The Effect of Vitamin D Supplementation on Hepcidin, Iron Status, and Inflammation in Pregnant Women in the United Kingdom
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design and Participants
2.2. Anthropometry and Characteristics
2.3. Biochemical Analysis
2.4. Statistical Analysis
3. Results
3.1. Early Pregnancy (Pre-Supplementation)
3.2. Late Pregnancy
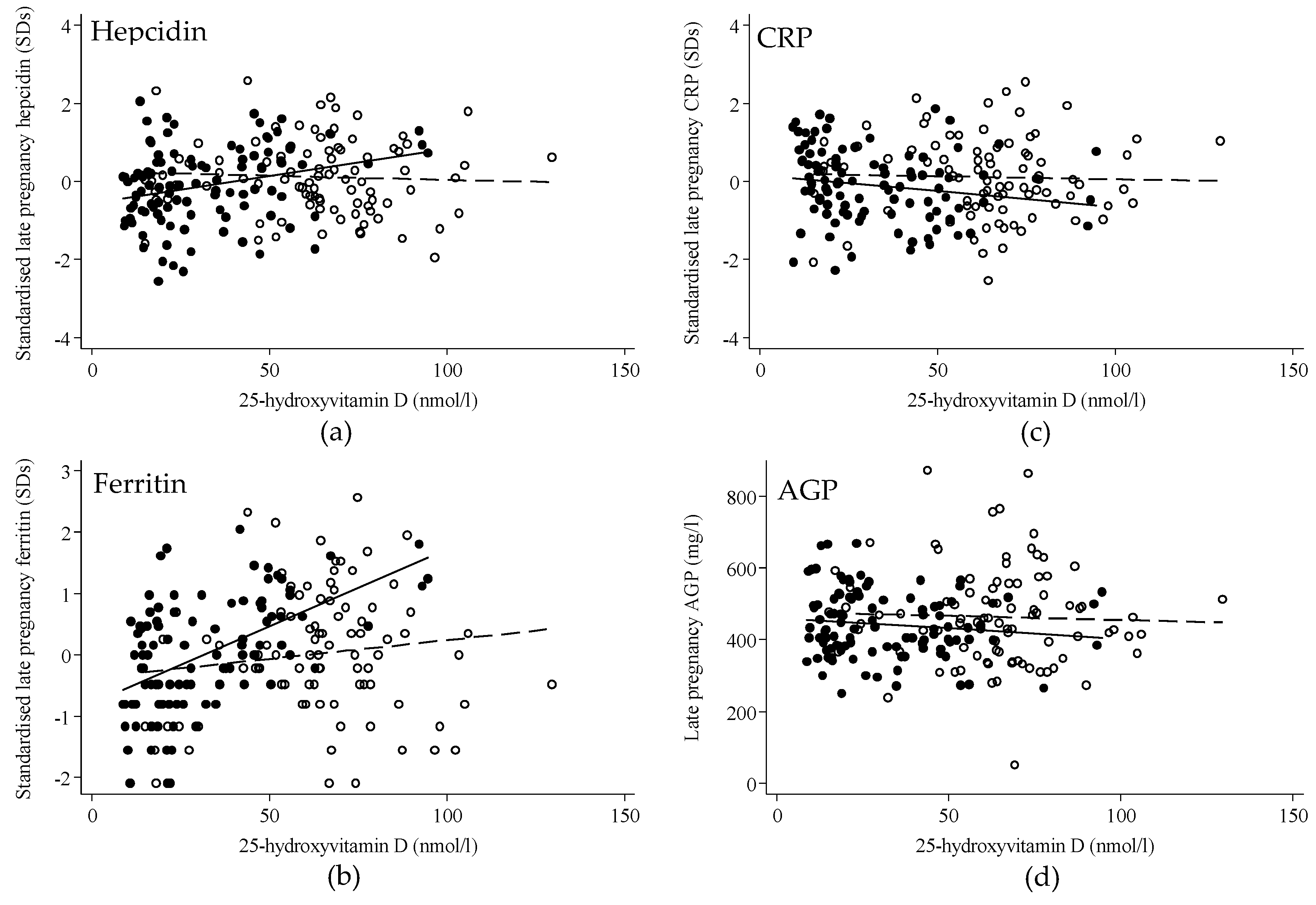
3.3. Regression Analysis
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Guillet, R.; O’Brien, K.; Thomas, C.E.; Queenan, R.A.; Cooper, E.M.; Kent, T.R.; Pressman, E.K.; Vermeylen, F.M.; Roberson, M.S.; O’Brien, K.O. Vitamin D status is inversely associated with anemia and serum erythropoietin during pregnancy. Am. J. Clin. Nutr. 2015, 102, 1088–1095. [Google Scholar]
- Nikooyeh, B.; Neyestani, T.R. Poor vitamin D status increases the risk of anemia in school children: National Food and Nutrition Surveillance. Nutrition 2018, 47, 69–74. [Google Scholar] [CrossRef] [PubMed]
- Frelut, M.-L.; Girardet, J.-P.; Bocquet, A.; Briend, A.; Chouraqui, J.-P.; Darmaun, D.; Dupont, C.; Feillet, F.; Hankard, R.; Rozé, J.-C.; et al. Impact of obesity on biomarkers of iron and vitamin D status in children and adolescents: The risk of misinterpretation. Archives de Pédiatrie 2018, 25, 3–5. [Google Scholar] [CrossRef] [PubMed]
- Michalski, E.S.; Nguyen, P.H.; Gonzalez-Casanova, I.; Nguyen, S.V.; Martorell, R.; Tangpricha, V.; Ramakrishnan, U. Serum 25-hydroxyvitamin D but not dietary vitamin D intake is associated with hemoglobin in women of reproductive age in rural northern Vietnam. J. Clin. Transl. Endocrinol. 2017, 8, 41–48. [Google Scholar] [CrossRef] [PubMed]
- Malczewska-Lenczowska, J.; Sitkowski, D.; Surała, O.; Orysiak, J.; Szczepańska, B.; Witek, K. The Association between Iron and Vitamin D Status in Female Elite Athletes. Nutrients 2018, 10, 167. [Google Scholar] [CrossRef] [PubMed]
- Ganz, T.; Nemeth, E. Hepcidin and iron homeostasis. Biochimica et Biophysica Acta (BBA)-Mol. Cell Res. 2012, 1823, 1434–1443. [Google Scholar] [CrossRef] [PubMed]
- Zughaier, S.M.; Alvarez, J.A.; Sloan, J.H.; Konrad, R.J.; Tangpricha, V. The role of vitamin D in regulating the iron-hepcidin-ferroportin axis in monocytes. J. Clin. Transl. Endocrinol. 2014, 1, e19–e25. [Google Scholar] [CrossRef]
