A Sustainable Wholesome Foodstuff; Health Effects and Potential Dietotherapy Applications of Yacon
Abstract
1. Introduction
2. Yacon, and Nutritional and Chemical Composition
3. Health Effects (Usually Benefits) of Yacon and Its Bioactive Components
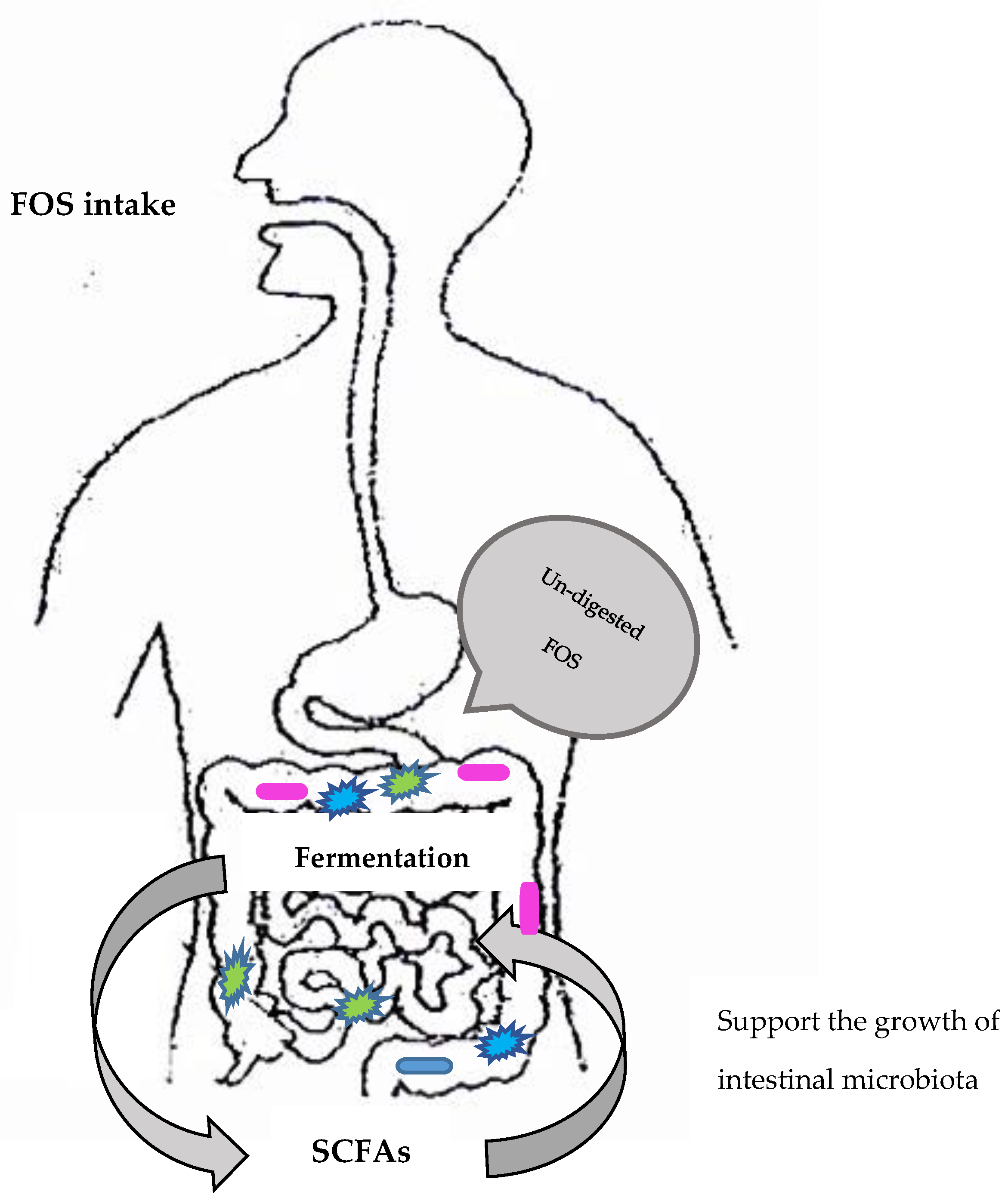
3.1. Health Effects of Fructooligosaccharides (FOS)
3.2. Health Effects of Inulin
3.3. Health Effects of Phenolic Compounds
3.4. Health Effects of Yacon Leaf and Tuber
3.5. Others
4. Potential Dietotherapy Applications: How to Use Yacon
4.1. Yacon Tea
4.2. Yacon Syrup
4.3. Yacon Powder
4.4. Yacon Capsules
4.5. Yacon Used in Infant Formula
4.6. Other Uses of Yacon
5. Yacon New Zealand: A Case Study
6. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Küster-Boluda, I.; Vidal-Capilla, I. Consumer attitudes in the election of functional foods. Span. J. Mark. ESIC 2017, 21, 65–79. [Google Scholar] [CrossRef]
- Bigliardi, B.; Galati, F. Innovation trends in the food industry: The case of functional foods. Trends Food Sci. Technol. 2013, 31, 118–129. [Google Scholar] [CrossRef]
- Doyon, M.; Labrecque, J. Functional foods: A conceptual definition. Br. Food J. 2008, 110, 1133–1149. [Google Scholar] [CrossRef]
- Delgado, G.T.; Tamashiro, W.M.; Marostica Junior, M.R.; Pastore, G.M. Yacon (Smallanthus sonchifolius): A functional food. Plant Foods Hum. Nutr. 2013, 68, 222–228. [Google Scholar] [CrossRef]
- De Almeida Paula, H.A.; Abranches, M.V.; de Luces Fortes Ferreira, C.L. Yacon (Smallanthus sonchifolius): A food with multiple functions. Crit. Rev. Food Sci. Nutr. 2015, 55, 32–40. [Google Scholar] [CrossRef]
- Valentová, K.; Ulrichová, J. Smallanthus sonchifolius and lepidium meyenii—Prospective andean crops for the prevention of chronic disease. Biomed. Pap. 2003, 147, 119–130. [Google Scholar] [CrossRef]
- Caetano, B.F.; de Moura, N.A.; Almeida, A.P.; Dias, M.C.; Sivieri, K.; Barbisan, L.F. Yacon (Smallanthus sonchifolius) as a food supplement: Health-promoting benefits of fructooligosaccharides. Nutrients 2016, 8. [Google Scholar] [CrossRef]
- Cao, Y.; Ma, Z.F.; Zhang, H.; Jin, Y.; Zhang, Y.; Hayford, F. Phytochemical properties and nutrigenomic implications of yacon as a potential source of prebiotic: Current evidence and future directions. Foods 2018, 7. [Google Scholar] [CrossRef]
- Balasubramanian, T.; Karthikeyan, M.; Muhammed Anees, K.P.; Kadeeja, C.P.; Jaseela, K. Antidiabetic and antioxidant potentials of amaranthus hybridus in streptozotocin-induced diabetic rats. J. Diet. Suppl. 2017, 14, 395–410. [Google Scholar] [CrossRef]
- Cocato, M.L.; Lobo, A.R.; Azevedo-Martins, A.K.; Filho, J.M.; de Sa, L.R.M.; Colli, C. Effects of a moderate iron overload and its interaction with yacon flour, and/or phytate, in the diet on liver antioxidant enzymes and hepatocyte apoptosis in rats. Food Chem. 2019, 285, 171–179. [Google Scholar] [CrossRef]
- Park, J.-S.; Yang, J.-S.; Hwang, B.-Y.; Yoo, B.-K.; Han, K. Hypoglycemic effect of yacon tuber extract and its constituent, chlorogenic acid, in streptozotocin-induced diabetic rats. Biomol. Ther. 2009, 17, 256–262. [Google Scholar] [CrossRef][Green Version]
- Dos Santos, K.C.; Bueno, B.G.; Pereira, L.F.; Francisqueti, F.V.; Braz, M.G.; Bincoleto, L.F.; da Silva, L.X.; Ferreira, A.L.A.; Nakamune, A.; Chen, C.O.; et al. Yacon (Smallanthus sonchifolius) leaf extract attenuates hyperglycemia and skeletal muscle oxidative stress and inflammation in diabetic rats. Evid. Based Complement. Altern. Med. 2017, 2017, 6418048. [Google Scholar] [CrossRef] [PubMed]
- De Ford, C.; Ulloa, J.L.; Catalan, C.A.N.; Grau, A.; Martino, V.S.; Muschietti, L.V.; Merfort, I. The sesquiterpene lactone polymatin B from Smallanthus sonchifolius induces different cell death mechanisms in three cancer cell lines. Phytochemistry 2015, 117, 332–339. [Google Scholar] [CrossRef] [PubMed]
- Khajehei, F.; Merkt, N.; Claupein, W.; Graeff-Hoenninger, S. Yacon (Smallanthus sonchifolius Poepp. & Endl.) as a novel source of health promoting compounds: Antioxidant activity, phytochemicals and sugar content in flesh, peel, and whole tubers of seven cultivars. Molecules 2018, 23. [Google Scholar] [CrossRef]
