Actigraphic Sleep and Dietary Macronutrient Intake in Children Aged 6–9 Years Old: A Pilot Study
Abstract
:1. Introduction
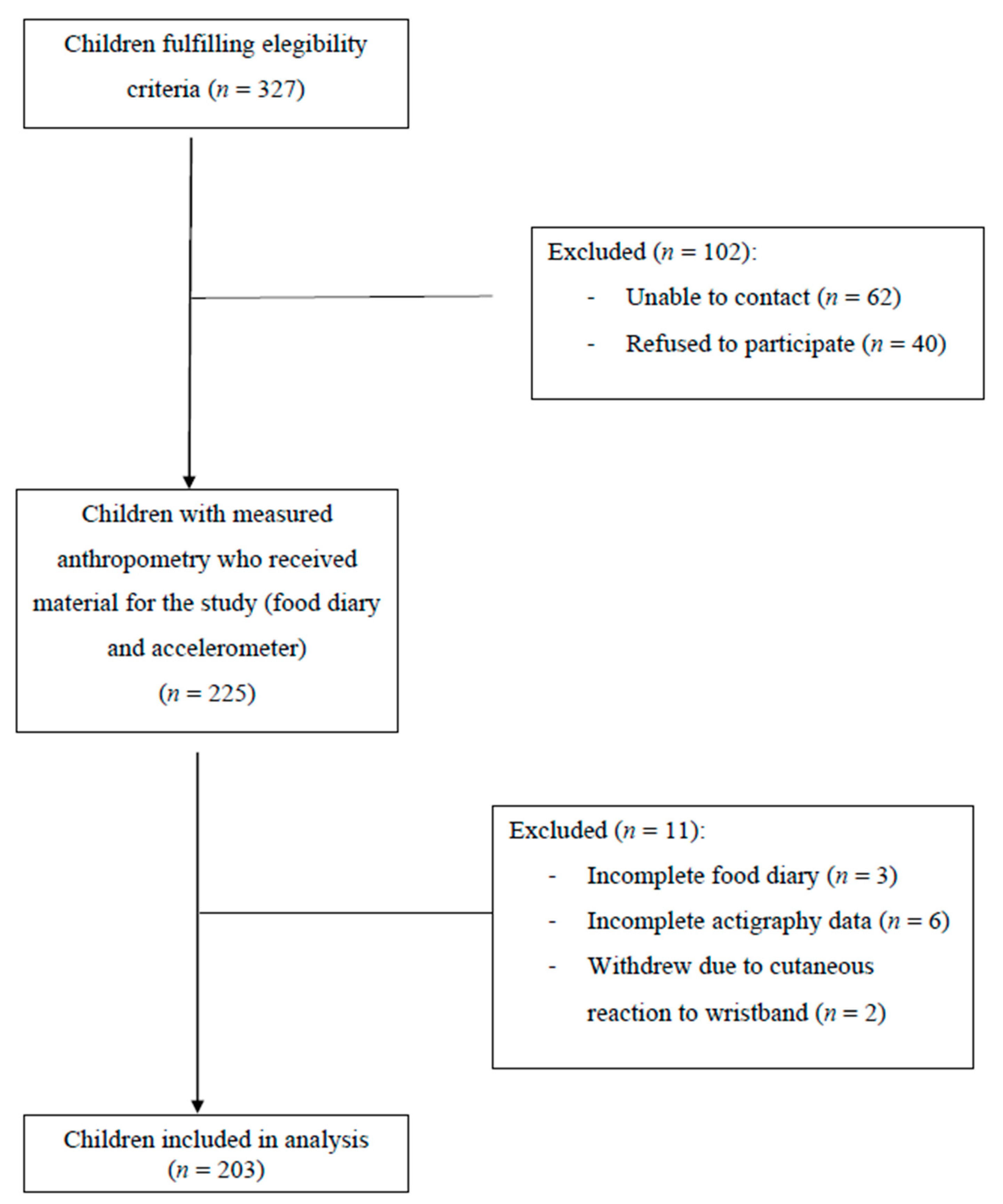
2. Materials and Methods
2.1. Study Design and Population
2.2. Sleep Assessment
2.3. Dietary Intake
2.4. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Kleiser, C.; Mensink, G.B.; Scheidt-Nave, C.; Kurth, B. HuSKY: A Healthy Nutrition Score Based on Food Intake of Children and Adolescents in Germany. Br. J. Nutr. 2009, 102, 610–618. [Google Scholar] [CrossRef] [PubMed]
- Magriplis, E.; Farajian, P.; Risvas, G.; Panagiotakos, D.; Zampelas, A. Newly Derived Children-Based Food Index. An Index that may Detect Childhood Overweight and Obesity. Int. J. Food Sci. Nutr. 2015, 66, 623–632. [Google Scholar] [CrossRef] [PubMed]
- Fatima, Y.; Doi, S.A.R.; Mamun, A.A. Sleep Quality and Obesity in Young Subjects: a Meta-Analysis. Obes. Rev. 2016, 17, 1154–1166. [Google Scholar] [CrossRef] [PubMed]
- Cappuccio, F.P.; Taggart, F.M.; Kandala, N.; Currie, A.; Peile, E.; Stranges, S.; Miller, M.A. Meta-Analysis of Short Sleep Duration and Obesity in Children and Adults. Sleep 2008, 31, 619–626. [Google Scholar] [CrossRef] [Green Version]
- Hogenkamp, P.S.; Nilsson, E.; Nilsson, V.C.; Chapman, C.D.; Vogel, H.; Lundberg, L.S.; Zarei, S.; Cedernaes, J.; Rångtell, F.H.; Broman, J. Acute Sleep Deprivation Increases Portion Size and Affects Food Choice in Young Men. Psychoneuroendocrinology 2013, 38, 1668–1674. [Google Scholar] [CrossRef]
- Tatone-Tokuda, F.; Dubois, L.; Ramsay, T.; Girard, M.; Touchette, E.; Petit, D.; Montplaisir, J.Y. Sex Differences in the Association between Sleep Duration, Diet and Body Mass Index: A Birth Cohort Study. J. Sleep Res. 2012, 21, 448–460. [Google Scholar] [CrossRef]
- Matricciani, L.; Olds, T.; Petkov, J. In Search of Lost Sleep: Secular Trends in the Sleep Time of School-Aged Children and Adolescents. Sleep Med. Rev. 2012, 16, 203–211. [Google Scholar] [CrossRef]
- Chen, X.; Beydoun, M.A.; Wang, Y. Is Sleep Duration Associated with Childhood Obesity? A Systematic Review and Meta-analysis. Obesity 2008, 16, 265–274. [Google Scholar] [CrossRef]
- Nielsen, L.; Danielsen, K.; Sørensen, T. Short Sleep Duration as a Possible Cause of Obesity: Critical Analysis of the Epidemiological Evidence. Obes. Rev. 2011, 12, 78–92. [Google Scholar] [CrossRef]
- Chaput, J.; Gray, C.E.; Poitras, V.J.; Carson, V.; Gruber, R.; Olds, T.; Weiss, S.K.; Connor Gorber, S.; Kho, M.E.; Sampson, M. Systematic Review of the Relationships between Sleep Duration and Health Indicators in School-Aged Children and Youth. Appl. Physiol. Nutr. Metab. 2016, 41, S266–S282. [Google Scholar] [CrossRef]
- Golley, R.K.; Maher, C.; Matricciani, L.; Olds, T. Sleep Duration or Bedtime? Exploring the Association between Sleep Timing Behaviour, Diet and BMI in Children and Adolescents. Int. J. Obes. 2013, 37, 546. [Google Scholar] [CrossRef]
- Blumfield, M.L.; Bei, B.; Zimberg, I.Z.; Cain, S.W. Dietary Disinhibition Mediates the Relationship between Poor Sleep Quality and Body Weight. Appetite 2018, 120, 602–608. [Google Scholar] [CrossRef] [PubMed]
- Kjeldsen, J.S.; Hjorth, M.F.; Andersen, R.; Michaelsen, K.F.; Tetens, I.; Astrup, A.; Chaput, J.; Sjödin, A. Short Sleep Duration and Large Variability in Sleep Duration are Independently Associated with Dietary Risk Factors for Obesity in Danish School Children. Int. J. Obes. 2014, 38, 32. [Google Scholar] [CrossRef] [PubMed]