- Bacchetta, J.; Zaritsky, J.J.; Sea, J.L.; Chun, R.F.; Lisse, T.S.; Zavala, K.; Nayak, A.; Wesseling-Perry, K.; Westerman, M.; Hollis, B.W.; et al. Suppression of Iron-Regulatory Hepcidin by Vitamin D. J. Am. Soc. Nephrol. 2013, 25, 564–572. [Google Scholar] [CrossRef]
- Smith, E.M.; Alvarez, J.A.; Kearns, M.D.; Hao, L.; Sloan, J.H.; Konrad, R.J.; Ziegler, T.R.; Zughaier, S.M.; Tangpricha, V. High-dose vitamin D 3 reduces circulating hepcidin concentrations: A pilot, randomized, double-blind, placebo-controlled trial in healthy adults. Clin. Nutr. 2017, 36, 980–985. [Google Scholar] [CrossRef]
- Laird, E.; McNulty, H.; Ward, M.; Hoey, L.; McSorley, E.; Wallace, J.M.W.; Carson, E.; Molloy, A.M.; Healy, M.; Casey, M.C.; et al. Vitamin D Deficiency Is Associated with Inflammation in Older Irish Adults. J. Clin. Endocrinol. Metab. 2014, 99, 1807–1815. [Google Scholar] [CrossRef]
- Calton, E.K.; Keane, K.N.; Newsholme, P.; Zhao, Y.; Soares, M.J. The impact of cholecalciferol supplementation on the systemic inflammatory profile: A systematic review and meta-analysis of high-quality randomized controlled trials. Eur. J. Clin. Nutr. 2017, 71, 931–943. [Google Scholar] [CrossRef] [PubMed]
- Vanherwegen, A.-S.; Gysemans, C.; Mathieu, C. Vitamin D endocrinology on the cross-road between immunity and metabolism. Mol. Cell. Endocrinol. 2017, 453, 52–67. [Google Scholar] [CrossRef] [PubMed]
- WHO. The Global Prevalence of Anaemia 2011; WHO: Geneva, Switzerland, 2015. [Google Scholar]
- Stevens, G.A.; Finucane, M.M.; De-Regil, L.M.; Paciorek, C.J.; Flaxman, S.R.; Branca, F.; Peña-Rosas, J.P.; Bhutta, Z.A.; Ezzati, M. Global, regional, and national trends in haemoglobin concentration and prevalence of total and severe anaemia in children and pregnant and non-pregnant women for 1995–2011: A systematic analysis of population-representative data. Lancet Glob. Health 2013, 1, e16–e25. [Google Scholar] [CrossRef]
- Allen, L.H. Anemia and iron deficiency: Effects on pregnancy outcome. Am. J. Clin. Nutr. 2000, 71, 1280S–1284S. [Google Scholar] [CrossRef] [PubMed]
- Haider, B.A.; Olofin, I.; Wang, M.; Spiegelman, D.; Ezzati, M.; Fawzi, W.W. Anaemia, prenatal iron use, and risk of adverse pregnancy outcomes: Systematic review and meta-analysis. BMJ 2013, 346, f3443. [Google Scholar] [CrossRef] [PubMed]