- Vaz-Tostes, M.; Viana, M.L.; Grancieri, M.; Luz, T.C.; Paula, H.; Pedrosa, R.G.; Costa, N.M. Yacon effects in immune response and nutritional status of iron and zinc in preschool children. Nutrition 2014, 30, 666–672. [Google Scholar] [CrossRef]
- Scheid, M.M.; Genaro, P.S.; Moreno, Y.M.; Pastore, G.M. Freeze-dried powdered yacon: Effects of FOS on serum glucose, lipids and intestinal transit in the elderly. Eur. J. Nutr. 2014, 53, 1457–1464. [Google Scholar] [CrossRef]
- Genta, S.; Cabrera, W.; Habib, N.; Pons, J.; Carillo, I.M.; Grau, A.; Sanchez, S. Yacon syrup: Beneficial effects on obesity and insulin resistance in humans. Clin. Nutr. 2009, 28, 182–187. [Google Scholar] [CrossRef]
- Nidhi, J.; Mini, S. FOS intake improves atherogenic profile and body weight in young obese adults of uban vadodara. World J. Pharm. Res. 2014, 4, 1094–1109. [Google Scholar]
- Machado, A.M.; da Silva, N.B.M.; Chaves, J.B.P.; Alfenas, R.C.G. Consumption of yacon flour improves body composition and intestinal function in overweight adults: A randomized, double-blind, placebo-controlled clinical trial. Clin. Nutr. ESPEN 2019, 29, 22–29. [Google Scholar] [CrossRef]
- Gomes da Silva, M.F.; Dionísio, A.P.; Ferreira Carioca, A.A.; Silveira Adriano, L.; Pinto, C.O.; Pinto de Abreu, F.A.; Wurlitzer, N.J.; Araújo, I.M.; dos Santos Garruti, D.; Ferreira Pontes, D. Yacon syrup: Food applications and impact on satiety in healthy volunteers. Food Res. Int. 2017, 100, 460–467. [Google Scholar] [CrossRef]
- Gusso, A.P.; Mattanna, P.; Richards, N. Yacon: Health benefits and technological applications. Rural Sci. 2015, 45, 912–919. [Google Scholar] [CrossRef][Green Version]
- Pedreschi, R.; Campos, D.; Noratto, G.; Chirinos, R.; Cisneros-Zevallos, L. Andean yacon root (Smallanthus sonchifolius poepp. endl) fructooligosaccharides as a potential novel source of prebiotics. J. Agric. Food Chem. 2003, 51, 5278–5284. [Google Scholar] [CrossRef] [PubMed]
- Ojansivu, I.; Ferreira, C.L.; Salminen, S. Yacon, a new source of prebiotic oligosaccharides with a history of safe use. Trends Food Sci. Technol. 2011, 22, 40–46. [Google Scholar] [CrossRef]
- Niness, K.R. Inulin and oligofructose: What are they? J. Nutr. 1999, 129, 1402–1406. [Google Scholar] [CrossRef] [PubMed]
- Qiu, Y.K.; Kang, T.G.; Dou, D.Q.; Liang, L.; Dong, F. Three novel compounds from the leaves of Smallanthus sonchifolius. J. Asian Nat. Prod. Res. 2008, 10, 1109–1115. [Google Scholar] [CrossRef] [PubMed]
- Valentova, K.; Cvak, L.; Muck, A.; Ulrichova, J.; Simanek, V. Antioxidant activity of extracts from the leaves of Smallanthus sonchifolius. Eur. J. Nutr. 2003, 42, 61–66. [Google Scholar] [CrossRef]
- Russo, D.; Valentao, P.; Andrade, P.B.; Fernandez, E.C.; Milella, L. Evaluation of antioxidant, antidiabetic and anticholinesterase activities of Smallanthus sonchifolius Landraces and correlation with their phytochemical profiles. Int. J. Mol. Sci. 2015, 16, 17696–17718. [Google Scholar] [CrossRef]
- Valentova, K.; Lebeda, A.; Dolezalova, I.; Jirovsky, D.; Simonovska, B.; Vovk, I.; Kosina, P.; Gasmanova, N.; Dziechciarkova, M.; Ulrichova, J. The biological and chemical variability of yacon. J. Agric. Food Chem. 2006, 54, 1347–1352. [Google Scholar] [CrossRef]
- Simonovska, B.; Vovk, I.; Andrenšek, S.; Valentová, K.; Ulrichová, J. Investigation of phenolic acids in yacon (Smallanthus sonchifolius) leaves and tubers. J. Chromatogr. A 2003, 1016, 89–98. [Google Scholar] [CrossRef]
- Douglas, J.A.; Follett, J.M.; Douglas, M.H.; Deo, B.; Scheffer, J.J.C.; Littler, R.A.; Manley-Harris, M. Effect of environment and time of planting on the production and quality of yacon (Smallanthus sonchifolius) storage roots. N. Z. J. Crop Hortic. Sci. 2007, 35, 107–116. [Google Scholar] [CrossRef]
- Santana, I.; Cardoso, M.H. Yacon tuberous root (Smallanthus sonchifolius): Cultivation potentialities, technological and nutritional aspects. Ciênc. Rural 2008, 38, 898–905. [Google Scholar] [CrossRef]
