Gluten-Free Diet: Gaps and Needs for a Healthier Diet
Abstract
1. Introduction
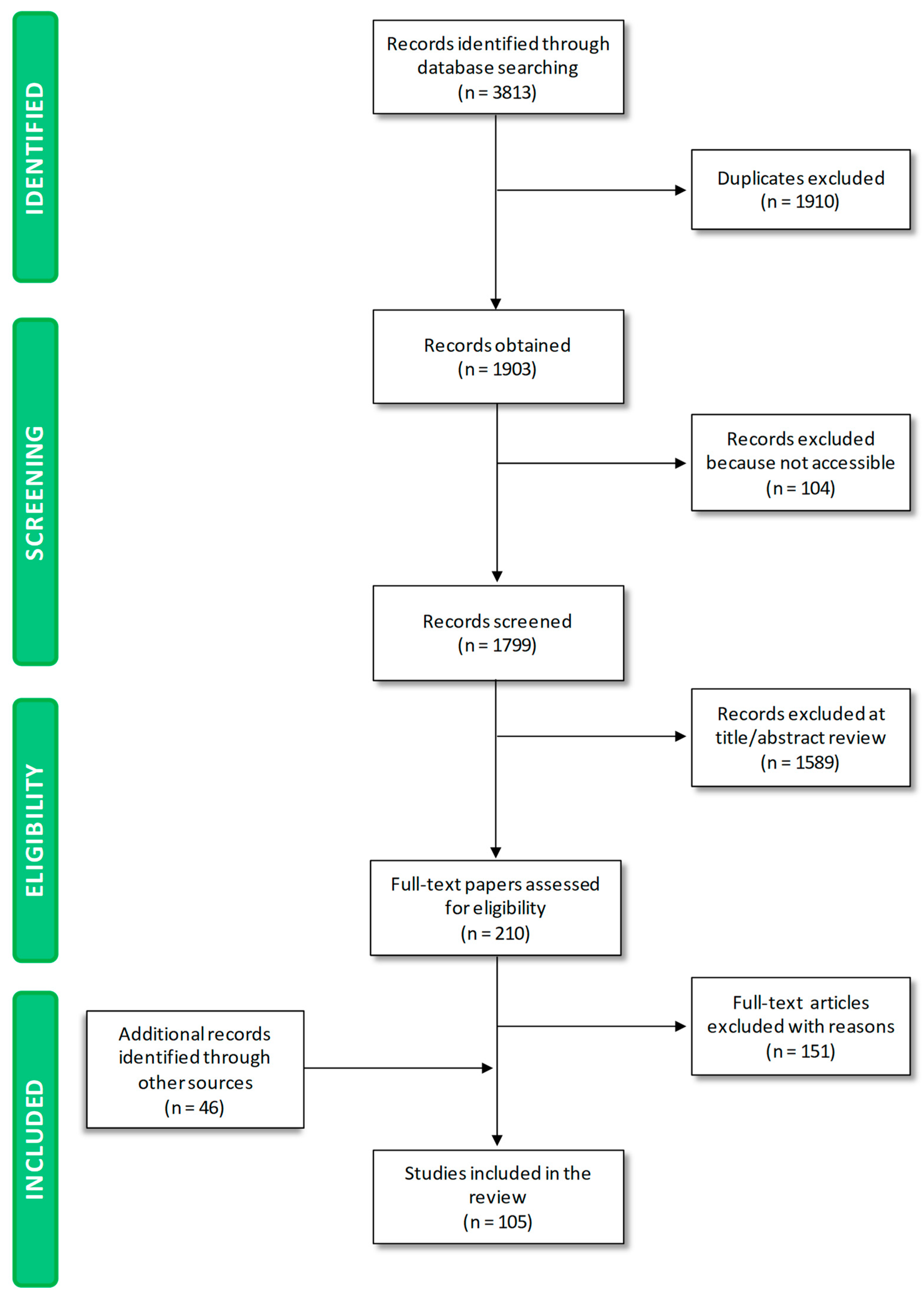
2. Materials and Methods
2.1. Literature Search
2.2. Including and Excluding Criteria
2.3. Software
3. Results and Discussion
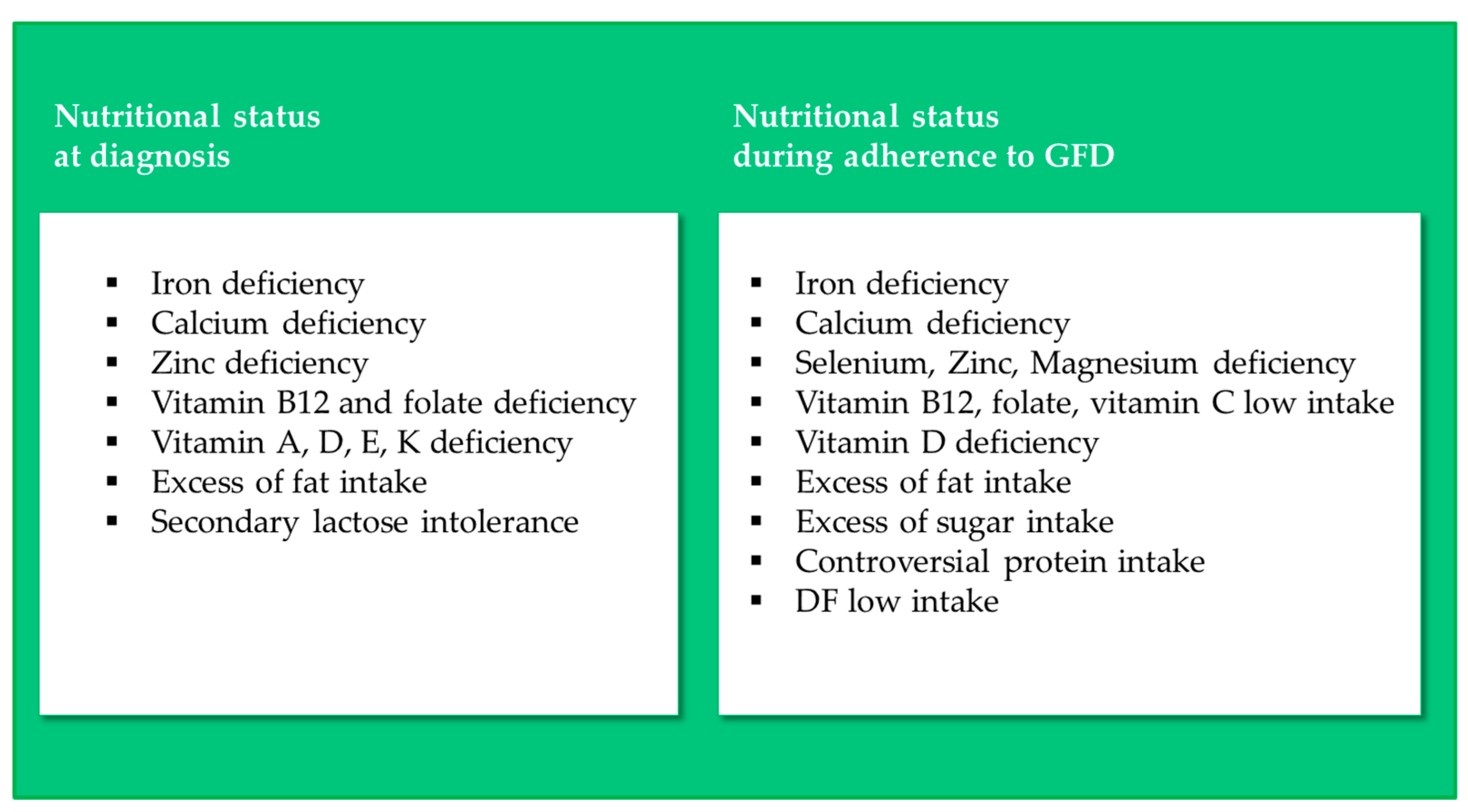
3.1. Nutritional Status of CD Patients at Diagnosis
3.2. Nutritional Status of CD Patients Adhering to a GFD
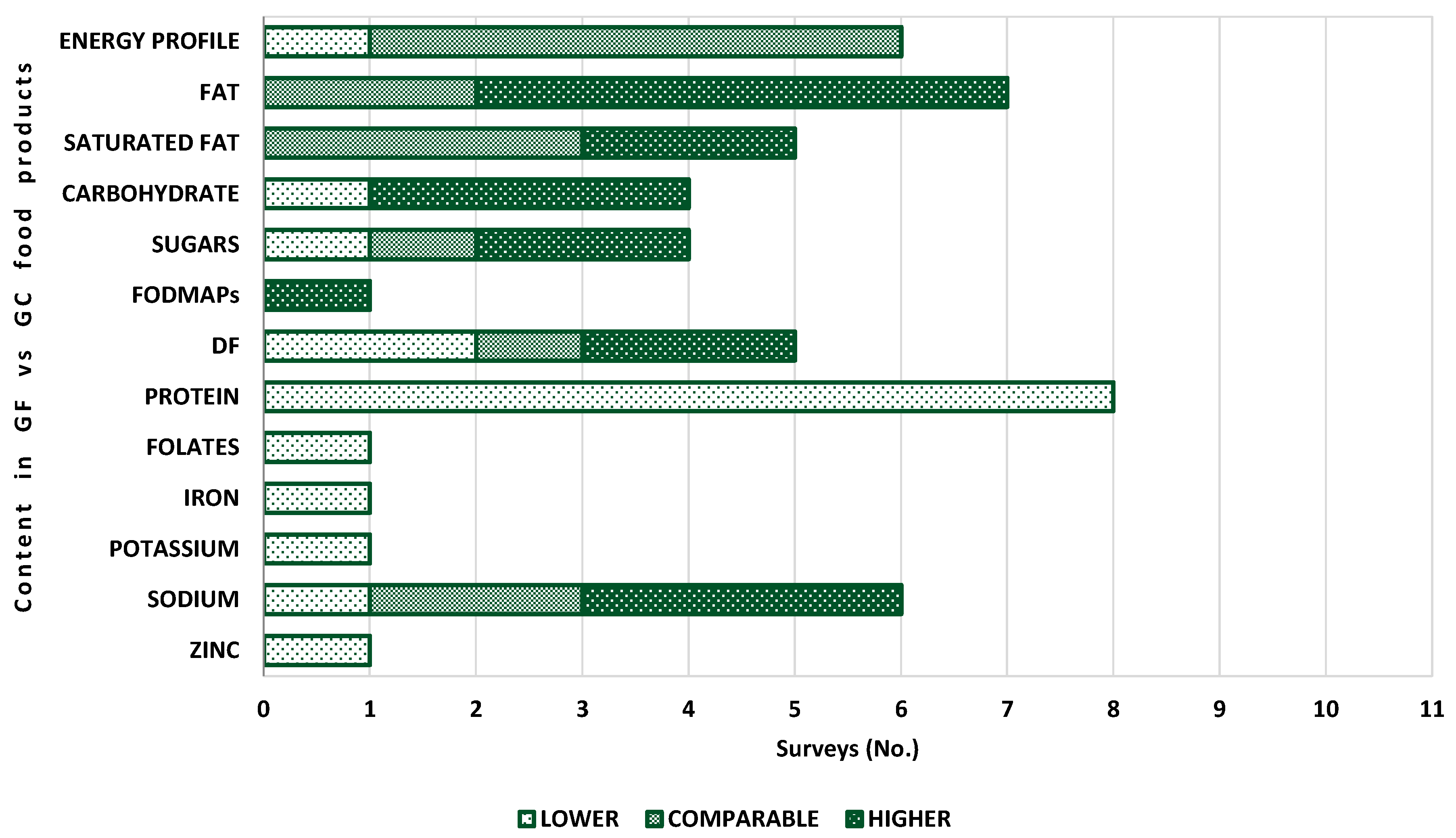
3.3. Nutritional Profile of Gluten-Containing and GF Food Products
3.4. GFD Adherence, Cardiovascular Disease and Metabolic Syndrome
3.5. Improving GFD for Gut Microbiota Recovery
3.6. Limitations of the Review
