Effects of Coffee and Tea Consumption on Glucose Metabolism: A Systematic Review and Network Meta-Analysis
Abstract
1. Introduction
2. Materials and Methods
2.1. Literature Search
2.2. Study Selection
2.3. Data Extraction
2.4. Risk of Bias Assessment
2.5. Strategy for Data Synthesis
2.6. Subgroup or Subset Analysis
3. Results
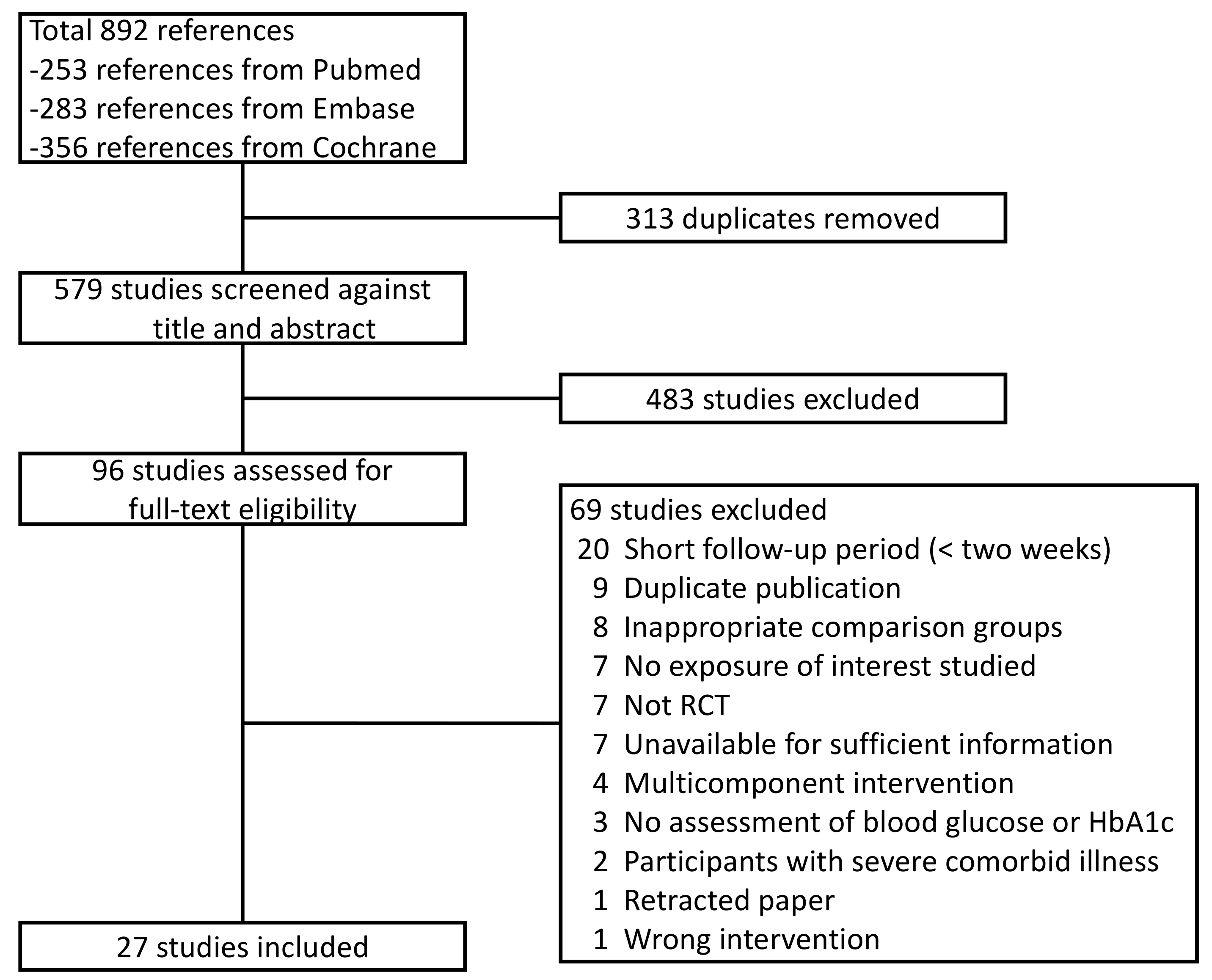
3.1. Study Selection and Characteristics
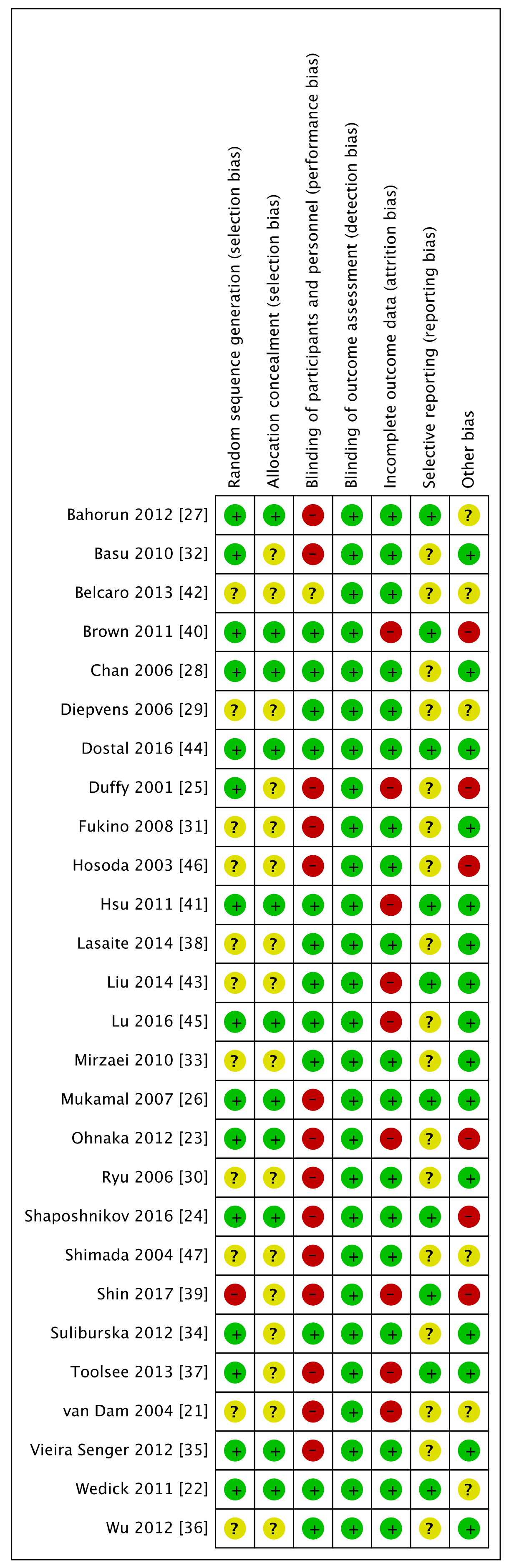
3.2. Risk of Bias for Included Studies
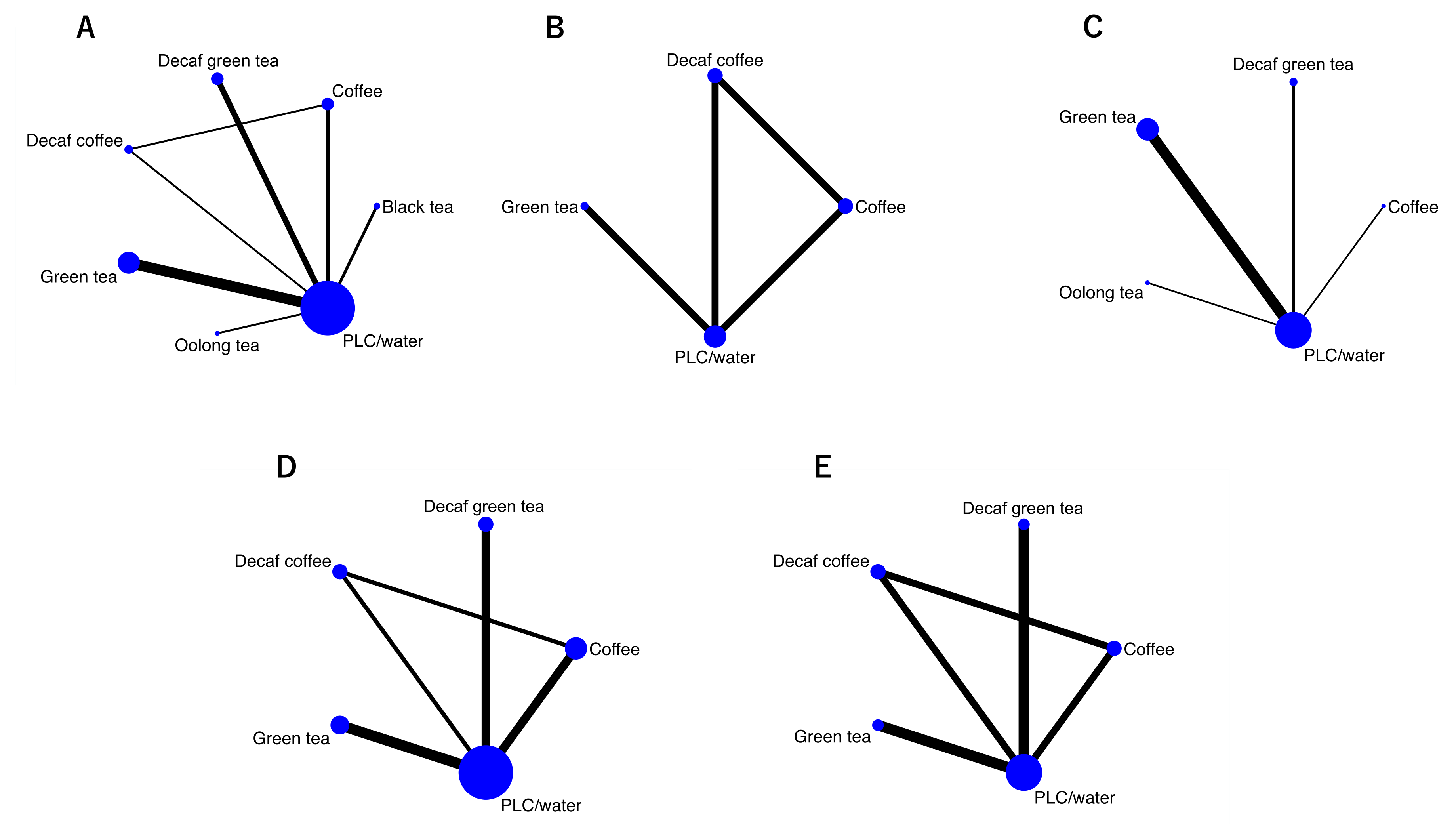
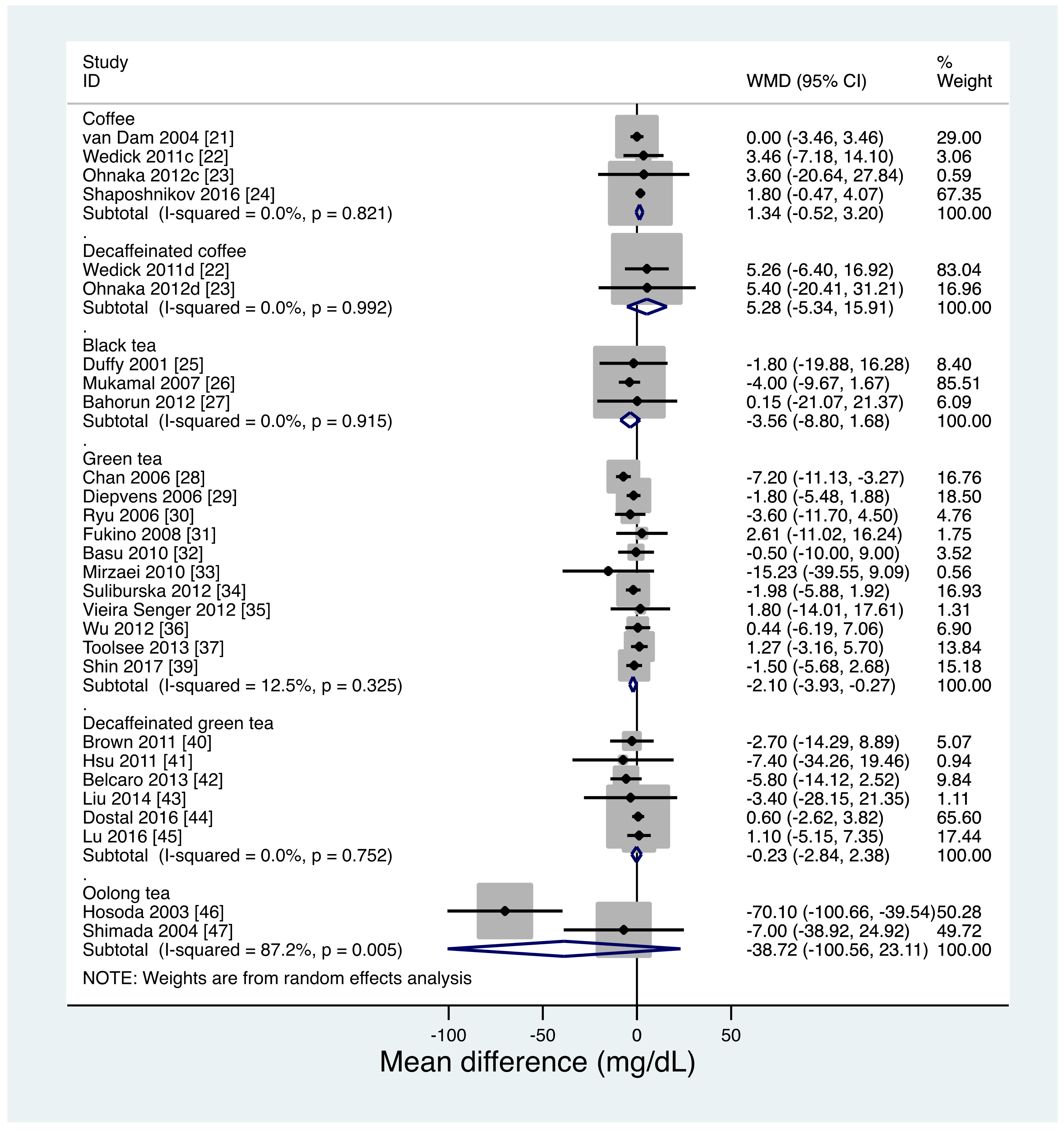
3.3. Primary Endpoint: FBG
3.4. Secondary Endpoints:
3.4.1. 75-g OGTT 2h-PG
3.4.2. HbA1c
3.4.3. Fasting IRI
3.4.4. HOMA-IR
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Ogurtsova, K.; da Rocha Fernandes, J.D.; Huang, Y.; Linnenkamp, U.; Guariguata, L.; Cho, N.H.; Cavan, D.; Shaw, J.E.; Makaroff, L.E. IDF Diabetes Atlas: Global estimates for the prevalence of diabetes for 2015 and 2040. Diabetes Res. Clin. Pract. 2017, 128, 40–50. [Google Scholar] [CrossRef] [PubMed]
- Bommer, C.; Heesemann, E.; Sagalova, V.; Manne-Goehler, J.; Atun, R.; Barnighausen, T.; Vollmer, S. The global economic burden of diabetes in adults aged 20–79 years: A cost-of-illness study. Lancet Diabetes Endocrinol. 2017, 5, 423–430. [Google Scholar] [CrossRef]
- Van Dam, R.M.; Feskens, E.J. Coffee consumption and risk of type 2 diabetes mellitus. Lancet 2002, 360, 1477–1478. [Google Scholar] [CrossRef]
- Salazar-Martinez, E.; Willett, W.C.; Ascherio, A.; Manson, J.E.; Leitzmann, M.F.; Stampfer, M.J.; Hu, F.B. Coffee consumption and risk for type 2 diabetes mellitus. Ann. Intern. Med. 2004, 140, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Odegaard, A.O.; Pereira, M.A.; Koh, W.P.; Arakawa, K.; Lee, H.P.; Yu, M.C. Coffee, tea, and incident type 2 diabetes: The Singapore Chinese Health Study. Am. J. Clin. Nutr. 2008, 88, 979–985. [Google Scholar] [CrossRef] [PubMed]
- Iso, H.; Date, C.; Wakai, K.; Fukui, M.; Tamakoshi, A. The relationship between green tea and total caffeine intake and risk for self-reported type 2 diabetes among Japanese adults. Ann. Intern. Med. 2006, 144, 554–562. [Google Scholar] [CrossRef] [PubMed]
- Liu, K.; Zhou, R.; Wang, B.; Chen, K.; Shi, L.Y.; Zhu, J.D.; Mi, M.T. Effect of green tea on glucose control and insulin sensitivity: A meta-analysis of 17 randomized controlled trials. Am. J. Clin. Nutr. 2013, 98, 340–348. [Google Scholar] [CrossRef] [PubMed]
- Duncan, M.J.; Stanley, M.; Parkhouse, N.; Cook, K.; Smith, M. Acute caffeine ingestion enhances strength performance and reduces perceived exertion and muscle pain perception during resistance exercise. Eur. J. Sport Sci. 2013, 13, 392–399. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Wang, C.; Huai, Q.; Guo, F.; Liu, L.; Feng, R.; Sun, C. Effects of tea or tea extract on metabolic profiles in patients with type 2 diabetes mellitus: A meta-analysis of ten randomized controlled trials. Diabetes Metab. Res. Rev. 2016, 32, 2–10. [Google Scholar] [CrossRef] [PubMed]
