Protective Effect of Spirulina platensis Extract against Dextran-Sulfate-Sodium-Induced Ulcerative Colitis in Rats
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Preparation of Spirulina platensis Extract
2.2.1. Sample Preparation for High-Performance Thin-Layer Chromatography (HPTLC) Analysis
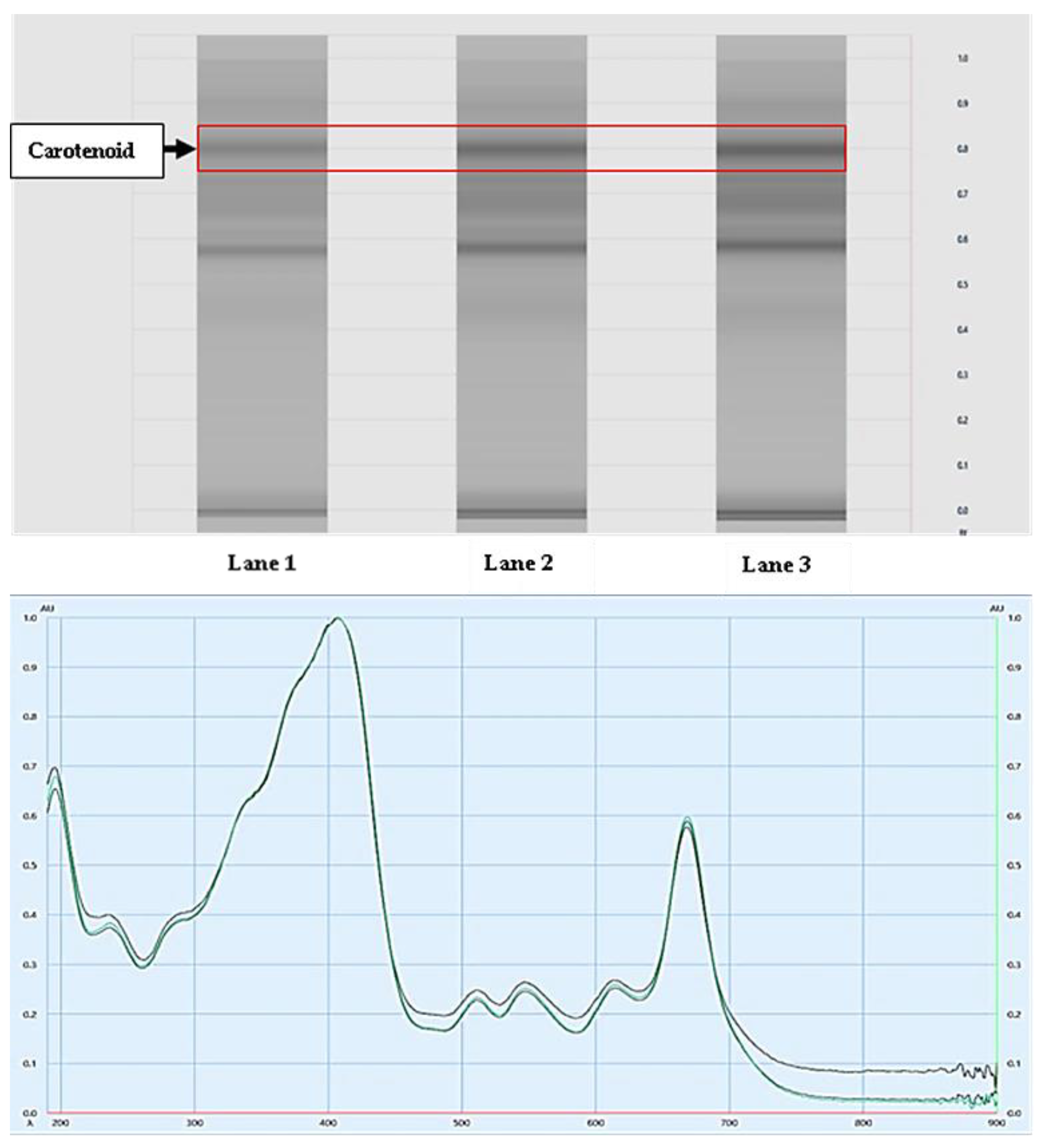
2.2.2. Analysis of Carotenoids Using HPTLC
2.2.3. Sample Preparation for Gas Chromatography–Mass Spectrometry (GC-MS)
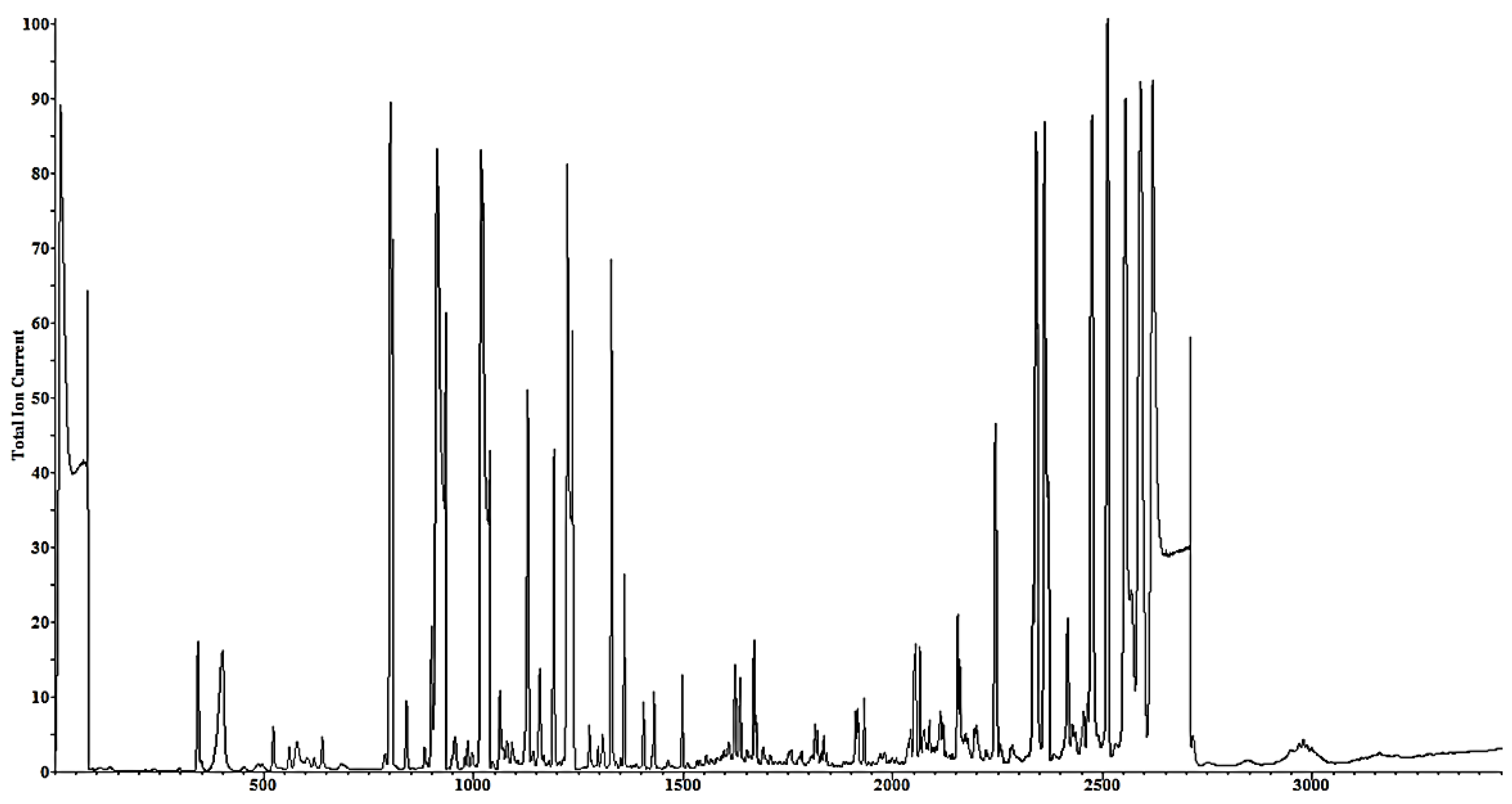
2.2.4. GC-MS Analysis
2.2.5. Metabolite Identification in GC-MS
2.3. Animals
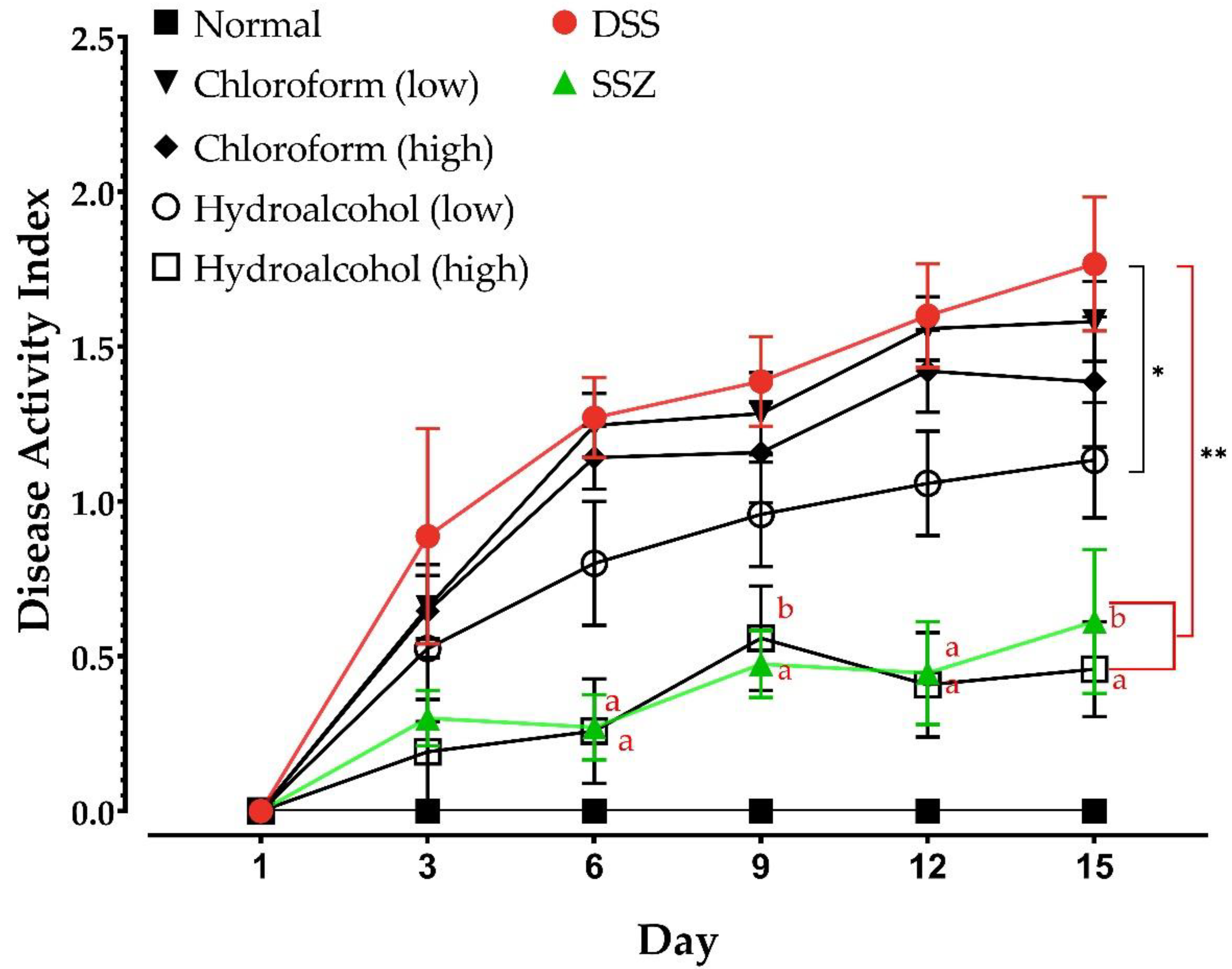
2.3.1. Monitoring of Animal Weight and Disease Activity Index (DAI)
2.3.2. Blood and Tissue Sample Collection
2.3.3. Measurement of Cytokine Levels
2.3.4. Measurement of MPO Activity
2.4. Statistical Analysis
3. Results
3.1. Active Ingredients and Metabolite Profile in the Spirulina platensis Extract
3.2. Effect of DSS and Various Treatments on Animal Body Weight and DAI Scores
3.3. Effect of DSS-Induced UC and Various Treatments on Blood Inflammatory Markers
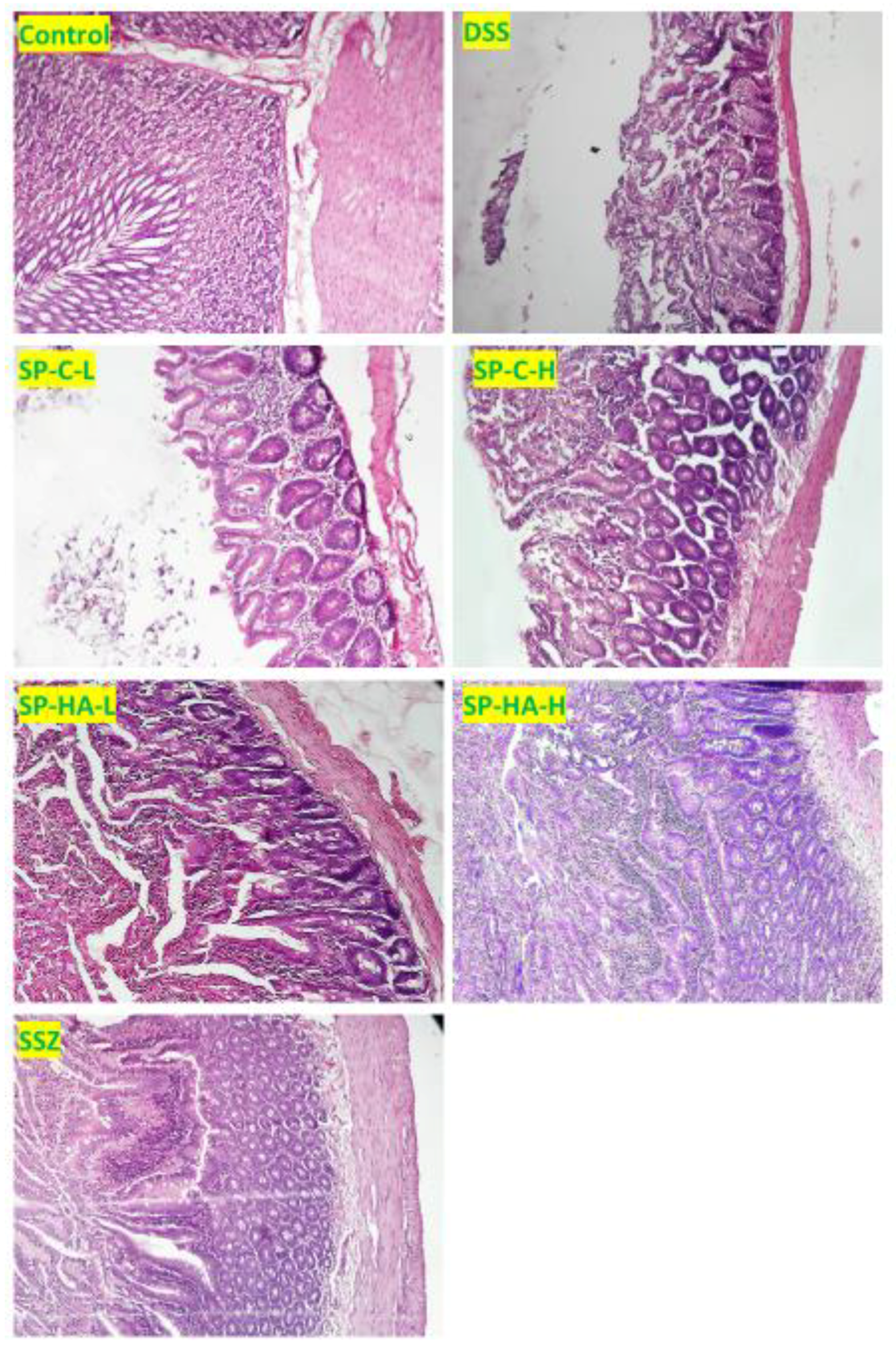
3.4. Effect of DSS and Various Treatments on Tissue Inflammation
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Beaugerie, L.; Itzkowitz, S.H. Cancers Complicating Inflammatory Bowel Disease. N. Engl. J. Med. 2015, 372, 1441–1452. [Google Scholar] [CrossRef] [PubMed]
- Ng, S.C.; Shi, H.Y.; Hamidi, N.; Underwood, F.E.; Tang, W.; Benchimol, E.I.; Panaccione, R.; Ghosh, S.; Wu, J.C.Y.; Chan, F.K.L.; et al. Worldwide incidence and prevalence of inflammatory bowel disease in the 21st century: A systematic review of population-based studies. Lancet 2017, 390, 2769–2778. [Google Scholar] [CrossRef]
- Liu, T.-C.; Stappenbeck, T.S. Genetics and pathogenesis of inflammatory bowel disease. Annu. Rev. Pathol. Mech. Dis. 2016, 11, 127–148. [Google Scholar] [CrossRef] [PubMed]
- Loddo, I.; Romano, C. Inflammatory Bowel Disease: Genetics, Epigenetics, and Pathogenesis. Front. Immunol. 2015, 6, 293. [Google Scholar] [CrossRef] [PubMed]
- Anderson, C.A.; Boucher, G.; Lees, C.W.; Franke, A.; D’Amato, M.; Taylor, K.D.; Lee, J.C.; Goyette, P.; Imielinski, M.; Latiano, A.; et al. Meta-analysis identifies 29 additional ulcerative colitis risk loci, increasing the number of confirmed associations to 47. Nat. Genet. 2011, 43, 246–252. [Google Scholar] [CrossRef] [PubMed]
- Jostins, L.; Ripke, S.; Weersma, R.K.; Duerr, R.H.; McGovern, D.P.; Hui, K.Y.; Lee, J.C.; Schumm, L.P.; Sharma, Y.; Anderson, C.A.; et al. Host-microbe interactions have shaped the genetic architecture of inflammatory bowel disease. Nature 2012, 491, 119–124. [Google Scholar] [CrossRef]
- Atreya, R.; Neurath, M.F. IBD pathogenesis in 2014: Molecular pathways controlling barrier function in IBD. Nat. Rev. Gastroenterol. Hepatol. 2015, 12, 67. [Google Scholar] [CrossRef]
- Mitsuyama, K.; Sasaki, E.; Toyonaga, A.; Ikeda, H.; Tsuruta, O.; Irie, A.; Arima, N.; Oriishi, T.; Harada, K.; Fujisaki, K.; et al. Colonic Mucosal Interleukin-6 in Inflammatory Bowel Disease. Digestion 1991, 50, 104–111. [Google Scholar] [CrossRef]
