The Antimicrobial and Antiviral Activity of Polyphenols from Almond (Prunus dulcis L.) Skin
Abstract
1. Introduction
2. Materials and Methods
2.1. Preparation of Polyphenols Mix
2.2. Microbial Strains and Culture Conditions
2.3. Susceptibility Assay
2.4. Cell Cultures and Virus
2.5. Cell Viability Assay
2.6. Plaque Reduction Assay
2.7. HSV-1 Infection on Vero Cells
2.8. Antibodies
2.9. Protein Extractions and Immunoblot Analysis
2.10. DNA Extraction and Quantitative Real-Time RT-PCR
2.11. Statistical Analysis
3. Results
3.1. Antimicrobial Activity of Polyphenols
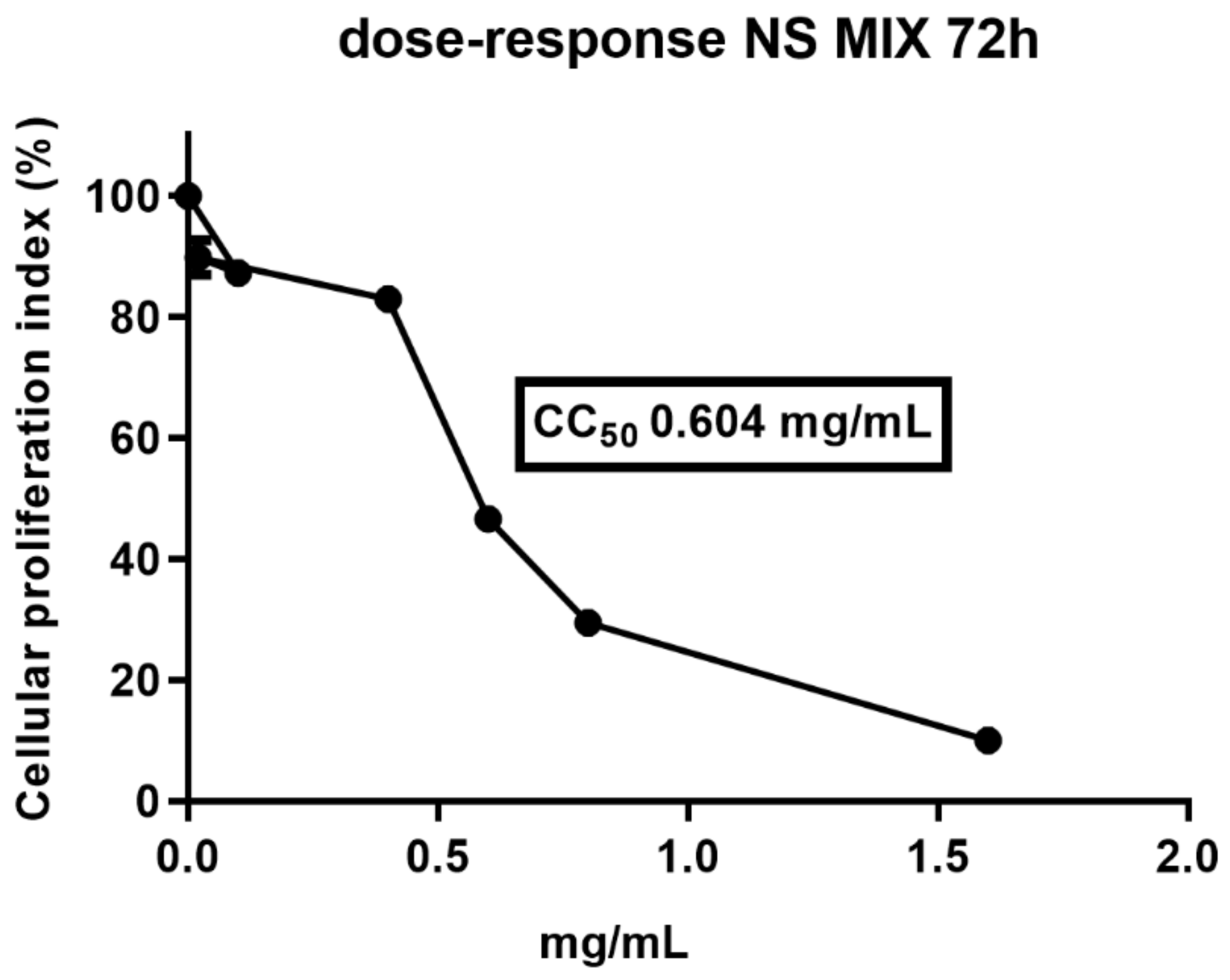
3.2. Cytotoxicity and Antiviral Activity Tests
Viability Assay
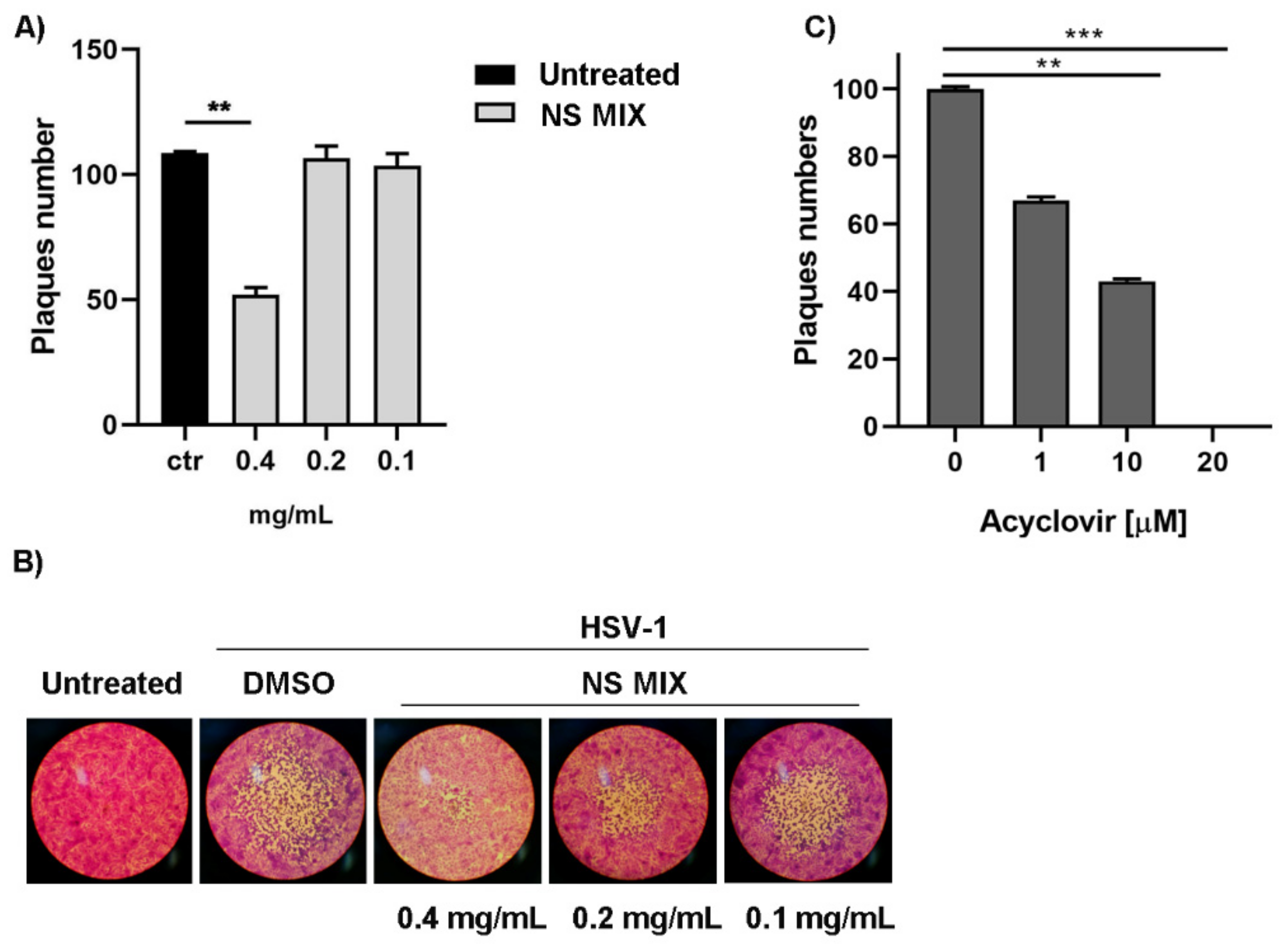
3.3. Antiviral Activity of Polyphenols Against HSV-1
3.3.1. Plaque Assay
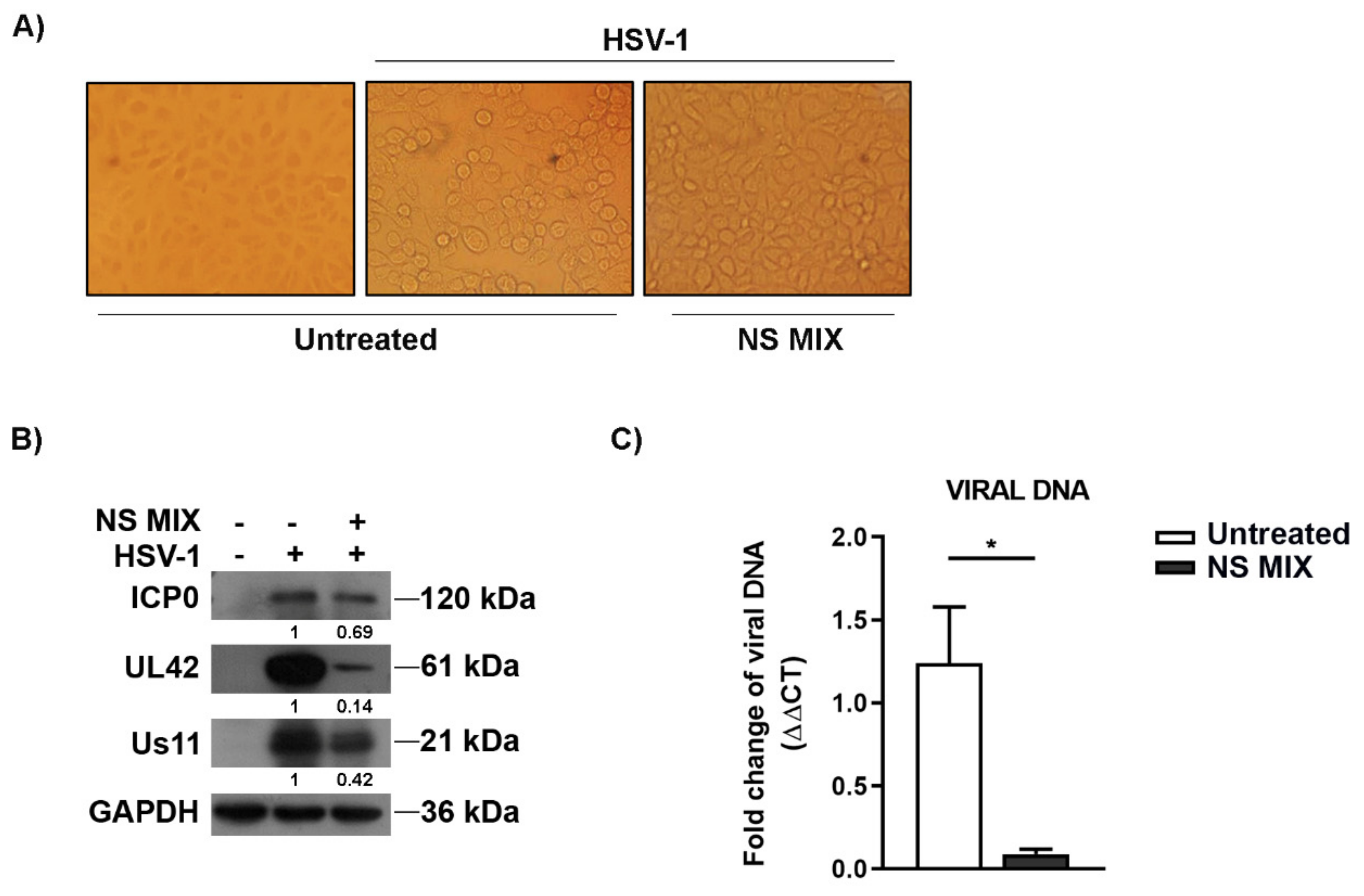
3.3.2. Expression of Viral Antigens and Quantification of Viral DNA
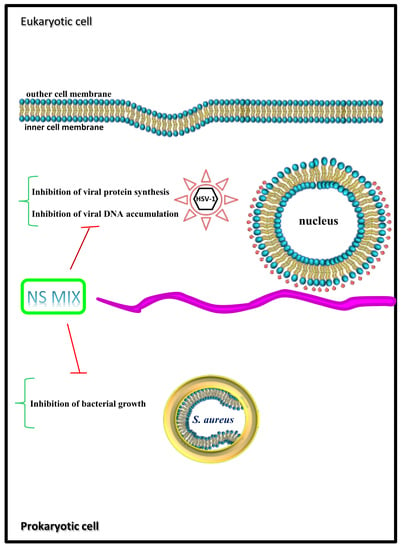
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Haider, S.; Batool, Z.; Haleem, D.J. Nootropic and hyphofagic effects following long term intake of almonds (Prunus amygdalus) in rats. Nutr. Hosp. 2012, 27, 2109–2115. [Google Scholar] [PubMed]
- U.S. Food and Drug Administration. Qualified Health Claims: Letter of Enforcement Discretion-Nuts and Coronary Heart Disease (Docket No. 02P-0505). Office of Nutritional Products, Labeling and Dietary Supplements; 14 July 2003. Available online: http://www.cfsan.fda.gov/~dms/qhcnuts2.html (accessed on 1 September 2019).
