Dietary Fiber, Whole Grains, and Head and Neck Cancer Prognosis: Findings from a Prospective Cohort Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design and Recruitment
2.2. Measurements
2.2.1. Predictors: Dietary Intake of Fiber and Whole Grains
2.2.2. Covariates
2.2.3. Outcomes: Recurrence and Mortality
2.3. Statistical Analysis
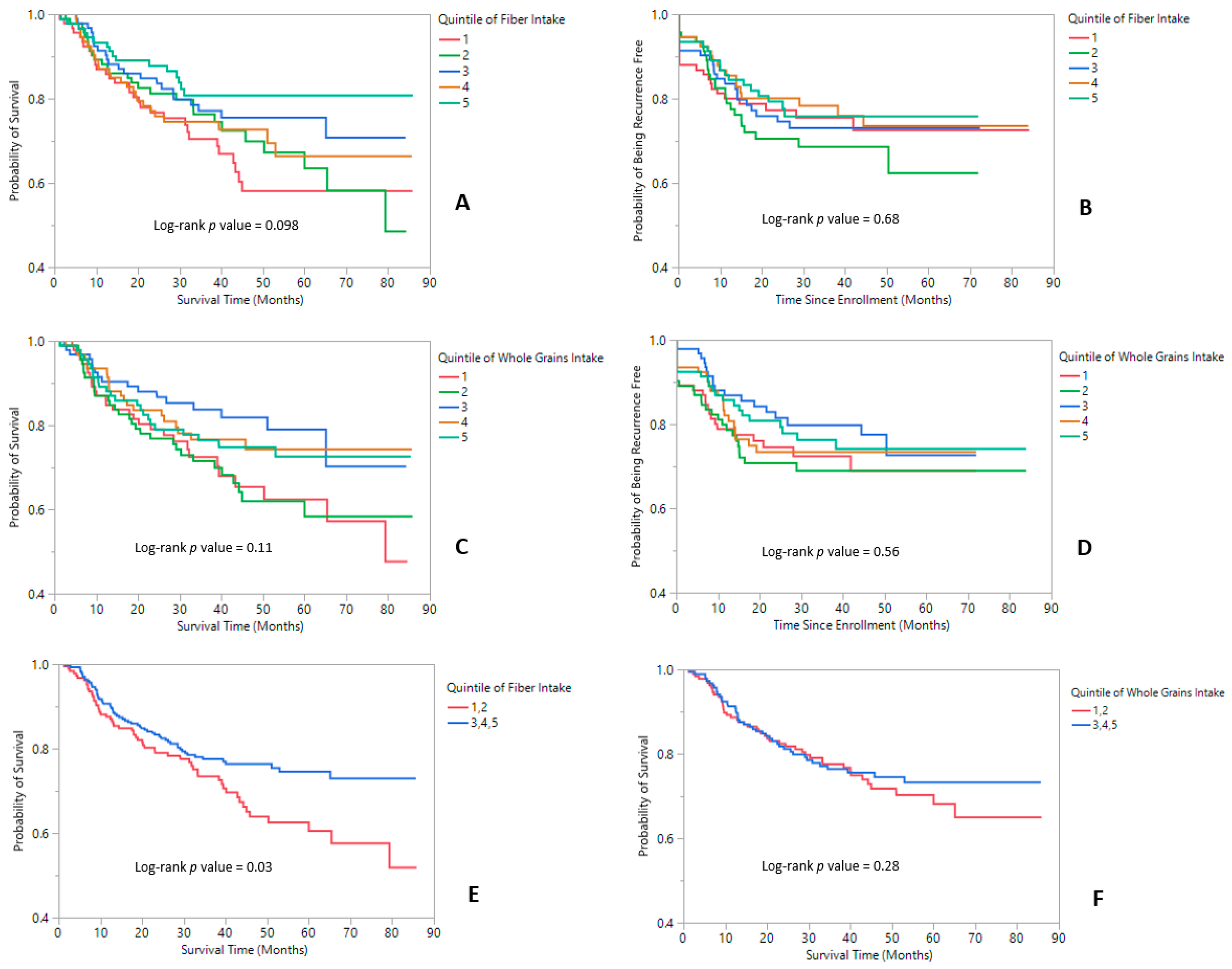
3. Results
3.1. Participant Characteristics
3.2. All-Cause and Cancer-Specific Mortality
3.3. Recurrence
3.4. Subgroup Analyses
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Argiris, A.; Karamouzis, M.V.; Raben, D.; Ferris, R.L. Head and neck cancer. Lancet Lond. Engl. 2008, 371, 1695–1709. [Google Scholar] [CrossRef]
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer statistics, 2019. CA Cancer J. Clin. 2019, 69, 7–34. [Google Scholar] [CrossRef] [PubMed]
- ACS. Cancer Facts & Figures 2019; American Cancer Society: Atlanta, GA, USA, 2019. [Google Scholar]
- Maino Vieytes, C.A.; Taha, H.M.; Burton-Obanla, A.A.; Douglas, K.G.; Arthur, A.E. Carbohydrate Nutrition and the Risk of Cancer. Curr. Nutr. Rep. 2019, 8, 230–239. [Google Scholar] [CrossRef] [PubMed]
- O’Keefe, S.J.D. Diet, microorganisms and their metabolites, and colon cancer. Nat. Rev. Gastroenterol. Hepatol. 2016, 13, 691–706. [Google Scholar] [CrossRef] [PubMed]
- Ray, A.; Alalem, M.; Ray, B.K. Insulin signaling network in cancer. Indian J. Biochem. Biophys. 2014, 51, 493–498. [Google Scholar]
- Probst-Hensch, N.M.; Wang, H.; Goh, V.H.H.; Seow, A.; Lee, H.-P.; Yu, M.C. Determinants of circulating insulin-like growth factor I and insulin-like growth factor binding protein 3 concentrations in a cohort of Singapore men and women. Cancer Epidemiol. Biomark. Prev. 2003, 12, 739–746. [Google Scholar]
- Ma, Y.; Hébert, J.R.; Li, W.; Bertone-Johnson, E.R.; Olendzki, B.; Pagoto, S.L.; Tinker, L.; Rosal, M.C.; Ockene, I.S.; Ockene, J.K.; et al. Association between dietary fiber and markers of systemic inflammation in the Women’s Health Initiative Observational Study. Nutrition 2008, 24, 941–949. [Google Scholar] [CrossRef]
- Jacobs, L.R. Relationship between Dietary Fiber and Cancer: Metabolic, Physiologic, and Cellular Mechanisms. Exp. Biol. Med. 1986, 183, 299–310. [Google Scholar] [CrossRef]