- Parisinos, C.A.; Serghiou, S.; Katsoulis, M.; George, M.J.; Patel, R.S.; Hemingway, H.; Hingorani, A.D. Variation in Interleukin 6 Receptor Gene Associates with Risk of Crohn’s Disease and Ulcerative Colitis. Gastroenterology 2018, 155, 303–306. [Google Scholar] [CrossRef]
- Fujii, M.; Nishida, A.; Imaeda, H.; Ohno, M.; Nishino, K.; Sakai, S.; Inatomi, O.; Bamba, S.; Kawahara, M.; Shimizu, T.; et al. Expression of Interleukin-26 is upregulated in inflammatory bowel disease. World J. Gastroenterol. 2017, 23, 5519–5529. [Google Scholar] [CrossRef]
- Zheng, C.; Huang, Y.; Hu, W.; Shi, J.; Ye, Z.; Qian, X.; Huang, Z.; Xue, A.; Wang, Y.; Lu, J.; et al. Phenotypic Characterization of Very Early-Onset Inflammatory Bowel Disease with Interleukin-10 Signaling Deficiency: Based on a Large Cohort Study. Inflamm. Bowel Dis. 2018, 25, 756–766. [Google Scholar] [CrossRef] [PubMed]
- Mohammed, K.; Shaik, N.A.; Al-Abbasi, F.A. Novel de Novo Mutations of the Interleukin-10 Receptor Gene Lead to Infantile Onset Inflammatory Bowel Disease: A Correction. J. Crohns Colitis 2017, 11, 1398–1399. [Google Scholar] [CrossRef] [PubMed]
- McDonald, B.S.; Narayanan, S.; Elawad, M.; Kiparissi, F.; Cullup, T.; Batta, K. Interleukin-10 receptor mutation presenting with severe nappy ulceration and infantile inflammatory bowel disease. Clin. Exp. Dermatol. 2017, 42, 771–773. [Google Scholar] [CrossRef] [PubMed]
- Ananthakrishnan, A.N.; Khalili, H.; Song, M.; Higuchi, L.M.; Richter, J.M.; Nimptsch, K.; Wu, K.; Chan, A.T. High School Diet and Risk of Crohn’s Disease and Ulcerative Colitis. Inflamm. Bowel Dis. 2015, 21, 2311–2319. [Google Scholar] [PubMed]
- Lewis, J.D.; Abreu, M.T. Diet as a Trigger or Therapy for Inflammatory Bowel Diseases. Gastroenterology 2017, 152, 398–414. [Google Scholar] [CrossRef] [PubMed]
- Durchschein, F.; Petritsch, W.; Hammer, H.F. Diet therapy for inflammatory bowel diseases: The established and the new. World J. Gastroenterol. 2016, 22, 2179–2194. [Google Scholar] [CrossRef]
- Forbes, A.; Escher, J.; Hébuterne, X.; Kłęk, S.; Krznaric, Z.; Schneider, S.; Shamir, R.; Stardelova, K.; Wierdsma, N.; Wiskin, A.E.; et al. ESPEN guideline: Clinical nutrition in inflammatory bowel disease. Clin. Nutr. 2017, 36, 321–347. [Google Scholar] [CrossRef] [PubMed]
- Elmaliklis, I.N.; Liveri, A.; Ntelis, B.; Paraskeva, K.; Goulis, I.; Koutelidakis, A.E. Increased Functional Foods’ Consumption and Mediterranean Diet Adherence May Have a Protective Effect in the Appearance of Gastrointestinal Diseases: A Case-Control Study. Medicines (Basel) 2019, 6, 50. [Google Scholar] [CrossRef] [PubMed]
- Andrade, L.; Andrade, C.; Dias, M.; Nascimento, C.; Mendes, M. Chlorella and Spirulina Microalgae as Sources of Functional Foods. MOJ Food Process Technol. 2018, 6(1), 45–58. [Google Scholar] [CrossRef]
- Yousefi, R.; Saidpour, A.; Mottaghi, A. The effects of Spirulina supplementation on metabolic syndrome components, its liver manifestation and related inflammatory markers: A systematic review. Complement. Ther. Med. 2019, 42, 137–144. [Google Scholar] [CrossRef]
- Masuda, K.; Chitundu, M. Multiple micronutrient supplementation using spirulina platensis and infant growth, morbidity, and motor development: Evidence from a randomized trial in Zambia. PLoS ONE 2019, 14, e0211693. [Google Scholar] [CrossRef] [PubMed]