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Healthy Diet. Available online: https://www.who.int/news-room/fact-sheets/detail/healthy-diet (accessed on 27 December 2018).
- Standards | CODEXALIMENTARIUS FAO-WHO. Available online: http://www.fao.org/fao-who-codexalimentarius/codex-texts/list-standards/jp/ (accessed on 12 December 2018).
- EU law—EUR-Lex. Available online: https://eur-lex.europa.eu/homepage.html (accessed on 12 December 2018).
- Leonard, M.M.; Sapone, A.; Catassi, C.; Fasano, A. Celiac Disease and Nonceliac Gluten Sensitivity: A Review. JAMA 2017, 318, 647–656. [Google Scholar] [CrossRef]
- Lionetti, E.; Gatti, S.; Pulvirenti, A.; Catassi, C. Celiac disease from a global perspective. Best Pract. Res. Clin. Gastroenterol. 2015, 29, 365–379. [Google Scholar] [CrossRef] [PubMed]
- Freeman, H.J. Adult Celiac Disease and Its Malignant Complications. Gut Liver 2009, 3, 237–246. [Google Scholar] [CrossRef] [PubMed]
- Halfdanarson, T.R.; Litzow, M.R.; Murray, J.A. Hematologic manifestations of celiac disease. Blood 2007, 109, 412–421. [Google Scholar] [CrossRef] [PubMed]
- McPhillips, J. Understanding coeliac disease: Symptoms and long-term risks. Br. J. Nurs. 2000, 9, 479–483. [Google Scholar] [CrossRef] [PubMed]
- Manufacturers and Retailers. Available online: https://www.coeliac.org.uk/food-industry-professionals/manufacturers-and-retailers/ (accessed on 12 December 2018).