- Kawakita, D.; Lee, Y.-C.A.; Turati, F.; Parpinel, M.; Decarli, A.; Serraino, D.; Matsuo, K.; Olshan, A.F.; Zevallos, J.P.; Winn, D.M.; et al. Dietary fiber intake and head and neck cancer risk: A pooled analysis in the International Head and Neck Cancer Epidemiology consortium. Int. J. Cancer 2017, 141, 1811–1821. [Google Scholar] [CrossRef]
- Kawakita, D.; Lee, Y.-C.A.; Gren, L.H.; Buys, S.S.; La Vecchia, C.; Hashibe, M. Fiber intake and the risk of head and neck cancer in the prostate, lung, colorectal and ovarian (PLCO) cohort. Int. J. Cancer 2019, 145, 2342–2348. [Google Scholar] [CrossRef]
- De Vito, R.; Lee, Y.C.A.; Parpinel, M.; Serraino, D.; Olshan, A.F.; Zevallos, J.P.; Levi, F.; Zhang, Z.F.; Morgenstern, H.; Garavello, W.; et al. Shared and Study-specific Dietary Patterns and Head and Neck Cancer Risk in an International Consortium. Epidemiol. (Camb. MA) 2019, 30, 93–102. [Google Scholar] [CrossRef]
- Lam, T.K.; Cross, A.J.; Freedman, N.; Park, Y.; Hollenbeck, A.R.; Schatzkin, A.; Abnet, C. Dietary fiber and grain consumption in relation to head and neck cancer in the NIH-AARP Diet and Health Study. Cancer Causes Control CCC 2011, 22, 1405–1414. [Google Scholar] [CrossRef]
- Arthur, A.E.; Goss, A.M.; Demark-Wahnefried, W.; Mondul, A.M.; Fontaine, K.R.; Chen, Y.T.; Carroll, W.R.; Spencer, S.A.; Rogers, L.Q.; Rozek, L.S.; et al. Higher carbohydrate intake is associated with increased risk of all-cause and disease-specific mortality in head and neck cancer patients: Results from a prospective cohort study. Int. J. Cancer 2018, 143, 1105–1113. [Google Scholar] [CrossRef]
- Willett, W.C.; Sampson, L.; Browne, M.L.; Stampfer, M.J.; Rosner, B.; Hennekens, C.H.; Speizer, F.E. The use of a self-administered questionnaire to assess diet four years in the past. Am. J. Epidemiol. 1988, 127, 188–199. [Google Scholar] [CrossRef]
- Rimm, E.B.; Giovannucci, E.L.; Stampfer, M.J.; Colditz, G.A.; Litin, L.B.; Willett, W.C. Reproducibility and validity of an expanded self-administered semiquantitative food frequency questionnaire among male health professionals. Am. J. Epidemiol. 1992, 135, 1114–1126, discussion 1127–1136. [Google Scholar] [CrossRef]
- McNutt, S.; Zimmerman, T.P.; Hull, S.G. Development of food composition databases for food frequency questionnaires (FFQ). J. Food Compos. Anal. 2008, 21, S20–S26. [Google Scholar] [CrossRef]
- DeVries, J.W.; Rader, J.I. Historical perspective as a guide for identifying and developing applicable methods for dietary fiber. J. AOAC Int. 2005, 88, 1349–1366. [Google Scholar]
