The Association between Alcohol Consumption and Serum Metabolites and the Modifying Effect of Smoking
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants and Study Design
2.2. Assessment of Alcohol Consumption
2.3. Assessment of Cigarette Smoking
2.4. Assessment of Diet and other Lifestyle Variables
2.5. Measurement of Serum Metabolites
2.6. Statistical Analysis
2.7. Identification of Metabolite Patterns
2.8. Multivariable Adjusted Linear Regression
3. Results
3.1. Characteristics of the Study Population
3.2. Identification of Metabolite Patterns
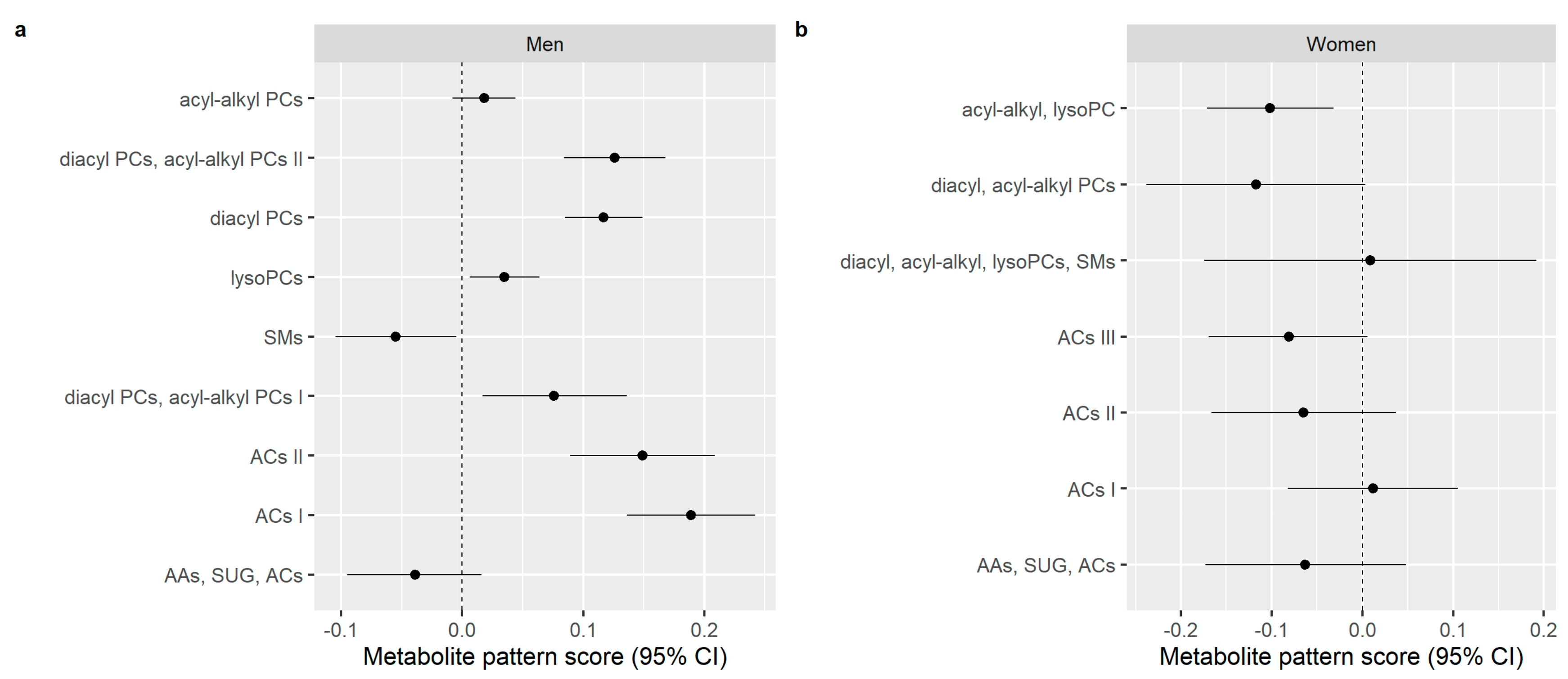
3.3. Multivariable Analyses of the Association between Alcohol Consumption and Metabolite Patterns
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- French, M.T.; Zavala, S.K. The health benefits of moderate drinking revisited: Alcohol use and self-reported health status. Am. J. Health Promot. 2007, 21, 484–491. [Google Scholar] [CrossRef] [PubMed]
- Matsumoto, C.; Miedema, M.D.; Ofman, P.; Gaziano, J.M.; Sesso, H.D. An expanding knowledge of the mechanisms and effects of alcohol consumption on cardiovascular disease. J. Cardiopulm. Rehabil. Prev. 2014, 34, 159–171. [Google Scholar] [CrossRef] [PubMed]
- Mostofsky, E.; Mukamal, K.J.; Giovannucci, E.L.; Stampfer, M.J.; Rimm, E.B. Key Findings on Alcohol Consumption and a Variety of Health Outcomes From the Nurses’ Health Study. Am. J. Public Health 2016, 106, 1586–1591. [Google Scholar] [CrossRef] [PubMed]
- Dumitrescu, R.G. Alcohol-Induced Epigenetic Changes in Cancer. Methods Mol. Biol. 2018, 1856, 157–172. [Google Scholar] [CrossRef] [PubMed]
- Cai, L. Alcoholic cardiomyopathy: Acetaldehyde, insulin insensitization and ER stress. J. Mol. Cell. Cardiol. 2008, 44, 979–982. [Google Scholar] [CrossRef] [PubMed]
- López-Larramona, G.; Lucendo, A.J.; González-Delgado, L. Alcoholic liver disease and changes in bone mineral density. Rev. Esp. Enferm. Dig. 2013, 105, 609–621. [Google Scholar] [CrossRef] [PubMed]
- Holmes, E.; Loo, R.L.; Stamler, J.; Bictash, M.; Yap, I.K.S.; Chan, Q.; Ebbels, T.; Iorio, M.d.; Brown, I.J.; Veselkov, K.A.; et al. Human metabolic phenotype diversity and its association with diet and blood pressure. Nature 2008, 453, 396–400. [Google Scholar] [CrossRef]
- Heinzmann, S.S.; Merrifield, C.A.; Rezzi, S.; Kochhar, S.; Lindon, J.C.; Holmes, E.; Nicholson, J.K. Stability and robustness of human metabolic phenotypes in response to sequential food challenges. J. Proteome Res. 2012, 11, 643–655. [Google Scholar] [CrossRef]
- Slupsky, C.M.; Rankin, K.N.; Wagner, J.; Fu, H.; Chang, D.; Weljie, A.M.; Saude, E.J.; Lix, B.; Adamko, D.J.; Shah, S.; et al. Investigations of the effects of gender, diurnal variation, and age in human urinary metabolomic profiles. Anal. Chem. 2007, 79, 6995–7004. [Google Scholar] [CrossRef]
