1. Introduction
Obesity is a major public health issue with approximately 60% of adults and nearly 30% of children aged 2–15 years in the UK being overweight or obese [
1]. There is a paucity of strategies proven to be effective for preventing, or treating, overweight or obesity. Fast eating is associated with excess body weight [
2] and reducing rate of eating appears to reduce energy intake [
3]. These systematic reviews have highlighted that further work is needed to ascertain the mechanism underlying effects of eating rate on weight control and whether eating rate influences self-reported appetite [
4]. For example, a recent study found that participants who consumed a test meal more slowly reported a greater increase in fullness, yet with reduced enjoyment and satisfaction from the meal [
5].
Research investigating the physiological basis of the effect of eating rate on satiety has yielded inconsistent results for gastrointestinal (GI) hormone responses. One study utilising a cross-over design (
N = 17) found greater post-prandial peptide YY (PYY) response after slowing eating rate (30 min vs. 5 min meal) however no effect was seen on ghrelin suppression [
6]. Similar findings were elicited by another study examining the effects of eating rate and eating density on GI hormone responses (
N = 20), where slow eating rate (20 g/min vs. 80 g/min) led to greater PYY response but no effect on ghrelin suppression [
7]. By contrast, one cross-over study (
N = 25), found no effect of eating rate by comparing 7 min, 14 min and 28 min meals on PYY release post-prandially [
8]. A study of overweight adolescents (
N = 27) utilising a Mandometer (device providing real-time feedback about consumption of meal by weight of plate) to slow eating rate vs. control, found slowing eating rate led to increased PYY response and greater ghrelin suppression post-prandially [
9]. Thus, there is no current consensus regarding the effect of eating rate on satiety hormone response and further research is needed.
Other work has explored whether the mechanism underlying differences in food intake following different eating rates may relate to enhancing or disrupting memory for recent eating episodes [
10,
11]. There is accumulating evidence that memory for recent eating plays a pivotal role in the control of energy intake [
12,
13,
14]. A recent study (
N = 40) found that participants who consumed a meal more slowly remembered eating a larger portion but eating more slowly was not found to affect self-reported memory vividness [
10], as was found in another study [
11]. Memory for recent eating is assumed to involve brain regions classically associated with memory, such as the hippocampus [
15,
16]. More recent functional magnetic resonance imaging (fMRI) studies utilising a “What, Where, When” paradigm of episodic memory [
17,
18] suggest that, in addition to the hippocampus and other medial temporal lobe regions, frontal and parietal brain regions are also important for memory-related processes.
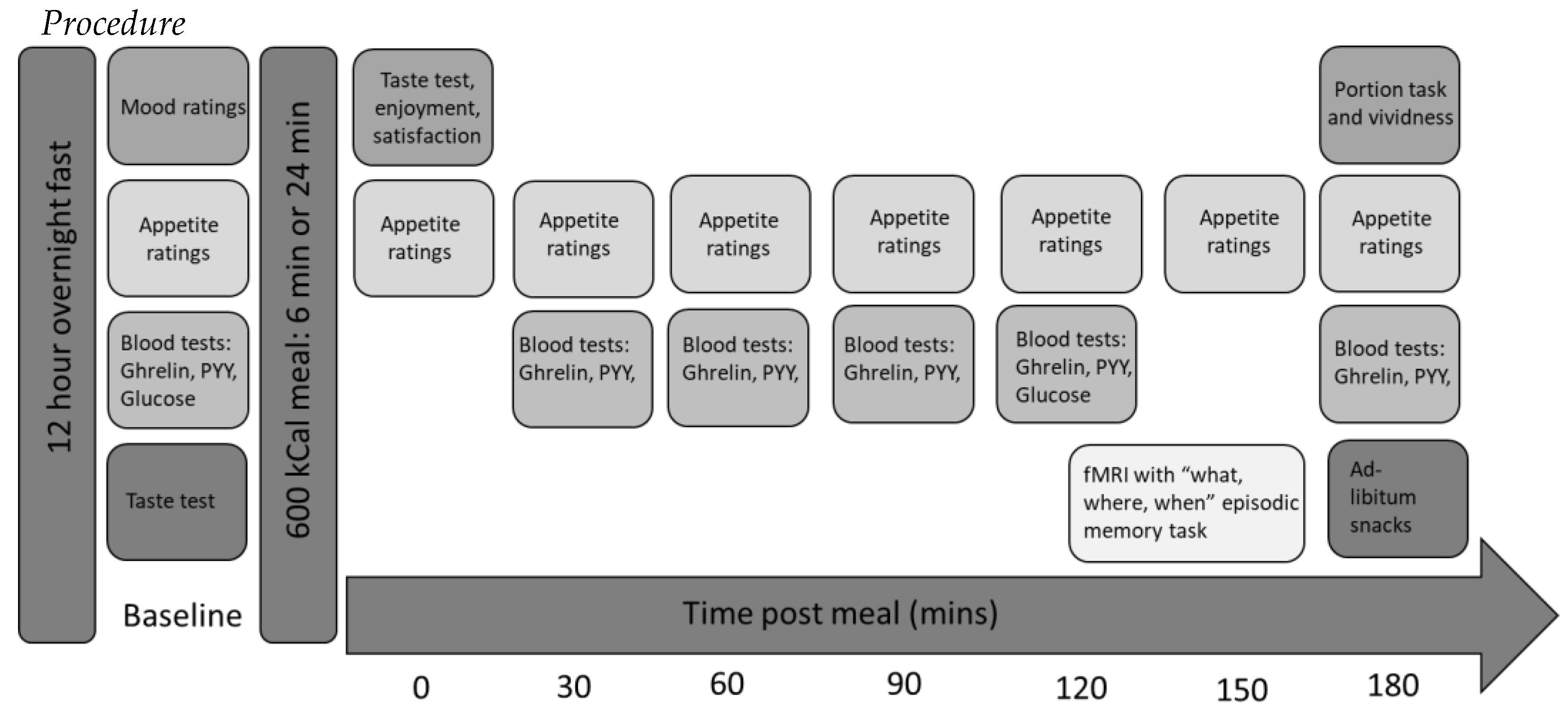