- Shi, X.; Xue, W.; Liang, S.; Zhao, J.; Zhang, X. Acute caffeine ingestion reduces insulin sensitivity in healthy subjects: A systematic review and meta-analysis. Nutr. J. 2016, 15, 103. [Google Scholar] [CrossRef] [PubMed]
- Guyatt, G.; Oxman, A.D.; Akl, E.A.; Kunz, R.; Vist, G.; Brozek, J.; Norris, S.; Falck-Ytter, Y.; Glasziou, P.; DeBeer, H.; et al. GRADE guidelines: 1. Introduction-GRADE evidence profiles and summary of findings tables. J. Clin. Epidemiol. 2011, 64, 383–394. [Google Scholar] [CrossRef] [PubMed]
- Efthimiou, O.; Debray, T.P.; van Valkenhoef, G.; Trelle, S.; Panayidou, K.; Moons, K.G.; Reitsma, J.B.; Shang, A.; Salanti, G.; GetReal Methods Review Group. GetReal in network meta-analysis: A review of the methodology. Res. Synth. Methods 2016, 7, 236–263. [Google Scholar] [CrossRef] [PubMed]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G.; PRISMA Group. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. J. Clin. Epidemiol. 2009, 62, 1006–1012. [Google Scholar] [CrossRef] [PubMed]
- Hutton, B.; Salanti, G.; Caldwell, D.M.; Chaimani, A.; Schmid, C.H.; Cameron, C.; Ioannidis, J.P.; Straus, S.; Thorlund, K.; Jansen, J.P.; et al. The PRISMA extension statement for reporting of systematic reviews incorporating network meta-analyses of health care interventions: Checklist and explanations. Ann. Intern. Med. 2015, 162, 777–784. [Google Scholar] [CrossRef] [PubMed]
- Higgins, J.P.; Altman, D.G.; Gotzsche, P.C.; Juni, P.; Moher, D.; Oxman, A.D.; Savovic, J.; Schulz, K.F.; Weeks, L.; Sterne, J.A.; et al. The Cochrane Collaboration’s tool for assessing risk of bias in randomised trials. BMJ 2011, 343, d5928. [Google Scholar] [CrossRef] [PubMed]
- Furukawa, T.A.; Barbui, C.; Cipriani, A.; Brambilla, P.; Watanabe, N. Imputing missing standard deviations in meta-analyses can provide accurate results. J. Clin. Epidemiol. 2006, 59, 7–10. [Google Scholar] [CrossRef] [PubMed]
- Higgins, J.P.; Jackson, D.; Barrett, J.K.; Lu, G.; Ades, A.E.; White, I.R. Consistency and inconsistency in network meta-analysis: Concepts and models for multi-arm studies. Res. Synth. Methods 2012, 3, 98–110. [Google Scholar] [CrossRef] [PubMed]
- Harris, R.; Bradburn, M.; Deeks, J.; Harbord, R.; Altman, D.; Sterne, J. Metan: Fixed- and random-effects meta-analysis. Stata J. 2008, 8, 3–28. [Google Scholar] [CrossRef]
- Harbord, R.M.; Higgins, J.P.T. Meta-regression in Stata. Stata J. 2008, 8, 493–519. [Google Scholar] [CrossRef]
- White, I.R. Network meta-analysis. Stata J. 2015, 15, 951–985. [Google Scholar] [CrossRef]
- van Dam, R.M.; Pasman, W.J.; Verhoef, P. Effects of coffee consumption on fasting blood glucose and insulin concentrations: Randomized controlled trials in healthy volunteers. Diabetes Care 2004, 27, 2990–2992. [Google Scholar] [CrossRef] [PubMed]
- Wedick, N.M.; Brennan, A.M.; Sun, Q.; Hu, F.B.; Mantzoros, C.S.; van Dam, R.M. Effects of caffeinated and decaffeinated coffee on biological risk factors for type 2 diabetes: A randomized controlled trial. Nutr. J. 2011, 10, 93. [Google Scholar] [CrossRef] [PubMed]
