Optimal Assessment and Quantification of Iodine Nutrition in Pregnancy and Lactation: Laboratory and Clinical Methods, Controversies and Future Directions
Abstract
1. Introduction
2. Methods for Measuring and Assessing Iodine Nutrition
2.1. Measurement of Urine Iodine Concentration (UIC) and Quality Assurance (QA)
2.2. Urine Iodine Concentration (UIC) and Total 24 h Urine Iodine Excretion (UIE) as Proxy Measures of Iodine Intake
2.3. Role of Neonatal TSH in Assessment and Monitoring of Mild to Moderate Iodine Deficiency
3. Definition and Justification of Mild, Moderate and Severe Iodine Deficiency Status
Classification of Iodine Deficiency
4. Mild to Moderate Gestational Iodine Deficiency and Impaired Neurodevelopment in the Offspring
4.1. Neurodevelopmental Impairment in Children Resulting from Gestational Mild to Moderate Maternal Iodine Deficiency
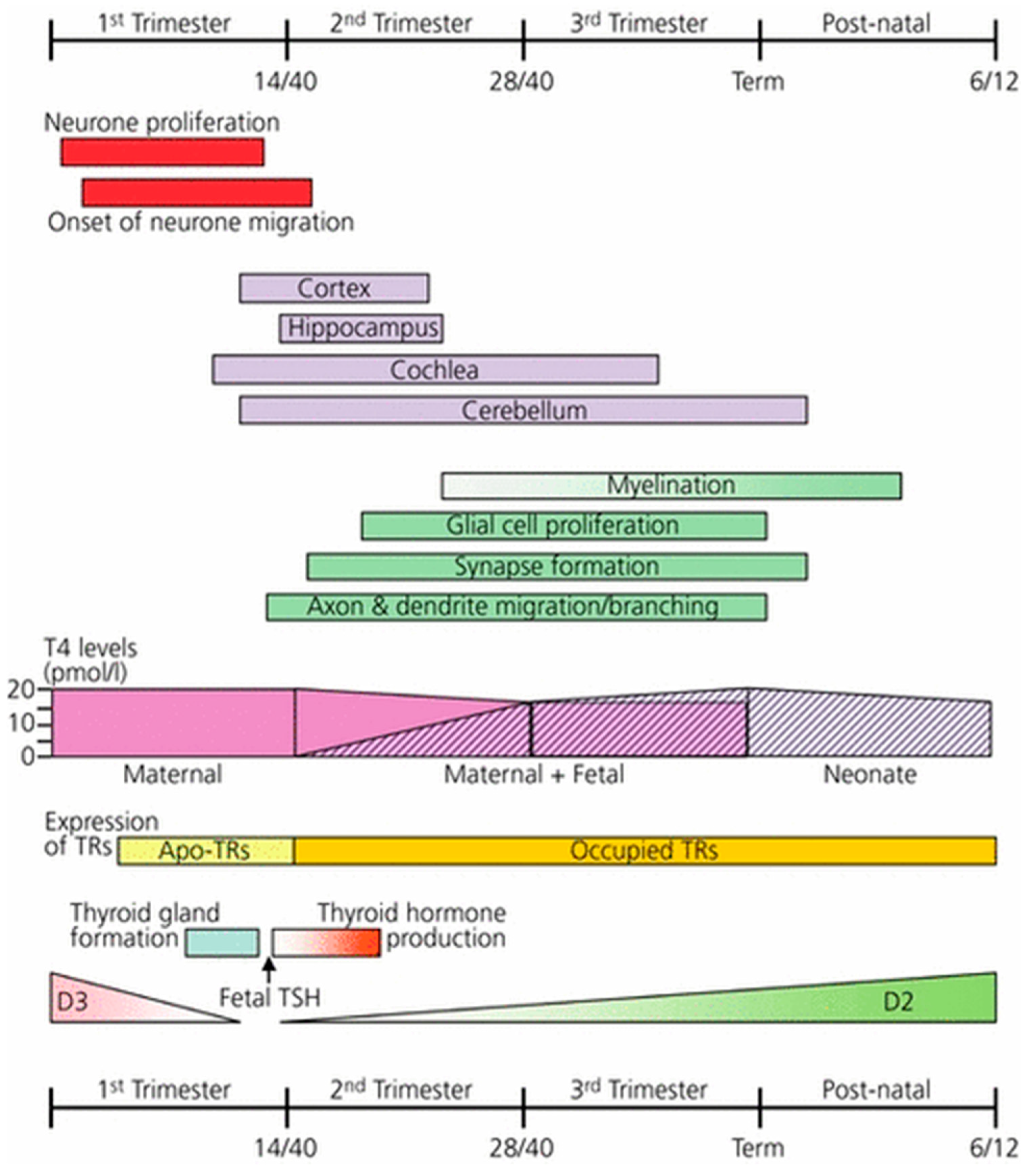
4.2. Mechanism of Neurological Damage in Mild to Moderate Gestational Iodine Deficiency
4.3. Controversies and Unanswered Questions
Isolated Hypothyroxinaemia
4.4. Attention Deficit Hyperactivity Disorders (ADHDs), Autism and Other Behavioural Disorders
5. Iodine Supplementation during Pregnancy and Lactation: Why, How Much and When?
5.1. Is Iodine Supplementation Safe during Pregnancy?
5.2. Iodine Supplementation during Lactation
6. Conclusions and Future Directions
6.1. Diagnosis and Classification of Iodine Deficiency
6.2. Paradigm Shift
6.3. Pathogenesis of Neurodevelopmental Disorders in the Foetus and Infant
6.4. Iodine Supplementation to Prevent Gestational Iodine Deficiency Disorders
6.5. Key Messages
- More precise definitions and measurements of gestational iodine deficiency, beyond a spot UIC, need to be developed.
- Agreement on the testing instruments and standardization of processes and procedures for IQ and psychomotor tests need to be reached in the medical and scientific community, so that valid comparisons can be made among studies of gestational iodine deficiency neurocognitive outcomes.
- Clinical studies need to be designed to address the pathogenesis of neurodevelopmental impairments in the foetus and infant.
- The safety and the efficacy of iodine supplementation in mild gestational iodine deficiency need to be established and excess intake of iodine during pregnancy is to be avoided.