- Piano, M.R. Alcohol’s Effects on the Cardiovascular System. Alcohol Res. 2017, 38, 219–241. [Google Scholar]
- Bagnardi, V.; Blangiardo, M.; Corrao, G.; Vecchia, C.L. Alcohol Consumption and the Risk of Cancer: A Meta-Analysis. Alcohol Res. Health 2001, 25, 263–270. [Google Scholar] [PubMed]
- Askgaard, G.; Grønbæk, M.; Kjær, M.S.; Tjønneland, A.; Tolstrup, J.S. Alcohol drinking pattern and risk of alcoholic liver cirrhosis: A prospective cohort study. J. Hepatol. 2015, 62, 1061–1067. [Google Scholar] [CrossRef] [PubMed]
- Wu, D.M.; Pai, L.; Sun, P.K.; Hsu, L.L.; Sun, C.A. Joint effects of alcohol consumption and cigarette smoking on atherogenic lipid and lipoprotein profiles: Results from a study of Chinese male population in Taiwan. Eur. J. Epidemiol. 2001, 17, 629–635. [Google Scholar] [CrossRef] [PubMed]
- Whitehead, T.P.; Robinson, D.; Allaway, S.L. The effects of cigarette smoking and alcohol consumption on blood lipids: A dose-related study on men. Ann. Clin. Biochem. 1996, 33 Pt 2, 99–106. [Google Scholar] [CrossRef]
- Lacruz, M.E.; Kluttig, A.; Tiller, D.; Medenwald, D.; Giegling, I.; Rujescu, D.; Prehn, C.; Adamski, J.; Frantz, S.; Greiser, K.H.; et al. Cardiovascular Risk Factors Associated With Blood Metabolite Concentrations and Their Alterations During a 4-Year Period in a Population-Based Cohort. Circ. Cardiovasc. Genet. 2016, 9, 487–494. [Google Scholar] [CrossRef] [PubMed]
- Pallister, T.; Jennings, A.; Mohney, R.P.; Yarand, D.; Mangino, M.; Cassidy, A.; MacGregor, A.; Spector, T.D.; Menni, C. Characterizing Blood Metabolomics Profiles Associated with Self-Reported Food Intakes in Female Twins. PLoS ONE 2016, 11, e0158568. [Google Scholar] [CrossRef]
- van Roekel, E.H.; Trijsburg, L.; Assi, N.; Carayol, M.; Achaintre, D.; Murphy, N.; Rinaldi, S.; Schmidt, J.A.; Stepien, M.; Kaaks, R.; et al. Circulating Metabolites Associated with Alcohol Intake in the European Prospective Investigation into Cancer and Nutrition Cohort. Nutrients 2018, 10, e654. [Google Scholar] [CrossRef] [PubMed]
- Jaremek, M.; Yu, Z.; Mangino, M.; Mittelstrass, K.; Prehn, C.; Singmann, P.; Xu, T.; Dahmen, N.; Weinberger, K.M.; Suhre, K.; et al. Alcohol-induced metabolomic differences in humans. Transl. Psychiatry 2013, 3, e276. [Google Scholar] [CrossRef]
- McDonald, J.H. Handbook of Biological Statistics, 3rd ed.; Sparky House Publishing: Baltimore, MD, USA, 2014. [Google Scholar]
- Förster, J. Treelet Transform for Untargeted Metabolomics Data. Ph.D. Thesis, Technische Universität Berlin, Fakultät VII—Wirtschaft und Management, Berlin/Heidelberg, Germany, 5 August 2014. [Google Scholar]