- Panagiotou, S.; Kontogianni, M.D. The economic burden of gluten-free products and gluten-free diet: A cost estimation analysis in Greece. J. Hum. Nutr. Diet. 2017, 30, 746–752. [Google Scholar] [CrossRef]
- Panagiotou, S.; Kontogianni, M. A cost comparison between gluten – free and conventional foods. Clin. Nutr. ESPEN 2016, 13, e56. [Google Scholar] [CrossRef]
- Lee, A.R.; Lebwohl, B.; Ciaccio, E.; Wolf, R.; Ellis, E.; Green, P.H.R. Persistent Economic Burden of the Gluten Free Diet. Gastroenterology 2017, 152, S265–S266. [Google Scholar] [CrossRef]
- Pourhoseingholi, M.A.; Rostami-Nejad, M.; Barzegar, F.; Rostami, K.; Volta, U.; Sadeghi, A.; Honarkar, Z.; Salehi, N.; Asadzadeh-Aghdaei, H.; Baghestani, A.R.; et al. Economic burden made celiac disease an expensive and challenging condition for Iranian patients. Gastroenterol. Hepatol. Bed Bench 2017, 10, 258–262. [Google Scholar]
- Burden, M.; Mooney, P.D.; Blanshard, R.J.; White, W.L.; Cambray-Deakin, D.R.; Sanders, D.S. Cost and availability of gluten-free food in the UK: In store and online. Postgrad. Med. J. 2015, 91, 622–626. [Google Scholar] [CrossRef]
- Silvester, J.A.; Weiten, D.; Graff, L.A.; Walker, J.R.; Duerksen, D.R. Living gluten-free: Adherence, knowledge, lifestyle adaptations and feelings towards a gluten-free diet. J. Hum. Nutr. Diet. 2016, 29, 374–382. [Google Scholar] [CrossRef] [PubMed]
- Theethira, T.; Dennis, M.; Leffler, D. Nutritional consequences of celiac disease and the gluten-free diet. Expert Rev. Gastroenterol. Hepatol. 2014, 8, 123–129. [Google Scholar] [CrossRef] [PubMed]
- Welstead, L. The Gluten-Free Diet in the 3rd Millennium: Rules, Risks and Opportunities. Diseases 2015, 3, 136–149. [Google Scholar] [CrossRef]
- Grace-Farfaglia, P. Bones of Contention: Bone Mineral Density Recovery in Celiac Disease—A Systematic Review. Nutrients 2015, 7, 3347–3369. [Google Scholar] [CrossRef] [PubMed]
- Krupa-Kozak, U. Pathologic bone alterations in celiac disease: Etiology, epidemiology, and treatment. Nutrition 2014, 30, 16–24. [Google Scholar] [CrossRef] [PubMed]
- Wierdsma, N.J.; van Bokhorst-de van der Schueren, M.A.E.; Berkenpas, M.; Mulder, C.J.J.; van Bodegraven, A.A. Vitamin and Mineral Deficiencies Are Highly Prevalent in Newly Diagnosed Celiac Disease Patients. Nutrients 2013, 5, 3975–3992. [Google Scholar] [CrossRef] [PubMed]
- Dickey, W.; Ward, M.; Whittle, C.R.; Kelly, M.T.; Pentieva, K.; Horigan, G.; Patton, S.; McNulty, H. Homocysteine and related B-vitamin status in coeliac disease: Effects of gluten exclusion and histological recovery. Scand. J. Gastroenterol. 2008, 43, 682–688. [Google Scholar] [CrossRef]
- Valente, F.X.; do Nascimento Campos, T.; de Sousa Moraes, L.F.; Hermsdorff, H.H.M.; de Morais Cardoso, L.; Pinheiro-Sant’Ana, H.M.; Gilberti, F.A.B.; Peluzio, M.D.C.G. B vitamins related to homocysteine metabolism in adults celiac disease patients: A cross-sectional study. Nutr. J. 2015, 14, 110. [Google Scholar] [CrossRef]
- Gopalsamy, G.L.; Alpers, D.H.; Binder, H.J.; Tran, C.D.; Ramakrishna, B.S.; Brown, I.; Manary, M.; Mortimer, E.; Young, G.P. The Relevance of the Colon to Zinc Nutrition. Nutrients 2015, 7, 572–583. [Google Scholar] [CrossRef]