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Hetzel, B. Iodine deficiency disorders (IDD) and their eradication. Lancet 1983, 2, 1126–1129. [Google Scholar] [CrossRef]
- Zimmermann, M.; Jooste, P.; Pandav, C. Iodine-deficiency disorders. Lancet 2008, 372, 1251–1262. [Google Scholar] [CrossRef]
- Eastman, C.J.; Li, M. Mild to moderate iodine deficiency. In IDD and Their Elimination; Pearce, E., Ed.; Springer Nature: Basel, Switzerland, 2017; pp. 39–74. [Google Scholar]
- Zimmermann, M.B. The impact of iodised salt or iodine supplements on iodine status during pregnancy, lactation and infancy. Public Health Nutr. 2007, 10, 1584–1595. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Eastman, C. The changing epidemiology of iodine deficiency. Nat. Rev. Endocrinol. 2012, 8, 434–440. [Google Scholar] [CrossRef] [PubMed]
- Hynes, K.L.; Otahal, P.; Hay, I.; Burgess, J.R. Mild iodine deficiency during pregnancy is associated with reduced educational outcomes in the offspring: 9-year follow-up of the gestational iodine cohort. J. Clin. Endocrinol. Metab. 2013, 98, 1954–1962. [Google Scholar] [CrossRef] [PubMed]
- Bath, S.C.; Steer, C.D.; Golding, J.; Emmett, P.; Rayman, M.P. Effect of inadequate iodine status in UK pregnant women on cognitive outcomes in their children: Results from the Avon Longitudinal Study of Parents and Children (ALSPAC). Lancet 2013, 382, 331–337. [Google Scholar] [CrossRef]
- Abel, M.H.; Caspersen, I.H.; Meltzer, H.M.; Haugen, M.; Brandlistuen, R.E.; Aase, H.; Alexander, J.; Torheim, L.E.; Brantsæter, A.-L. Suboptimal Maternal Iodine Intake Is Associated with Impaired Child Neurodevelopment at 3 Years of Age in the Norwegian Mother and Child Cohort Study. J. Nutr. 2017, 147, 1314–1324. [Google Scholar] [CrossRef] [PubMed]
- van Mil, N.H.; Tiemeier, H.; Bongers-Schokking, J.J.; Ghassabian, A.; Hofman, A.; Hooijkaas, H.; Steegers, E.A.P.; Visser, T.J.; Visser, W.; Ross, H.A.; et al. Low urinary iodine excretion during early pregnancy is associated with alterations in executive functioning in children. J. Nutr. 2012, 142, 2167–2174. [Google Scholar] [CrossRef]
- Murcia, M.; Rebagliato, M.; Iniguez, C.; Lopez-Espinosa, M.J.; Estarlich, M.; Plaza, B.; Barona-Vilar, C.; Espada, M.; Vioque, J.; Ballester, F. Effect of iodine supplementation during pregnancy on infant neurodevelopment at 1 year of age. Am. J. Epidemiol. 2011, 173, 804–812. [Google Scholar] [CrossRef]