- Wilsnack, R.W.; Wilsnack, S.C.; Kristjanson, A.F.; Vogeltanz-Holm, N.D.; Gmel, G. Gender and alcohol consumption: Patterns from the multinational GENACIS project. Addiction 2009, 104, 1487–1500. [Google Scholar] [CrossRef]
- Mittelstrass, K.; Ried, J.S.; Yu, Z.; Krumsiek, J.; Gieger, C.; Prehn, C.; Roemisch-Margl, W.; Polonikov, A.; Peters, A.; Theis, F.J.; et al. Discovery of sexual dimorphisms in metabolic and genetic biomarkers. PLoS Genet. 2011, 7, e1002215. [Google Scholar] [CrossRef]
- Schühlen, H. Pre-specified vs. post-hoc subgroup analyses: Are we wiser before or after a trial has been performed? Eur. Heart J. 2014, 35, 2055–2057. [Google Scholar] [CrossRef] [PubMed]
- Boeing, H.; Wahrendorf, J.; Becker, N. EPIC-Germany—A source for studies into diet and risk of chronic diseases. European Investigation into Cancer and Nutrition. Ann. Nutr. Metab. 1999, 43, 195–204. [Google Scholar] [CrossRef] [PubMed]
- Ford, E.S.; Schulze, M.B.; Bergmann, M.M.; Thamer, C.; Joost, H.-G.; Boeing, H. Liver enzymes and incident diabetes: Findings from the European Prospective Investigation into Cancer and Nutrition (EPIC)-Potsdam Study. Diabetes Care 2008, 31, 1138–1143. [Google Scholar] [CrossRef] [PubMed]
- Bohlscheid-Thomas, S.; Hoting, I.; Boeing, H.; Wahrendorf, J. Reproducibility and relative validity of food group intake in a food frequency questionnaire developed for the German part of the EPIC project. European Prospective Investigation into Cancer and Nutrition. Int. J. Epidemiol. 1997, 26 (Suppl. 1), S59–S70. [Google Scholar] [CrossRef]
- Zukunft, S.; Prehn, C.; Röhring, C.; Möller, G.; Hrabě de Angelis, M.; Adamski, J.; Tokarz, J. High-throughput extraction and quantification method for targeted metabolomics in murine tissues. Metabolomics 2017, 14, 18. [Google Scholar] [CrossRef] [PubMed]
- Römisch-Margl, W.; Prehn, C.; Bogumil, R.; Röhring, C.; Suhre, K.; Adamski, J. Procedure for tissue sample preparation and metabolite extraction for high-throughput targeted metabolomics. Metabolomics 2012, 8, 133–142. [Google Scholar] [CrossRef]
- Fukai, K.; Harada, S.; Iida, M.; Kurihara, A.; Takeuchi, A.; Kuwabara, K.; Sugiyama, D.; Okamura, T.; Akiyama, M.; Nishiwaki, Y.; et al. Metabolic Profiling of Total Physical Activity and Sedentary Behavior in Community-Dwelling Men. PLoS ONE 2016, 11, e0164877. [Google Scholar] [CrossRef] [PubMed]
- Hefni, M.E.; Witthöft, C.M.; Moazzami, A.A. Plasma metabolite profiles in healthy women differ after intervention with supplemental folic acid v. folate-rich foods. J. Nutr. Sci. 2018, 7, e32. [Google Scholar] [CrossRef]
- Auro, K.; Joensuu, A.; Fischer, K.; Kettunen, J.; Salo, P.; Mattsson, H.; Niironen, M.; Kaprio, J.; Eriksson, J.G.; Lehtimaki, T.; et al. A metabolic view on menopause and ageing. Nat. Commun. 2014, 5, 4708. [Google Scholar] [CrossRef]