- Masuda, K.; Chitundu, M. Multiple Micronutrient Supplementation Using Spirulina platensis during the First 1000 Days is Positively Associated with Development in Children under Five Years: A Follow up of A Randomized Trial in Zambia. Nutrients 2019, 11, 730. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, C.H.; Balachandran, P.; Pugh, N.D.; Sufka, K.J.; Walsted, A.; Schjørring-Thyssen, M.; Pasco, D.S.; Christensen, O.; Tamta, H.; Wu, X.; et al. Enhancement of Natural Killer Cell Activity in Healthy Subjects by Immulina®, a Spirulina Extract Enriched for Braun-Type Lipoproteins. Planta Medica 2010, 76, 1802–1808. [Google Scholar] [CrossRef] [PubMed]
- Martinez-Samano, J.; Torres-Montes de Oca, A.; Luqueno-Bocardo, O.I.; Torres-Duran, P.V.; Juarez-Oropeza, M.A. Spirulina maxima Decreases Endothelial Damage and Oxidative Stress Indicators in Patients with Systemic Arterial Hypertension: Results from Exploratory Controlled Clinical Trial. Mar. Drugs 2018, 16, 496. [Google Scholar] [CrossRef] [PubMed]
- Szulinska, M.; Gibas-Dorna, M.; Miller-Kasprzak, E.; Suliburska, J.; Miczke, A.; Walczak-Galezewska, M.; Stelmach-Mardas, M.; Walkowiak, J.; Bogdanski, P. Spirulina maxima improves insulin sensitivity, lipid profile, and total antioxidant status in obese patients with well-treated hypertension: A randomized double-blind placebo-controlled study. Eur. Rev. Med. Pharmacol. Sci. 2017, 21, 2473–2481. [Google Scholar]
- Hynstova, V.; Sterbova, D.; Klejdus, B.; Hedbavny, J.; Huska, D.; Adam, V. Separation, identification and quantification of carotenoids and chlorophylls in dietary supplements containing Chlorella vulgaris and Spirulina platensis using High Performance Thin Layer Chromatography. J. Pharm. Biomed. Anal. 2018, 148, 108–118. [Google Scholar] [CrossRef] [PubMed]
- Madhyastha, H.; Vatsala, T.; Madhyastha, H. Pigment production in Spirulina fussiformis in different photophysical conditions. Biomol. Eng. 2007, 24, 301–305. [Google Scholar] [CrossRef] [PubMed]
- Glabska, D.; Guzek, D.; Zakrzewska, P.; Wlodarek, D.; Lech, G. Lycopene, Lutein and Zeaxanthin May Reduce Faecal Blood, Mucus and Pus but not Abdominal Pain in Individuals with Ulcerative Colitis. Nutrients 2016, 8, 613. [Google Scholar] [CrossRef]
- Shirazi, K.M.; Nikniaz, Z.; Shirazi, A.M.; Rohani, M. Vitamin A supplementation decreases disease activity index in patients with ulcerative colitis: A randomized controlled clinical trial. Complement. Ther. Med. 2018, 41, 215–219. [Google Scholar] [CrossRef] [PubMed]
- Galal, M.K.; Elleithy, E.M.; Abdrabou, M.I.; Yasin, N.A.; Shaheen, Y.M. Modulation of caspase-3 gene expression and protective effects of garlic and spirulina against CNS neurotoxicity induced by lead exposure in male rats. NeuroToxicology 2019, 72, 15–28. [Google Scholar] [CrossRef]
- Mahmoud, Y.I.; El-Ghffar, E.A.A. Spirulina ameliorates aspirin-induced gastric ulcer in albino mice by alleviating oxidative stress and inflammation. Biomed. Pharmacother. 2019, 109, 314–321. [Google Scholar] [CrossRef] [PubMed]
- Khafaga, A.F.; El-Sayed, Y.S. Spirulina ameliorates methotrexate hepatotoxicity via antioxidant, immune stimulation, and proinflammatory cytokines and apoptotic proteins modulation. Life Sci. 2018, 196, 9–17. [Google Scholar] [CrossRef] [PubMed]