- Rebagliato, M.; Murcia, M.; Alvarez-Pedrerol, M.; Espada, M.; Fernandez-Somoano, A.; Lertxundi, N.; Navarrete-Muñoz, E.-M.; Forns, J.; Aranbarri, A.; Llop, S.; et al. Iodine supplementation during pregnancy and infant neuropsychological development. INMA Mother and Child Cohort Study. Am. J. Epidemiol. 2013, 177, 944–953. [Google Scholar] [CrossRef]
- Williams, G.R. Neurodevelopmental and neurophysiological actions of thyroid hormone. J. Neuroendocrinol. 2008, 20, 784–794. [Google Scholar] [CrossRef]
- Corcino, C.M.; Berbara, T.; Saraiva, D.A.; Morais, N.; Schtscherbyna, A.; Gertrudes, L.N.; Teixeira, P.d.F.d.S.; Vaisman, M. Variation of iodine status during pregnancy and its associations with thyroid function in women from Rio de Janeiro, Brazil. Public Health Nutr. 2019, 22, 1232–1240. [Google Scholar] [CrossRef]
- Dunn, J.T.; Crutchfield, H.E.; Gutekunst, R.; Dunn, A.D. Two simple methods for measuring iodine in urine. Thyroid Off. J. Am. Thyroid Assoc. 1993, 3, 119–123. [Google Scholar] [CrossRef]
- Jooste, P.L.; Strydom, E. Methods for determination of iodine in urine and salt. Best Pract. Res. Clin. Endocrinol. Metab. 2010, 24, 77–88. [Google Scholar] [CrossRef]
- Pino, S.; Fang, S.L.; Braverman, L.E. Ammonium persulfate: A safe alternative oxidizing reagent for measuring urinary iodine. Clin. Chem. 1996, 42, 239–243. [Google Scholar]
- World Health Organisation, United Nations Children’s Fund. International Council for the Control. of Iodine Deficiency Disorders. Assessment of Iodine Deficiency Disorders and Monitoring Their Elimination: A Guide for Programme Managers, 3rd ed.; WHO: Geneva, Switzerland, 2007. [Google Scholar]
- Ohashi, T.; Yamaki, M.; Pandav, C.S.; Karmarkar, M.G.; Irie, M. Simple microplate method for determination of urinary iodine. Clin. Chem. 2000, 46, 529–536. [Google Scholar]
- Macours, P.; Aubry, J.C.; Hauquier, B.; Boeynaems, J.M.; Goldman, S.; Moreno-Reyes, R. Determination of urinary iodine by inductively coupled plasma mass spectrometry. J. Trace Elem. Med. Biol. 2008, 22, 162–165. [Google Scholar] [CrossRef]
- Shelor, C.P.; Dasgupta, P.K. Review of analytical methods for the quantification of iodine in complex matrices. Anal. Chim. Acta 2011, 702, 16–36. [Google Scholar] [CrossRef]
- Centers for Disease Control and Prevention. Ensuring the Quality of Urinary Iodine Procedures (EQUIP). Available online: https://www.cdc.gov./labstandards/equip.html (accessed on 16 July 2019).