- Ohnaka, K.; Ikeda, M.; Maki, T.; Okada, T.; Shimazoe, T.; Adachi, M.; Nomura, M.; Takayanagi, R.; Kono, S. Effects of 16-week consumption of caffeinated and decaffeinated instant coffee on glucose metabolism in a randomized controlled trial. J. Nutr. Metab. 2012, 2012, 207426. [Google Scholar] [CrossRef] [PubMed]
- Shaposhnikov, S.; Hatzold, T.; Yamani, N.; Stavro, P.; Lorenzo, Y.; Dusinska, M.; Reus, A.; Pasman, W.; Collins, A. Coffee and oxidative stress: A human intervention study. Eur. J. Nutr. 2016. [Google Scholar] [CrossRef] [PubMed]
- Duffy, S.J.; Keaney, J.F., Jr.; Holbrook, M.; Gokce, N.; Swerdloff, P.L.; Frei, B.; Vita, J.A. Short- and long-term black tea consumption reverses endothelial dysfunction in patients with coronary artery disease. Circulation 2001, 104, 151–156. [Google Scholar] [CrossRef] [PubMed]
- Mukamal, K.J.; MacDermott, K.; Vinson, J.A.; Oyama, N.; Manning, W.J.; Mittleman, M.A. A 6-month randomized pilot study of black tea and cardiovascular risk factors. Am. Heart J. 2007, 154, 724.e1–724.e6. [Google Scholar] [CrossRef] [PubMed]
- Bahorun, T.; Luximon-Ramma, A.; Neergheen-Bhujun, V.S.; Gunness, T.K.; Googoolye, K.; Auger, C.; Crozier, A.; Aruoma, O.I. The effect of black tea on risk factors of cardiovascular disease in a normal population. Prev. Med. 2012, 54, S98–S102. [Google Scholar] [CrossRef] [PubMed]
- Chan, C.C.; Koo, M.W.; Ng, E.H.; Tang, O.S.; Yeung, W.S.; Ho, P.C. Effects of Chinese green tea on weight, and hormonal and biochemical profiles in obese patients with polycystic ovary syndrome—A randomized placebo-controlled trial. J. Soc. Gynecol. Investig. 2006, 13, 63–68. [Google Scholar] [CrossRef]
- Diepvens, K.; Kovacs, E.M.; Vogels, N.; Westerterp-Plantenga, M.S. Metabolic effects of green tea and of phases of weight loss. Physiol. Behav. 2006, 87, 185–191. [Google Scholar] [CrossRef]
- Ryu, O.H.; Lee, J.; Lee, K.W.; Kim, H.Y.; Seo, J.A.; Kim, S.G.; Kim, N.H.; Baik, S.H.; Choi, D.S.; Choi, K.M. Effects of green tea consumption on inflammation, insulin resistance and pulse wave velocity in type 2 diabetes patients. Diabetes Res. Clin. Pract. 2006, 71, 356–358. [Google Scholar] [CrossRef]
- Fukino, Y.; Ikeda, A.; Maruyama, K.; Aoki, N.; Okubo, T.; Iso, H. Randomized controlled trial for an effect of green tea-extract powder supplementation on glucose abnormalities. Eur. J. Clin. Nutr. 2008, 62, 953–960. [Google Scholar] [CrossRef] [PubMed]
- Basu, A.; Sanchez, K.; Leyva, M.J.; Wu, M.; Betts, N.M.; Aston, C.E.; Lyons, T.J. Green tea supplementation affects body weight, lipids, and lipid peroxidation in obese subjects with metabolic syndrome. J. Am. Coll. Nutr. 2010, 29, 31–40. [Google Scholar] [CrossRef] [PubMed]
- Mirzaei, K.; Hosseinzadeh-Attar, M.J.; Hossein-Nezhad, A.; Karimi, M.; Kamalinejad, M.; Jafari, N.; Najmafshar, A.; Karimi, F.; Eshraghian, M.R.; Mohammadzadeh, N.; et al. Visfatin genotype may influence the effects of green tea extract (Camellia sinensis) in Type 2 diabetes patients. Double blind placebo-controlled clinical trial study. Gazz. Med. Ital. 2010, 169, 137–147. [Google Scholar]