- Gargouri, M.; Hamed, H.; Akrouti, A.; Dauvergne, X.; Magne, C.; El Feki, A. Effects of Spirulina platensis on lipid peroxidation, antioxidant defenses, and tissue damage in kidney of alloxan-induced diabetic rats. Appl. Physiol. Nutr. Metab. 2018, 43, 345–354. [Google Scholar] [CrossRef] [PubMed]
- Nasirian, F.; Dadkhah, M.; Moradi-Kor, N.; Obeidavi, Z. Effects of Spirulina platensis microalgae on antioxidant and anti-inflammatory factors in diabetic rats. Diabetes Metab. Syndr. Obes. Targets Ther. 2018, 11, 375–380. [Google Scholar] [CrossRef] [PubMed]
- El-Tantawy, W.H. Antioxidant effects of Spirulina supplement against lead acetate-induced hepatic injury in rats. J. Tradit. Complement. Med. 2016, 6, 327–331. [Google Scholar] [CrossRef] [PubMed]
- Srividhya, S.; Jeyakumar, P. HPTLC quantification of β-carotene in papaya (Carica papaya L.) fruit treated with 1- methylcyclopropene (1-MCP). Int. J. Chem. Stud. 2017, 5, 486–489. [Google Scholar]
- Chassaing, B.; Aitken, J.D.; Malleshappa, M.; Vijay-Kumar, M. Dextran Sulfate Sodium (DSS)-Induced Colitis in Mice. Curr. Protoc. Immunol. 2014, 104. [Google Scholar] [CrossRef]
- Okayasu, I.; Hatakeyama, S.; Yamada, M.; Ohkusa, T.; Inagaki, Y.; Nakaya, R. A novel method in the induction of reliable experimental acute and chronic ulcerative colitis in mice. Gastroenterology 1990, 98, 694–702. [Google Scholar] [CrossRef]
- Naito, Y.; Takagi, T.; Kuroda, M.; Katada, K.; Ichikawa, H.; Kokura, S.; Yoshida, N.; Okanoue, T.; Yoshikawa, T. An orally active matrix metalloproteinase inhibitor, ONO-4817, reduces dextran sulfate sodium-induced colitis in mice. Inflamm. Res. 2004, 53, 462–468. [Google Scholar] [CrossRef]
- Senol, A.; Isler, M.; Sutcu, R.; Akin, M.; Cakir, E.; Ceyhan, B.M.; Kockar, M.C. Kefir treatment ameliorates dextran sulfate sodium-induced colitis in rats. World J. Gastroenterol. 2015, 21, 13020–13029. [Google Scholar] [CrossRef]
- Kreuter, J.; Mattson, B.; Wang, B.; You, Z.-B.; Hope, B.; Hope, B. Cocaine-induced Fos expression in rat striatum is blocked by chloral hydrate or urethane. Neuroscience 2004, 127, 233–242. [Google Scholar] [CrossRef] [PubMed]
- Ramachandran, R.; Hyun, E.; Zhao, L.; Lapointe, T.K.; Chapman, K.; Hirota, C.L.; Ghosh, S.; McKemy, D.D.; Vergnolle, N.; Beck, P.L.; et al. TRPM8 activation attenuates inflammatory responses in mouse models of colitis. Proc. Natl. Acad. Sci. USA 2013, 110, 7476. [Google Scholar] [CrossRef] [PubMed]
- Smith, P.; Krohn, R.; Hermanson, G.; Mallia, A.; Gartner, F.; Provenzano, M.; Fujimoto, E.; Goeke, N.; Olson, B.; Klenk, D. Measurement of protein using bicinchoninic acid. Anal. Biochem. 1985, 150, 76–85. [Google Scholar] [CrossRef]
- Nikfar, S.; Rahimi, R.; Rezaie, A.; Abdollahi, M. A meta-analysis of the efficacy of sulfasalazine in comparison with 5-aminosalicylates in the induction of improvement and maintenance of remission in patients with ulcerative colitis. Dig. Dis. Sci. 2009, 54, 1157–1170. [Google Scholar] [CrossRef] [PubMed]
- Eichele, D.D.; Kharbanda, K.K. Dextran sodium sulfate colitis murine model: An indispensable tool for advancing our understanding of inflammatory bowel diseases pathogenesis. World J. Gastroenterol. 2017, 23, 6016–6029. [Google Scholar] [CrossRef]