- Institute of Medicine, Academy of Sciences. Dietary Reference Intakes for Vitamin A, Vitamin K, Arsenic, Boron, Chromium, Copper, Iodine, Iron, Manganese, Molybdenum, Nickel, Silicon, Vanadium, and Zinc; National Academy Press: Washington, DC, USA, 2001. [Google Scholar]
- Andersson, M.; de Benoist, B.; Delange, F.; Zupan, J. Prevention and control of iodine deficiency in pregnant and lactating women and in children less than 2-years-old: Conclusions and recommendations of the Technical Consultation. Public Health Nutr. 2007, 10, 1606–1611. [Google Scholar]
- Konig, F.; Andersson, M.; Hotz, K.; Aeberli, I.; Zimmermann, M.B. Ten repeat collections for urinary iodine from spot samples or 24-hour samples are needed to reliably estimate individual iodine status in women. J. Nutr. 2011, 141, 2049–2054. [Google Scholar] [CrossRef]
- Perrine, C.G.; Cogswell, M.E.; Swanson, C.A.; Sullivan, K.M.; Chen, T.C.; Carriquiry, A.L.; Dodd, K.W.; Caldwelle, K.L.; Wang, C.Y. Comparison of population iodine estimates from 24-hour urine and timed-spot urine samples. Thyroid Off. J. Am. Thyroid Assoc. 2014, 24, 748–757. [Google Scholar] [CrossRef]
- Knudsen, N.; Christiansen, E.; Brandt-Christensen, M.; Nygaard, B.; Perrild, H. Age- and sex-adjusted iodine/creatinine ratio. A new standard in epidemiological surveys? Evaluation of three different estimates of iodine excretion based on casual urine samples and comparison to 24 h values. Eur. J. Clin. Nutr. 2000, 54, 361–363. [Google Scholar] [CrossRef]
- Andersen, S.; Pedersen, K.M.; Pedersen, I.B.; Laurberg, P. Variations in urinary iodine excretion and thyroid function. A 1-year study in healthy men. Eur. J. Endocrinol. 2001, 144, 461–465. [Google Scholar] [CrossRef]
- Chen, W.; Wu, Y.; Lin, L.; Tan, L.; Shen, J.; Pearce, E.N.; Guo, X.; Wang, W.; Bian, J.; Jiang, W.; et al. 24-Hour Urine Samples Are More Reproducible Than Spot Urine Samples for Evaluation of Iodine Status in School-Age Children. J. Nutr. 2016, 146, 142–146. [Google Scholar] [CrossRef]
- Li, C.; Peng, S.; Zhang, X.; Xie, X.; Wang, D.; Mao, J.; Teng, X.; Shan, Z.; Teng, W. The Urine Iodine to Creatinine as an Optimal Index of Iodine During Pregnancy in an Iodine Adequate Area in China. J. Clin. Endocrinol. Metab. 2016, 101, 1290–1298. [Google Scholar] [CrossRef]
- Dussault, J.H. Relation of basic and clinical research on foetal and neonatal thyroid pathology to neonatal thyroid screening. Acta Paediatr. 1999, 88, 15–17. [Google Scholar] [CrossRef]
- Delange, F. Neonatal thyroid screening as a monitoring tool for the control of iodine deficiency. Acta Paediatr. 1999, 88, 21–24. [Google Scholar] [CrossRef]
- Li, M.; Eastman, C.J. Neonatal TSH screening: Is it a sensitive and reliable tool for monitoring iodine status in populations? Best Pract. Res. Clin. Endocrinol. Metab. 2010, 24, 63–75. [Google Scholar] [CrossRef]
- Vandevijvere, S.; Coucke, W.; Vanderpas, J.; Trumpff, C.; Fauvart, M.; Meulemans, A.; Marie, S.; Vincent, M.-F.; Schoos, R.; Boemer, F.; et al. Neonatal thyroid-stimulating hormone concentrations in Belgium: A useful indicator for detecting mild iodine deficiency? PLoS ONE 2012, 7, e47770. [Google Scholar] [CrossRef]