- Sabater-Molina, M.; Larque, E.; Torrella, F.; Zamora, S. Dietary fructooligosaccharides and potential benefits on health. J. Physiol. Biochem. 2009, 65, 315–328. [Google Scholar] [CrossRef] [PubMed]
- Whelan, K. Mechanisms and effectiveness of prebiotics in modifying the gastrointestinal microbiota for the management of digestive disorders. Proc. Nutr. Soc. 2013, 72, 288–298. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; O’Riordan, M.X. Regulation of bacterial pathogenesis by intestinal short-chain Fatty acids. Adv. Appl. Microbiol. 2013, 85, 93–118. [Google Scholar] [CrossRef] [PubMed]
- Wong, J.M.; de Souza, R.; Kendall, C.W.; Emam, A.; Jenkins, D.J. Colonic health: Fermentation and short chain fatty acids. J. Clin. Gastroenterol. 2006, 40, 235–243. [Google Scholar] [CrossRef]
- Tang, Y.; Chen, Y.; Jiang, H.; Nie, D. The role of short-chain fatty acids in orchestrating two types of programmed cell death in colon cancer. Autophagy 2011, 7, 235–237. [Google Scholar] [CrossRef]
- Respondek, F.; Gerard, P.; Bossis, M.; Boschat, L.; Bruneau, A.; Rabot, S.; Wagner, A.; Martin, J.C. Short-chain fructo-oligosaccharides modulate intestinal microbiota and metabolic parameters of humanized gnotobiotic diet induced obesity mice. PLoS ONE 2013, 8, e71026. [Google Scholar] [CrossRef]
- Raman, M.; Ambalam, P.; Kondepudi, K.K.; Pithva, S.; Kothari, C.; Patel, A.T.; Purama, R.K.; Dave, J.M.; Vyas, B.R. Potential of probiotics, prebiotics and synbiotics for management of colorectal cancer. Gut Microbes 2013, 4, 181–192. [Google Scholar] [CrossRef]
- Campos, D.; Betalleluz-Pallardel, I.; Chirinos, R.; Aguilar-Galvez, A.; Noratto, G.; Pedreschi, R. Prebiotic effects of yacon (Smallanthus sonchifolius Poepp. & Endl), a source of fructooligosaccharides and phenolic compounds with antioxidant activity. Food Chem. 2012, 135, 1592–1599. [Google Scholar] [CrossRef]
- Delgado, G.T.C.; Tamashiro, W.M.S.C.; Pastore, G.M. Immunomodulatory effects of fructans. Food Res. Int. 2010, 43, 1231–1236. [Google Scholar] [CrossRef]
- Delgado, G.T.; Thome, R.; Gabriel, D.L.; Tamashiro, W.M.; Pastore, G.M. Yacon (Smallanthus sonchifolius)-derived fructooligosaccharides improves the immune parameters in the mouse. Nutr. Res. 2012, 32, 884–892. [Google Scholar] [CrossRef] [PubMed]
- Velez, E.; Castillo, N.; Meson, O.; Grau, A.; Bibas Bonet, M.E.; Perdigon, G. Study of the effect exerted by fructo-oligosaccharides from yacon (Smallanthus sonchifolius) root flour in an intestinal infection model with Salmonella Typhimurium. Br. J. Nutr. 2013, 109, 1971–1979. [Google Scholar] [CrossRef] [PubMed]
- Hoseinifar, S.H.; Soleimani, N.; Ringo, E. Effects of dietary fructo-oligosaccharide supplementation on the growth performance, haemato-immunological parameters, gut microbiota and stress resistance of common carp (Cyprinus carpio) fry. Br. J. Nutr. 2014, 112, 1296–1302. [Google Scholar] [CrossRef] [PubMed]
- Slevin, M.M.; Allsopp, P.J.; Magee, P.J.; Bonham, M.P.; Naughton, V.R.; Strain, J.J.; Duffy, M.E.; Wallace, J.M.; Mc Sorley, E.M. Supplementation with calcium and short-chain fructo-oligosaccharides affects markers of bone turnover but not bone mineral density in postmenopausal women. J. Nutr. 2014, 144, 297–304. [Google Scholar] [CrossRef] [PubMed]
- Meksawan, K.; Chaotrakul, C.; Leeaphorn, N.; Gonlchanvit, S.; Eiam-Ong, S.; Kanjanabuch, T. Effects of fructo-oligosaccharide supplementation on constipation in elderly continuous ambulatory peritoneal dialysis patients. Perit. Dial. Int. 2016, 36, 60–66. [Google Scholar] [CrossRef] [PubMed]
- Carabin, I.G.; Flamm, W.G. Evaluation of safety of inulin and oligofructose as dietary fiber. Regul. Toxicol. Pharmacol. 1999, 30, 268–282. [Google Scholar] [CrossRef]
- Closa-Monasterolo, R.; Gispert-Llaurado, M.; Luque, V.; Ferre, N.; Rubio-Torrents, C.; Zaragoza-Jordana, M.; Escribano, J. Safety and efficacy of inulin and oligofructose supplementation in infant formula: Results from a randomized clinical trial. Clin. Nutr. 2013, 32, 918–927. [Google Scholar] [CrossRef]