- Carayol, M.; Leitzmann, M.F.; Ferrari, P.; Zamora-Ros, R.; Achaintre, D.; Stepien, M.; Schmidt, J.A.; Travis, R.C.; Overvad, K.; Tjønneland, A.; et al. Blood metabolic signatures of body mass index: A targeted metabolomics study in the EPIC cohort. J. Proteome Res. 2017, 16, 3137–3146. [Google Scholar] [CrossRef] [PubMed]
- Britton, A.; Ben-Shlomo, Y.; Benzeval, M.; Kuh, D.; Bell, S. Life course trajectories of alcohol consumption in the United Kingdom using longitudinal data from nine cohort studies. BMC Med. 2015, 13, 47. [Google Scholar] [CrossRef] [PubMed]
- Kleiner, K.D.; Gold, M.S.; Frost-Pineda, K.; Lenz-Brunsman, B.; Perri, M.G.; Jacobs, W.S. Body mass index and alcohol use. J. Addict. Dis. 2004, 23, 105–118. [Google Scholar] [CrossRef] [PubMed]
- Werneck, A.O.; Oyeyemi, A.L.; Szwarcwald, C.L.; Silva, D.R. Association between physical activity and alcohol consumption: Sociodemographic and behavioral patterns in Brazilian adults. J. Public Health 2018. [Google Scholar] [CrossRef]
- Johnson, B.A.; Seneviratne, C. Alcohol-medical drug interactions. Handb. Clin. Neurol. 2014, 125, 543–559. [Google Scholar] [CrossRef] [PubMed]
- Collins, S.E. Associations between Socioeconomic Factors and Alcohol Outcomes. Alcohol Res. 2016, 38, 83–94. [Google Scholar]
- Rachdaoui, N.; Sarkar, D.K. Effects of alcohol on the endocrine system. Endocrinol. Metab. Clin. N. Am. 2013, 42, 593–615. [Google Scholar] [CrossRef]
- Cheung, W.; Keski-Rahkonen, P.; Assi, N.; Ferrari, P.; Freisling, H.; Rinaldi, S.; Slimani, N.; Zamora-Ros, R.; Rundle, M.; Frost, G.; et al. A metabolomic study of biomarkers of meat and fish intake. Am. J. Clin. Nutr. 2017, 105, 600–608. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, J.A.; Rinaldi, S.; Ferrari, P.; Carayol, M.; Achaintre, D.; Scalbert, A.; Cross, A.J.; Gunter, M.J.; Fensom, G.K.; Appleby, P.N.; et al. Metabolic profiles of male meat eaters, fish eaters, vegetarians, and vegans from the EPIC-Oxford cohort. Am. J. Clin. Nutr. 2015, 102, 1518–1526. [Google Scholar] [CrossRef]
- Ruf, T.; Nagel, G.; Altenburg, H.-P.; Miller, A.B.; Thorand, B. Food and nutrient intake, anthropometric measurements and smoking according to alcohol consumption in the EPIC Heidelberg study. Ann. Nutr. Metab. 2005, 49, 16–25. [Google Scholar] [CrossRef]
- Fawehinmi, T.O.; Ilomäki, J.; Voutilainen, S.; Kauhanen, J. Alcohol Consumption and Dietary Patterns: The FinDrink Study. PLoS ONE 2012, 7, e38607. [Google Scholar] [CrossRef]
- Kesse, E.; Clavel-Chapelon, F.; Slimani, N.; van Liere, M.; E3N Group. Do eating habits differ according to alcohol consumption? Results of a study of the French cohort of the European Prospective Investigation into Cancer and Nutrition (E3N-EPIC). Am. J. Clin. Nutr. 2001, 74, 322–327. [Google Scholar] [CrossRef] [PubMed]