- Morgan, M.E.; Zheng, B.; Koelink, P.J.; Van De Kant, H.J.G.; Haazen, L.C.J.M.; Van Roest, M.; Garssen, J.; Folkerts, G.; Kraneveld, A.D. New Perspective on Dextran Sodium Sulfate Colitis: Antigen-Specific T Cell Development during Intestinal Inflammation. PLoS ONE 2013, 8, e69936. [Google Scholar] [CrossRef] [PubMed]
- Zhu, L.; Gu, P.; Shen, H. Protective effects of berberine hydrochloride on DSS-induced ulcerative colitis in rats. Int. Immunopharmacol. 2019, 68, 242–251. [Google Scholar] [CrossRef]
- Wirtz, S.; Neufert, C.; Weigmann, B.; Neurath, M.F. Chemically induced mouse models of intestinal inflammation. Nat. Protoc. 2007, 2, 541–546. [Google Scholar] [CrossRef]
- Van Der Logt, E.M.; Blokzijl, T.; Van Der Meer, R.; Faber, K.N.; Dijkstra, G. Westernized high-fat diet accelerates weight loss in dextran sulfate sodium-induced colitis in mice, which is further aggravated by supplementation of heme. J. Nutr. Biochem. 2013, 24, 1159–1165. [Google Scholar] [CrossRef]
- Zhang, Y.; Chen, D.; Wang, F.; Li, X.; Xue, X.; Jiang, M.; Xu, B.; Chu, Y.; Wang, W.; Wu, K.; et al. Comparison of the efficiency of different enemas on patients with distal ulcerative colitis. Cell Prolif. 2019, 52, e12559. [Google Scholar] [CrossRef]
- Cioffi, M.; De Rosa, A.; Serao, R.; Picone, I.; Vietri, M.T. Laboratory markers in ulcerative colitis: Current insights and future advances. World J. Gastrointest. Pathophysiol. 2015, 6, 13–22. [Google Scholar] [CrossRef]
- Göpfert, C.; Andreas, N.; Weber, F.; Häfner, N.; Yakovleva, T.; Gaestel, M.; Kamradt, T.; Drube, S. The p38-MK2/3 Module Is Critical for IL-33–Induced Signaling and Cytokine Production in Dendritic Cells. J. Immunol. 2018, 200, 1198–1206. [Google Scholar] [CrossRef] [PubMed]
- Delgado, M.E.; Brunner, T. The many faces of tumor necrosis factor signaling in the intestinal epithelium. Genes Immun. 2019, 1. [Google Scholar] [CrossRef] [PubMed]
- Kaser, A.; Blumberg, R.S. Paneth cells and inflammation dance together in Crohn’s disease. Cell Res. 2008, 18, 1160–1162. [Google Scholar] [CrossRef] [PubMed]
- Schmitt, M.; Schewe, M.; Sacchetti, A.; Feijtel, D.; Van De Geer, W.S.; Teeuwssen, M.; Sleddens, H.F.; Joosten, R.; Van Royen, M.E.; Van De Werken, H.J.; et al. Paneth Cells Respond to Inflammation and Contribute to Tissue Regeneration by Acquiring Stem-like Features through SCF/c-Kit Signaling. Cell Rep. 2018, 24, 2312–2328. [Google Scholar] [CrossRef]
- Natarajan, K.; Abraham, P.; Kota, R.; Isaac, B. NF-κB-iNOS-COX2-TNF α inflammatory signaling pathway plays an important role in methotrexate induced small intestinal injury in rats. Food Chem. Toxicol. 2018, 118, 766–783. [Google Scholar] [CrossRef]
- Souza, M.H.L.P.; Lemos, H.P.; Oliveira, R.B.; Cunha, F.Q. Gastric damage and granulocyte infiltration induced by indomethacin in tumour necrosis factor receptor 1 (TNF-R1) or inducible nitric oxide synthase (iNOS) deficient mice. Gut 2004, 53, 791–796. [Google Scholar] [CrossRef] [PubMed]
- Yamada, T.; Takahashi, S.; Masuda, K.; Ohara, H.; Nakazawa, T.; Sano, H.; Ando, T.; Nakamura, S.; Kobayashi, S.; Kuno, A.; et al. Kupffer-cell depletion attenuates colonic and extracolonic granulomatous inflammation in chronic colitis. J. Lab. Clin. Med. 2003, 142, 268–277. [Google Scholar] [CrossRef]