- Bah, A.; Pasricha, S.-R.; Jallow, M.W.; A Sise, E.; Wegmuller, R.; E Armitage, A.; Drakesmith, H.; Moore, S.E.; Prentice, A.M. Serum Hepcidin Concentrations Decline during Pregnancy and May Identify Iron Deficiency: Analysis of a Longitudinal Pregnancy Cohort in The Gambia. J. Nutr. 2017, 147, 1131–1137. [Google Scholar] [CrossRef] [PubMed]
- Watts, D.H.; Krohn, M.A.; Wener, M.H.; Eschenbach, D.A. C-Reactive Protein in Normal Pregnancy. Obstet. Gynecol. 1991, 77, 176–180. [Google Scholar] [CrossRef] [PubMed]
- Koenig, M.D.; Tussing-Humphreys, L.; Day, J.; Cadwell, B.; Nemeth, E. Hepcidin and Iron Homeostasis during Pregnancy. Nutrients 2014, 6, 3062–3083. [Google Scholar] [CrossRef]
- Harvey, N.C.; Javaid, K.; Bishop, N.; Kennedy, S.; Papageorghiou, A.T.; Fraser, R.; Gandhi, S.V.; Schoenmakers, I.; Prentice, A.; Cooper, C.; et al. MAVIDOS Maternal Vitamin D Osteoporosis Study: Study protocol for a randomized controlled trial. The MAVIDOS Study Group. Trials 2012, 13, 13. [Google Scholar] [CrossRef]
- Cooper, C.; Harvey, N.C.; Bishop, N.J.; Kennedy, S.; Papageorghiou, A.T.; Schoenmakers, I.; Fraser, R.; Gandhi, S.V.; Carr, A.; D’Angelo, S.; et al. Maternal gestational vitamin D supplementation and offspring bone health (MAVIDOS): A multicentre, double-blind, randomised placebo-controlled trial. Lancet Diabetes Endocrinol. 2016, 4, 393–402. [Google Scholar] [CrossRef]
- Webb, A.; Kazantzidis, A.; Kift, R.; Farrar, M.; Wilkinson, J.; Rhodes, L. Colour Counts: Sunlight and Skin Type as Drivers of Vitamin D Deficiency at UK Latitudes. Nutrients 2018, 10, 457. [Google Scholar] [CrossRef] [PubMed]
- Armitage, P.; Berry, G.; Matthews, J.N.S. Statistical Methods in Medical Research; Wiley: Hoboken, NJ, USA, 2002. [Google Scholar]
- Namaste, S.M.; Rohner, F.; Huang, J.; Bhushan, N.L.; Flores-Ayala, R.; Kupka, R.; Mei, Z.; Rawat, R.; Williams, A.M.; Raiten, D.J.; et al. Adjusting ferritin concentrations for inflammation: Biomarkers Reflecting Inflammation and Nutritional Determinants of Anemia (BRINDA) project. Am. J. Clin. Nutr. 2017, 106, 359–371. [Google Scholar]
- WHO. Serum Ferritin Concentrations for the Assessment of Iron Status and Iron Deficiency in Populations. Vitamin and Mineral Nutrition Information System 2011 [cited 2017 24/10/2017]. Available online: http://www.who.int/vmnis/indicators/serum_ferritin/en (accessed on 16 January 2019).