- Wu, H.; Flint, A.J.; Qi, Q.; van Dam, R.M.; Sampson, L.A.; Rimm, E.B.; Holmes, M.D.; Willett, W.C.; Hu, F.B.; Sun, Q. Association between dietary whole grain intake and risk of mortality: Two large prospective studies in US men and women. JAMA Intern. Med. 2015, 175, 373–384. [Google Scholar] [CrossRef]
- Murphy, N.; Norat, T.; Ferrari, P.; Jenab, M.; Bueno-de-Mesquita, B.; Skeie, G.; Dahm, C.C.; Overvad, K.; Olsen, A.; Tjønneland, A.; et al. Dietary fibre intake and risks of cancers of the colon and rectum in the European prospective investigation into cancer and nutrition (EPIC). PLoS ONE 2012, 7, e39361. [Google Scholar] [CrossRef]
- Willett, W.C.; Howe, G.R.; Kushi, L.H. Adjustment for total energy intake in epidemiologic studies. Am. J. Clin. Nutr. 1997, 65, 1220S–1228S, discussion 1229S–1231S. [Google Scholar] [CrossRef]
- Rosner, B. Percentage Points for a Generalized ESD Many-Outlier Procedure. Technometrics 1983, 25, 165–172. [Google Scholar] [CrossRef]
- Crowder, S.L.; Douglas, K.G.; Yanina Pepino, M.; Sarma, K.P.; Arthur, A.E. Nutrition impact symptoms and associated outcomes in post-chemoradiotherapy head and neck cancer survivors: A systematic review. J. Cancer Surviv. Res. Pract. 2018, 12, 479–494. [Google Scholar] [CrossRef]
- Olmos, A.; Priyalatha, G. A practical guide for using propensity score weighting in R. Pract. Assess. Res. Eval. 2015, 20. [Google Scholar]
- Austin, P.C. An Introduction to Propensity Score Methods for Reducing the Effects of Confounding in Observational Studies. Multivar. Behav. Res. 2011, 46, 399–424. [Google Scholar] [CrossRef]
- DuGoff, E.H.; Schuler, M.; Stuart, E.A. Generalizing Observational Study Results: Applying Propensity Score Methods to Complex Surveys. Health Serv. Res. 2014, 49, 284–303. [Google Scholar] [CrossRef]
- Li, R.; Hertzmark, E.; Louie, M.; Chen, L.; Spiegelman, D. The SAS LGTPHCURV9 Macro; Channing Laboratory: Boston, MA, USA, 2011. [Google Scholar]
- Maslowski, K.M.; Vieira, A.T.; Ng, A.; Kranich, J.; Sierro, F.; Yu, D.; Schilter, H.C.; Rolph, M.S.; Mackay, F.; Artis, D.; et al. Regulation of inflammatory responses by gut microbiota and chemoattractant receptor GPR43. Nature 2009, 461, 1282–1286. [Google Scholar] [CrossRef]
- Vinolo, M.A.R.; Rodrigues, H.G.; Nachbar, R.T.; Curi, R. Regulation of inflammation by short chain fatty acids. Nutrients 2011, 3, 858–876. [Google Scholar] [CrossRef]