- St-Onge, M.; Roberts, A.L.; Chen, J.; Kelleman, M.; O’Keeffe, M.; RoyChoudhury, A.; Jones, P.J. Short Sleep Duration Increases Energy Intakes but Does Not Change Energy Expenditure in Normal-Weight Individuals. Am. J. Clin. Nutr. 2011, 94, 410–416. [Google Scholar] [CrossRef] [PubMed]
- Heath, G.; Roach, G.D.; Dorrian, J.; Ferguson, S.A.; Darwent, D.; Sargent, C. The Effect of Sleep Restriction on Snacking Behaviour during a Week of Simulated Shiftwork. Accid. Anal. Prev. 2012, 45, 62–67. [Google Scholar] [CrossRef]
- Bel, S.; Michels, N.; De Vriendt, T.; Patterson, E.; Cuenca-García, M.; Diethelm, K.; Gutin, B.; Grammatikaki, E.; Manios, Y.; Leclercq, C. Association between Self-Reported Sleep Duration and Dietary Quality in European Adolescents. Br. J. Nutr. 2013, 110, 949–959. [Google Scholar] [CrossRef]
- Grandner, M.A.; Kripke, D.F.; Naidoo, N.; Langer, R.D. Relationships among Dietary Nutrients and Subjective Sleep, Objective Sleep, and Napping in Women. Sleep Med. 2010, 11, 180–184. [Google Scholar] [CrossRef]
- Magriplis, E.; Farajian, P.; Panagiotakos, D.B.; Risvas, G.; Zampelas, A. The Relationship between Behavioral Factors, Weight Status and a Dietary Pattern in Primary School Aged Children: The GRECO Study. Clin. Nutr. 2019, 38, 310–316. [Google Scholar] [CrossRef]
- Pérez-Farinós, N.; Villar-Villalba, C.; Sobaler, A.M.L.; Saavedra, M.Á.D.R.; Aparicio, A.; Sanz, S.S.; de Dios, T.R.; Castrodeza-Sanz, J.J.; Anta, R.M.O. The Relationship between Hours of Sleep, Screen Time and Frequency of Food and Drink Consumption in Spain in the 2011 and 2013 ALADINO: A Cross-Sectional Study. BMC Public Health 2017, 17, 33. [Google Scholar]
- Chaput, J. Sleep Patterns, Diet Quality and Energy Balance. Physiol. Behav. 2014, 134, 86–91. [Google Scholar] [CrossRef]
- Westerlund, L.; Ray, C.; Roos, E. Associations between Sleeping Habits and Food Consumption Patterns among 10–11-Year-Old Children in Finland. Br. J. Nutr. 2009, 102, 1531–1537. [Google Scholar] [CrossRef] [PubMed]
- Taylor, R.W.; Gray, A.R.; Heath, A.M.; Galland, B.C.; Lawrence, J.; Sayers, R.; Healey, D.; Tannock, G.W.; Meredith-Jones, K.A.; Hanna, M. Sleep, Nutrition, and Physical Activity Interventions to Prevent Obesity in Infancy: Follow-Up of the Prevention of Overweight in Infancy (POI) Randomized Controlled Trial at Ages 3.5 and 5 Y. Am. J. Clin. Nutr. 2018, 108, 228–236. [Google Scholar] [CrossRef] [PubMed]
- Paul, I.M.; Savage, J.S.; Anzman, S.L.; Beiler, J.S.; Marini, M.E.; Stokes, J.L.; Birch, L.L. Preventing Obesity during Infancy: A Pilot Study. Obesity 2011, 19, 353–361. [Google Scholar] [CrossRef] [PubMed]
- Sadeh, A. The Role and Validity of Actigraphy in Sleep Medicine: An Update. Sleep Med. Rev. 2011, 15, 259–267. [Google Scholar] [CrossRef]
- Bélanger, M.; Bernier, A.; Paquet, J.; Simard, V.; Carrier, J. Validating Actigraphy as a Measure of Sleep for Preschool Children. J. Clin. Sleep Med. 2013, 9, 701–706. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hyde, M.; O’DRISCOLL, D.M.; Binette, S.; Galang, C.; Tan, S.K.; Verginis, N.; Davey, M.J.; Horne, R.S. Validation of Actigraphy for Determining Sleep and Wake in Children with Sleep Disordered Breathing. J. Sleep Res. 2007, 16, 213–216. [Google Scholar] [CrossRef]
- Meltzer, L.J.; Montgomery-Downs, H.E.; Insana, S.P.; Walsh, C.M. Use of Actigraphy for Assessment in Pediatric Sleep Research. Sleep Med. Rev. 2012, 16, 463–475. [Google Scholar] [CrossRef]