- Suliburska, J.; Bogdanski, P.; Szulinska, M.; Stepien, M.; Pupek-Musialik, D.; Jablecka, A. Effects of green tea supplementation on elements, total antioxidants, lipids, and glucose values in the serum of obese patients. Biol. Trace Elem. Res. 2012, 149, 315–322. [Google Scholar] [CrossRef] [PubMed]
- Vieira Senger, A.E.; Schwanke, C.H.; Gomes, I.; Valle Gottlieb, M.G. Effect of green tea (Camellia sinensis) consumption on the components of metabolic syndrome in elderly. J. Nutr. Health Aging 2012, 16, 738–742. [Google Scholar] [CrossRef] [PubMed]
- Wu, A.H.; Spicer, D.; Stanczyk, F.Z.; Tseng, C.C.; Yang, C.S.; Pike, M.C. Effect of 2-month controlled green tea intervention on lipoprotein cholesterol, glucose, and hormone levels in healthy postmenopausal women. Cancer Prev. Res. 2012, 5, 393–402. [Google Scholar] [CrossRef] [PubMed]
- Toolsee, N.A.; Aruoma, O.I.; Gunness, T.K.; Kowlessur, S.; Dambala, V.; Murad, F.; Googoolye, K.; Daus, D.; Indelicato, J.; Rondeau, P.; et al. Effectiveness of green tea in a randomized human cohort: Relevance to diabetes and its complications. Biomed. Res. Int. 2013, 2013, 412379. [Google Scholar] [CrossRef]
- Lasaite, L.; Spadiene, A.; Savickiene, N.; Skesters, A.; Silova, A. The effect of Ginkgo biloba and Camellia sinensis extracts on psychological state and glycemic control in patients with type 2 diabetes mellitus. Nat. Prod. Commun. 2014, 9, 1345–1350. [Google Scholar]
- Shin, C.M.; Lee, D.H.; Seo, A.Y.; Lee, H.J.; Kim, S.B.; Son, W.-C.C.; Kim, Y.K.; Lee, S.J.; Park, S.-H.H.; Kim, N.; et al. Green tea extracts for the prevention of metachronous colorectal polyps among patients who underwent endoscopic removal of colorectal adenomas: A randomized clinical trial. Clin. Nutr. 2017. [Google Scholar] [CrossRef]
- Brown, A.L.; Lane, J.; Holyoak, C.; Nicol, B.; Mayes, A.E.; Dadd, T. Health effects of green tea catechins in overweight and obese men: A randomised controlled cross-over trial. Br. J. Nutr. 2011, 106, 1880–1889. [Google Scholar] [CrossRef]
- Hsu, C.H.; Liao, Y.L.; Lin, S.C.; Tsai, T.H.; Huang, C.J.; Chou, P. Does supplementation with green tea extract improve insulin resistance in obese type 2 diabetics? A randomized, double-blind, and placebo-controlled clinical trial. Altern. Med. Rev. 2011, 16, 157–163. [Google Scholar] [CrossRef] [PubMed]
- Belcaro, G.; Ledda, A.; Hu, S.; Cesarone, M.R.; Feragalli, B.; Dugall, M. Greenselect phytosome for borderline metabolic syndrome. Evid. Based Complement. Alternat. Med. 2013, 2013, 869061. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.Y.; Huang, C.J.; Huang, L.H.; Chen, I.J.; Chiu, J.P.; Hsu, C.H. Effects of green tea extract on insulin resistance and glucagon-like peptide 1 in patients with type 2 diabetes and lipid abnormalities: A randomized, double-blinded, and placebo-controlled trial. PLoS ONE 2014, 9, e91163. [Google Scholar] [CrossRef] [PubMed]