- Cummings, J.H.; Pomare, E.W.; Branch, W.J.; Naylor, C.P.; Macfarlane, G.T. Short chain fatty acids in human large intestine, portal, hepatic and venous blood. Gut 1987, 28, 1221–1227. [Google Scholar] [CrossRef]
- McLoughlin, R.F.; Berthon, B.S.; Jensen, M.E.; Baines, K.J.; Wood, L.G. Short-chain fatty acids, prebiotics, synbiotics, and systemic inflammation: A systematic review and meta-analysis. Am. J. Clin. Nutr. 2017, 106, 930–945. [Google Scholar] [CrossRef]
- McNabney, S.M.; Henagan, T.M. Short Chain Fatty Acids in the Colon and Peripheral Tissues: A Focus on Butyrate, Colon Cancer, Obesity and Insulin Resistance. Nutrients 2017, 9, 1348. [Google Scholar] [CrossRef]
- Limesand, K.H.; Chibly, A.M.; Fribley, A. Impact of targeting insulin-like growth factor signaling in head and neck cancers. Growth Horm. IGF Res. 2013, 23, 135–140. [Google Scholar] [CrossRef] [PubMed]
- Wu, X. Serum Levels of Insulin Growth Factor (IGF-I) and IGF-Binding Protein Predict Risk of Second Primary Tumors in Patients with Head and Neck Cancer. Clin. Cancer Res. 2004, 10, 3988–3995. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Canfora, E.E.; Jocken, J.W.; Blaak, E.E. Short-chain fatty acids in control of body weight and insulin sensitivity. Nat. Rev. Endocrinol. 2015, 11, 577–591. [Google Scholar] [CrossRef] [PubMed]
- Liese, A.D.; Schulz, M.; Fang, F.; Wolever, T.M.S.; D’Agostino, R.B.; Sparks, K.C.; Mayer-Davis, E.J. Dietary glycemic index and glycemic load, carbohydrate and fiber intake, and measures of insulin sensitivity, secretion, and adiposity in the Insulin Resistance Atherosclerosis Study. Diabetes Care 2005, 28, 2832–2838. [Google Scholar] [CrossRef] [PubMed]
- Lau, C.; Faerch, K.; Glümer, C.; Tetens, I.; Pedersen, O.; Carstensen, B.; Jørgensen, T.; Borch-Johnsen, K. Inter99 study Dietary glycemic index, glycemic load, fiber, simple sugars, and insulin resistance: The Inter99 study. Diabetes Care 2005, 28, 1397–1403. [Google Scholar] [CrossRef]
- Thompson, S.V.; Hannon, B.A.; An, R.; Holscher, H.D. Effects of isolated soluble fiber supplementation on body weight, glycemia, and insulinemia in adults with overweight and obesity: A systematic review and meta-analysis of randomized controlled trials. Am. J. Clin. Nutr. 2017, 106, 1514–1528. [Google Scholar] [CrossRef]