- Morgenthaler, T.; Alessi, C.; Friedman, L.; Owens, J.; Kapur, V.; Boehlecke, B.; Brown, T.; Chesson Jr, A.; Coleman, J.; Lee-Chiong, T. Practice Parameters for the use of Actigraphy in the Assessment of Sleep and Sleep Disorders: An Update for 2007. Sleep 2007, 30, 519–529. [Google Scholar] [CrossRef]
- De Onis, M.; Lobstein, T. Defining Obesity Risk Status in the General Childhood Population: Which Cut-Offs should we use? Int. J. Pediatr. Obes. 2010, 5, 458–460. [Google Scholar] [CrossRef]
- De Onis, M.; Onyango, A.W.; Borghi, E.; Siyam, A.; Nishida, C.; Siekmann, J. Development of a WHO Growth Reference for School-Aged Children and Adolescents. Bull. World Health Organ. 2007, 85, 660–667. [Google Scholar] [CrossRef]
- Activinsights Ltd. GENEActiv Original; Activinsights Ltd.: Kimbolton, UK, 2019. [Google Scholar]
- Van Hees, V.T.; Sabia, S.; Jones, S.E.; Wood, A.R.; Anderson, K.N.; Kivimäki, M.; Frayling, T.M.; Pack, A.I.; Bucan, M.; Trenell, M. Estimating Sleep Parameters using an Accelerometer without Sleep Diary. Sci. Rep. 2018, 8, 12975. [Google Scholar] [CrossRef] [PubMed]
- Te Lindert, B.H.; Van Someren, E.J. Sleep Estimates using Microelectromechanical Systems (MEMS). Sleep 2013, 36, 781–789. [Google Scholar] [CrossRef] [PubMed]
- Phillips, L.R.; Parfitt, G.; Rowlands, A.V. Calibration of the GENEA Accelerometer for Assessment of Physical Activity Intensity in Children. J. Sci. Med. Sport 2013, 16, 124–128. [Google Scholar] [CrossRef] [PubMed]
- Duncan, M.J.; Wilson, S.; Tallis, J.; Eyre, E. Validation of the Phillips Et Al. GENEActiv Accelerometer Wrist Cut-Points in Children Aged 5–8 Years Old. Eur. J. Pediatr. 2016, 175, 2019–2021. [Google Scholar] [CrossRef] [PubMed]
- Ohayon, M.; Wickwire, E.M.; Hirshkowitz, M.; Albert, S.M.; Avidan, A.; Daly, F.J.; Dauvilliers, Y.; Ferri, R.; Fung, C.; Gozal, D. National Sleep Foundation’s Sleep Quality Recommendations: First Report. Sleep Health 2017, 3, 6–19. [Google Scholar] [CrossRef]
- Sociedad Valenciana de Pediatría. Cuántas Horas De Sueño Son Las Recomendadas En Función De La Edad De Los Niños; Sociedad Valenciana de Pediatría: Valencia, Spain, 2019. [Google Scholar]
- Hirshkowitz, M.; Whiton, K.; Albert, S.M.; Alessi, C.; Bruni, O.; DonCarlos, L.; Hazen, N.; Herman, J.; Katz, E.S.; Kheirandish-Gozal, L. National Sleep Foundation’s Sleep Time Duration Recommendations: Methodology and Results Summary. Sleep Health 2015, 1, 40–43. [Google Scholar] [CrossRef]
- Paruthi, S.; Brooks, L.J.; D’Ambrosio, C.; Hall, W.A.; Kotagal, S.; Lloyd, R.M.; Malow, B.A.; Maski, K.; Nichols, C.; Quan, S.F. Consensus Statement of the American Academy of Sleep Medicine on the Recommended Amount of Sleep for Healthy Children: Methodology and Discussion. J. Clin. Sleep Med. 2016, 12, 1549–1561. [Google Scholar] [CrossRef]
- Galland, B.C.; Short, M.A.; Terrill, P.; Rigney, G.; Haszard, J.J.; Coussens, S.; Foster-Owens, M.; Biggs, S.N. Establishing Normal Values for Pediatric Nighttime Sleep Measured by Actigraphy: A Systematic Review and Meta-Analysis. Sleep 2018, 41, zsy017. [Google Scholar] [CrossRef]
- Department of Nutrition (UCM) y Alce Ingeniería, S.L. DIAL Software for Assessing Diets and Food Calculations; Universidad Complutense de Madrid: Madrid, Spain, 2016. [Google Scholar]
- Institute of Medicine of the National Academies. Dietary Reference Intakes: Macronutrients; Institute of Medicine of The National Academies: Washington, DC, USA, 2019. [Google Scholar]
- St-Onge, M. The Role of Sleep Duration in the Regulation of Energy Balance: Effects on Energy Intakes and Expenditure. J. Clin. Sleep Med. 2013, 9, 73–80. [Google Scholar] [CrossRef]