- Mandalari, G.; Tomaino, A.; Arcoraci, T.; Martorana, M.; Lo Turco, V.; Cacciola, F.; Rich, G.T.; Bisignano, C.; Saija, A.; Dugo, P.; et al. Characterization of polyphenols, lipids and dietary fibre from skins of almonds (Amygdalus communis L.). J. Food Comp. Anal. 2010, 23, 166–174. [Google Scholar] [CrossRef]
- Milbury, P.E.; Chen, C.-Y.; Dolnikowski, G.G.; Blumberg, J.B. Determination of Flavonoids and Phenolics and Their Distribution in Almonds. J. Agric. Food Chem. 2006, 54, 5027–5033. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.-Y.O.; Blumberg, J.B. In Vitro Activity of Almond Skin Polyphenols for Scavenging Free Radicals and Inducing Quinone Reductase. J. Agric. Food Chem. 2008, 56, 4427–4434. [Google Scholar] [CrossRef] [PubMed]
- Mandalari, G.; Bisignano, C.; D’Arrigo, M.; Ginestra, G.; Arena, A.; Tomaino, A.; Wickham, M.; Bisignano, G. Antimicrobial potential of polyphenols extracted from almond skins. Lett. Appl. Microbiol. 2010, 51, 83–89. [Google Scholar] [CrossRef] [PubMed]
- Mandalari, G.; Faulks, R.M.; Bisignano, C.; Waldron, K.W.; Narbad, A.; Wickham, M.S. In vitro evaluation of the prebiotic properties of almond skins (Amygdalus communis L.). FEMS Microbiol. Lett. 2010, 304, 116–122. [Google Scholar] [CrossRef] [PubMed]
- Bisignano, C.; Filocamo, A.; La Camera, E.; Zummo, S.; Fera, M.T.; Mandalari, G. Antibacterial activities of almond skins on cagA-positive and-negative clinical isolates of Helicobacter pylori. BMC Microbiol. 2013, 13, 103. [Google Scholar] [CrossRef]
- Lowy, F.D. Staphylococcus aureus infections. N. Engl. J. Med. 1998, 339, 520–532. [Google Scholar] [CrossRef] [PubMed]
- Arena, A.; Bisignano, C.; Stassi, G.; Mandalari, G.; Wickham, M.S.; Bisignano, G. Immunomodulatory and antiviral activity of almond skins. Immunol. Lett. 2010, 132, 18–23. [Google Scholar] [CrossRef]
- Bisignano, C.; Mandalari, G.; Smeriglio, A.; Trombetta, D.; Pizzo, M.M.; Pennisi, R.; Sciortino, M.T. Almond Skin Extracts Abrogate HSV-1 Replication by Blocking Virus Binding to the Cell. Viruses 2017, 9, 178. [Google Scholar] [CrossRef]
- Arena, A.; Bisignano, C.; Stassi, G.; Filocamo, A.; Mandalari, G. Almond Skin Inhibits HSV-2 Replication in Peripheral Blood Mononuclear Cells by Modulating the Cytokine Network. Molecules 2015, 20, 8816–8822. [Google Scholar] [CrossRef] [PubMed]
- Mandalari, G.; Genovese, T.; Bisignano, C.; Mazzon, E.; Wickham, M.; Di Paola, R.; Bisignano, G.; Cuzzocrea, S. Neuroprotective effects of almond skins in experimental spinal cord injury. Clin. Nutr. 2011, 30, 221–233. [Google Scholar] [CrossRef] [PubMed]
- Clinical and Laboratory Standards Institute (CLSI). Performance Standards for Antimicrobial Susceptibility Testing; Twentieth Informational Supplement; Clinical and Laboratory Standards Institute: Wayne, PA, USA, 2012. [Google Scholar]
- Sciortino, M.T.; Parisi, T.; Siracusano, G.; Mastino, A.; Taddeo, B.; Roizman, B. The virion host shutoff RNase plays a key role in blocking the activation of protein kinase R in cells infected with herpes simplex virus 1. J. Virol. 2013, 87, 3271–3276. [Google Scholar] [CrossRef] [PubMed]