- Food Standards Australia New Zealand. Addition of Inulin, FOS and GOS to Food; Food Standards Australia New Zealand: Canberra, Australia, 2007. [Google Scholar]
- Franck, A. Technological functionality of inulin and oligofructose. Br. J. Nutr. 2002, 87, 287–291. [Google Scholar] [CrossRef]
- Meyer, D.; Bayarri, S.; Tárrega, A.; Costell, E. Inulin as texture modifier in dairy products. Food Hydrocoll. 2011, 25, 1881–1890. [Google Scholar] [CrossRef]
- Mensink, M.A.; Frijlink, H.W.; van der Voort Maarschalk, K.; Hinrichs, W.L. Inulin, a flexible oligosaccharide I: Review of its physicochemical characteristics. Carbohydr. Polym. 2015, 130, 405–419. [Google Scholar] [CrossRef]
- Roberfroid, M.B. Inulin-type fructans: Functional food ingredients. J. Nutr. 2007, 137, 2493–2502. [Google Scholar] [CrossRef] [PubMed]
- Kalyani Nair, K.; Kharb, S.; Thompkinson, D.K. Inulin dietary fiber with functional and health attributes—A review. Food Rev. Int. 2010, 26, 189–203. [Google Scholar] [CrossRef]
- Liu, F.; Prabhakar, M.; Ju, J.; Long, H.; Zhou, H.W. Effect of inulin-type fructans on blood lipid profile and glucose level: A systematic review and meta-analysis of randomized controlled trials. Eur. J. Clin. Nutr. 2017, 71, 9–20. [Google Scholar] [CrossRef] [PubMed]
- Bacchetta, J.; Villard, F.; Vial, T.; Dubourg, L.; Bouvier, R.; Kassai, B.; Cochat, P. ‘Renal hypersensitivity’ to inulin and IgA nephropathy. Pediatr. Nephrol. 2008, 23, 1883–1885. [Google Scholar] [CrossRef] [PubMed]
- Lin, D.; Xiao, M.; Zhao, J.; Li, Z.; Xing, B.; Li, X.; Kong, M.; Li, L.; Zhang, Q.; Liu, Y.; et al. An overview of plant phenolic compounds and their importance in human nutrition and management of type 2 diabetes. Molecules 2016, 21. [Google Scholar] [CrossRef] [PubMed]
- Giada, M.L.R. Food phenolic compounds: Main classes, sources and their antioxidant power. In Oxidative Stress and Chronic Degenerative Diseases—A Role for Antioxidants; Morales-Gonzalez, J.A., Ed.; IntechOpen Ltd.: London, UK, 2013. [Google Scholar] [CrossRef]
- Minatel, I.O.; Borges, C.V.; Ferreira, M.I.; Gomez, H.A.G.; Chen, C.-Y.O.; Lima, G.P.P. Phenolic compounds: Functional properties, impact of processing and bioavailability. In Phenolic Compounds—Biological Activity; Soto-Hernandez, M., Ed.; IntechOpen Ltd.: London, UK, 2017. [Google Scholar] [CrossRef]
- Pandey, K.B.; Rizvi, S.I. Plant polyphenols as dietary antioxidants in human health and disease. Oxid. Med. Cell. Longev. 2009, 2, 270–278. [Google Scholar] [CrossRef]
- Vauzour, D.; Rodriguez-Mateos, A.; Corona, G.; Oruna-Concha, M.J.; Spencer, J.P. Polyphenols and human health: Prevention of disease and mechanisms of action. Nutrients 2010, 2, 1106–1131. [Google Scholar] [CrossRef]
- Ambriz-Perez, D.L.; Leyva-Lopez, N.; Gutierrez-Grijalva, E.P.; Heredia, J.B.; Yildiz, F. Phenolic compounds: Natural alternative in inflammation treatment. A review. Cogent Food Agric. 2016, 2. [Google Scholar] [CrossRef]
- Hussain, T.; Tan, B.; Yin, Y.; Blachier, F.; Tossou, M.C.; Rahu, N. Oxidative stress and inflammation: What polyphenols can do for us? Oxid. Med. Cell. Longev. 2016, 2016, 7432797. [Google Scholar] [CrossRef]
- GutiErrez-Grijalva, E.P.; Ambriz-Pere, D.L.; Leyva-Lopez, N.; Castillo-Lopez, R.I.; Heiedia, J.B. Review: Dietary phenolic compounds, health benefits and bioaccessibility. Arch. Latinoam. Nutr. 2016, 66, 87–100. [Google Scholar]
- Roleira, F.M.; Tavares-da-Silva, E.J.; Varela, C.L.; Costa, S.C.; Silva, T.; Garrido, J.; Borges, F. Plant derived and dietary phenolic antioxidants: Anticancer properties. Food Chem. 2015, 183, 235–258. [Google Scholar] [CrossRef] [PubMed]
- dos Santos, K.C.; Cury, S.S.; Ferraz, A.; Corrente, J.E.; Goncalves, B.M.; de Araujo Machado, L.H.; Carvalho, R.F.; de Melo Stevanato Nakamune, A.C.; Fabro, A.T.; Freire, P.P.; et al. Recovery of cardiac remodeling and dysmetabolism by pancreatic islet injury improvement in diabetic rats after yacon leaf extract treatment. Oxid. Med. Cell. Longev. 2018, 2018, 1821359. [Google Scholar] [CrossRef] [PubMed]