- Dostal, A.M.; Samavat, H.; Espejo, L.; Arikawa, A.Y.; Stendell-Hollis, N.R.; Kurzer, M.S. Green Tea Extract and Catechol-O-Methyltransferase Genotype Modify Fasting Serum Insulin and Plasma Adiponectin Concentrations in a Randomized Controlled Trial of Overweight and Obese Postmenopausal Women. J. Nutr. 2016, 146, 38–45. [Google Scholar] [CrossRef]
- Lu, P.H.; Hsu, C.H. Does supplementation with green tea extract improve acne in post-adolescent women? A randomized, double-blind, and placebo-controlled clinical trial. Complement. Ther. Med. 2016, 25, 159–163. [Google Scholar] [CrossRef]
- Hosoda, K.; Wang, M.F.; Liao, M.L.; Chuang, C.K.; Iha, M.; Clevidence, B.; Yamamoto, S. Antihyperglycemic effect of oolong tea in type 2 diabetes. Diabetes Care 2003, 26, 1714–1718. [Google Scholar] [CrossRef]
- Shimada, K.; Kawarabayashi, T.; Tanaka, A.; Fukuda, D.; Nakamura, Y.; Yoshiyama, M.; Takeuchi, K.; Sawaki, T.; Hosoda, K.; Yoshikawa, J. Oolong tea increases plasma adiponectin levels and low-density lipoprotein particle size in patients with coronary artery disease. Diabetes Res. Clin. Pract. 2004, 65, 227–234. [Google Scholar] [CrossRef]
- Eng, Q.Y.; Thanikachalam, P.V.; Ramamurthy, S. Molecular understanding of Epigallocatechin gallate (EGCG) in cardiovascular and metabolic diseases. J. Ethnopharmacol. 2018, 210, 296–310. [Google Scholar] [CrossRef]
- Waltner-Law, M.E.; Wang, X.L.; Law, B.K.; Hall, R.K.; Nawano, M.; Granner, D.K. Epigallocatechin gallate, a constituent of green tea, represses hepatic glucose production. J. Biol. Chem. 2002, 277, 34933–34940. [Google Scholar] [CrossRef]
- Ortsater, H.; Grankvist, N.; Wolfram, S.; Kuehn, N.; Sjoholm, A. Diet supplementation with green tea extract epigallocatechin gallate prevents progression to glucose intolerance in db/db mice. Nutr. Metab. 2012, 9, 11. [Google Scholar] [CrossRef]
- Ueda, M.; Nishiumi, S.; Nagayasu, H.; Fukuda, I.; Yoshida, K.; Ashida, H. Epigallocatechin gallate promotes GLUT4 translocation in skeletal muscle. Biochem. Biophys. Res. Commun. 2008, 377, 286–290. [Google Scholar] [CrossRef] [PubMed]
- Fu, Z.; Zhen, W.; Yuskavage, J.; Liu, D. Epigallocatechin gallate delays the onset of type 1 diabetes in spontaneous non-obese diabetic mice. Br. J. Nutr. 2011, 105, 1218–1225. [Google Scholar] [CrossRef] [PubMed]
- U.K. Prospective Diabetes Study Group. U.K. Prospective Diabetes Study 16: Overview of 6 Years’ Therapy of Type II Diabetes: A Progressive Disease. Diabetes 1995, 44, 1249–1258. [Google Scholar] [CrossRef]
- Saisho, Y. Beta-cell dysfunction: Its critical role in prevention and management of type 2 diabetes. World J. Diabetes 2015, 6, 109–124. [Google Scholar] [CrossRef] [PubMed]
- Higdon, J.V.; Frei, B. Coffee and health: A review of recent human research. Crit. Rev. Food Sci. Nutr. 2006, 46, 101–123. [Google Scholar] [CrossRef] [PubMed]