- Chen, J.; Lin, S.; Liu, C. Sulfasalazine for ankylosing spondylitis. Cochrane Database Syst. Rev. 2014, CD004800. [Google Scholar] [CrossRef]
- Plosker, G.L.; Croom, K.F. Sulfasalazine: A review of its use in the management of rheumatoid arthritis. Drugs 2005, 65, 1825–1849. [Google Scholar] [CrossRef]
- Tekeli, I.O.; Atessahin, A.; Sakin, F.; Aslan, A.; Çeribaşı, S.; Yipel, M. Protective effects of conventional and colon-targeted lycopene and linalool on ulcerative colitis induced by acetic acid in rats. Inflammopharmacology 2018, 27, 313–322. [Google Scholar] [CrossRef]
- Zhou, J.; Tan, L.; Xie, J.; Lai, Z.; Huang, Y.; Qu, C.; Luo, D.; Lin, Z.; Huang, P.; Su, Z.; et al. Characterization of brusatol self-microemulsifying drug delivery system and its therapeutic effect against dextran sodium sulfate-induced ulcerative colitis in mice. Drug Deliv. 2017, 24, 1667–1679. [Google Scholar] [CrossRef] [PubMed]
- El-Ashmawy, N.E.; Khedr, N.F.; El-Bahrawy, H.A.; El-Adawy, S.A. Downregulation of iNOS and elevation of cAMP mediate the anti-inflammatory effect of glabridin in rats with ulcerative colitis. Inflammopharmacology 2018, 26, 551–559. [Google Scholar] [CrossRef] [PubMed]
- Shabana, E.F.; Gabr, M.A.; Moussa, H.R.; El-Shaer, E.A.; Ismaiel, M.M. Biochemical composition and antioxidant activities of Arthrospira (Spirulina) platensis in response to gamma irradiation. Food Chem. 2017, 214, 550–555. [Google Scholar] [CrossRef] [PubMed]
- Bashandy, S.A.; El Awdan, S.A.; Ebaid, H.; Alhazza, I.M. Antioxidant Potential of Spirulina platensis Mitigates Oxidative Stress and Reprotoxicity Induced by Sodium Arsenite in Male Rats. Oxid. Med. Cell. Longev. 2016, 2016, 7174351. [Google Scholar] [CrossRef] [PubMed]
- Rosas, V.T.; Monserrat, J.M.; Bessonart, M.; Magnone, L.; Romano, L.A.; Tesser, M.B. Fish oil and meal replacement in mullet (Mugil liza) diet with Spirulina (Arthrospira platensis) and linseed oil. Comp. Biochem. Physiol. Part C Toxicol. Pharmacol. 2019, 218, 46–54. [Google Scholar] [CrossRef]
- Glabska, D.; Guzek, D.; Zakrzewska, P.; Lech, G. Intake of Lutein and Zeaxanthin as a Possible Factor Influencing Gastrointestinal Symptoms in Caucasian Individuals with Ulcerative Colitis in Remission Phase. J. Clin. Med. 2019, 8, 77. [Google Scholar] [CrossRef] [PubMed]
- Coskun, Z.K.; Kerem, M.; Gurbuz, N.; Omeroglu, S.; Pasaoglu, H.; Demirtas, C.; Lortlar, N.; Salman, B.; Pasaoglu, O.T.; Turgut, H.B. The study of biochemical and histopathological effects of spirulina in rats with TNBS-induced colitis. Bratisl. Lek. List. 2011, 112, 235–243. [Google Scholar]
- Elson, C.O.; Sartor, R.; Tennyson, G.S.; Riddell, R.H. Experimental models of inflammatory bowel disease. Gastroenterology 1995, 109, 1344–1367. [Google Scholar] [CrossRef]
- Randhawa, P.K.; Singh, K.; Singh, N.; Jaggi, A.S. A Review on Chemical-Induced Inflammatory Bowel Disease Models in Rodents. Korean J. Physiol. Pharmacol. 2014, 18, 279–288. [Google Scholar] [CrossRef]
- Abdel-Daim, M.M.; Farouk, S.M.; Madkour, F.F.; Azab, S.S. Anti-inflammatory and immunomodulatory effects ofSpirulina platensisin comparison toDunaliella salinain acetic acid-induced rat experimental colitis. Immunopharmacol. Immunotoxicol. 2015, 37, 126–139. [Google Scholar] [CrossRef]