- Nedeltcheva, A.V.; Kilkus, J.M.; Imperial, J.; Kasza, K.; Schoeller, D.A.; Penev, P.D. Sleep Curtailment is Accompanied by Increased Intake of Calories from Snacks. Am. J. Clin. Nutr. 2008, 89, 126–133. [Google Scholar] [CrossRef]
- Crispim, C.A.; Zimberg, I.Z.; dos Reis, B.G.; Diniz, R.M.; Tufik, S.; de Mello, M.T. Relationship between Food Intake and Sleep Pattern in Healthy Individuals. J. Clin. Sleep Med. 2011, 7, 659–664. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hart, C.N.; Carskadon, M.A.; Considine, R.V.; Fava, J.L.; Lawton, J.; Raynor, H.A.; Jelalian, E.; Owens, J.; Wing, R. Changes in Children’s Sleep Duration on Food Intake, Weight, and Leptin. Pediatrics 2013, 132, 1473–1480. [Google Scholar] [CrossRef] [PubMed]
- Dashti, H.S.; Zuurbier, L.A.; de Jonge, E.; Voortman, T.; Jacques, P.F.; Lamon-Fava, S.; Scheer, F.A.; Kiefte-De Jong, J.C.; Hofman, A.; Ordovás, J.M. Actigraphic Sleep Fragmentation, Efficiency and Duration Associate with Dietary Intake in the Rotterdam Study. J. Sleep Res. 2016, 25, 404–411. [Google Scholar] [CrossRef] [PubMed]
- Garaulet, M.; Ortega, F.; Ruiz, J.; Rey-Lopez, J.; Beghin, L.; Manios, Y.; Cuenca-Garcia, M.; Plada, M.; Diethelm, K.; Kafatos, A. Short Sleep Duration is Associated with Increased Obesity Markers in European Adolescents: Effect of Physical Activity and Dietary Habits. The HELENA Study. Int. J. Obes. 2011, 35, 1308. [Google Scholar] [CrossRef] [PubMed]
- Matricciani, L.; Paquet, C.; Galland, B.; Short, M.; Olds, T. Children’s Sleep and Health: A Meta-Review. Sleep Med. Rev. 2019, 46, 136–150. [Google Scholar] [CrossRef] [PubMed]
- Galland, B.; Meredith-Jones, K.; Terrill, P.; Taylor, R. Challenges and Emerging Technologies within the Field of Pediatric Actigraphy. Front. Psychiatry 2014, 5, 99. [Google Scholar] [CrossRef]
- Ronnlund, H.; Elovainio, M.; Virtanen, I.; Matomaki, J.; Lapinleimu, H. Poor Parental Sleep and the Reported Sleep Quality of their Children. Pediatrics 2016, 137, e20153425. [Google Scholar] [CrossRef]
- Kovács, E.; Siani, A.; Konstabel, K.; Hadjigeorgiou, C.; De Bourdeaudhuij, I.; Eiben, G.; Lissner, L.; Gwozdz, W.; Reisch, L.; Pala, V. Adherence to the Obesity-Related Lifestyle Intervention Targets in the IDEFICS Study. Int. J. Obes. 2014, 38, S144. [Google Scholar] [CrossRef]
- Fuligni, A.J.; Hardway, C. Daily Variation in Adolescents’ Sleep, Activities, and Psychological Well-being. J. Res. Adolesc. 2006, 16, 353–378. [Google Scholar] [CrossRef]
- St-Onge, M.; McReynolds, A.; Trivedi, Z.B.; Roberts, A.L.; Sy, M.; Hirsch, J. Sleep Restriction Leads to Increased Activation of Brain Regions Sensitive to Food Stimuli. Am. J. Clin. Nutr. 2012, 95, 818–824. [Google Scholar] [CrossRef]
- Rothausen, B.W.; Matthiessen, J.; Andersen, L.F.; Brockhoff, P.B.; Tetens, I. Dietary Patterns on Weekdays and Weekend Days in 4–14-Year-Old Danish Children. Br. J. Nutr. 2013, 109, 1704–1713. [Google Scholar] [CrossRef]
- Rothausen, B.W.; Matthiessen, J.; Hoppe, C.; Brockhoff, P.B.; Andersen, L.F.; Tetens, I. Differences in Danish Children’s Diet Quality on Weekdays V. Weekend Days. Public Health Nutr. 2012, 15, 1653–1660. [Google Scholar] [CrossRef]
- Haines, P.S.; Hama, M.Y.; Guilkey, D.K.; Popkin, B.M. Weekend Eating in the United States Is Linked with Greater Energy, Fat, and Alcohol Intake. Obes. Res. 2003, 11, 945–949. [Google Scholar] [CrossRef]