- Valentova, K.; Moncion, A.; de Waziers, I.; Ulrichova, J. The effect of Smallanthus sonchifolius leaf extracts on rat hepatic metabolism. Cell Biol. Toxicol. 2004, 20, 109–120. [Google Scholar] [CrossRef] [PubMed]
- Baroni, S.; Suzuki-Kemmelmeier, F.; Caparroz-Assef, S.M.; Cuman, R.K.N.; Bersani-Amado, C.A. Effect of crude extracts of leaves of Smallanthus sonchifolius (yacon) on glycemia in diabetic rats. Braz. J. Pharm. Sci. 2008, 44, 521–530. [Google Scholar] [CrossRef]
- Genta, S.B.; Cabrera, W.M.; Mercado, M.I.; Grau, A.; Catalan, C.A.; Sanchez, S.S. Hypoglycemic activity of leaf organic extracts from Smallanthus sonchifolius: Constituents of the most active fractions. Chem. Biol. Interact. 2010, 185, 143–152. [Google Scholar] [CrossRef] [PubMed]
- Raga, D.D.; Alimboyoguen, A.B.; del Fierro, R.S.; Ragasa, C.Y. Hypoglycaemic effects of tea extracts and ent-kaurenoic acid from Smallanthus sonchifolius. Nat. Prod. Res. 2010, 24, 1771–1782. [Google Scholar] [CrossRef]
- Baroni, S.; da Rocha, B.A.; Oliveira de Melo, J.; Comar, J.F.; Caparroz-Assef, S.M.; Bersani-Amado, C.A. Hydroethanolic extract of Smallanthus sonchifolius leaves improves hyperglycemia of streptozotocin induced neonatal diabetic rats. Asian Pac. J. Trop. Med. 2016, 9, 432–436. [Google Scholar] [CrossRef]
- Joung, H.; Kwon, D.Y.; Choi, J.G.; Shin, D.Y.; Chun, S.S.; Yu, Y.B.; Shin, D.W. Antibacterial and synergistic effects of Smallanthus sonchifolius leaf extracts against methicillin-resistant Staphylococcus aureus under light intensity. J. Nat. Med. 2010, 64, 212–215. [Google Scholar] [CrossRef]
- Siriwan, D.; Miyawaki, C.; Miyamoto, T.; Naruse, T.; Okazaki, K.; Tamura, H. Chemopreventive activity of sesquiterpene lactones (SLs) from yacon against TPA-induced Raji cells deformation. Pak. J. Biol. Sci. 2011, 14, 605–609. [Google Scholar] [CrossRef]
- Siriwan, D.; Naruse, T.; Tamura, H. Effect of epoxides and alpha-methylene-gamma-lactone skeleton of sesquiterpenes from yacon (Smallanthus sonchifolius) leaves on caspase-dependent apoptosis and NF-kappaB inhibition in human cercival cancer cells. Fitoterapia 2011, 82, 1093–1101. [Google Scholar] [CrossRef]
- Kitai, Y.; Hayashi, K.; Otsuka, M.; Nishiwaki, H.; Senoo, T.; Ishii, T.; Sakane, G.; Sugiura, M.; Tamura, H. New sesquiterpene lactone dimer, uvedafolin, extracted from eight yacon leaf varieties (Smallanthus sonchifolius): Cytotoxicity in HeLa, HL-60, and murine B16-F10 melanoma cell lines. J. Agric. Food Chem. 2015, 63, 10856–10861. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, R.B.; Chagas-Paula, D.A.; Secatto, A.; Gasparoto, T.H.; Faccioli, L.H.; Campanelli, A.P.; Da Costa, F.B. Topical anti-inflammatory activity of yacon leaf extracts. Rev. Bras. Farmacogn. 2013, 23, 497–505. [Google Scholar] [CrossRef]
- Yan, X.; Suzuki, M.; Ohnishi-Kameyama, M.; Sada, Y.; Nakanishi, T.; Nagata, T. Extraction and identification of antioxidants in the roots of yacon (Smallanthus sonchifolius). J. Agric. Food Chem. 1999, 47, 4711–4713. [Google Scholar] [CrossRef] [PubMed]
- Da Silva Almeida, A.P.; Avi, C.M.; Barbisan, L.F.; de Moura, N.A.; Caetano, B.F.R.; Romualdo, G.R.; Sivieri, K. Yacon (Smallanthus sonchifolius) and Lactobacillus acidophilus CRL 1014 reduce the early phases of colon carcinogenesis in male Wistar rats. Food Res. Int. 2015, 74, 48–54. [Google Scholar] [CrossRef]
- Habib, N.C.; Honore, S.M.; Genta, S.B.; Sanchez, S.S. Hypolipidemic effect of Smallanthus sonchifolius (yacon) roots on diabetic rats: Biochemical approach. Chem. Biol. Interact. 2011, 194, 31–39. [Google Scholar] [CrossRef]
- Habib, N.C.; Serra-Barcellona, C.; Honore, S.M.; Genta, S.B.; Sanchez, S.S. Yacon roots (Smallanthus sonchifolius) improve oxidative stress in diabetic rats. Pharm. Biol. 2015, 53, 1183–1193. [Google Scholar] [CrossRef]
- Oliveira, G.O.; Braga, C.P.; Fernandes, A.A. Improvement of biochemical parameters in type 1 diabetic rats after the roots aqueous extract of yacon [Smallanthus sonchifolius (Poepp.& Endl.)] treatment. Food Chem. Toxicol. 2013, 59, 256–260. [Google Scholar] [CrossRef]