The objectives of this feasibility study were two-fold: first, to assess the feasibility of conducting a further, full-powered study combining a range of different measures (appetite ratings, satiety hormones, fMRI and subsequent consumption) to assess the effect of manipulating eating rate on these measures relating to energy intake. Feasibility outcomes were (i) combining novel and technically challenging measures (how many full data sets were obtained), (ii) a novel fMRI task (were the participants able to perform the task), (iii) ability to measure imaging signal in the brain regions of interest using fMRI, (iv) the practicality and acceptability of the blood sampling protocol (number of samples obtained) and (v) whether participants were aware of the aim of the study. Neuroimaging, a novel memory for recent eating task and GI hormones measurements were combined to ascertain whether top-down cognitive mechanisms or bottom-up physiological mechanisms are driving the effect of slowing eating rate. We developed a new “what, where, when” task to assess for memory for recent eating to be utilised during fMRI, as no such paradigm was previously available. Secondary objectives were to provide preliminary data using measures of appetite, hormones, fMRI and ad-libitum consumption to gain a greater understanding of the mechanisms underlying the effect of eating rate on energy intake.
To our knowledge, this is the first study to apply neuroimaging to investigate the effects of eating rate. The extensive fMRI literature on the satiating effects of food and food cues predicts a “network” of brain regions whose response might be influenced by eating rate, including insula [
19] and orbitofrontal cortex (OFC) [
20], in addition to subcortical reward regions [
21,
22,
23]. We predicted that slowing eating rate would lead to greater signal change during a memory for recent eating task in memory-, satiety- and reward-responsive brain areas, including hippocampal, frontal and parietal regions. We also hypothesised that experimentally slowing eating rate would lead to reduced enjoyment and satisfaction, a greater feeling of fullness post-meal and improved memory of the meal. In relation to GI hormones, we hypothesised that slowing eating rate would lead to increased ghrelin suppression and increased PYY secretion post-meal. We predicted that the combined effect of these mechanisms would lead to a reduced subsequent food intake at an
ad-libitum snack meal.
4. Discussion
In this study, we sought to assess the feasibility of our novel paradigm that comprised unique and technical challenging measures. Our objectives addressed the feasibility of several aspects of the paradigm; all of which were shown to have positive outcomes, and therefore can be used to guide the design of a future, fully powered study. Moreover, this paradigm was designed to gain a greater understanding of mechanisms underlying the effect of eating rate on energy intake, both cognitive and physiological. This combination of measures, tested in this feasibility study, provided preliminary data to show that slower eating led to a greater feeling of fullness, increased ghrelin suppression and a more vivid and accurate memory of the meal, yet in the context of reduced enjoyment and satisfaction from that meal. Importantly, these effects were associated with a 25% reduction in
ad-libitum intake for those who ate their previous fixed meal more slowly. This corroborates one previous study with a similar design [
11], but is contrary to other studies of similar design [
7,
8,
45], which may in part be due to differing percentage manipulation of eating rate. This is, therefore a relatively novel effect, as many of the previous studies in this field have measured the effect of eating rate within a single meal or by comparing different food textures [
3,
46,
47,
48].