- Monteiro, L.S.; Hassan, B.K.; Estima, C.C.P.; Souza, A.d.M.; Verly Junior, E.; Sichieri, R.; Pereira, R.A. Food Consumption according to the Days of the Week–National Food Survey, 2008–2009. Rev. Saude Publica 2017, 51, 93. [Google Scholar] [CrossRef]
- Baron, K.G.; Reid, K.J.; Van Horn, L.; Zee, P.C. Contribution of Evening Macronutrient Intake to Total Caloric Intake and Body Mass Index. Appetite 2013, 60, 246–251. [Google Scholar] [CrossRef]
- Spiegel, K.; Leproult, R.; L’Hermite-Balériaux, M.; Copinschi, G.; Penev, P.D.; Van Cauter, E. Leptin Levels are Dependent on Sleep Duration: Relationships with Sympathovagal Balance, Carbohydrate Regulation, Cortisol, and Thyrotropin. J. Clin. Endocrinol. Metab. 2004, 89, 5762–5771. [Google Scholar] [CrossRef]
- Xiao, Q.; Garaulet, M.; Scheer, F.A. Meal Timing and Obesity: Interactions with Macronutrient Intake and Chronotype. Int. J. Obes. 2019, 43, 1701–1711. [Google Scholar] [CrossRef]
- Frank, S.; Gonzalez, K.; Lee-Ang, L.; Young, M.C.; Tamez, M.; Mattei, J. Diet and Sleep Physiology: Public Health and Clinical Implications. Front. Neurol. 2017, 8, 393. [Google Scholar] [CrossRef]
- Lundahl, A.; Nelson, T.D. Sleep and Food Intake: A Multisystem Review of Mechanisms in Children and Adults. J. Health Psychol. 2015, 20, 794–805. [Google Scholar] [CrossRef]
- Taheri, S.; Lin, L.; Austin, D.; Young, T.; Mignot, E. Short Sleep Duration is Associated with Reduced Leptin, Elevated Ghrelin, and Increased Body Mass Index. PLoS Med. 2004, 1, e62. [Google Scholar] [CrossRef]
- St-Onge, M.; Wolfe, S.; Sy, M.; Shechter, A.; Hirsch, J. Sleep Restriction Increases the Neuronal Response to Unhealthy Food in Normal-Weight Individuals. Int. J. Obes. 2014, 38, 411. [Google Scholar] [CrossRef]
- Goel, N.; Rao, H.; Durmer, J.S.; Dinges, D.F. Neurocognitive Consequences of Sleep Deprivation. Semin. Neurol. 2009, 29, 320–339. [Google Scholar] [CrossRef] [Green Version]
- Lemola, S.; Ledermann, T.; Friedman, E.M. Variability of Sleep Duration Is Related to Subjective Sleep Quality and Subjective Well-being: An Actigraphy Study. PLoS ONE 2013, 8, e71292. [Google Scholar] [CrossRef]
- Kant, A.K.; Graubard, B.I. Association of Self-Reported Sleep Duration with Eating Behaviors of American Adults: NHANES 2005–2010. Am. J. Clin. Nutr. 2014, 100, 938–947. [Google Scholar] [CrossRef]
| All (n = 203) | Male (n = 102) | Female (n = 101) | P Value | |
|---|---|---|---|---|
| General Characteristics | ||||
| Age (years) | 7.4 (1.1) | 7.2 (1.1) | 7.5 (1.1) | 0.025 |
| 6 years old n (%) | 59 (29.1) | 36 (34.6) | 23 (22.8) | 0.134 |
| 7 years old n (%) | 60 (29.6) | 31 (30.4) | 29 (28.7) | |
| 8 years old n (%) | 39 (19.2) | 16 (15.7) | 23 (22.8) | |
| 9 years old n (%) | 46 (22.7) | 19 (18.6) | 27 (26.7) | |
| BMI Z-score | 0.8 (1.3) | 0.9 (1.5) | 0.7 (1.1) | 0.159 |
| Major Obese n (%) | 6 (3.0) | 4 (3.9) | 2 (2.0) | 0.342 |
| Obese n (%) | 30 (14.8) | 19 (18.8) | 11 (10.8) | |
| Overweight n (%) | 51 (25.1) | 23 (22.8) | 28 (27.5) | |
| Normal n (%) | 115 (56.7) | 55 (54.5) | 60 (58.8) | |
| Wasted n (%) | 1 (0.5) | 0 (0) | 1 (1) | |
| Body fat (%) | 15.5 (6.5) | 13.7 (7 | 17.4 (5.3) | 0.001 |
| WHR (cm) | 0.88 (0.05) | 0.89 (0.46) | 0.87 (0.05) | 0.001 |
| Actigraphic Sleep Parameters | ||||
| Sleep duration week (h and min) | 7 h 44.1 min (54.6 min) | 7 h 38.9 min (57.6 min) | 7 h 49.1 min (51.3 min) | 0.187 |
| ≥9 h n (%) | 11 (5.4) | 5 (4.9) | 6 (5.9) | |
| <9 h n (%) | 192 (94.6) | 96 (95.0) | 95 (94.1) | 0.507 |