- Evans, C.; Barry Nix, A.; Hillier, S.; Moat, S.J. Neonatal blood TSH concentration in Wales (UK): An indicator of iodine sufficiency. Clin. Endocrinol. 2014, 81, 606–609. [Google Scholar] [CrossRef]
- Caylan, N.; Tezel, B.; Ozbas, S.; Sahin, N.; Aydin, S.; Acican, D.; Keskinkılıç, B. Neonatal Thyroid-Stimulating Hormone Screening as a Monitoring Tool for Iodine Deficiency in Turkey. J. Clin. Res. Pediatric Endocrinol. 2016, 8, 187–191. [Google Scholar] [CrossRef]
- Mehran, L.; Yarahmadi, S.; Khalili, D.; Nazeri, P.; Delshad, H.; Abdollahi, Z.; Azhang, N.; Azizi, F. The Impact of Iodine Status on the Recall Rate of the Screening Program for Congenital Hypothyroidism: Findings from Two National Studies in Iran. Nutrients 2017, 9, 1194. [Google Scholar] [CrossRef]
- Mullan, K.; Patterson, C.; Doolan, K.; Cundick, J.; Hamill, L.; McKeeman, G.; McMullan, P.; Smyth, P.; Young, L.; Woodside, J.V. Neonatal TSH levels in Northern Ireland from 2003 to 2014 as a measure of population iodine status. Clin. Endocrinol. 2018, 89, 849–855. [Google Scholar] [CrossRef]
- Hutchings, N.; Tovmasyan, I.; Hovsepyan, M.; Qefoyan, M.; Baghdasaryan, S.; Bilezikian, J.P. Neonatal thyrotropin (TSH) screening as a tool for monitoring iodine nutrition in Armenia. Eur. J. Clin. Nutr. 2018, 73, 905–909. [Google Scholar] [CrossRef]
- Trumpff, C.; Vandevijvere, S.; Moreno-Reyes, R.; Vanderpas, J.; Tafforeau, J.; Van Oyen, H.; Schepper, J.D. Neonatal thyroid-stimulating hormone level is influenced by neonatal, maternal, and pregnancy factors. Nutr. Res. (New York, NY, USA) 2015, 35, 975–981. [Google Scholar] [CrossRef]
- Zhang, Y.; Du, C.; Wang, W.; Chen, W.; Shao, P.; Wang, C.; Leng, J.; Shen, J.; Tan, L.; Zhang, W. Effect of maternal and neonatal factors on neonatal thyroid stimulating hormone: Results from a population-based prospective cohort study in China. J. Trace Elem. Med. Biol. 2018, 49, 151–156. [Google Scholar] [CrossRef]
- Ehrenkranz, J.; Fualal, J.; Ndizihiwe, A.; Clarke, I.; Alder, S. Neonatal age and point of careTSH testing in the monitoring of iodine deficiency disorders: Findings from western Uganda. Thyroid Off. J. Am. Thyroid Assoc. 2011, 21, 183–188. [Google Scholar] [CrossRef]
- Laurberg, P.; Cerqueira, C.; Ovesen, L.; Rasmussen, L.B.; Perrild, H.; Andersen, S.; Pedersen, L.B.; Carlé, A. Iodine intake as a determinant of thyroid disorders in populations. Best Pract. Res. Clin. Endocrinol. Metab. 2010, 24, 13–27. [Google Scholar] [CrossRef]
- Glinoer, D. The regulation of thyroid function in pregnancy: Pathways of endocrine adaptation from physiology to pathology. Endocr. Rev. 1997, 18, 404–433. [Google Scholar] [CrossRef]
- Boyages, S.C.; Collins, J.K.; Maberly, G.F.; Jupp, J.J.; Morris, J.; Eastman, C.J. Iodine deficiency impairs intellectual and neuromotor development in apparently-normal persons. A study of rural inhabitants of north-central China. Med. J. Aust. 1989, 150, 676–682. [Google Scholar]
- Boyages, S.C.; Halpern, J.P.; Maberly, G.F.; Eastman, C.J.; Morris, J.; Collins, J.; Jupp, J.J.; Jin, C.; Wang, Z.; You, C. A comparative study of neurological and myxedematous endemic cretinism in western China. J. Clin. Endocrinol. Metab. 1988, 67, 1262–1271. [Google Scholar] [CrossRef]