- Rischin, D.; Young, R.J.; Fisher, R.; Fox, S.B.; Le, Q.-T.; Peters, L.J.; Solomon, B.; Choi, J.; O’Sullivan, B.; Kenny, L.M.; et al. Prognostic significance of p16INK4A and human papillomavirus in patients with oropharyngeal cancer treated on TROG 02.02 phase III trial. J. Clin. Oncol. 2010, 28, 4142–4148. [Google Scholar] [CrossRef]
- Ang, K.K.; Harris, J.; Wheeler, R.; Weber, R.; Rosenthal, D.I.; Nguyen-Tân, P.F.; Westra, W.H.; Chung, C.H.; Jordan, R.C.; Lu, C.; et al. Human papillomavirus and survival of patients with oropharyngeal cancer. N. Engl. J. Med. 2010, 363, 24–35. [Google Scholar] [CrossRef]
- Posner, M.R.; Lorch, J.H.; Goloubeva, O.; Tan, M.; Schumaker, L.M.; Sarlis, N.J.; Haddad, R.I.; Cullen, K.J. Survival and human papillomavirus in oropharynx cancer in TAX 324: A subset analysis from an international phase III trial. Ann. Oncol. 2011, 22, 1071–1077. [Google Scholar] [CrossRef]
- Crowder, S.L.; Sarma, K.P.; Mondul, A.M.; Chen, Y.T.; Li, Z.; Pepino, M.Y.; Zarins, K.R.; Wolf, G.; Rozek, L.S.; Arthur, A.E. Pre-treatment dietary patterns are associated with the presence of nutrition impact symptoms 1-year after diagnosis in patients with head and neck cancer. Cancer Epidemiol. Biomak. Prev. 2019. [Google Scholar] [CrossRef]
- Kalantar-Zadeh, K.; Kopple, J.D.; Deepak, S.; Block, D.; Block, G. Food intake characteristics of hemodialysis patients as obtained by food frequency questionnaire. J. Ren. Nutr. 2002, 12, 17–31. [Google Scholar] [CrossRef]
- Grooms, K.N.; Ommerborn, M.J.; Pham, D.Q.; Djoussé, L.; Clark, C.R. Dietary Fiber Intake and Cardiometabolic Risks among US Adults, NHANES 1999-2010. Am. J. Med. 2013, 126, 1059–1067.e4. [Google Scholar] [CrossRef]
- McGill, C.; Iii, V.; Devareddy, L. Ten-Year Trends in Fiber and Whole Grain Intakes and Food Sources for the United States Population: National Health and Nutrition Examination Survey 2001–2010. Nutrients 2015, 7, 1119–1130. [Google Scholar] [CrossRef]
| Characteristic | Survivors # (%) |
|---|---|
| Age (year) | |
| Mean ± SD | 61.1 ± 11.3 |
| Min/Max | 25/95 |
| Sex b | |
| Male | 344 (74.6) |
| Female | 117 (25.4) |
| Education c | |
| High school or less | 160 (34.8) |
| Some college or more | 300 (65.2) |
| Race d | |
| Non-Hispanic white | 434 (94.8) |
| Other | 24 (5.2) |
| Body Mass Index (BMI) (kg/m2) | |
| Underweight (<18.5) | 20 (4.1) |