- van Dam, R.M.; Willett, W.C.; Manson, J.E.; Hu, F.B. Coffee, caffeine, and risk of type 2 diabetes: A prospective cohort study in younger and middle-aged U.S. women. Diabetes Care 2006, 29, 398–403. [Google Scholar] [CrossRef] [PubMed]
- Ding, M.; Bhupathiraju, S.N.; Chen, M.; van Dam, R.M.; Hu, F.B. Caffeinated and decaffeinated coffee consumption and risk of type 2 diabetes: A systematic review and a dose-response meta-analysis. Diabetes Care 2014, 37, 569–586. [Google Scholar] [CrossRef] [PubMed]
- Lofvenborg, J.E.; Andersson, T.; Carlsson, P.O.; Dorkhan, M.; Groop, L.; Martinell, M.; Rasouli, B.; Storm, P.; Tuomi, T.; Carlsson, S. Coffee consumption and the risk of latent autoimmune diabetes in adults—Results from a Swedish case-control study. Diabetes Med. 2014, 31, 799–805. [Google Scholar] [CrossRef]
| Study ID | Study Design | Population | Country | Interventions | Outcomes | Total Sample Size | Duration of Intervention (weeks) | Age | Male Sex (%) | BMI (kg/m2) | Baseline FBG (mg/dL) |
|---|---|---|---|---|---|---|---|---|---|---|---|
| van Dam 2004 [21] | Crossover | Healthy volunteers | Netherlands | Coffee/no coffee | FBG, F-IRI | 26 | 4 | 37.0 | 38 | 23.0 | NR |
| Wedick 2011 [22] | Parallel | Overweight | US | Coffee/decaf coffee/no coffee | FBG, 2hPG, F-IRI, IR | 45 | 8 | 40.6 | 35 | 29.5 | 86.4 |
| Ohnaka 2012 [23] | Parallel | Overweight men with IFG | Japan | Coffee/decaf coffee/water | FBG, 2hPG, F-IRI, IR | 43 | 16 | 52.7 | 100 | 25-30 | 107.6 |
| Shaposhnikov 2016 [24] | Parallel | Healthy volunteers | Germany | Coffee/water | FBG, A1c, F-IRI, | 160 | 8 | 51.0 | NR | 26.5 | 97.2 |
| Duffy 2001 [25] | Crossover | CAD | US | Black tea/water | FBG | 50 | 4 | 55.0 | 78 | 29.7 | 121.5 |
| Mukamal 2007 [26] | Parallel | Diabetes or 2 other cardiovascular risk factors | US | Black tea/water | FBG | 28 | 26 | 65.8 | 36 | 29.1 | 97.0 |
| Bahorun 2012 [27] | Parallel | Healthy subjects | Mauritius | Black tea/water | FBG | 77 | 12 | 25–74 | 55 | NR | 125.5 |
| Chan 2006 [28] | Parallel | Obese women with polycystic ovary syndrome | China | Green tea extract/placebo | FBG, 2hPG, F-IRI | 34 | 13 | 25–40 | 0 | 30.1 | 92.6 |
| Diepvens 2006 [29] | Parallel | Overweight female | Netherlands | Green tea extract/placebo | FBG | 46 | 12 | 41.7 | 0 | 27.7 | 93.6 |
| Ryu 2006 [30] | Crossover | T2D | Korea | Green tea/water | FBG, F-IRI, IR | 55 | 4 | 53.9 | 56 | 25.0 | NR |
| Fukino 2008 [31] | Crossover | Prediabetes | Japan | Green tea extract/water | FBG, A1c, F-IRI, IR | 60 | 9 | 53.6 | 85 | 25.7 | 137.7 |
| Basu 2010 [32] | Parallel | Obesity & MetS | US | Green tea/green tea extract/water | FBG, A1c, IR | 35 | 8 | 42.5 | 49 | 36.2 | 88.2 |
| Mirzaei 2010 [33] | Parallel | T2D | Iran | Green tea extract/placebo | FBG, 2hPG, A1c, F-IRI | 102 | 8 | 54.6 | 21 | 29.2 | 172.1 |
| Suliburska 2012 [34] | Crossover | Obese | Poland | Green tea extract/placebo | FBG | 46 | 13 | 50.4 | 50 | 32.8 | 101.8 |
| Vieira Senger 2012 [35] | Parallel | MetS | Brazil | Green tea/no green tea | FBG | 45 | 9 | ≥60 | 16 | 30.5 | 119.0 |