| <7 h, n (%) | 36 (17.7) | 25 (24.8) | 11 (10.8) | 0.078 |
| ≥7 to <8 h n (%) | 76 (37.4) | 34 (33.7) | 42 (41.2) | |
| ≥8 to <9 h n (%) | 80 (39.4) | 37 (36.6) | 43 (42.3) | |
| Sleep duration weekend (h and min) | 7 h 44.8 min (1 h 5.8 min) | 7 h 36.0 min (1 h 9.9 min) | 7 h 53.6 min (1 h 0.4 min) | 0.057 |
| ≥9 h n (%) | 19 (9.4) | 9 (8.9) | 10 (9.8) | |
| <9 h n (%) | 183 (90.1) | 93 (62.1) | 90 (88.2) | 0.358 |
| <7 h n (%) | 45 (22.2) | 29 (28.7) | 16 (15.7) | 0.065 |
| ≥7 to <8 h n (%) | 72 (35.5) | 37 (36.6) | 35 (34.3) | |
| ≥8 to <9 h n (%) | 67 (33) | 26 (25.7) | 41 (40.2) | |
| Sleep Efficiency weekday (%) | 80.5 (8.3) | 78.9 (8.4) | 82.1 (7.8) | 0.005 |
| ≥85% n (%) | 66 (32.5) | 30 (29.7) | 36 (35.3) | 0.242 |
| <85% n (%) | 137 (67.5) | 71 (70.3) | 66 (64.7) | |
| Sleep Efficiency weekend (%) | 79.4 (9.4) | 78.9 (9.8) | 79.9 (8.9) | 0.454 |
| ≥85% n (%) | 64 (31.5) | 28 (27.7) | 36 (35.3) | 0.291 |
| <85%, n (%) | 139 (68.5) | 73 (72.3) | 66 (64.7) | |
| Habitual sleep variability (min) | 55.8 (29.2) | 57.4 (32.1) | 54 (26.2) | 0.429 |
| Dietary intake | ||||
| Energy (kcal/day) | 2396 (712) | 2429 (839) | 2363 (560) | 0.512 |
| 6–8 years: Boys 1400–1700 kcal/day; Girls: 1300–1600 kcal/day * | ||||
| 9 years: Boys 1800–2300 kcal/day; Girls: 1700–2000 kcal/day * | ||||
| Protein (g/day) | 109.4 (60.7) | 113.2 (79.2) | 105.6 (33.1) | 0.377 |
| 6–8 years: 19 g/day * | ||||
| 9 years: 34 g/day * | ||||
| Fat (g/day) | 108.8 (48.9) | 110 (58.2) | 107.7 (35.1) | 0.746 |
| 6–8 years: 25 g/day * | ||||
| 9 years: 35 g/day * | ||||
| Carbohydrate (g/day) | 234.2 (41.5) | 236.9 (39.1) | 231.5 (43.8) | 0.357 |
| 6–9 years: 130 g/day * |
| Energy, kcal 2396 (712) | P Value | Fat, g 109.4 (60.7) | P Value | Protein, g 108.8 (48.9) | P Value | Carbohydrate, g 234.2 (41.5) | P Value | |
|---|---|---|---|---|---|---|---|---|
| Sleep Duration | ||||||||
| Weekday Continuous | 2397.7 (714.2) | 0.002 | 109.0 (48.0) | 0.002 | 109.5 (60.9) | 0.003 | 234.2 (41.6) | 0.329 |
| Weekday Categorical | ||||||||
| ≥9 h | 2136.6 (177.1) | 90.7 (10.9) | 90.4 (21.1) | 230.5 (34.4) | ||||
| <9 h | 2412.6 (730.5) | 0.005 | 110.1 (49.1) | 0.006 | 110.6 (62.2) | 0.007 | 234.4 (42.0) | 0.351 |
| <7 h | 2619.1 (1328.3) | 0.152 | 122.8 (94.1) | 0.215 | 127.2 (128.4) | 0.186 | 241.0 (37.3) | 0.754 |
| ≥7 to <8 h | 2371.8 (399.3) | 107.8 (23.3) | 107.3 (25.1) | 233.2 (39.5) | ||||
| ≥8 to <9 h | 2358.4 (577.1) | 106.5 (36.2) | 106.2 (36.0) | 232.6 (46.4) | ||||
| Weekday Continuous | 2397.7 (714.2) | 0.503 | 109.0 (48.0) | 0.719 | 109.5 (60.9) | 0.506 | 234.2 (41.6) | 0.436 |
| Weekend Categorical | ||||||||
| ≥9 h | 2395.1 (714.1) | 109.0 (48.0) | 109.5 (60.9) | 232.4 (41.0) | ||||
| <9 h | 2404.2 (746.7) | 0.655 | 109.7 (50.2) | 0.933 | 110.1 (63.7) | 0.613 | 109.7 (50.2) | 0.933 |
| <7 h | 2642.9 (1180.8) | 0.940 | 126.3 (82.8) | 0.809 | 123.9 (115.2) | 0.785 | 242.6 (38.9) | 0.766 |
| ≥7 to <8 h | 2257.8 (422.4) | 100.1 (23.9) | 98.1 (22.6) | 230.7 (43.5) | ||||
| ≥8 to <9 h | 2384.0 (563.8) | 108.1 (37.3) | 108.9 (35.34) | 232.7 (41.7) | ||||
| Sleep Efficiency | ||||||||
| Weekday | 2397.7 (714.2) | 0.971 | 109.0 (48.0) | 0.776 | 109.5 (60.9) | 0.660 | 234.2 (41.6) | 0.195 |
| ≥85% | 2383.7 (1030.6) | 0.842 | 109.7 (72.0) | 0.942 | 114.1 (97.0) | 0.817 | 225.8 (35.7) | 0.226 |
| <85% | 2404.4 (499.8) | 0.914 | 108.7 (30.9) | 0.638 | 107.3 (31.5) | 0.704 | 238.2 (43.7) | 0.936 |
| Weekend | 2397.7 (714.2) | 0.914 | 109.0 (48.0) | 0.629 | 109.5 (60.9) | 0.508 | 234.2 (41.6) | 0.015 |
