partial hepatectomy in dogs. J. Vet. Sci. 2003, 4, 83–92. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, F.; Mikkelsen, B.B.; Nielsen, J.B.; Andersen, H.R.; Grandjean, P. Plasma malondialdehyde as biomarker for oxidative stress: Reference interval and effects of life-style factors. Clin. Chem. 1997, 43, 1209–1214. [Google Scholar] [PubMed]
- Reiter, R.J.; Tan, D.X.; Osuna, C.; Gitto, E. Actions of melatonin in the reduction of oxidative stress. A review. J. Biomed. Sci. 2000, 7, 444–458. [Google Scholar] [CrossRef] [PubMed]
- Jung, K.; Henke, W. Developmental changes of antioxidant enzymes in kidney and iver from rats. Free Radic. Biol. Med. 1996, 20, 613–617. [Google Scholar] [CrossRef]


| Organ | Lesions | Group | |||||
|---|---|---|---|---|---|---|---|
| N | CCl4 | Sily | Taxifolin (mg/kg) | ||||
| 100 | 150 | 200 | |||||
| Vacuolization1 | 0 *** | 1.4 ± 0.18 | 0.8 ± 0.13 ** | 0.8 ± 0.14 | 1 ± 0.15 * | 0.5 ± 0.17 *** | |
| Coagulative necrosis | 0 *** | 4 | 3.0 ± 0.31 * | 3.9 ± 0.10 | 3.5 ± 0.17 | 2.6 ± 0.43 * | |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yang, C.-L.; Lin, Y.-S.; Liu, K.-F.; Peng, W.-H.; Hsu, C.-M. Hepatoprotective Mechanisms of Taxifolin on Carbon Tetrachloride-Induced Acute Liver Injury in Mice. Nutrients 2019, 11, 2655. https://doi.org/10.3390/nu11112655
Yang C-L, Lin Y-S, Liu K-F, Peng W-H, Hsu C-M. Hepatoprotective Mechanisms of Taxifolin on Carbon Tetrachloride-Induced Acute Liver Injury in Mice. Nutrients. 2019; 11(11):2655. https://doi.org/10.3390/nu11112655
Chicago/Turabian StyleYang, Chao-Lin, Yu-Shih Lin, Keng-Fan Liu, Wen-Huang Peng, and Chih-Ming Hsu. 2019. "Hepatoprotective Mechanisms of Taxifolin on Carbon Tetrachloride-Induced Acute Liver Injury in Mice" Nutrients 11, no. 11: 2655. https://doi.org/10.3390/nu11112655