| Wu 2012 [36] | Parallel | Postmenopausal women | US | Green tea extract/placebo | FBG, A1c, F-IRI, | 103 | 9 | 59.8 | 0 | 29.3 | 99.7 |
| Toolsee 2013 [37] | Parallel | Prediabetes | Mauritius | Green tea/water | FBG, A1c, | 117 | 14 | 48.3 | 51 | 25.6 | 91.6 |
| Lasaite 2014 [38] | Parallel | T2D | Lithuania | Green tea extract/placebo | A1c | 31 | 78 | 57.0 | 38 | NR | NR |
| Shin 2017 [39] | Parallel | After endoscopic polypectomy | Korea | Green tea extract/no Green tea extract | FBG | 143 | 52 | 59.7 | 68 | 23.9 | 101.7 |
| Brown 2011 [40] | Crossover | Healthy overweight and obese men | UK | Decaf green tea extract/placebo | FBG, F-IRI | 66 | 6 | 49.4 | 100 | 31.5 | 107.1 |
| Hsu 2011 [41] | Parallel | T2D | Taiwan | Decaf green tea extract/placebo | FBG, A1c, F-IRI, IR | 68 | 16 | 51.3 | 35 | 29.8 | 173.0 |
| Belcaro 2013 [42] | Parallel | MetS | Italia | Decaf green tea extract/placebo | FBG | 98 | 24 | 46.5 | 50 | 31.0 | 115.9 |
| Liu 2014 [43] | Parallel | T2D with dyslipidemia | Taiwan | Decaf green tea extract/placebo | FBG, A1c, F-IRI, IR | 77 | 16 | 54.3 | 42 | 26.3 | 145.6 |
| Dostal 2016 [44] | Parallel | Obese women | US | Decaf green tea extract/placebo | FBG, F-IRI, IR | 237 | 52 | 60.7 | 0 | 28.2 | 97.4 |
| Lu 2016 [45] | Parallel | Women with acne | Taiwan | Decaf green tea extract/placebo | FBG | 64 | 4 | 29.1 | 0 | 21.2 | 87.6 |
| Hosoda 2003 [46] | Crossover | T2D | Taiwan | Oolong tea/water | FBG | 20 | 4 | 61.2 | 50 | 22.6 | NR |
| Shimada 2004 [47] | Crossover | CAD | Japan | Oolong tea/water | FBG, A1c | 22 | 4 | 64.3 | 77 | 23.0 | 170.5 |
| Intervention | Number of Studies in Pairwise Comparison | Number of Participants in Pairwise Comparison | Mean Difference (95% CI, mg/dL) | I2 (%) | Quality of Evidence |
|---|---|---|---|---|---|
| Coffee (pairwise) | 4 | 247 | 1.34 (−0.52 to 3.20) | 0.0 | Low a |
| Coffee (NMA) | 1.27 (−1.18 to 3.71) | ||||
| Decaffeinated coffee (pairwise) | 2 | 55 | 5.28 (−5.34 to 15.91) | 0.0 | Low b |
| Decaffeinated coffee (NMA) | 4.12 (−5.41 to 13.65) | ||||
| Black tea (pairwise) | 3 | 155 | −3.56 (−8.80 to 1.68) | 0.0 | Low c |
| Black tea (NMA) | −3.51 (−9.09 to 2.07) | ||||
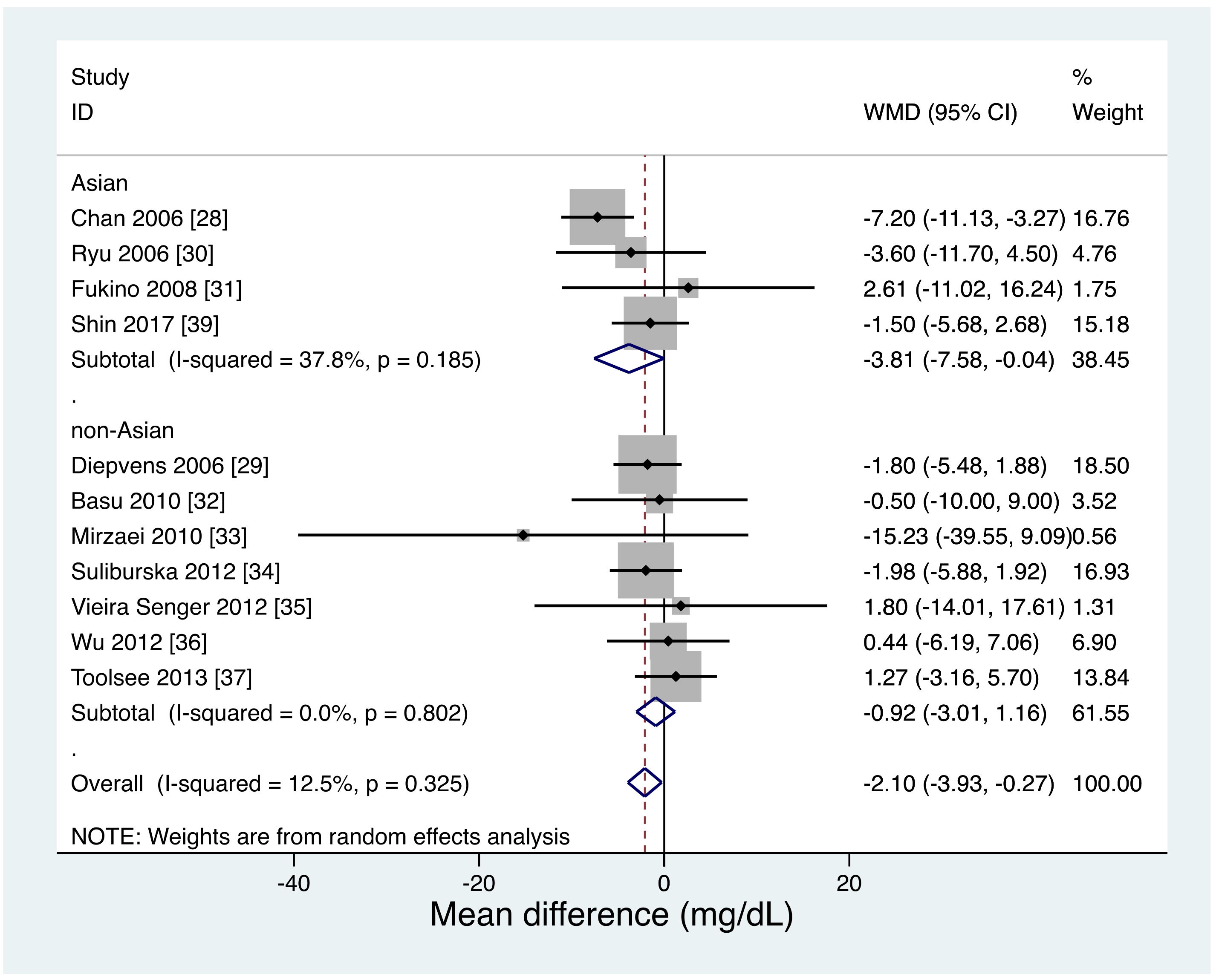
| Green tea (pairwise) | 11 | 786 | −2.10 (−3.93 to −0.27) | 12.5 | Moderate d |
| Green tea (NMA) | −2.09 (−3.96 to −0.24) | ||||
| Decaffeinated green tea (pairwise) | 6 | 610 | −0.23 (−2.84 to 2.38) | 0.0 | Low e |
| Decaffeinated green tea (NMA) | −0.44 (−3.53 to 2.64) | ||||
| Oolong tea (pairwise) | 2 | 42 | −38.72 (−100.56 to 23.11) | 87.2 | Very low f |
| Oolong tea (NMA) | −39.91 (−62.04 to −17.78) |
| Intervention | Number of Studies | Number of Intervention | Mean Difference (95% CI) (mg/dL) | I2 (%) | Quality of Evidence |
|---|---|---|---|---|---|
| Coffee (pairwise) | 2 | 61 | −23.99 (−63.78 to 15.81) | 66.4 | Very low a |
| Coffee (NMA) | −17.89 (−44.95 to 9.18) | ||||
| Decaffeinated coffee (pairwise) | 2 | 55 | 12.27 (−8.52 to 33.07) | 0.0 | Low b |