| ≥85% | 2470.1 (1132.7) | 0.403 | 115.8 (78.5) | 0.517 | 120.1 (102.3) | 0.599 | 227.3 (38.0) | 0.052 |
| <85% | 2364.3 (396.8) | 0.023 | 106.1 (23.4) | 0.166 | 104.6 (23.9) | 0.061 | 237.4 (42.9) | 0.032 |
| Habitual Sleep Variabity | 2398.9 (715.8) | 0.106 | 109.1 (48.2) | 0.132 | 109.6 (61.0) | 0.024 | 234.2 (41.7) | 0.757 |
| Energy, kcal | Fat, g | Protein, g | Carbohydrate, g | |||||
|---|---|---|---|---|---|---|---|---|
| β (95% CI) * | P Value | β (95% CI) * | P Value | β (95% CI) * | P Value | β (95% CI) * | P Value | |
| Sleep Duration (h) | ||||||||
| Weekday Continuous | −170.17 (−276.59, −63.76) | 0.002 | −11.43 (−18.59, −4.20) | 0.002 | −13.84 (−22.90, −4.75) | 0.003 | −3.14 (−9.47, 3.10) | 0.329 |
| Weekday Categorical | ||||||||
| ≥9 h | 15.79 (−459.29, 487.87) | 0.941 | 4.75 (−24.16, 33.66) | 0.713 | −9.38 (−65.27, 46.51) | 0.719 | 1.21 (−90.57, 92.80) | 0.978 |
| <9 h | −172.76 (−293.62, −51.90) | 0.001 | −11.53 (−19.66, −3.39) | 0.006 | −14.28 (−24.59, 3.97) | 0.007 | −3.35 (−10.43, 3.72) | 0.351 |
| <7 h | −883.42 (−1621.78, −145.00) | 0.020 | −62.82 (−115.09, −10.54) | 0.020 | −80.11 (−152.24, −7.98) | 0.031 | 0.86 (−21.58, 23.31) | 0.938 |
| ≥7 to <8 h | 35.81 (−286.26, 357.88) | 0.825 | 0.209 (−18.58, 19.00) | 0.982 | 4.10 (−16.14, 24.35) | 0.687 | 1.26 (−30.57, 33.11) | 0.937 |
| ≥8 to <9 h | −144.70 (−639.38, 349.97) | 0.562 | −8.23 (−39.24, 22.76) | 0.598 | −2.38 (−33.33, 28.56) | 0.878 | −11.84 (−51.58, 27.89) | 0.555 |
| Weekend Continuous | −25.57 (−100.67, 49.52) | 0.503 | −0.92 (−5.98, 4.13) | 0.506 | −2.16 (−8.56, 4.23) | 0.719 | −1.73 (−6.10, 2.63) | 0.436 |
| Weekday Categorical | ||||||||
| ≥9 h | −98.18 (−327.09, 130.72) | 0.378 | −4.72 (−17.97, 8.53) | 0.463 | −7.36 (−25.48, 10.75) | 0.403 | −7.11 (−27.39, 13.15) | 0.469 |
| <9 h | −25.60 (−104.37, 53.17) | 0.522 | −0.671 (−5.97, 4.63) | 0.803 | −1.92 (−8.63, 4.78) | 0.573 | −2.57 (−7.01, 1.95) | 0.263 |
| <7 h | −68.91 (−262.39, 124.56) | 0.476 | −3.27 (−16.88, 10.33) | 0.630 | −3.70 (−22.65, 15.25) | 0.696 | −5.36 (−11.57, 0.83) | 0.088 |
| ≥7 to <8 h | −2.73 (−80.43, 74.97) | 0.944 | 0.48 (−3.91, 4.88) | 0.826 | −1.02 (−5.18, 3.13) | 0.625 | −0.786 (−8.76, 7.19) | 0.845 |
| ≥8 to <9 h | 56.03 (−88.53, 200.61) | 0.442 | 3.26 (−6.29, 12.82) | 0.497 | 0.12 (−8.97, 9.23) | 0.978 | 7.17 (−3.41, 17.75) | 0.181 |
| Sleep Efficiency (%) | ||||||||
| Weekday | −1.49 (−13.39–10.41) | 0.805 | 0.13 (−0.67–0.93) | 0.753 | 0.14 (−0.86–1.16) | 0.775 | −0.72 (−1.41–0.04) | 0.039 |
| ≥85% | 1088.11 (−9798.81–11,975.04) | 0.842 | −27.79 (−788.26, 732.67) | 0.942 | 119.36 (−904.90, 1143.63) | 0.817 | 228.19 (−144.37, 600.75) | 0.226 |
| <85% | 63.47 (−1098.00, 1225.55) | 0.914 | 17.09 (−54.61, 88.79) | 0.638 | −14.08 (−87.27, 59.11) | 0.704 | −4.51 (−105.00, 97.43) | 0.936 |
| Weekend | −57.32 (−1113.77, −999.14) | 0.915 | 22.43 (−48.57, 93.43) | 0.534 | 27.32 (−62.62, 117.27) | 0.550 | −82.57 (−143.00–22.14) | 0.008 |
| ≥85% | 4254.11 (−5837.14–14,345.37) | 0.403 | 228.55 (−475.77, 929.87) | 0.517 | 241.58 (−672.59, 1155.76) | 0.599 | 327.31 (−3.11, 657.73) | 0.052 |
| <85% | −811.38 (−1509.00, 113.55) | 0.023 | −29.02 (−70.21, 12.17) | 0.166 | −40.48 (−82.83, 1.87) | 0.061 | −82.70 (−158.27, −7.12) | 0.032 |
| Habitual Sleep Variabity (min) | 0.46 (−2.93, 3.84) | 0.791 | −0.01 (−0.24, 0.22) | 0.917 | 0.17 (−0.11, 0.46) | 0.234 | −0.02 (−0.22, 0.18) | 0.917 |
| Energy, kcal | Fat, g | Protein, g | Carbohydrate, g | |||||