- Bisignano, C.; Ginestra, G.; Smeriglio, A.; La Camera, E.; Crisafi, G.; Franchina, F.A.; Tranchida, P.Q.; Alibrandi, A.; Trombetta, D.; Mondello, L.; et al. Study of the Lipid Profile of ATCC and Clinical Strains of Staphylococcus aureus in Relation to Their Antibiotic Resistance. Molecules 2019, 24, 1276. [Google Scholar] [CrossRef] [PubMed]
- Duda-Chodak, A.; Tarko, T.; Satora, P.; Sroka, P. Interaction of dietary compounds, especially polyphenols, with the intestinal microbiota: A review. Eur. J. Nutr. 2015, 54, 325–341. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.-H.; Wang, M.-S.; Zeng, X.-A.; Xu, X.-M.; Brennan, C.S. Membrane and genomic DNA dual-targeting of citrus flavonoid naringenin against Staphylococcus aureus. Integr. Biol. 2017, 9, 820–829. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.-H.; Huang, C.-C.; Chen, C.-C.; Yang, K.-J.; Huang, C.-Y. Inhibition of Staphylococcus aureus PriA Helicase by Flavonol Kaempferol. Protein J. 2015, 34, 169–172. [Google Scholar] [CrossRef] [PubMed]
- Ming, D.; Wang, D.; Cao, F.; Xiang, H.; Mu, D.; Cao, J.; Li, B.; Zhong, L.; Dong, X.; Zhong, X.; et al. Kaempferol Inhibits the Primary Attachment Phase of Biofilm Formation in Staphylococcus aureus. Front. Microbiol. 2017, 8, 2263. [Google Scholar] [CrossRef]
- Lin, R.-D.; Chin, Y.-P.; Hou, W.-C.; Lee, M.-H. The Effects of Antibiotics Combined with Natural Polyphenols against Clinical Methicillin-Resistant Staphylococcus aureus (MRSA). Planta Medica 2008, 74, 840–846. [Google Scholar] [CrossRef]
- Jiang, L.; Li, H.; Wang, L.; Song, Z.; Shi, L.; Li, W.; Deng, X.; Wang, J. Isorhamnetin Attenuates Staphylococcus aureus-Induced Lung Cell Injury by Inhibiting Alpha-Hemolysin Expression. J. Microbiol. Biotechnol. 2016, 26, 596–602. [Google Scholar] [CrossRef]
- Khan, M.T.H.; Ather, A.; Thompson, K.D.; Gambari, R. Extracts and molecules from medicinal plants against herpes simplex viruses. Antivir. Res. 2005, 67, 107–119. [Google Scholar] [CrossRef] [PubMed]
- Son, M.; Lee, M.; Sung, G.-H.; Lee, T.; Shin, Y.S.; Cho, H.; Lieberman, P.M.; Kang, H. Bioactive activities of natural products against herpesvirus infection. J. Microbiol. 2013, 51, 545–551. [Google Scholar] [CrossRef] [PubMed]
- Hassan, S.T.; Masarčíková, R.; Berchová, K. Bioactive natural products with anti-herpes simplex virus properties. J. Pharm. Pharmacol. 2015, 67, 1325–1336. [Google Scholar] [CrossRef] [PubMed]
- Schneider, S.; Reichling, J.; Stintzing, F.; Carle, R.; Schnitzler, P. Efficacy of an aqueous Pelargonium sidoides extract against herpesvirus. Planta Med. 2008, 74, PA322. [Google Scholar] [CrossRef]
- Ren, Z.; Zhang, C.-H.; Wang, L.-J.; Cui, Y.-X.; Qi, R.-B.; Yang, C.-R.; Zhang, Y.-J.; Wei, X.-Y.; Lu, D.-X.; Wang, Y.-F. In vitro anti-viral activity of the total alkaloids from Tripterygium hypoglaucum against herpes simplex virus type 1. Virol. Sin. 2010, 25, 107–114. [Google Scholar] [CrossRef]