- Qian, M.; Wang, D.; Watkins, W.; Gebski, V.; Yan, Y.; Li, M.; Chen, Z. The effects of iodine on intelligence in children: A meta-analysis of studies conducted in China. Asia Pac. J. Clin. Nutr. 2005, 14, 32–42. [Google Scholar]
- Hynes, K.L.; Otahal, P.; Burgess, J.R.; Oddy, W.H.; Hay, I. Reduced Educational Outcomes Persist into Adolescence Following Mild Iodine Deficiency in Utero, Despite Adequacy in Childhood: 15-Year Follow-Up of the Gestational Iodine Cohort Investigating Auditory Processing Speed and Working Memory. Nutrients 2017, 9, 1354. [Google Scholar] [CrossRef]
- Levie, D.; Korevaar, T.I.M.; Bath, S.C.; Murcia, M.; Dineva, M.; Llop, S.; Espada, M.; van Herwaarden, A.E.; de Rijke, Y.B.; Ibarluzea, J.M.; et al. Association of maternal iodine status with child IQ: A meta-analysis of individual-participant data. J. Clin. Endocrinol. Metab. 2019. [Google Scholar] [CrossRef]
- Velasco, I.; Bath, S.C.; Rayman, M.P. Iodine as Essential Nutrient during the First 1000 Days of Life. Nutrients 2018, 10, 290. [Google Scholar] [CrossRef]
- Melse-Boonstra, A.; Jaiswal, N. Iodine deficiency in pregnancy, infancy and childhood and its consequences for brain development. Best Pract. Res. Clin. Endocrinol. Metab. 2010, 24, 29–38. [Google Scholar] [CrossRef]
- Boyages, S.C.; Halpern, J.P. Endemic cretinism: Toward a unifying hypothesis. Thyroid Off. J. Am. Thyroid Assoc. 1993, 3, 59–69. [Google Scholar] [CrossRef]
- Halpern, J.P.; Boyages, S.C.; Maberly, G.F.; Collins, J.K.; Eastman, C.J.; Morris, J.G. The neurology of endemic cretinism. A study of two endemias. Brain A J. Neurol. 1991, 114 Pt 2, 825–841. [Google Scholar] [CrossRef]
- Cao, X.Y.; Jiang, X.M.; Dou, Z.H.; Rakeman, M.A.; Zhang, M.L.; O’Donnell, K.; Ma, T.; Amette, K.; DeLong, N.; DeLong, G.R. Timing of vulnerability of the brain to iodine deficiency in endemic cretinism. N. Engl. J. Med. 1994, 331, 1739–1744. [Google Scholar] [CrossRef]
- Levie, D.; Korevaar, T.I.M.; Bath, S.C.; Dalmau-Bueno, A.; Murcia, M.; Espada, M.; Dineva, M.; Ibarluzea, J.M.; Sunyer, J.; Tiemeier, H.; et al. Thyroid Function in Early Pregnancy, Child IQ and Autistic Traits: A Meta-Analysis of Individual Participant Data. J. Clin. Endocrinol. Metab. 2018, 103, 2967–2979. [Google Scholar] [CrossRef]
- Rohner, F.; Zimmermann, M.; Jooste, P.; Pandav, C.; Caldwell, K.; Raghavan, R.; Raiten, D.J. Biomarkers of nutrition for development—Iodine review. J. Nutr. 2014, 144, 1322S–1342S. [Google Scholar] [CrossRef]
- Pop, V.J.; Kuijpens, J.L.; van Baar, A.L.; Verkerk, G.; van Son, M.M.; de Vijlder, J.J.; Vulsma, T.; Wiersinga, W.M.; Drexhage, H.A.; Vader, H.L. Low maternal free thyroxine concentrations during early pregnancy are associated with impaired psychomotor development in infancy. Clin. Endocrinol. 1999, 50, 149–155. [Google Scholar] [CrossRef]
- De Groot, L.; Abalovich, M.; Alexander, E.K.; Amino, N.; Barbour, L.; Cobin, R.H.; Eastman, C.J.; Lazarus, J.H.; Luton, D.; Mandel, S.J.; et al. Management of thyroid dysfunction during pregnancy and postpartum: An Endocrine Society clinical practice guideline. J. Clin. Endocrinol. Metab. 2012, 97, 2543–2565. [Google Scholar] [CrossRef]