In terms of feasibility, the study protocol was acceptable for participants and none of the participants guessed the actual aim of the study, therefore successfully avoiding a potential source of bias. It proved feasible to combine (i) behavioural measures, such as consuming a test meal, and regular appetite ratings, with (ii) physiological measures, such as gastrointestinal hormones and fMRI, in addition to (ii) cognitive measures, such as the memory for recent eating task. The blood sampling protocol was acceptable to participants and demonstrated that young, adult, healthy volunteers were prepared to have blood samples taken for research. The two conditions (normal and slow eating rates) for consuming the meal were acceptable to participants and all participants finished the test meal within the allocated time. The novel ‘what, where, when’ memory paradigm utilised to evaluate memory for recent eating was understood by participants and responses were not at ceiling. Performance on the “what” and “when” memory trials were similar, with a relative reduced accuracy and longer response times on the ‘where’ memory trials. Trial types may not have been well matched in terms of difficulty or spatial memory for a meal may not be as relevant as ‘what’ and ‘when’ in relation to memory for a recent eating episode. These issues will be taken forward into developing the task for future studies.
There are a number of potential limitations of this feasibility study that should be considered in the design of future, fully powered studies. First, we acknowledge that the small sample size renders the effect size estimations imprecise and therefore we have been cautious in our interpretation of the preliminary results. This will be borne in mind when calculating the sample size for future larger studies.
A priori, it was decided not to apply null hypothesis significance testing (NHST) to this data set. While currently this may be unconventional in this field, this study was designed with feasibility objectives in mind and therefore, in line with CONSORT guidelines for pilot and feasibility studies [
49], it would not be valid to perform NHST on the initial data due to the sample size. Secondly, combining the memory and physiological measures was not without difficulties. A between-subjects design was employed (as per [
10]) to enable memory for recent eating measures to be included and to avoid possible carry over effects of manipulating the meal within-subjects. However, this design may have been disadvantageous for the hormonal measures, which can vary between individuals. Importantly, all factors were measured relative to an individual’s baseline to account for within-session variability. Careful design considerations are necessary to overcome complications of including cognitive and physiological measures in one design.
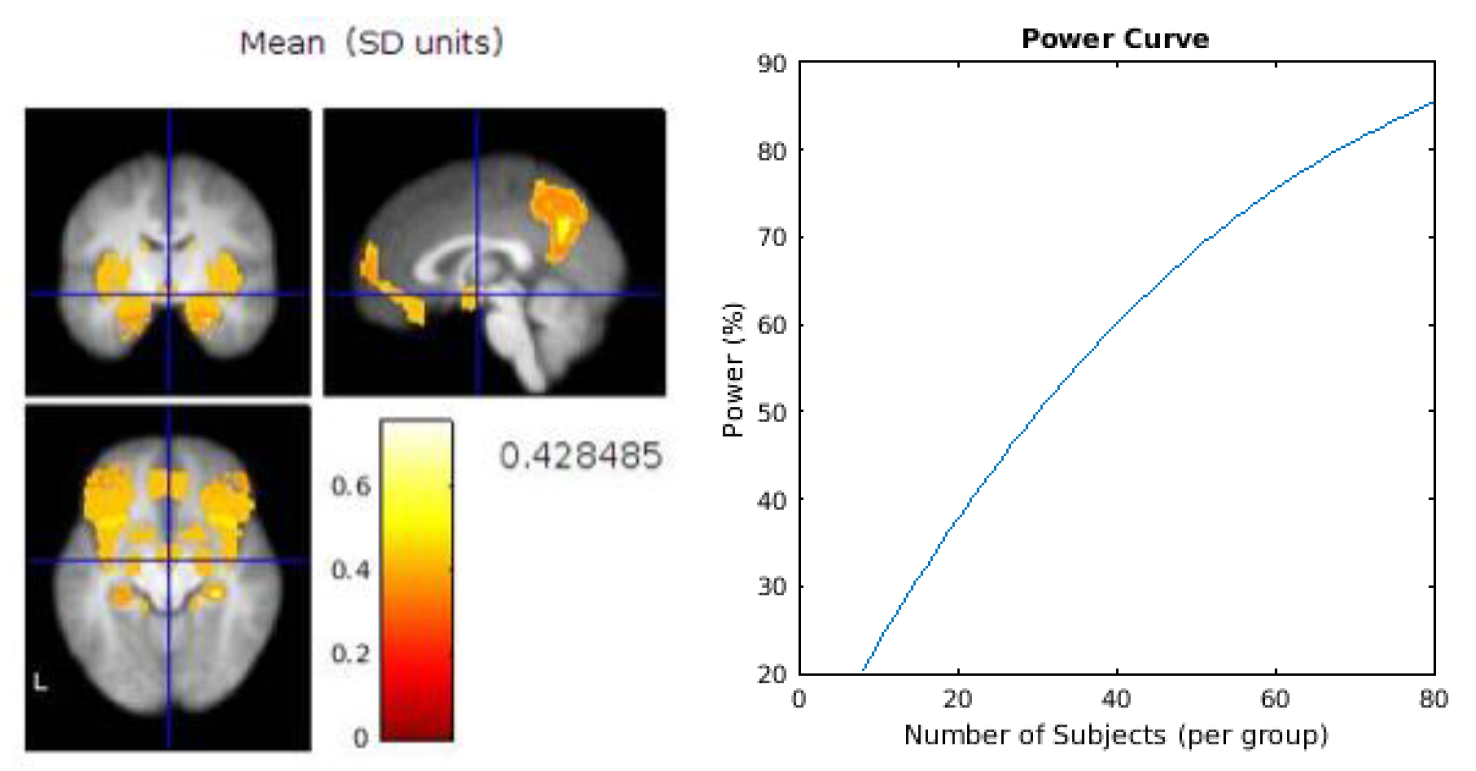
Based on our feasibility assessment above, therefore, the recommendations for a future design would be to first perform a series of power calculations for the main outcome measures to gauge the level of sensitivity required for each measure and therefore to ascertain the number of participants required to fully-power those measures. An example power calculation for two brain regions of interest is provided in