- Lee, K.P.; Choi, N.H.; Kim, J.T.; Park, I.S. The effect of yacon (Samallanthus sonchifolius) ethanol extract on cell proliferation and migration of C6 glioma cells stimulated with fetal bovine serum. Nutr. Res. Pract. 2015, 9, 256–261. [Google Scholar] [CrossRef]
- Sousa, S.; Pinto, J.; Rodrigues, C.; Giao, M.; Pereira, C.; Tavaria, F.; Malcata, F.X.; Gomes, A.; Bertoldo Pacheco, M.T.; Pintado, M. Antioxidant properties of sterilized yacon (Smallanthus sonchifolius) tuber flour. Food Chem. 2015, 188, 504–509. [Google Scholar] [CrossRef]
- De Moura, N.A.; Caetano, B.F.; Sivieri, K.; Urbano, L.H.; Cabello, C.; Rodrigues, M.A.; Barbisan, L.F. Protective effects of yacon (Smallanthus sonchifolius) intake on experimental colon carcinogenesis. Food Chem. Toxicol. 2012, 50, 2902–2910. [Google Scholar] [CrossRef]
- Lobo, A.R.; Cocato, M.L.; Borelli, P.; Gaievski, E.H.S.; Crisma, A.R.; Nakajima, K.; Nakano, E.Y.; Colli, C. Iron bioavailability from ferric pyrophosphate in rats fed with fructan-containing yacon (Smallanthus sonchifolius) flour. Food Chem. 2011, 126, 885–891. [Google Scholar] [CrossRef]
- Lobo, A.R.; Colli, C.; Alvares, E.P.; Filisetti, T.M. Effects of fructans-containing yacon (Smallanthus sonchifolius Poepp and Endl.) flour on caecum mucosal morphometry, calcium and magnesium balance, and bone calcium retention in growing rats. Br. J. Nutr. 2007, 97, 776–785. [Google Scholar] [CrossRef] [PubMed]
- Honore, S.M.; Grande, M.V.; Gomez Rojas, J.; Sanchez, S.S. Smallanthus sonchifolius (yacon) flour improves visceral adiposity and metabolic parameters in high-fat-diet-fed rats. J. Obes. 2018, 2018, 5341384. [Google Scholar] [CrossRef] [PubMed]
- Roselino, M.N.; Pauly-Silveira, N.D.; Cavallini, D.C.; Celiberto, L.S.; Pinto, R.A.; Vendramini, R.C.; Rossi, E.A. A potential synbiotic product improves the lipid profile of diabetic rats. Lipids Health Dis. 2012, 11, 114. [Google Scholar] [CrossRef] [PubMed]
- Utami, N.W.; Sone, T.; Tanaka, M.; Nakatsu, C.H.; Saito, A.; Asano, K. Comparison of yacon (Smallanthus sonchifolius) tuber with commercialized fructo-oligosaccharides (FOS) in terms of physiology, fermentation products and intestinal microbial communities in rats. Biosci. Microbiota Food Health 2013, 32, 167–178. [Google Scholar] [CrossRef]
- An, L.; Yang, J.C.; Yin, H.; Xue, R.; Wang, Q.; Sun, Y.C.; Zhang, Y.Z.; Yang, M. Inulin-type oligosaccharides extracted from yacon produce antidepressant-like effects in behavioral models of depression. Phytother. Res. 2016, 30, 1937–1942. [Google Scholar] [CrossRef] [PubMed]
- Genta, S.B.; Cabrera, W.M.; Grau, A.; Sanchez, S.S. Subchronic 4-month oral toxicity study of dried Smallanthus sonchifolius (yacon) roots as a diet supplement in rats. Food Chem. Toxicol. 2005, 43, 1657–1665. [Google Scholar] [CrossRef]
- De Oliveira, R.B.; de Paula, D.A.; Rocha, B.A.; Franco, J.J.; Gobbo-Neto, L.; Uyemura, S.A.; dos Santos, W.F.; Da Costa, F.B. Renal toxicity caused by oral use of medicinal plants: The yacon example. J. Ethnopharmacol. 2011, 133, 434–441. [Google Scholar] [CrossRef]
- Yun, E.Y.; Kim, H.S.; Kim, Y.E.; Kang, M.K.; Ma, J.E.; Lee, G.D.; Cho, Y.J.; Kim, H.C.; Lee, J.D.; Hwang, Y.S.; et al. A case of anaphylaxis after the ingestion of yacon. Allergy Asthma Immunol. Res. 2010, 2, 149–152. [Google Scholar] [CrossRef]
- Jimenez, M.E.; Rossi, A.; Samman, N. Health properties of oca (Oxalis tuberosa) and yacon (Smallanthus sonchifolius). Food Funct. 2015, 6, 3266–3274. [Google Scholar] [CrossRef]
- Sugahara, S.; Ueda, Y.; Fukuhara, K.; Kamamuta, Y.; Matsuda, Y.; Murata, T.; Kuroda, Y.; Kabata, K.; Ono, M.; Igoshi, K.; et al. Antioxidant effects of herbal tea leaves from yacon (Smallanthus sonchifolius) on multiple free radical and reducing power assays, especially on different superoxide anion radical generation systems. J. Food Sci. 2015, 80, 2420–2429. [Google Scholar] [CrossRef] [PubMed]
- Satoh, H.; Audrey Nguyen, M.T.; Kudoh, A.; Watanabe, T. Yacon diet (Smallanthus sonchifolius, Asteraceae) improves hepatic insulin resistance via reducing Trb3 expression in Zucker fa/fa rats. Nutr. Diabetes 2013, 3, e70. [Google Scholar] [CrossRef] [PubMed]