| Normal weight (18.5–24.9) | 140 (30.2) |
| Overweight (25–29.9) | 177 (38.2) |
| Obese (30+) | 126 (27.2) |
| Site a | |
| Oral cavity | 173 (37.5) |
| Oropharynx | 184 (39.8) |
| Hypopharynx | 11 (2.4) |
| Larynx | 94 (20.4) |
| Stage | |
| 0, I, II | 145 (31.3) |
| III, IV | 318 (68.7) |
| HPV status a | |
| HPV-negative | 150 (32.5) |
| HPV-positive | 73 (15.8) |
| Unknown | 239 (51.7) |
| Treatment a | |
| Surgery only | 116 (25.1) |
| Radiation only | 34 (7.4) |
| Surgery + adjuvant radiation or chemo | 83 (18.0) |
| Chemotherapy + radiation | 186 (40.3) |
| Chemotherapy only | 14 (3.0) |
| Palliative or unknown | 29 (6.3) |
| Smoking Status a | |
| Current | 168 (36.3) |
| Former | 162 (35.1) |
| Never | 132 (28.6) |
| Drinking status a | |
| Current | 319 (69.1) |
| Former | 108 (23.4) |
| Never | 35 (7.6) |
| Fiber Intake Quintile (g/day) | Q1 12.94 | Q2 12.94–15.87 | Q3 15.90–19.00 | Q4 19.05–22.91 | Q5 >22.91 |
| Mean fiber intake (g) | 10.5 | 14.6 | 17.4 | 20.8 | 27.6 |
| Age | 57.65 | 60.72 | 60.58 | 62.62 | 63.76 |
| Females (%) | 16 (17.4) | 18 (19.4) | 26 (28.0 | 27 (29.0) | 30 (32.3) |
| Some college or more (%) | 51 (56.0) | 55 (59.1) | 55 (60.4) | 66 (71.0) | 73 (79.3) |
| Stages III, IV (%) | 65 (70.9) | 66 (71.0) | 66 (71.0) | 63 (67.7) | 58 (63.0) |
| Current smoker (%) | 49 (53.3) | 41 (44.1) | 38 (40.9) | 26 (28.3) | 14 (15.2) |
| Current drinker (%) | 72 (78.3) | 61 (65.6) | 69 (74.2) | 55 (59.8) | 62 (67.4) |
| Body Mass Index (kg/m2) | 26.5 | 28.5 | 26.8 | 28.5 | 27.7 |
| Total caloric intake (kcal) | 1926.9 | 1971.3 | 1936.6 | 1947.4 | 1940.9 |
| Glycemic load | 116.1 | 118.4 | 119.6 | 127.9 | 125.0 |
| Fruit/vegetable consumption (servings/day) | 1.7 | 2.6 | 3.2 | 4.3 | 6.3 |
| Total fat consumption (g) | 68.8 | 76.9 | 76.1 | 71.0 | 67.4 |
| Whole grain intake quintile (g/day) | 1 13.70 | 2 13.71–23.41 | 3 23.43–32.95 | 4 32.96–44.29 | 5 >44.29 |
| Mean whole grain intake (g) | 8.5 | 18.5 | 27.9 | 38.2 | 61.1 |
| Age | 60.15 | 62.44 | 59.02 | 61.26 | 62.31 |
| Females (%) | 18 (19.6) | 28 (30.4) | 21 (22.6) | 25 (27.2) | 24 (26.1) |
| Some college or more (%) | 43 (47.3) | 55 (60.4) | 62 (67.4) | 66 (71.7) | 72 (78.3) |
| Stages III, IV (%) | 58 (63.0) | 64 (69.6) | 71 (76.3) | 67 (72.8) | 81 (88.0) |
| Current smoker (%) | 48 (52.2) | 37 (40.2) | 32 (34.4) | 29 (31.9) | 22 (23.9) |
| Current drinker (%) | 66 (71.7) | 60 (65.2) | 70 (75.3) | 65 (71.4) | 58 (63.0) |