- Bergmann, M.M.; Rehm, J.; Klipstein-Grobusch, K.; Boeing, H.; Schütze, M.; Drogan, D.; Overvad, K.; Tjønneland, A.; Halkjær, J.; Fagherazzi, G.; et al. The association of pattern of lifetime alcohol use and cause of death in the European prospective investigation into cancer and nutrition (EPIC) study. Int. J. Epidemiol. 2013, 42, 1772–1790. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.J.; Wang, J.Y.; Hertervig, E.; Cheng, Y.; Nilsson, A.; Duan, R.D. Activation of neutral sphingomyelinase participates in ethanol-induced apoptosis in Hep G2 cells. Alcohol Alcohol. 2000, 35, 569–573. [Google Scholar] [CrossRef] [PubMed]
- Deaciuc, I.V.; Nikolova-Karakashian, M.; Fortunato, F.; Lee, E.Y.; Hill, D.B.; McClain, C.J. Apoptosis and dysregulated ceramide metabolism in a murine model of alcohol-enhanced lipopolysaccharide hepatotoxicity. Alcohol. Clin. Exp. Res. 2000, 24, 1557–1565. [Google Scholar] [CrossRef] [PubMed]
- Saito, M.; Saito, M.; Cooper, T.B.; Vadasz, C. Ethanol-induced changes in the content of triglycerides, ceramides, and glucosylceramides in cultured neurons. Alcohol. Clin. Exp. Res. 2005, 29, 1374–1383. [Google Scholar] [CrossRef] [PubMed]
- Thomasson, H.R. Gender differences in alcohol metabolism. Physiological responses to ethanol. Recent Dev. Alcohol. 1995, 12, 163–179. [Google Scholar] [PubMed]
- Altman, D.G.; Royston, P. The cost of dichotomising continuous variables. BMJ 2006, 332, 1080. [Google Scholar] [CrossRef]
- Liang, Y.; Harris, F.L.; Brown, L.A.S. Alcohol induced mitochondrial oxidative stress and alveolar macrophage dysfunction. BioMed Res. Int. 2014, 2014, 371593. [Google Scholar] [CrossRef] [PubMed]
- McGill, M.R.; Li, F.; Sharpe, M.R.; Williams, C.D.; Curry, S.C.; Ma, X.; Jaeschke, H. Circulating acylcarnitines as biomarkers of mitochondrial dysfunction after acetaminophen overdose in mice and humans. Arch. Toxicol. 2014, 88, 391–401. [Google Scholar] [CrossRef]
- Bjørndal, B.; Alterås, E.K.; Lindquist, C.; Svardal, A.; Skorve, J.; Berge, R.K. Associations between fatty acid oxidation, hepatic mitochondrial function, and plasma acylcarnitine levels in mice. Nutr. Metab. 2018, 15, 10. [Google Scholar] [CrossRef]
- Perkins, K.A. Metabolic effects of cigarette smoking. J. Appl. Physiol. 1992, 72, 401–409. [Google Scholar] [CrossRef] [PubMed]
- Ionescu, N.; de Freitas, C.; Bueno, A.A. Perturbations in blood phosphatidylcholine and sphingomyelin Fatty Acid composition in a sample population of cigarette smokers. Indian J. Clin. Biochem. 2013, 28, 361–367. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Würtz, P.; Cook, S.; Wang, Q.; Tiainen, M.; Tynkkynen, T.; Kangas, A.J.; Soininen, P.; Laitinen, J.; Viikari, J.; Kähönen, M.; et al. Metabolic profiling of alcohol consumption in 9778 young adults. Int. J. Epidemiol. 2016, 45, 1493–1506. [Google Scholar] [CrossRef] [PubMed]