- Guzmán-Gómez, O.; García-Rodríguez, R.V.; Quevedo-Corona, L.; Pérez-Pastén-Borja, R.; Rivero-Ramírez, N.L.; Ríos-Castro, E.; Pérez-Gutiérrez, S.; Perez-Ramos, J.; Chamorro-Cevallos, G.A. Amelioration of Ethanol-Induced Gastric Ulcers in Rats Pretreated with Phycobiliproteins of Arthrospira (Spirulina) Maxima. Nutrients 2018, 10, 763. [Google Scholar] [CrossRef] [PubMed]
- Fajardo, A.R.; Cerdán, L.E.; Medina, A.R.; Fernández, F.G.A.; Moreno, P.A.G.; Grima, E.M. Lipid extraction from the microalgaPhaeodactylum tricornutum. Eur. J. Lipid Sci. Technol. 2007, 109, 120–126. [Google Scholar] [CrossRef]
- Tavanandi, H.A.; Vanjari, P.; Raghavarao, K. Synergistic method for extraction of high purity Allophycocyanin from dry biomass of Arthrospira platensis and utilization of spent biomass for recovery of carotenoids. Sep. Purif. Technol. 2019, 225, 97–111. [Google Scholar] [CrossRef]
- Mitchell, G.V.; Grundel, E.; Jenkins, M.; Blakely, S.R. Effects of Graded Dietary Levels of Spirulina maxima on Vitamins A and E in Male Rats. J. Nutr. 1990, 120, 1235–1240. [Google Scholar] [CrossRef] [PubMed]
- Teimouri, M.; Amirkolaie, A.K.; Yeganeh, S. The effects of dietary supplement of Spirulina platensis on blood carotenoid concentration and fillet color stability in rainbow trout (Oncorhynchus mykiss). Aquaculture 2013, 414, 224–228. [Google Scholar] [CrossRef]
- Scoparo, C.T.; Borato, D.G.; Souza, L.M.; Dartora, N.; Silva, L.M.; Maria-Ferreira, D.; Sassaki, G.L.; Gorin, P.A.; Baggio, C.H.; Iacomini, M. Gastroprotective bio-guiding fractionation of hydro-alcoholic extracts from green- and black-teas (Camellia sinensis). Food Res. Int. 2014, 64, 577–586. [Google Scholar] [CrossRef] [PubMed]
- Ambrozova, J.V.; Mišurcová, L.; Vicha, R.; Machů, L.; Samek, D.; Baron, M.; Mlček, J.; Sochor, J.; Jurikova, T. Influence of Extractive Solvents on Lipid and Fatty Acids Content of Edible Freshwater Algal and Seaweed Products, the Green Microalga Chlorella kessleri and the Cyanobacterium Spirulina platensis. Molecules 2014, 19, 2344–2360. [Google Scholar] [CrossRef]
| Chemical Nature | Total Number |
|---|---|
| Amino acids | 4 |
| Secondary metabolites | 3 |
| Organic acids | 7 |
| Terpenes | 1 |
| Fatty acids | 5 |
| Others | 4 |
| Compound Name | RT | TMS Derivative | KEGG ID/Chem ID | Qualification Ions | |
|---|---|---|---|---|---|
| 1 | Methylamine | 7.973 | 2 TMS | C00218 | 175, 160 |
| 2 | 4-Hydroxybutanoic acid | 8.451 | 2 TMS | C01089 | 248, 247 |
| 3 | L-Alanine | 9.474 | 1 TMS | C00041 | 161, 146 |
| 4 | Ethylene glycol | 10.296 | 2 TMS | C01380 | 206, 191 |
| 5 | Dimethylglycine | 10.456 | 1 TMS | C01026 | 175, 160 |
| 6 | L-Valine | 11.705 | 2 TMS | C00183 | 261, 246 |
| 7 | Lactic acid | 11.806 | 2 TMS | C00186 | 234, 219 |
| 8 | Acetic acid | 12.142 | 2 TMS | C00033 | 220, 205 |
| 9 | Glycerol | 12.754 | 3 TMS | C00116 | 308, 293 |
| 10 | Succinic acid | 14.147 | 2 TMS | C00042 | 262, 247 |
| 11 | Glyceric acid | 14.558 | 3 TMS | C00258 | 322, 307 |
| 12 | Uracil | 14.801 | 2 TMS | C00106 | 256, 255 |
| 13 | L-Isoleucine | 15.799 | 2 TMS | C00407 | 275, 260 |