| Decaffeinated coffee (NMA) | 12.20 (−11.33 to 35.73) | ||||
| Green tea (pairwise) | 2 | 138 | −8.25 (−27.11 to 10.61) | 0.0 | Moderate c |
| Green tea (NMA) | −8.35 (−29.40 to 12.70) |
| Intervention | Number of Studies | Number of Interventions | Mean Difference (95% CI) (mg/dL) | I2 (%) | Quality of Evidence |
|---|---|---|---|---|---|
| Green tea (pairwise) | 6 | 504 | 0.00 (−0.15 to 0.16) | 23.8 | Low a |
| Green tea (NMA) | 0.02 (−0.09 to 0.12) | ||||
| Decaffeinated green tea (pairwise) | 2 | 145 | −0.08 (−0.67 to 0.51) | 0.0 | Moderate b |
| Decaffeinated green tea (NMA) | −0.08 (−0.67 to 0.51) |
| Intervention | Number of Studies | Number of Interventions | Mean Difference (95% CI) (mg/dL) | I2 (%) | Quality of Evidence |
|---|---|---|---|---|---|
| Coffee (pairwise) | 4 | 273 | 1.10 (0.17 to 2.03) | 0.0 | Low a |
| Coffee (NMA) | 1.10 (0.17 to 2.04) | ||||
| Decaffeinated coffee (pairwise) | 2 | 55 | 0.00 (−4.99 to 5.00) | 46.9 | Very low b |
| Decaffeinated coffee (NMA) | 1.24 (−1.70 to 4.18) | ||||
| Green tea (pairwise) | 5 | 469 | −0.11 (−0.67 to 0.45) | 0.0 | Low c |
| Green tea (NMA) | −0.11 (−0.67 to 0.45) | ||||
| Decaffeinated green tea (pairwise) | 4 | 510 | −0.51 (−2.16 to 1.13) | 45.7 | Moderate d |
| Decaffeinated green tea (NMA) | −0.02 (−0.83 to 0.80) |
| Intervention | Number of Studies | Number of Interventions | Mean Difference (95% CI) | I2 (%) | Quality of Evidence |
|---|---|---|---|---|---|
| Coffee (pairwise) | 2 | 61 | 0.04 (−0.75 to 0.83) | 0.0 | Low a |
| Coffee (NMA) | 0.05 (−0.79 to 0.89) | ||||
| Decaffeinated coffee (pairwise) | 2 | 55 | 0.14 (−1.13 to 1.41) | 35.0 | Very low b |
| Decaffeinated coffee (NMA) | 0.18 (−0.83 to 1.20) | ||||
| Green tea (pairwise) | 3 | 265 | −0.11 (−0.62 to 0.39) | 0.0 | Low c |
| Green tea (NMA) | −0.10 (−0.68 to 0.48) | ||||
| Decaffeinated green tea (pairwise) | 3 | 382 | −0.08 (−1.12 to 0.95) | 63.3 | Very low d |
| Decaffeinated green tea (NMA) | 0.01 (−0.63 to 0.66) |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kondo, Y.; Goto, A.; Noma, H.; Iso, H.; Hayashi, K.; Noda, M. Effects of Coffee and Tea Consumption on Glucose Metabolism: A Systematic Review and Network Meta-Analysis. Nutrients 2019, 11, 48. https://doi.org/10.3390/nu11010048
Kondo Y, Goto A, Noma H, Iso H, Hayashi K, Noda M. Effects of Coffee and Tea Consumption on Glucose Metabolism: A Systematic Review and Network Meta-Analysis. Nutrients. 2019; 11(1):48. https://doi.org/10.3390/nu11010048
Chicago/Turabian StyleKondo, Yoshinobu, Atsushi Goto, Hisashi Noma, Hiroyasu Iso, Kunihiko Hayashi, and Mitsuhiko Noda. 2019. "Effects of Coffee and Tea Consumption on Glucose Metabolism: A Systematic Review and Network Meta-Analysis" Nutrients 11, no. 1: 48. https://doi.org/10.3390/nu11010048
APA StyleKondo, Y., Goto, A., Noma, H., Iso, H., Hayashi, K., & Noda, M. (2019). Effects of Coffee and Tea Consumption on Glucose Metabolism: A Systematic Review and Network Meta-Analysis. Nutrients, 11(1), 48. https://doi.org/10.3390/nu11010048