- Roth, D.E.; Abrams, S.A.; Aloia, J.; Bergeron, G.; Bourassa, M.W.; Brown, K.H.; Calvo, M.S.; Cashman, K.D.; Combs, G.; De-Regil, L.M.; et al. Global prevalence and disease burden of vitamin D deficiency: A roadmap for action in low- and middle-income countries. Ann. N. Y. Acad. Sci. 2018, 1430, 44–79. [Google Scholar] [CrossRef] [PubMed]
- Mwangi, M.N.; Roth, J.M.; Smit, M.R.; Trijsburg, L.; Mwangi, A.M.; Demir, A.Y.; Wielders, J.P.; Mens, P.F.; Verweij, J.J.; Cox, S.E.; et al. Effect of Daily Antenatal Iron Supplementation on Plasmodium Infection in Kenyan Women: A Randomized Clinical Trial. JAMA 2015, 314, 1009–1020. [Google Scholar] [CrossRef] [PubMed]
- Cao, C.; O’Brien, K. Pregnancy and iron homeostasis: An update. Nutr. Rev. 2013, 71, 35–51. [Google Scholar] [CrossRef] [PubMed]
- Jones, K.S.; Assar, S.; Prentice, A.; Schoenmakers, I. Vitamin D expenditure is not altered in pregnancy and lactation despite changes in vitamin D metabolite concentrations. Sci. Rep. 2016, 6, 26795. [Google Scholar] [CrossRef] [PubMed]
- Schoenmakers, I.; Jones, K.S. Chapter 37—Pharmacology and Pharmacokinetics, in Vitamin D, 4th ed.; Feldman, D., Ed.; Academic Press: Cambridge, MA, USA, 2018; pp. 635–661. [Google Scholar]
- Byg, K.-E.; Milman, N.; Hansen, S.; Agger, A.O. Serum Ferritin is a Reliable, Non-invasive Test for Iron Status in Pregnancy: Comparison of Ferritin with Other Iron Status Markers in a Longitudinal Study on Healthy Pregnant Women; Erythropoiesis. Hematology 2000, 5, 319–325. [Google Scholar] [CrossRef] [PubMed]
- Daru, J.; Colman, K.; Stanworth, S.J.; De La Salle, B.; Wood, E.M.; Pasricha, S.-R. Serum ferritin as an indicator of iron status: What do we need to know? Am. J. Clin. Nutr. 2017, 106, 1634S–1639S. [Google Scholar] [CrossRef]
| Measures in Early Pregnancy | All | Placebo | Vitamin D3 |
|---|---|---|---|
| n = (195) | n = (102) | n = (93) | |
| White ethnicity, n (%) | 182 (94%) | 95 (93%) | 87 (95%) |
| Previous children, median (IQR) | 1 (0, 1) | 1 (0, 1) | 1 (0, 1) |
| Nulliparous, n (%) | 91 (47%) | 49 (49%) | 42 (46%) |
| Age (years), mean (SD) | 30.4 (5.2) | 30.8 (5.2) | 29.9 (5.2) |
| Gestation (weeks), mean (SD) | 15.7 (1.0) | 15.8 (1.0) | 15.6 (1.0) |
| Weight (kg), median (IQR) | 69.6 (61.5, 80.0) | 70.5 (62.4, 79.6) | 68.0 (60.7, 81.0) |
| Height (m), mean (SD) | 1.66 (0.07) | 1.66 (0.07) | 1.66 (0.07) |
| BMI (kg/m2), median (IQR) | 24.7 (22.1, 28.7) | 24.9 (22.0, 28.8) | 24.6 (22.1, 28.6) |
| Supplement use, n (%) | 178 (92%) | 92 (90%) | 86 (93%) |