- Hirabayashi, K.; Iwata, S.; Matsumoto, H.; Mori, T.; Shibata, S.; Baba, M.; Ito, M.; Shigeta, S.; Nakashima, H.; Yamamoto, N. Antiviral activities of glycyrrhizin and its modified compounds against human immunodeficiency virus type 1(HIV-1) and herpes simplex virus type 1(HSV-1) in vitro. Chem. Pharm. Bull. 1991, 39, 112–115. [Google Scholar] [CrossRef] [PubMed]
| NS MIX | Vancomycin | Teicoplanin | ||||
|---|---|---|---|---|---|---|
| S. aureus Strain | MIC | MBC | MIC | MBC | MIC | MBC |
| ATCC 6538P | 0.31 | >1.25 | 0.31 | 0.62 | 0.15 | 0.15 |
| 43300 | 1.25 | >1.25 | 0.31 | 0.62 | 0.31 | 0.31 |
| 526 | 1.25 | >1.25 | 0.31 | 0.61 | 0.31 | 0.62 |
| 530 | >1.25 | >1.25 | 0.31–0.62 | 0.62 | 0.31–0.62 | 0.62 |
| 808 | 1.25 | >1.25 | 0.31 | 0.31 | 0.15 | 0.15 |
| 814 | 1.25 | >1.25 | 0.62 | 0.62 | 0.62 | 1.25 |
| 8 | >1,25 | >1.25 | 0.62–1.25 | 1.25 | 0.31 | 0.31 |
| 14 | >1.25 | >1.25 | 0.31 | 0.31 | 0.15–0.31 | 0.31 |
| 6 | 0.62 | >1.25 | 0.31 | 0.62 | 1.25 | 1.25 |
| 84 | >1.25 | >1.25 | 0.62 | 0.62 | 0.62 | 1.25 |
| 32 | >1.25 | >1.25 | 0.62 | 0.62 | 0.31 | 0.31 |
| S. aureus Strain | N-7-O-g | K-3-O-g | I-3-O-r | I-3-O-g | Cat | Epic |
|---|---|---|---|---|---|---|
| ATCC 6538P | 1.25–0.62 | 0.62 | 0.31–0.15 | 1.25 | 0.15 | 0.15–0.078 |
| 43300 | >1.25 | >1.25 | 1.25 | >1.25 | >1.25 | >1.25 |
| 526 | >1.25 | 0.62 | 0.62 | >1.25 | >1.25 | >1.25 |
| 530 | >1.25 | >1.25 | 0.62 | >1.25 | >1.25 | >1.25 |
| 808 | >1.25 | >1.25 | 1.25-0.62 | >1.25 | >1.25 | >1.25 |
| 814 | >1.25 | >1.25 | >1.25 | >1.25 | >1.25 | >1.25 |
| 8 | >1.25 | >1.25 | >1.25 | >1.25 | >1.25 | >1.25 |
| 14 | >1.25 | 0.62 | 1.25–0.62 | >1.25 | >1.25 | >1.25 |
| 6 | 1.25 | 0.62 | 0.62–0,31 | >1.25 | >1.25 | >1.25 |
| 84 | 1.25–0.62 | 0.62 | 1,25–0.62 | >1.25 | >1.25 | >1.25 |
| 32 | 1.25 | 0.62 | 0.31–0.15 | >1.25 | >1.25 | >1.25 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Musarra-Pizzo, M.; Ginestra, G.; Smeriglio, A.; Pennisi, R.; Sciortino, M.T.; Mandalari, G. The Antimicrobial and Antiviral Activity of Polyphenols from Almond (Prunus dulcis L.) Skin. Nutrients 2019, 11, 2355. https://doi.org/10.3390/nu11102355
Musarra-Pizzo M, Ginestra G, Smeriglio A, Pennisi R, Sciortino MT, Mandalari G. The Antimicrobial and Antiviral Activity of Polyphenols from Almond (Prunus dulcis L.) Skin. Nutrients. 2019; 11(10):2355. https://doi.org/10.3390/nu11102355
Chicago/Turabian StyleMusarra-Pizzo, Maria, Giovanna Ginestra, Antonella Smeriglio, Rosamaria Pennisi, Maria Teresa Sciortino, and Giuseppina Mandalari. 2019. "The Antimicrobial and Antiviral Activity of Polyphenols from Almond (Prunus dulcis L.) Skin" Nutrients 11, no. 10: 2355. https://doi.org/10.3390/nu11102355
APA StyleMusarra-Pizzo, M., Ginestra, G., Smeriglio, A., Pennisi, R., Sciortino, M. T., & Mandalari, G. (2019). The Antimicrobial and Antiviral Activity of Polyphenols from Almond (Prunus dulcis L.) Skin. Nutrients, 11(10), 2355. https://doi.org/10.3390/nu11102355