- Dahm, C.C.; Ostergaard, J.N.; Gorst-Rasmussen, A.; Jakobsen, M.U.; Schmidt, E.B.; Tjonneland, A.; Overvad, K. P1-17 The treelet transform—A novel method for determining patterns in adipose tissue fatty acids. J. Epidemiol. Commun. Health 2011, 65, A71–A72. [Google Scholar] [CrossRef]
- Del Boca, F.K.; Darkes, J. The validity of self-reports of alcohol consumption: State of the science and challenges for research. Addiction 2003, 98 (Suppl. 2), 1–12. [Google Scholar] [CrossRef]
- Kalapatapu, R.K.; Chambers, R. Novel Objective Biomarkers of Alcohol Use: Potential Diagnostic and Treatment Management Tools in Dual Diagnosis Care. J. Dual Diagn. 2009, 5, 57–82. [Google Scholar] [CrossRef]
| Number of Participants | Total | Men | Women | p-Value |
|---|---|---|---|---|
| 2433 | 934 | 1499 | ||
| Participant characteristics | ||||
| Age1, years | 50.46 (8.89) | 52.61 (7.87) | 49.13 (9.22) | <0.001 |
| BMI1, kg/m2 | 26.14 (4.33) | 26.80 (3.63) | 25.73 (4.68) | <0.001 |
| WC1, cm | 85.90 (12.87) | 94.18 (9.98) | 80.74 (11.73) | <0.001 |
| Education, university2 | 929 (38.2) | 490 (52.5) | 439 (29.3) | <0.001 |
| Full time employment (≥35 h/week)2 | 1435 (59.0) | 607 (65.0) | 828 (55.2) | <0.001 |
| Physically active, moderately inactive2a | 956 (39.3) | 348 (37.3) | 608 (40.6) | 0.157 |
| Alcohol consumption3 | 8.54 (3.01, 20.6) | 19.62 (8.92, 33.82) | 5.16 (2.02, 10.76) | <0.001 |
| Alcohol consumption2 | <0.001 | |||
| Non-Consumersb | 72 (3.0) | 31 (3.3) | 41 (2.7) | |
| Lightc | 272 (11.2) | 61 (6.5) | 211 (14.0) | |
| Below recommended limitd | 1403 (57.7) | 479 (51.3) | 924 (61.6) | |
| Light to moderatee | 547 (22.5) | 296 (31.7) | 251 (16.7) | |
| Heavyf | 139 5.7) | 67 (7.2) | 72 (4.8) | |
| Smoking status2 | <0.001 | |||
| never smokerg | 1627 (66.9) | 553 (59.2) | 1074 (71.6) | |
| former smokerh | 352 (14.5) | 175 (18.7) | 177 (11.8) | |
| Current smoker | 454 (18.7) | 206 (22.1) | 248 (16.5) | |
| Number of cigarettes2 | <0.001 | |||
| ≤ 15, CPD | 291 (64.1) | 102 (49.5) | 189 (76.2) | |
| 16–24, CPD | 112 (24.7) | 63 (30.6) | 49 (19.8) | |
| ≥ 25, CPD | 51 (11.2) | 41 (19.9) | 10 (4.0) | |
| Prevalent Diseases2 | ||||
| Cancer | 127 (5.2) | 30 (3.2) | 97 (6.5) | 0.001 |
| Stroke | 27 (1.1) | 18 (1.9) | 9 (0.6) | 0.005 |
| Myocardial infarction | 54 (2.2) | 42 (4.5) | 12 (0.8) | <0.001 |
| Medication2 | ||||
| Lipid-lowering Drugs | 130 (5.3) | 65 (6.9) | 65 (4.3) | 0.007 |
| Antiphlogistika | 5 (0.2) | 4 (0.4) | 1 (0.1) | 0.149 |
| Diuretics | 57 (2.3) | 21 (2.2) | 36 (2.4) | 0.916 |
| Metabolite Patterns | Groups of Alcohol Consumers | Mean Score* |
|---|---|---|
| ACs I | Heavy | 0.952a |
| Light to moderate | 0.372b | |
| Below RL | −0.098c | |
| Light | −0.317c | |