Appendix B. The challenge for future studies combining diverse measures will lie in powering the whole study to the measure requiring the most sensitivity. The memory task would benefit from some improvements, for example by developing “spatial” trials which enabled a similar performance to the “what” and “when” trials. We would also recommend consideration as to whether to employ the same between subject design as this feasibility study, or to utilise a within subject design to enable potentially more accurate comparison of the two eating rates, whilst minimising the risk of carry over effects.
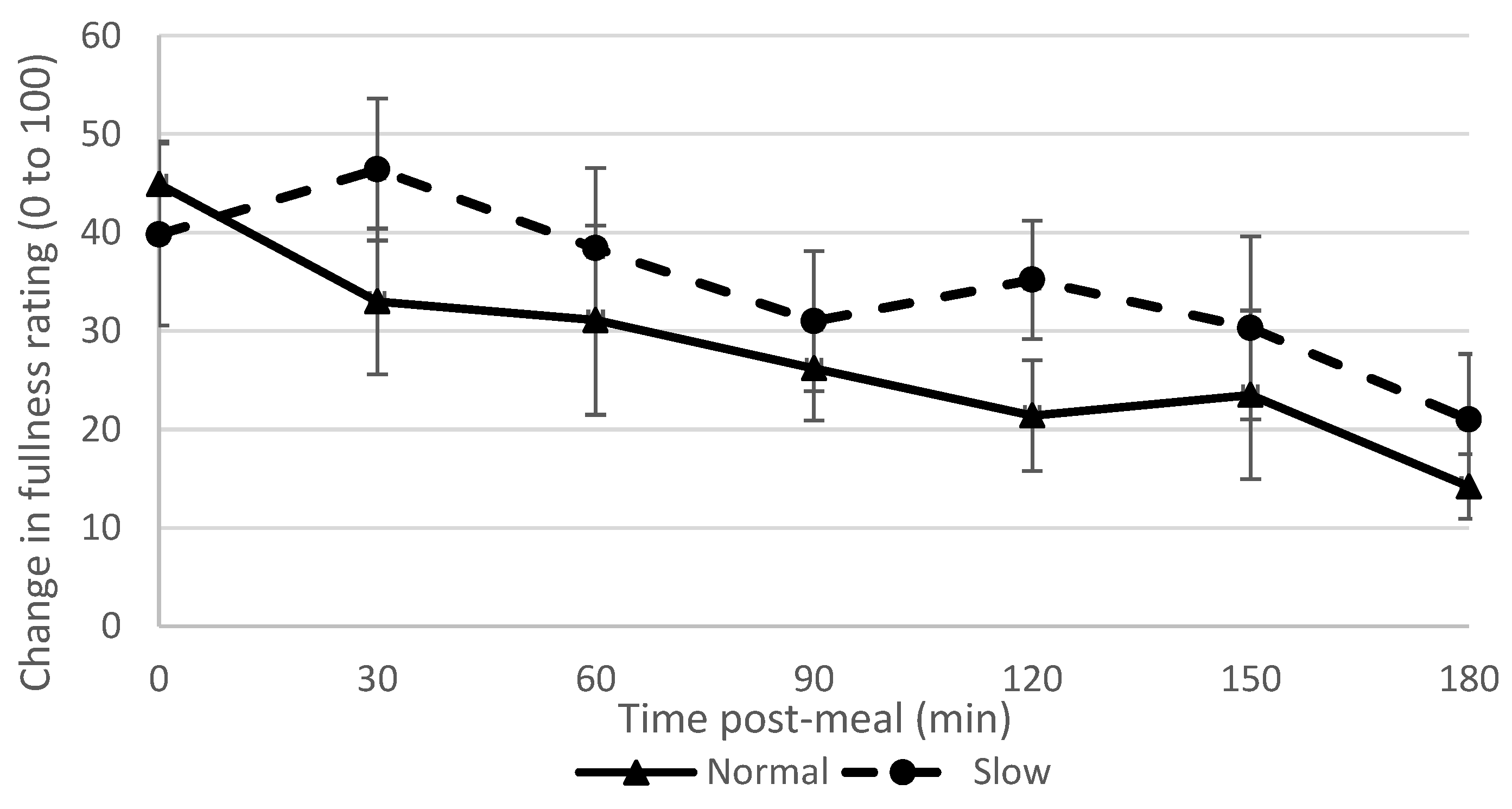
The slow rate group reported feeling fuller from 30 min post-meal and persisted for the full study duration of three hours. This supports our hypothesis and findings of previous studies [
6,
10,
50,
51], and provides evidence that was previously lacking regarding the effect of eating rate in a fixed meal on fullness ratings [
3]. Although the slow rate group consumed more water than the normal rate group, previous research has shown that this does not affect fullness or later ad-libitum consumption [
52]; however this effect cannot be ruled out [
46].
The normal rate group reported enjoying and feeling more satisfied by the meal, again supporting our hypothesis and replicating previous findings [