| Iron supplement use, n (% of those using supplements) | 106 (60%) | 49 (53%) | 57 (66%) |
| CRP (mg/L), median (IQR) | 5.4 (3.1, 8.3) | 4.2 (2.9, 7.8) | 6.2 (3.6, 11.5) |
| CRP > 5 mg/L, n (%) | 103 (53%) | 48 (47%) | 55 (60%) |
| AGP (mg/L), mean (SD) | 506 (115) | 500 (109) | 512 (122) |
| AGP > 500 mg/L, n (%) | 96 (49%) | 50 (49%) | 46 (49%) |
| Hepcidin (μg/L), median (IQR) | 7.3 (3.0, 16.7) | 7.4 (2.5, 16.5) | 6.9 (3.3, 16.8) |
| Ferritin (μg/L), median (IQR) | 38 (25, 60) | 43 (26, 62) | 35 (25, 55) |
| Ferritin < 15 μg/L, n (%) | 19 (9.7%) | 11 (10.8%) | 8 (8.6%) |
| 25-hydroxyvitamin D (nmol/L), mean (SD) | 44.1 (16.0) | 42.5 (15.8) | 45.7 (16.2) |
| 25-hydroxyvitamin D < 25 nmol/L, n (%) | 23 (12%) | 15 (15%) | 8 (9%) |
| 25-hydroxyvitamin D < 50 nmol/L, n (%) | 128 (66%) | 70 (69%) | 58 (62%) |
| Measures in Late Pregnancy | All | Placebo | Vitamin D3 | p-Value |
|---|---|---|---|---|
| n = (195) | n = (102) | n = (93) | ||
| Age (years), mean (SD) | 30.6 (5.3) | 31.2 (5.2) d | 30.0 (5.3) d | 0.13 |
| Gestation (weeks), mean (SD) | 34.7 (0.8) | 34.6 (0.6) d | 34.8 (0.9) d | 0.06 |
| Weight (kg), median (IQR) | 78.8 (71.1, 90.4) | 78.5 (73.2, 90.0) d | 79.2 (69.9, 92.8) d | 0.69 |
| BMI (kg/m2), median (IQR) | 28.4 (25.5, 32.9) | 28.6 (25.5, 32.5) d | 28.3 (25.4, 32.9) d | 0.71 |
| Supplement use, n (%) | 119 (62%) | 61 (61%) d | 58 (63%) d | 0.77 |
| Iron supplement use, n (% of those using supplements) | 111 (93%) | 54 (89%) a | 57 (98%) b | 0.03 |
| CRP (mg/L), median (IQR) | 3.9 (2.5, 7.3) | 3.8 (2.3, 6.4) c | 4.1 (2.6, 8.1) c | 0.16 |
| CRP > 5 mg/L, n (%) | 69 (38%) | 31 (32%) b | 38 (43%) b | 0.13 |
| AGP (mg/L), mean (SD) | 452 (115) | 442 (93) d | 463 (135) b | 0.20 |
| AGP > 500 mg/L, n (%) | 57 (29%) | 28 (27%) c | 29 (32%) b | 0.53 |
| Hepcidin (μg/L), median (IQR) | 0.97 (0.79, 1.99) | 0.93 (0.74, 1.57) d | 0.99 (0.84, 2.30) d | 0.19 |
| Ferritin (μg/L), median (IQR) | 10 (7, 18) | 10 (7, 17) d | 10 (8, 18) d | 0.94 |
| Ferritin < 15 μg/L, n (%) | 129 (67%) | 65 (64%) d | 64 (70%) d | 0.44 |
| 25-hydroxyvitamin D (nmol/L), median (IQR) | 47.1 (21.4, 66.8) | 25.2 (16.9, 45.8) d | 64.6 (52.0, 75.7) d | 0.0001 |
| 25-hydroxyvitamin D < 25 nmol/L, n (%) | 61 (31%) | 51 (50%) d | 10 (11%) | 0.001 |
| 25-hydroxyvitamin D < 50 nmol/L, n (%) | 107 (55%) | 85 (83%) b | 22 (24%) d | 0.001 |
| Change in Characteristic | All | Placebo | Vitamin D3 | p-Value |
|---|---|---|---|---|
| n = (195) | n = (102) | n = (93) | ||
| Maternal pregnancy weight (kg), mean (SD) | 9.8 (3.6) | 9.7 (3.5) | 9.8 (3.8) | 0.65 |