- Alexander, E.K.; Pearce, E.N.; Brent, G.A.; Brown, R.S.; Chen, H.; Dosiou, C.; Grobman, W.A.; Laurberg, P.; Lazarus, J.H.; Susan, J.; et al. 2017 Guidelines of the American Thyroid Association for the Diagnosis and Management of Thyroid Disease During Pregnancy and the Postpartum. Thyroid Off. J. Am. Thyroid Assoc. 2017, 27, 315–389. [Google Scholar] [CrossRef]
- Cooper, D.S.; Pearce, E.N. Subclinical Hypothyroidism and Hypothyroxinemia in Pregnancy—Still No Answers. N. Engl. J. Med. 2017, 376, 876–877. [Google Scholar] [CrossRef]
- Vermiglio, F.; Lo Presti, V.P.; Moleti, M.; Sidoti, M.; Tortorella, G.; Scaffidi, G.; Castagna, M.G.; Mattina, F.; Violi, M.A.; Crisà, A.; et al. Attention deficit and hyperactivity disorders in the offspring of mothers exposed to mild-moderate iodine deficiency: A possible novel iodine deficiency disorder in developed countries. J. Clin. Endocrinol. Metab. 2004, 89, 6054–6060. [Google Scholar] [CrossRef]
- Abel, M.H.; Ystrom, E.; Caspersen, I.H.; Meltzer, H.M.; Aase, H.; Torheim, L.E.; Askeland, R.B.; Reichborn-Kjennerud, T.; Brantsæter, A.L. Maternal Iodine Intake and Offspring Attention-Deficit/Hyperactivity Disorder: Results from a Large Prospective Cohort Study. Nutrients 2017, 9, 1239. [Google Scholar] [CrossRef]
- Abel, M.H.; Korevaar, T.I.M.; Erlund, I.; Villanger, G.D.; Caspersen, I.H.; Arohonka, P.; Alexander, J.; Meltzer, H.M.; Brantsæter, A.L. Iodine Intake is Associated with Thyroid Function in Mild to Moderately Iodine Deficient Pregnant Women. Thyroid Off. J. Am. Thyroid Assoc. 2018, 28, 1359–1371. [Google Scholar] [CrossRef]
- Mackerras, D.E.; Eastman, C.J. Estimating the iodine supplementation level to recommend for pregnant and breastfeeding women in Australia. Med. J. Aust. 2012, 197, 238–242. [Google Scholar] [CrossRef]
- Glinoer, D.; De Nayer, P.; Delange, F.; Lemone, M.; Toppet, V.; Spehl, M.; Grun, J.P.; Kinthaert, J.; Lejeune, B. A randomized trial for the treatment of mild iodine deficiency during pregnancy: Maternal and neonatal effects. J. Clin. Endocrinol. Metab. 1995, 80, 258–269. [Google Scholar]
- Zhou, S.; Anderson, A.; Gibson, R.; Makrides, M. Effect of iodine supplementation in pregnancy on child development and other clinical outcomes: A systematic review of randomized controlled trials. Am. J. Clin. Nutr. 2013, 98, 1241–1254. [Google Scholar] [CrossRef]
- Gowachirapant, S.; Jaiswal, N.; Melse-Boonstra, A.; Galetti, V.; Stinca, S.; Mackenzie, I.; Thomas, S.; Thomas, T.; Winichagoon, P.; Srinivasan, K.; et al. Effect of iodine supplementation in pregnant women on child neurodevelopment: A randomised, double-blind, placebo-controlled trial. Lancet Diabetes Endocrinol. 2017, 5, 853–863. [Google Scholar] [CrossRef]
- Bath, S.C. Iodine supplementation in pregnancy in mildly deficient regions. Lancet Diabetes Endocrinol. 2017, 5, 840–841. [Google Scholar] [CrossRef]
- Shi, X.; Han, C.; Li, C.; Mao, J.; Wang, W.; Xie, X.; Li, C.; Xu, B.; Meng, T.; Du, J.; et al. Optimal and safe upper limits of iodine intake for early pregnancy in iodine-sufficient regions: A cross-sectional study of 7190 pregnant women in China. J. Clin. Endocrinol. Metab. 2015, 100, 1630–1638. [Google Scholar] [CrossRef]
- Wolff, J.; Chaikoff, I.L.; Goldberg, R.C.; Meier, J.R. The temporary nature of the inhibitory action of excess iodine on organic iodine synthesis in the normal thyroid. Endocrinology 1949, 45, 504–513. [Google Scholar] [CrossRef]