5,
11]. This may explain in part why people consume more if they eat more quickly, as it is a more enjoyable activity. This is an interesting finding however, because it could be postulated that savouring one’s food rather than eating it more quickly might be expected to increase enjoyment and satisfaction. Participants in the slow rate group in the study may have found it frustrating being instructed to consume their food more slowly, and that undermined enjoyment, including the enjoyment component of satisfaction. It is possible that there may be an optimal eating rate for individuals, encompassed within a window of tolerance to change; and reducing eating rate beyond that, reduces enjoyment and satisfaction from the meal (as seen here with this experimentally slow condition) which was also suggested by a previous study [
53]. Exploiting the boundaries of this tolerance window may be important for the design of eating rate interventions. On the other hand, tolerance to a slower eating rate might increase with repeated experience, so that slower eating becomes the norm. This would worthy of investigation in future studies.
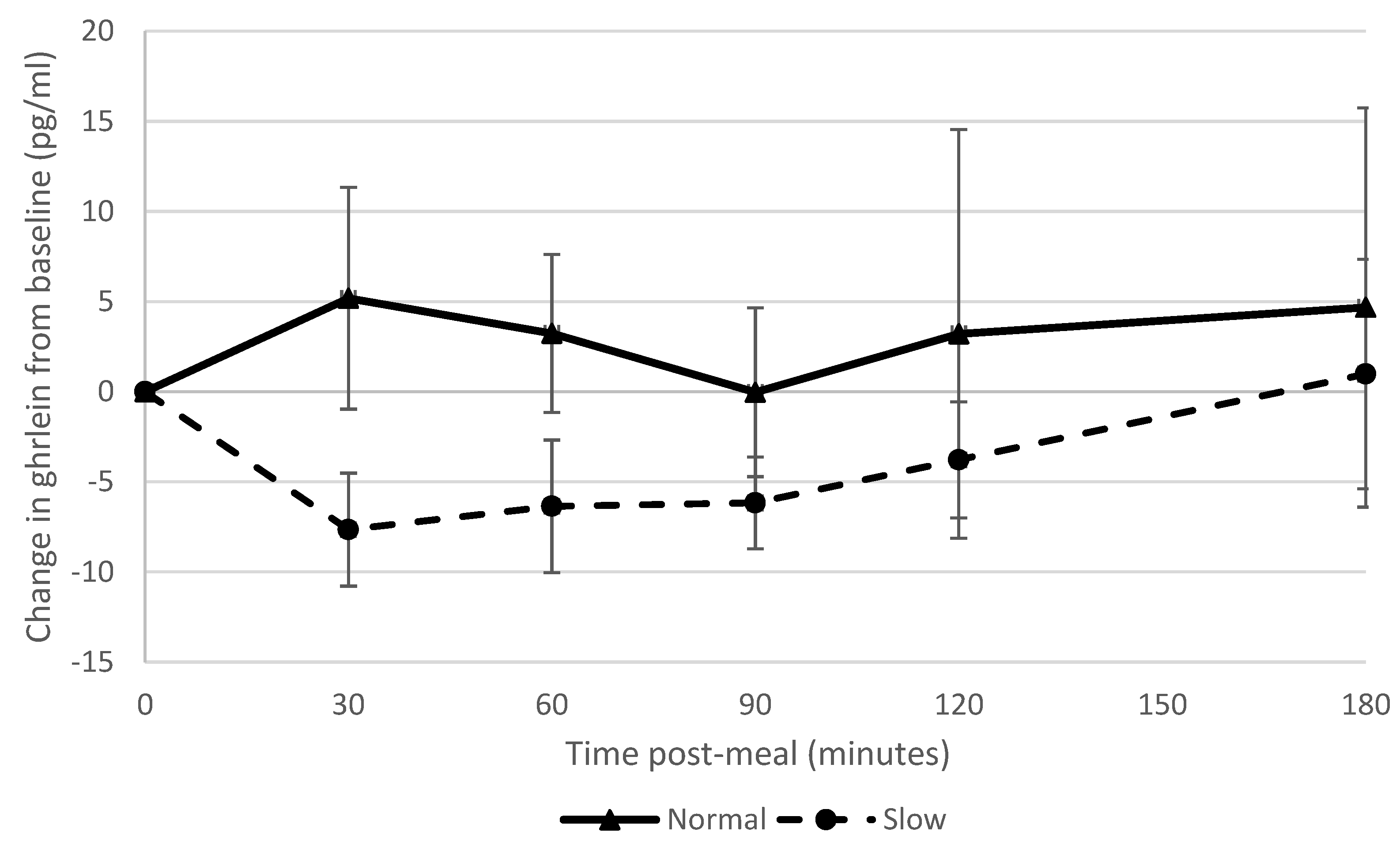
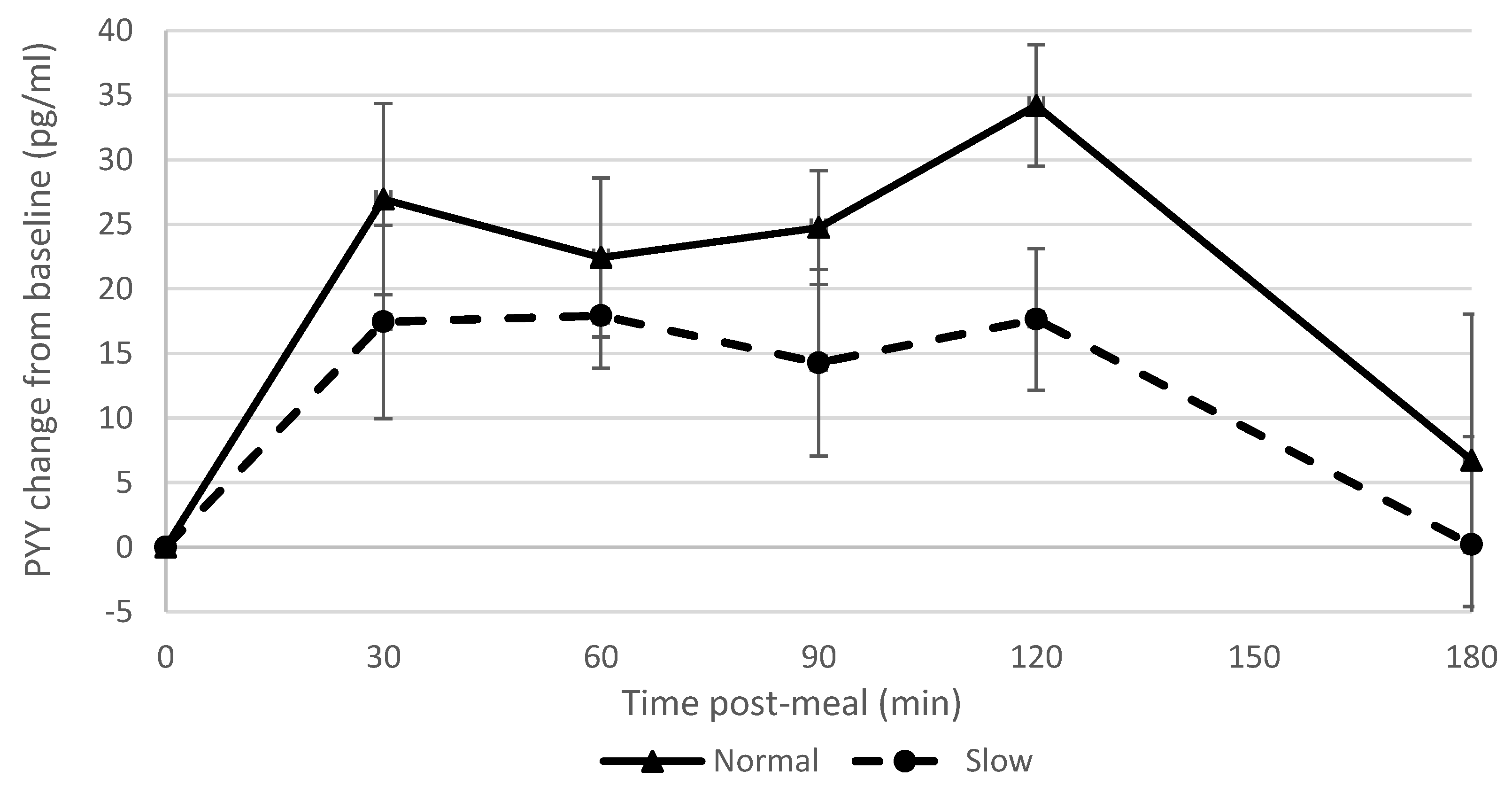
Slowing eating rate showed a large effect on ghrelin suppression, which supported our hypothesis and corroborated the findings of a previous study [
9]; however, other studies found no effect of manipulating eating rate on ghrelin suppression [
6,
7]. There was a strong correlation between blood ghrelin concentrations post-meal and ad libitum snack consumption. Accordingly, reduced appetite stimulation by ghrelin may in part explain why participants who had eaten more slowly consumed fewer snacks. This is also consistent with the finding that participants who had consumed the meal more slowly reported feeling fuller subsequently (i.e., ghrelin is effectively a signal for an empty stomach). The normal rate group showed a greater PYY response than the slow rate group which was in contrast to what we had hypothesised and findings of previous studies [
6,
7,
9] but in agreement with another study [
8]. These differences may in part be due to different methods used to manipulate eating rate [
8], different percentage alteration in eating rate and different hormone assays [
3], and this is clearly an area where further research is required.
At 3 h post meal, the slow rate group reported that they remembered the meal more vividly than the normal rate group. This finding was in contrast to previous studies which found no effect of eating rate on vividness [
10,
11], although the latter study showed that vividness of the memory of a meal was negatively associated with subsequent ad-libitum intake [
11]. Through the novel memory for recent eating task, the slow rate group demonstrated more accurate memory for portion size, and responded more quickly to the portion trials compared to the normal rate group; the latter of which has been previously associated with participants being more confident in their answers and relying on memory rather than guessing [
54,
55].