- Bonet, M.E.B.; Meson, O.; de LeBlanc, A.D.; Dogi, C.A.; Chaves, S.; Kortsarz, A.; Grau, A.; Perdigon, G. Prebiotic effect of yacon (Smallanthus sonchifolius) on intestinal mucosa using a mouse model. Food Agric. Immunol. 2010, 21, 175–189. [Google Scholar] [CrossRef]
- Geyer, M.; Manrique, I.; Degen, L.; Beglinger, C. Effect of yacon (Smallanthus sonchifolius) on colonic transit time in healthy volunteers. Digestion 2008, 78, 30–33. [Google Scholar] [CrossRef]
- Gardner, E. Alternative sugars: Yacon syrup (nectar). Br. Dent. J. 2017, 223, 625. [Google Scholar] [CrossRef]
- Einerhand, S. Infant formula brought closer to breast milk thanks to prebiotic oligosaccharides. Agro Food Ind. Hi Tech 2016, 27, 2. [Google Scholar]
- Akkerman, R.; Faas, M.M.; de Vos, P. Non-digestible carbohydrates in infant formula as substitution for human milk oligosaccharide functions: Effects on microbiota and gut maturation. Crit. Rev. Food Sci. Nutr. 2019, 59, 1486–1497. [Google Scholar] [CrossRef]
- Tormena, M.M.L.; de Medeiros, L.T.; de Lima, P.C.; Possebon, G.; Fuchs, R.H.B.; Bona, E. Application of multi-block analysis and mixture design with process variable for development of chocolate cake containing yacon (Smallanthus sonchifolius) and maca (Lepidium meyenii). J. Sci. Food Agric. 2017, 97, 3559–3567. [Google Scholar] [CrossRef]
- Manzoni, M.S.J.; Rossi, E.A.; Pauly-Silveira, N.D.; Pinto, R.A.; Roselino, M.N.; Carlos, I.Z.; Quilles, M.B.; de Abreu Gloria, M.B.; Cavallini, D.C.U. Consumption effect of a synbiotic beverage made from soy and yacon extracts containing Bifidobacterium animalis ssp. lactis BB-12 on the intestinal polyamine concentrations in elderly individuals. Food Res. Int. 2017, 99, 495–500. [Google Scholar] [CrossRef]
- Barcellona, C.S.; Cabrera, W.M.; Honore, S.M.; Mercado, M.I.; Sanchez, S.S.; Genta, S.B. Safety assessment of aqueous extract from leaf Smallanthus sonchifolius and its main active lactone, enhydrin. J. Ethnopharmacol. 2012, 144, 362–370. [Google Scholar] [CrossRef]
- International Organisation for Standardisation. Determination of the Glycaemic Index and Recommendation for Food Classification; The International Organisation for Standardisation: Geneva, Switzerland, 2010. [Google Scholar]

| Author(s) | Year | Stated Yacon Source | Component(s) Under Study | Study Design | Physiological Effect(s) | |
|---|---|---|---|---|---|---|
| Method | In Vivo (Human Study or Animal Model) | |||||
| Cocato et al. [10] | 2019 | Yacon root flour | FOS* | Rats (Wistar) | Improved mineral absorption | |
| Machado et al. [19] | 2019 | Yacon root flour | FOS | Human (BMI 30 ± 2.4 kg m-2) | Improved body composition | |
| dos Santos et al. [65] | 2018 | Yacon leaf extract | Rats (Wistar) | Significant reduction in glycemia | ||
| Gomes da Silva et al. [20] | 2017 | Yacon syrup | Human | Improved satiety effects | ||
| dos Santos et al. [12] | 2017 | Yacon leaf extract | Rats (Wistar, diabetic) | Improved glycemic control | ||
| An et al. [89] | 2016 | Yacon root extract | Inulin | Mice (Kuming); Rats (Sprague Dawley) | Antidepressant-like effects | |
| Baroni et al. [70] | 2016 | Yacon leaf extract | Phenolics | Rats (Wistar, diabetic) | Improved insulin sensitivity | |
| da Silva et al. [77] | 2015 | Yacon root extract | FOS | Rats (Wistar) | Anti-cancer (against colon carcinogenesis on the early phases) | |
| de Ford et al. [13] | 2015 | Yacon leaf extract | In vitro | Anti-cancer | ||
| Habib et al. [79] | 2015 | Yacon root flour | Rats (Wistar, diabetic) | Antidiabetic effects | ||
| Jimenez et al. [93] | 2015 | Yacon root | FOS | Rats (Wistar) | Improved intestinal microflora | |
| Lee et al. [81] | 2015 | Yacon extract | In vitro | Anti-cancer (inhibited cell proliferation) | ||
| Russo et al. [27] | 2015 | Yacon leaf extract | Phenolics | In vitro | Antioxidant effects | |
| Sousa et al. [82] | 2015 | Yacon tuber flour | Phenolics | In vitro | Antioxidant effects | |