| Maternal pregnancy BMI (kg/m2), mean (SD) | 3.6 (1.3) | 3.6 (1.2) | 3.5 (1.4) | 0.44 |
| CRP (mg/L), mean (SD) | −1.6 (10.3) | −1.8 (9.4) | −1.4 (11.2) | 0.12 |
| AGP (mg/L), mean (SD) | −54 (126) | −58 (101) | −50 (149) | 0.76 |
| Hepcidin (μg/L), median (IQR) | −5.1 (−14.1, −1.3) | −5.6 (−14.9, −1.1) | −4.3 (−11.0, −1.6) | 0.10 |
| Ferritin (μg/L), mean (SD) | −31.9 (58.5) | −36.5 (41.2) | −26.9 (72.9) | 0.67 |
| 25-hydroxyvitamin D (nmol/L), mean (SD) | 2.6 (27.1) | −10.7 (17.9) | 17.1 (28.0) | <0.0001 |
| All Women (n = 195) | Beta (95% CI) | p-Value | n | Interaction p-Value * |
|---|---|---|---|---|
| Early pregnancy: hepcidin (SDs) | 0.02 (−0.07, 0.11) | 0.67 | 195 | 0.99 |
| Late pregnancy: hepcidin (SDs) | 0.06 (0.01, 0.11) | 0.03 | 195 | 0.02 |
| Early pregnancy: ferritin (SDs) | 0.06 (−0.02, 0.15) | 0.16 | 195 | 0.85 |
| Late pregnancy: ferritin (SDs) | 0.09 (0.04, 0.15) | <0.001 | 193 | 0.003 |
| Early pregnancy: CRP (SDs) | −0.10 (−0.19, −0.02) | 0.02 | 193 | 0.23 |
| Late pregnancy: CRP (SDs) | −0.01 (−0.06, 0.05) | 0.85 | 184 | 0.32 |
| Early pregnancy: AGP (mg/L) | −5.3 (−15.4, 4.9) | 0.31 | 195 | 0.06 |
| Late pregnancy: AGP (mg/L) | −0.1 (6.4, 6.2) | 0.97 | 194 | 0.66 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Braithwaite, V.S.; Crozier, S.R.; D’Angelo, S.; Prentice, A.; Cooper, C.; Harvey, N.C.; Jones, K.S.; the MAVIDOS Trial Group. The Effect of Vitamin D Supplementation on Hepcidin, Iron Status, and Inflammation in Pregnant Women in the United Kingdom. Nutrients 2019, 11, 190. https://doi.org/10.3390/nu11010190
Braithwaite VS, Crozier SR, D’Angelo S, Prentice A, Cooper C, Harvey NC, Jones KS, the MAVIDOS Trial Group. The Effect of Vitamin D Supplementation on Hepcidin, Iron Status, and Inflammation in Pregnant Women in the United Kingdom. Nutrients. 2019; 11(1):190. https://doi.org/10.3390/nu11010190
Chicago/Turabian StyleBraithwaite, Vickie S., Sarah R. Crozier, Stefania D’Angelo, Ann Prentice, Cyrus Cooper, Nicholas C. Harvey, Kerry S. Jones, and the MAVIDOS Trial Group. 2019. "The Effect of Vitamin D Supplementation on Hepcidin, Iron Status, and Inflammation in Pregnant Women in the United Kingdom" Nutrients 11, no. 1: 190. https://doi.org/10.3390/nu11010190
APA StyleBraithwaite, V. S., Crozier, S. R., D’Angelo, S., Prentice, A., Cooper, C., Harvey, N. C., Jones, K. S., & the MAVIDOS Trial Group. (2019). The Effect of Vitamin D Supplementation on Hepcidin, Iron Status, and Inflammation in Pregnant Women in the United Kingdom. Nutrients, 11(1), 190. https://doi.org/10.3390/nu11010190