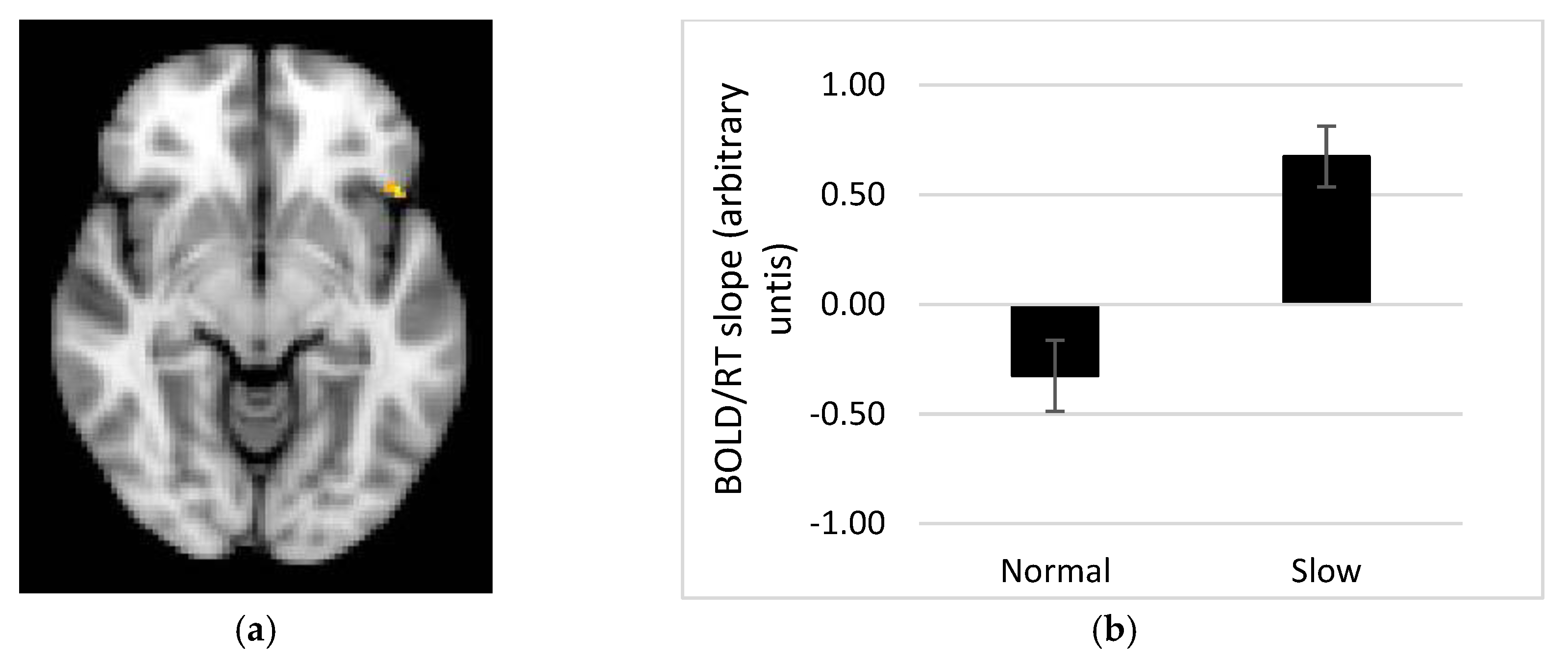
Our fMRI results demonstrate a linear relationship between the BOLD response and time to respond to portion size trials in several brain regions, whereby a steeper BOLD vs. RT relationship in the slow rate group might be linked to successful performance on the task. One alternative interpretation of this finding is that improved performance on the memory for recent eating task is associated with reward accompanying the sense of answering the trial correctly, and this might be reflected in activity within reward related brain regions e.g., OFC, amygdala. However, this explanation sits less well with activity in parietal cortex, which showed the same pattern. Memory for portion sizes recently consumed may therefore require recruitment of areas such as the OFC, insula and putamen, as well as precuneus, angular gyrus, supramarginal gyrus and middle frontal and temporal gyri. Those regions showing a direct relationship with response time have a stronger association with the memory processes recruited when recalling recent eating episodes. These regions are in keeping with the areas sub-serving the object recognition task of Kwok et al. [
17] (closest equivalent to the portion size task in this study), but not in keeping with the hippocampal activity found associated with the object task in Cheke et al. [
18]. The lack of hippocampal activity in response to the current memory for recent eating task maybe due to the lack of integration required [
18], as memory elements (portion size, interval and spatial aspects) were studied separately (as per design in [
17]).
The fMRI data also reveals brain regions subserving memory for the spatial environment, or context, in which the meal was consumed. Superior parietal and fusiform cortices were associated with the spatial trials, which is in keeping with a similar spatial task in Kwok et al. [
15]. Across both groups, a linear relationship between BOLD signal and response time for spatial trials was observed in the inferior and middle frontal cortices, suggesting that these regions are implicated in memory of this kind.
In terms of future applications of this work, investigating whether or not these effects of experimentally manipulating eating rate are also found in childhood would be beneficial, by repeating this study in both normal weight and obese children, ideally with known genetic variability [
56]. In view of alternative studies [
6,
7] that demonstrated responses for other satiety hormones (such as glucagon-like peptide 1 and cholecystokinin), it would be informative to incorporate a wider range of GI hormones into a further study to provide a more complete picture of the endocrine consequences of slowing eating rate. In order to apply the findings of this research, further work is needed in order to design effective interventions to manipulate eating rate on a long term basis, through a range of methods such as environmental modifications [
57], behavioural training from childhood [
58] and modification of food textures [
59,
60]. Our findings highlight the need to focus on preserving meal enjoyment and meal satisfaction when reducing eating rate in behavioural interventions.