- Connolly, R.J.; Vidor, G.I.; Stewart, J.C. Increase in thyrotoxicosis in endemic goitre area after iodation of bread. Lancet 1970, 1, 500–502. [Google Scholar] [CrossRef]
- Li, M.; Liu, D.; Qu, C.; Zhang, P.; Qian, Q.; Zhang, C.; Jia, Q.; Wang, H.; Eastman, C.J.; Boyages, S.C.; et al. Endemic goitre in central China caused by excessive iodine intake. Lancet 1987, 2, 257–259. [Google Scholar]
- Chen, W.; Zhang, Y.; Hao, Y.; Wang, W.; Tan, L.; Bian, J.; Pearce, E.N.; Zimmermann, M.B.; Shen, J.; Zhang, W. Adverse effects on thyroid of Chinese children exposed to long-term iodine excess: Optimal and safe Tolerable Upper Intake Levels of iodine for 7- to 14-y-old children. Am. J. Clin. Nutr. 2018, 107, 780–788. [Google Scholar] [CrossRef]
- World Health Organisation, United Nations Children’s Fund. Reaching Optimal Iodine Nutrition in Pregnant and Lactating Women and Young Children. 2005. Available online: https://www.whoint/nutrition/publications/WHOStatement__IDD_pregnancy pdf (accessed on 21 June 2019).
- Delange, F. Iodine requirements during pregnancy, lactation and the neonatal period and indicators of optimal iodine nutrition. Public Health Nutr. 2007, 10, 1571–1580. [Google Scholar] [CrossRef]
- Bouhouch, R.R.; Bouhouch, S.; Cherkaoui, M.; Aboussad, A.; Stinca, S.; Haldimann, M.; Andersson, M.; Zimmermann, M.B. Direct iodine supplementation of infants versus supplementation of their breastfeeding mothers: A double-blind, randomised, placebo-controlled trial. Lancet Diabetes Endocrinol. 2014, 2, 197–209. [Google Scholar] [CrossRef]
- Harding, K.B.; Pena-Rosas, J.P.; Webster, A.C.; Yap, C.M.; Payne, B.A.; Ota, E.; De-Regil, L.M. Iodine supplementation for women during the preconception, pregnancy and postpartum period. Cochrane Database Syst. Rev. 2017, 3, Cd011761. [Google Scholar] [CrossRef]
| Population Group | Median UIC (μg/L) | Category of Iodine Intake |
|---|---|---|
| Non-Pregnant women | <100 >100 | Insufficient Adequate |
| Pregnant women | <150 150–249 250–499 ≥500 | Insufficient Adequate More than adequate Excessive ‡ |
| Lactating women | <100 ≥100 | Insufficient Adequate |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Eastman, C.J.; Ma, G.; Li, M. Optimal Assessment and Quantification of Iodine Nutrition in Pregnancy and Lactation: Laboratory and Clinical Methods, Controversies and Future Directions. Nutrients 2019, 11, 2378. https://doi.org/10.3390/nu11102378
Eastman CJ, Ma G, Li M. Optimal Assessment and Quantification of Iodine Nutrition in Pregnancy and Lactation: Laboratory and Clinical Methods, Controversies and Future Directions. Nutrients. 2019; 11(10):2378. https://doi.org/10.3390/nu11102378
Chicago/Turabian StyleEastman, Creswell J, Gary Ma, and Mu Li. 2019. "Optimal Assessment and Quantification of Iodine Nutrition in Pregnancy and Lactation: Laboratory and Clinical Methods, Controversies and Future Directions" Nutrients 11, no. 10: 2378. https://doi.org/10.3390/nu11102378
APA StyleEastman, C. J., Ma, G., & Li, M. (2019). Optimal Assessment and Quantification of Iodine Nutrition in Pregnancy and Lactation: Laboratory and Clinical Methods, Controversies and Future Directions. Nutrients, 11(10), 2378. https://doi.org/10.3390/nu11102378