| Body Mass Index (kg/m2) | 26.6 | 27.1 | 28.0 | 28.3 | 28.0 |
| Total caloric intake (kcal) | 2005.0 | 1826.3 | 2011.1 | 2022.3 | 1884.7 |
| Glycemic load | 114.1 | 120.0 | 120.9 | 123.1 | 132.6 |
| Fruit/vegetable consumption (servings/day) | 2.7 | 3.4 | 3.8 | 4.1 | 4.3 |
| Total fat consumption (g) | 74.0 | 74.9 | 71.0 | 72.5 | 68.4 |
| Fiber Intake Quintile and Range (g/day) | 1 12.94 | 2 12.94–15.87 | 3 15.90–19.00 | 4 19.05–22.91 | 5 >22.91 | ptrend4 |
| All-cause mortality | ||||||
| 1 Model 1 | Referent | 0.65 (0.39–1.10) | 0.52 (0.30–0.90) * | 0.66 (0.39–1.12) | 0.34 (0.18–0.63) *** | 0.002 ** |
| 2 Model 2 | Referent | 0.79 (0.46–1.35) | 0.59 (0.34–1.05) | 0.76 (0.45–1.31) | 0.41 (0.21–0.78) ** | 0.014 * |
| 3 Model 3 | Referent | 0.83 (0.43–1.59) | 0.63 (0.32–1.25) | 0.68 (0.30–1.52) | 0.37 (0.14–0.95) * | 0.04 * |
| Fiber intake quintile and range (g/day) | 1 13.21 | 2 13.41–16.03 | 3 16.04–19.11 | 4 19.22–23.17 | 5 >23.20 | ptrend4 |
| Cancer-specific mortality | ||||||
| 1 Model 1 | Referent | 0.84 (0.43–1.66) | 0.73 (0.37–1.46) | 0.76 (0.38–1.52) | 0.48 (0.22–1.03) | 0.06 |
| 2 Model 2 | Referent | 1.01 (0.51–2.01) | 0.79 (0.39–1.63) | 0.83 (0.41–1.69) | 0.63 (0.28–1.40) | 0.22 |
| 3 Model 3 | Referent | 1.10 (0.48–2.51) | 0.80 (0.33–1.94) | 0.68 (0.24–1.93) | 0.46 (0.14–1.52) | 0.14 |
| Whole grain intake quintile and range (g/day) | 1 13.70 | 2 13.71–23.41 | 3 23.43–32.95 | 4 32.96–44.29 | 5 >44.29 | ptrend4 |
| All-cause mortality | ||||||
| 1 Model 1 | Referent | 0.96 (0.58–1.60) | 0.55 (0.30–1.00) * | 0.66 (0.37–1.15) | 0.65 (0.38–1.13) | 0.07 |
| 2 Model 2 | Referent | 0.88 (0.53–1.47) | 0.60 (0.33–1.10) | 0.71 (0.40–1.25) | 0.65 (0.37–1.15) | 0.12 |
| 3 Model 3 | Referent | 0.85 (0.50–1.46) | 0.63 (0.33–1.20) | 0.89 (0.47–1.68) | 0.64 (0.34–1.24) | 0.24 |
| Whole grain intake quintile and range (g/day) | 1 14.12 | 2 14.23–23.97 | 3 24.10–33.16 | 4 33.16–44.42 | 5 >44.60 | ptrend4 |
| Cancer-specific mortality | ||||||
| 1 Model 1 | Referent | 1.16 (0.59–2.28) | 0.84 (0.40–1.77) | 0.91 (0.45–1.88) | 0.80 (0.39–1.66) | 0.08 |
| 2 Model 2 | Referent | 1.12 (0.57–2.20) | 0.95 (0.45–2.03) | 0.97 (0.47–2.00) | 0.87 (0.41–1.87) | 0.24 |
| 3 Model 3 | Referent | 1.17 (0.57–2.39) | 0.92 (0.41–2.07) | 1.22 (0.54–2.75) | 0.83 (0.35–1.95) | 0.18 |