| 14 | Phenylacetic acid | 16.059 | 1 TMS | C07086 | 208, 193 |
| 15 | Butanoic acid | 16.881 | 3 TMS | C00246 | 336, 321 |
| 16 | Erythronic acid | 18.811 | 4 TMS | 2781043 | 424, 409 |
| 17 | Pyrogallol | 19.993 | 3 TMS | C01108 | 342, 327 |
| 18 | p-Hydroxyphenylacetic acid | 20.539 | 2 TMS | C00642 | 296, 281 |
| 19 | Myristic acid | 22.82 | 1 TMS | C06424 | 300, 385 |
| 20 | Palmitelaidic acid | 24.666 | 1 TMS | 5282745 | 326, 311 |
| 21 | Palmitic acid | 24.892 | 1 TMS | C00249 | 328, 313 |
| 22 | 10-Heptadecenoic acid | 25.689 | 1 TMS | C00249 | 340, 325 |
| 23 | Phytol | 26.167 | 1 TMS | C01389 | 368, 353 |
| 24 | Linoleic acid | 26.822 | 1 TMS | C01595 | 352, 337 |
| Day | Positive (DSS) | Normal | Standard (SSZ) | Chloroform (Low) | Chloroform (High) | HA (Low) | HA (High) |
|---|---|---|---|---|---|---|---|
| 1 | 220.33 ± 8.64 | 224.83 ± 10.26 | 217.50 ± 4.90 | 226.41 ± 14.67 | 231.52 ± 7.25 | 229.83 ± 8.62 | 226.54 ± 6.88 |
| 8 | 198.50 ± 7.69 c | 230.25 ± 7.08 a | 206.67 ± 7.03 | 208.00 ± 8.92 b | 220.13 ± 6.17 a | 220.83 ± 6.56 a | 228.83 ± 3.94 a |
| 15 | 174.00 ± 5.71 cd | 231.17 ± 5.83 a | 211.83 ± 8.42 a | 197.33 ± 8.18 ab | 215.33 ± 5.12 a | 222.50 ± 6.49 a | 229.96 ± 7.81 a |
| Parameter | Positive (DSS) | Normal Control | Standard (SSZ) | Chloroform (Low) | Chloroform (High) | HA (Low) | HA (High) |
|---|---|---|---|---|---|---|---|
| TNF-α (ng/mL) | 2.2 ± 0.118 # | 1.33 ± 0.124 | 1.14 ± 0.103 | 1.83 ± 0.115 *abc | 1.74 ± 0.154 *abc | 1.41 ± 0.136 | 1.16 ± 0.119 b |
| IL-6 (ng/mL) | 7.09 ± 0.187 # | 1.24 ± 0.116 | 1.79 ± 0.127 * | 5.20 ± 0.124 *ab | 3.11 ± 0.153 *ab | 2.07 ± 0.138 * | 1.68 ± 0.104 *b |
| ESR (mm/h) | 8.61 ± 0.93 | 2.71 ± 0.55 d | 5.04 ± 0.53 d | 5.79 ± 1.76 e | 5.55 ± 1.86 fg | 6.08 ± 1.47 eh | 3.72 ± 1.36 dg |
| Parameter | Positive (DSS) | Normal Control | Standard (SSZ) | Chloroform (Low) | Chloroform (High) | HA (Low) | HA (High) |
|---|---|---|---|---|---|---|---|
| MPO (ng/mg protein) | 40.50 ± 2.46 a | 25.23 ± 2.37 | 20.48 ± 2.68 # | 37.18 ± 2.62 ##@@ | 33.07 ± 2.88 ##@@ | 26.53 ± 1.92 @b | 24.00 ± 1.98 b |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Morsy, M.A.; Gupta, S.; Nair, A.B.; Venugopala, K.N.; Greish, K.; El-Daly, M. Protective Effect of Spirulina platensis Extract against Dextran-Sulfate-Sodium-Induced Ulcerative Colitis in Rats. Nutrients 2019, 11, 2309. https://doi.org/10.3390/nu11102309
Morsy MA, Gupta S, Nair AB, Venugopala KN, Greish K, El-Daly M. Protective Effect of Spirulina platensis Extract against Dextran-Sulfate-Sodium-Induced Ulcerative Colitis in Rats. Nutrients. 2019; 11(10):2309. https://doi.org/10.3390/nu11102309
Chicago/Turabian StyleMorsy, Mohamed A., Sumeet Gupta, Anroop B. Nair, Katharigatta N. Venugopala, Khaled Greish, and Mahmoud El-Daly. 2019. "Protective Effect of Spirulina platensis Extract against Dextran-Sulfate-Sodium-Induced Ulcerative Colitis in Rats" Nutrients 11, no. 10: 2309. https://doi.org/10.3390/nu11102309
APA StyleMorsy, M. A., Gupta, S., Nair, A. B., Venugopala, K. N., Greish, K., & El-Daly, M. (2019). Protective Effect of Spirulina platensis Extract against Dextran-Sulfate-Sodium-Induced Ulcerative Colitis in Rats. Nutrients, 11(10), 2309. https://doi.org/10.3390/nu11102309