| Non-consumers | −0.283c | |
| ACs II | Heavy | 0.715a |
| Light to moderate | 0.227ab | |
| Below RL | −0.106bc | |
| Light | −0.604c | |
| Non-consumers | −0.208bc | |
| SMs | Heavy | −0.240b |
| Light to moderate | 0.198ab | |
| Below RL | 0.261a | |
| Light | 0.066ab | |
| Non-consumers | 0.592a | |
| diacyl PCs | Heavy | 0.541a |
| Light to moderate | 0.228a | |
| Below RL | −0.008b | |
| Light | −0.015b | |
| Non-consumers | −0.183b | |
| diacyl PCs, acyl-alkyl PCs II | Heavy | 0.461a |
| Light to moderate | 0.254a | |
| Below RL | −0.009b | |
| Light | −0.194b | |
| Non-consumers | −0.581b |
| Metabolite Patterns | Never Smoker | Former Smoker | Current Smoker | |||
|---|---|---|---|---|---|---|
| β (CI) | p-Value | β (CI) | p-Value | β (CI) | p-Value | |
| Men 1 | ||||||
| ACs I | 0.206 (0.137–0.276) | <0.001 | 0.094 (−0.052–0.240) | 0.208 | 0.192 (0.072–0.312) | 0.002 |
| ACs II | 0.169 (0.084–0.254) | <0.001 | 0.026 (−0.120–0.173) | 0.726 | 0.141 (0.013–0.268) | 0.032 |
| diacyl PCs, acyl-alkyl PCs I | 0.097 (0.015–0.178) | 0.021 | 0.013 (−0.153–0.179) | 0.877 | 0.037 (−0.077–0.151) | 0.523 |
| SMs | −0.035 (−0.101–0.032) | 0.308 | −0.057 (−0.198–0.084) | 0.430 | −0.137 (−0.252–−0.023) | 0.020 |
| lysoPC | 0.043 (0.004–0.081) | 0.030 | −0.029 (−0.120–0.061) | 0.525 | 0.075 (0.016–0.134) | 0.014 |
| diacyl PCs | 0.127 (0.084–0.170) | <0.001 | 0.082 (−0.015–0.178) | 0.100 | 0.119 (0.054–0.185) | <0.001 |
| diacyl PCs, acyl-alkyl PCs II | 0.153 (0.096–0.210) | <0.001 | 0.121 (0.005–0.238) | 0.043 | 0.109 (0.026–0.192) | 0.011 |
| Women 2 | ||||||
| acyl-alkyl, lysoPC | −0.068 (−0.150–0.014) | 0.104 | −0.127 (−0.361–0.107) | 0.289 | −0.184 (−0.362–−0.007) | 0.043 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Langenau, J.; Boeing, H.; Bergmann, M.M.; Nöthlings, U.; Oluwagbemigun, K. The Association between Alcohol Consumption and Serum Metabolites and the Modifying Effect of Smoking. Nutrients 2019, 11, 2331. https://doi.org/10.3390/nu11102331
Langenau J, Boeing H, Bergmann MM, Nöthlings U, Oluwagbemigun K. The Association between Alcohol Consumption and Serum Metabolites and the Modifying Effect of Smoking. Nutrients. 2019; 11(10):2331. https://doi.org/10.3390/nu11102331
Chicago/Turabian StyleLangenau, Julia, Heiner Boeing, Manuela M. Bergmann, Ute Nöthlings, and Kolade Oluwagbemigun. 2019. "The Association between Alcohol Consumption and Serum Metabolites and the Modifying Effect of Smoking" Nutrients 11, no. 10: 2331. https://doi.org/10.3390/nu11102331
APA StyleLangenau, J., Boeing, H., Bergmann, M. M., Nöthlings, U., & Oluwagbemigun, K. (2019). The Association between Alcohol Consumption and Serum Metabolites and the Modifying Effect of Smoking. Nutrients, 11(10), 2331. https://doi.org/10.3390/nu11102331