|---|---|---|---|---|---|---|---|---|
| β (95% CI) * | P Value | β (95% CI) * | P Value | β (95% CI) * | P value | β (95% CI) * | P Value | |
| Sleep Duration (h) | ||||||||
| Weekday Continuous | −165,45 (−274.01, −56.88) | 0.003 | −11.14 (−18.44, −3.84) | 0.003 | −13.27 (−22.52, −4.02) | 0.005 | −3.13 (−9.57, 3.31) | 0.339 |
| Weekday Categorical | ||||||||
| ≥9 h | 214.5 (−414.25, 843.25) | 0.421 | 7.92 (−36.59, 52.44) | 0.666 | 3.93 (−79.18, 87.05) | 0.908 | 32.46 (−126.32, 191.25) | 0.339 |
| <9 h | −167.08 (−290.98, −43.18) | 0.008 | −11.11 (−19.44, −2.78) | 0.009 | −13.76 (−24.30, −3.22) | 0.011 | −3.33 (−10.56, 3.90) | 0.366 |
| Weekend Continuous | −23.66 (−100.16, 52.84) | 0.543 | −0.95 (−6.09, 4.20) | 0.717 | −1.88 (−8.39,46.61) | 0.567 | −1.37 (−5.82,3.07) | 0.543 |
| Weekday Categorical | ||||||||
| ≥9 h | −18.49 (−108.11, 71.13) | 0.665 | −1.91 (−7.36, 3.54) | 0.465 | −0.13 (−6.84, 6.58) | 0.967 | 0.86 (−6.75, 8.46) | 0.810 |
| <9 h | −23.92 (−147.39, 99.57) | 0.703 | 0.60 (−7.69, 8.90) | 0.886 | −2.37 (−12.88, 8.13) | 0.656 | −4.46 (−11.54, 2.61) | 0.215 |
| Sleep Efficiency (%) | ||||||||
| Weekday | −11.81 (−1177.82,1154.20) | 0.984 | 10.56 (−67.83, 88.95) | 0.791 | 23.59 (−75.42, 122.61) | 0.639 | −41.01 (−108.58, 26.56) | 0.233 |
| ≥85% | 2410.57 (−8937.42, 13,748.60) | 0.672 | 90.52 (−697.63, 878.68) | 0.819 | 267.11 (−797.83, 1332.05 | 0.618 | 145.94 (−223.97, 515.86) | 0.433 |
| <85% | 79.20 (−1090.53, 1248.94) | 0.894 | 18.34 (−52.79, 89.48) | 0.611 | −12.55 (−85.81, 60.72) | 0.735 | −4.47 (−106.80, 97.86) | 0.931 |
| Weekend | −433.78 (−1277.26, 409.69) | 0.311 | −10.66 (−62.17, 40.85) | 0.683 | −23.34 (−76.24, 29.57) | 0.385 | −52.57 (−126.09, 20.94) | 0.160 |
| ≥85% | 5294.27 (−4980.20, 15,568.74) | 0.307 | 301.40 (−411.49, 1014.30) | 0.401 | 335.35 (−593.30, 1264.01) | 0.473 | 337.05 (3.32, 670.77) | 0.048 |
| <85% | −847.43 (−1566.77, 128.09) | 0.021 | −32.17 (−74.56, 10.22) | 0.136 | −40.56 (−83.94, 2.82) | 0.067 | −83.96 (−161.76,-6.15) | 0.035 |
| Habitual Sleep Variabity (min) | 2.82 (−0.65, 6.30) | 0.111 | 0.18 (−0.05, 0.41) | 0.133 | 0.32 (0.03, 0.62) | 0.031 | −0.02 (−0.23, 0.18) | 0.814 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Coronado Ferrer, S.; Peraita-Costa, I.; Llopis-Morales, A.; Picó, Y.; Soriano, J.M.; Nieto, F.J.; Llopis-González, A.; Morales-Suarez-Varela, M. Actigraphic Sleep and Dietary Macronutrient Intake in Children Aged 6–9 Years Old: A Pilot Study. Nutrients 2019, 11, 2568. https://doi.org/10.3390/nu11112568
Coronado Ferrer S, Peraita-Costa I, Llopis-Morales A, Picó Y, Soriano JM, Nieto FJ, Llopis-González A, Morales-Suarez-Varela M. Actigraphic Sleep and Dietary Macronutrient Intake in Children Aged 6–9 Years Old: A Pilot Study. Nutrients. 2019; 11(11):2568. https://doi.org/10.3390/nu11112568
Chicago/Turabian StyleCoronado Ferrer, Silvia, Isabel Peraita-Costa, Agustín Llopis-Morales, Yolanda Picó, José Miguel Soriano, F. Javier Nieto, Agustín Llopis-González, and María Morales-Suarez-Varela. 2019. "Actigraphic Sleep and Dietary Macronutrient Intake in Children Aged 6–9 Years Old: A Pilot Study" Nutrients 11, no. 11: 2568. https://doi.org/10.3390/nu11112568
APA StyleCoronado Ferrer, S., Peraita-Costa, I., Llopis-Morales, A., Picó, Y., Soriano, J. M., Nieto, F. J., Llopis-González, A., & Morales-Suarez-Varela, M. (2019). Actigraphic Sleep and Dietary Macronutrient Intake in Children Aged 6–9 Years Old: A Pilot Study. Nutrients, 11(11), 2568. https://doi.org/10.3390/nu11112568