| Sugahara et al. [94] | 2015 | Yacon leaf extract | Phenolics | In vitro | Antioxidant effects | |
| Scheid et al. [16] | 2014 | Yacon root powder | FOS | Human (>60 years old) | Diabetes management, no side effects observed | |
| Vaz-Tostes et al. [15] | 2014 | Yacon root flour | FOS | Human (ages 2–5 years old) | Improved systemic immunity | |
| Oliverra et al. [75] | 2013 | Yacon leaf extract | In vitro | Anti-inflammatory | ||
| Oliverra et al. [80] | 2013 | Yacon root extract | Fructan | Rats (induced type 1 diabetic) | Antidiabetic effects | |
| Satoh et al. [95] | 2013 | Yacon tuber extract | Rats (Zucker fa/fa) | Improved insulin sensitivity | ||
| Utami et al. [88] | 2013 | Yacon tuber powder | FOS | Rats (Sprague Dawley) | Improved intestinal microflora | |
| Velez et al. [42] | 2013 | Yacon root flour | FOS | Mice (BALB/c) | Regulated intestinal microflora | |
| Campos et al. [39] | 2012 | Yacon root flour | FOS | Guinea pig | Improved intestinal microflora | |
| Delgado et al. [41] | 2012 | Yacon root flour | FOS | Mice (BALB/c) | Improved immunity efficiency | |
| de Moura et al. [83] | 2012 | Yacon root extract | FOS | Rats (Wistar) | Anti-cancer (against colon carcinogenesis on the early phases) | |
| Roselino et al. [87] | 2012 | Yacon root extract | Fructan | Rats (Wistar) | Improved lipid profiles | |
| Habib et al. [78] | 2011 | Yacon root flour | FOS | Rats (Wistar) | Antidiabetic and hypolipidemic effects | |
| Lobo et al. [84] | 2011 | Yacon root flour | Inulin | Rats (Wistar) | Improved mineral absorption | |
| Siriwan et al. [72] | 2011 | Yacon leaf extract | In vitro | Anti-cancer (inhibited cancer cell proliferation) | ||
| Siriwan et al. [73] | 2011 | Yacon leaf extract | In vitro | Anti-cancer (inhibited cancer cell proliferation) | ||
| Bonet et al. [96] | 2010 | Yacon root flour | Mice (BALB/c) | Improved intestinal microflora (growth of Bifidobacteria and Lactobacilli) | ||
| Genta et al. [68] | 2010 | Yacon leaf extract | Phenolics | Rats (Wistar) | Hypoglycemic effects | |
| Joung et al. [71] | 2010 | Yacon leaf extract | In vitro | Antimicrobial effects | ||
| Raga et al. [69] | 2010 | Yacon leaf extract | Phenolics | Mice (albino) | Hypoglycemic effects | |
| Yun et al. [92] | 2010 | Yacon root ingestion | Human (a 55-years-old woman) | Anaphylaxis1 | ||
| Genta et al. [17] | 2009 | Yacon syrup | Human (obese with mild dyslipidemia) | Improve insulin-resistance, and satiety effects | ||
| Park et al. [11] | 2009 | Yacon tuber extract | Rats (Sprague Dawley, diabetic) | Improved glycemic control | ||
| Baroni et al. [67] | 2008 | Yacon leaf extract | Phenolics | Rats (Wistar) | Hypoglycemic effects | |
| Geyer et al. [97] | 2008 | Yacon syrup | FOS | Human | Improved the colonic transit | |
| Lobo et al. [85] | 2007 | Yacon root flour | Rats (Wistar) | Improved mineral absorption | ||
| Genta et al. [90] | 2005 | Yacon root flour | FOS | Rats (Wistar) | No adverse side effects observed2 | |
| Valentova et al. [66] | 2004 | Yacon leaf extract | Rats (diabetic) | Anti-hyperglycemic effects | ||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yan, M.R.; Welch, R.; Rush, E.C.; Xiang, X.; Wang, X. A Sustainable Wholesome Foodstuff; Health Effects and Potential Dietotherapy Applications of Yacon. Nutrients 2019, 11, 2632. https://doi.org/10.3390/nu11112632
Yan MR, Welch R, Rush EC, Xiang X, Wang X. A Sustainable Wholesome Foodstuff; Health Effects and Potential Dietotherapy Applications of Yacon. Nutrients. 2019; 11(11):2632. https://doi.org/10.3390/nu11112632
Chicago/Turabian StyleYan, Mary R., Robert Welch, Elaine C. Rush, Xuesong Xiang, and Xin Wang. 2019. "A Sustainable Wholesome Foodstuff; Health Effects and Potential Dietotherapy Applications of Yacon" Nutrients 11, no. 11: 2632. https://doi.org/10.3390/nu11112632
APA StyleYan, M. R., Welch, R., Rush, E. C., Xiang, X., & Wang, X. (2019). A Sustainable Wholesome Foodstuff; Health Effects and Potential Dietotherapy Applications of Yacon. Nutrients, 11(11), 2632. https://doi.org/10.3390/nu11112632