| Fiber Intake Quintile and Range (g/day) | 1 12.94 | 2 12.94–15.87 | 3 15.90–19.00 | 4 19.05–22.91 | 5 >22.91 | ptrend2 |
|---|---|---|---|---|---|---|
| All-cause mortality | ||||||
| 1 Model 3 | Referent | 1.16 (0.72–1.85) | 0.83 (0.52–1.33) | 0.54 (0.31–0.95) * | 0.22 (0.10–0.48) *** | <0.0001 *** |
| Fiber Intake Quintile and Range (g/day) | 1 12.94 | 2 12.94–15.87 | 3 15.90–19.00 | 4 19.05–22.91 | 5 >22.91 | ptrend4 |
| Recurrence | ||||||
| 1 Model 1 | Referent | 1.07 (0.60–1.88) | 0.87 (0.48–1.57) | 0.74 (0.40–1.36) | 0.69 (0.37–1.28) | 0.10 |
| 2 Model 2 | Referent | 1.31 (0.74–2.34) | 0.97 (0.53–1.77) | 0.85 (0.45–1.58) | 0.93 (0.49–1.78) | 0.43 |
| 3 Model 3 | Referent | 1.42 (0.73–2.75) | 0.98 (0.49–1.98) | 0.74 (0.32–1.73) | 0.77 (0.30–1.97) | 0.33 |
| Whole grain intake quintile and range (g/day) | 1 13.70 | 2 13.71–23.41 | 3 23.43–32.95 | 4 32.96-44.29 | 5 >44.29 | ptrend4 |
| Recurrence | ||||||
| 1 Model 1 | Referent | 1.00 (0.58–1.75) | 0.70 (0.38–1.27) | 0.70 (0.38–1.27) | 0.72 (0.40–1.30) | 0.22 |
| 2 Model 2 | Referent | 0.96 (0.55–1.67) | 0.72 (0.39–1.33) | 0.87 (0.48–1.56) | 0.81 (0.44–1.48) | 0.53 |
| 3 Model 3 | Referent | 1.06 (0.59–1.92) | 0.77 (0.40–1.50) | 1.06 (0.56–2.04) | 0.76 (0.38–1.50) | 0.42 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Maino Vieytes, C.A.; Mondul, A.M.; Li, Z.; Zarins, K.R.; Wolf, G.T.; Rozek, L.S.; Arthur, A.E. Dietary Fiber, Whole Grains, and Head and Neck Cancer Prognosis: Findings from a Prospective Cohort Study. Nutrients 2019, 11, 2304. https://doi.org/10.3390/nu11102304
Maino Vieytes CA, Mondul AM, Li Z, Zarins KR, Wolf GT, Rozek LS, Arthur AE. Dietary Fiber, Whole Grains, and Head and Neck Cancer Prognosis: Findings from a Prospective Cohort Study. Nutrients. 2019; 11(10):2304. https://doi.org/10.3390/nu11102304
Chicago/Turabian StyleMaino Vieytes, Christian A., Alison M. Mondul, Zonggui Li, Katie R. Zarins, Gregory T. Wolf, Laura S. Rozek, and Anna E. Arthur. 2019. "Dietary Fiber, Whole Grains, and Head and Neck Cancer Prognosis: Findings from a Prospective Cohort Study" Nutrients 11, no. 10: 2304. https://doi.org/10.3390/nu11102304
APA StyleMaino Vieytes, C. A., Mondul, A. M., Li, Z., Zarins, K. R., Wolf, G. T., Rozek, L. S., & Arthur, A. E. (2019). Dietary Fiber, Whole Grains, and Head and Neck Cancer Prognosis: Findings from a Prospective Cohort Study. Nutrients, 11(10), 2304. https://doi.org/10.3390/nu11102304