- Walker, W.A.; Duggan, C.P.; Watkins, J.B. Nutrition in Pediatrics: Basic Science and Clinical Applications; PMPH-USA: Shelton, CT, USA, 2003; ISBN 978-1-55009-226-4. [Google Scholar]
- Kupper, C. Dietary guidelines and implementation for celiac disease. Gastroenterology 2005, 128, S121–S127. [Google Scholar] [CrossRef]
- Braamskamp, M.J.A.M.; Dolman, K.M.; Tabbers, M.M. Clinical practice. Eur. J. Pediatr. 2010, 169, 1179–1185. [Google Scholar] [CrossRef] [PubMed]
- Shepherd, S.J.; Gibson, P.R. Nutritional inadequacies of the gluten-free diet in both recently-diagnosed and long-term patients with coeliac disease. J. Hum. Nutr. Diet. 2013, 26, 349–358. [Google Scholar] [CrossRef] [PubMed]
- Ciacci, C.; Ciclitira, P.; Hadjivassiliou, M.; Kaukinen, K.; Ludvigsson, J.F.; McGough, N.; Sanders, D.S.; Woodward, J.; Leonard, J.N.; Swift, G.L. The gluten-free diet and its current application in coeliac disease and dermatitis herpetiformis. United Eur. Gastroenterol. J. 2015, 3, 121–135. [Google Scholar] [CrossRef] [PubMed]
- Sue, A.; Dehlsen, K.; Ooi, C.Y. Paediatric Patients with Coeliac Disease on a Gluten-Free Diet: Nutritional Adequacy and Macro- and Micronutrient Imbalances. Curr. Gastroenterol. Rep. 2018, 20, 2. [Google Scholar] [CrossRef] [PubMed]
- Larretxi, I.; Simon, E.; Benjumea, L.; Miranda, J.; Bustamante, M.A.; Lasa, A.; Eizaguirre, F.J.; Churruca, I. Gluten-free-rendered products contribute to imbalanced diets in children and adolescents with celiac disease. Eur. J. Nutr. 2018. [Google Scholar] [CrossRef] [PubMed]
- Babio, N.; Alcázar, M.; Castillejo, G.; Recasens, M.; Martínez-Cerezo, F.; Gutiérrez-Pensado, V.; Masip, G.; Vaqué, C.; Vila-Martí, A.; Torres-Moreno, M.; et al. Patients With Celiac Disease Reported Higher Consumption of Added Sugar and Total Fat Than Healthy Individuals. J. Pediatr. Gastroenterol. Nutr. 2017, 64, 63–69. [Google Scholar] [CrossRef]
- Martin, J.; Geisel, T.; Maresch, C.; Krieger, K.; Stein, J. Inadequate nutrient intake in patients with celiac disease: Results from a German dietary survey. Digestion 2013, 87, 240–246. [Google Scholar] [CrossRef]
- Öhlund, K.; Olsson, C.; Hernell, O.; Öhlund, I. Dietary shortcomings in children on a gluten-free diet. J. Hum. Nutr. Diet. 2010, 23, 294–300. [Google Scholar] [CrossRef]
- Hopman, E.G.D.; le Cessie, S.; von Blomberg, B.M.E.; Mearin, M.L. Nutritional Management of the Gluten-free Diet in Young People with Celiac Disease in The Netherlands. J. Pediatr. Gastroenterol. Nutr. 2006, 43, 102–108. [Google Scholar] [CrossRef]
- Bardella, M.T.; Fredella, C.; Prampolini, L.; Molteni, N.; Giunta, A.M.; Bianchi, P.A. Body composition and dietary intakes in adult celiac disease patients consuming a strict gluten-free diet. Am. J. Clin. Nutr. 2000, 72, 937–939. [Google Scholar] [CrossRef]
- Mariani, P.; Viti, M.G.; Montuori, M.; La Vecchia, A.; Cipolletta, E.; Calvani, L.; Bonamico, M. The gluten-free diet: A nutritional risk factor for adolescents with celiac disease? J. Pediatr. Gastroenterol. Nutr. 1998, 27, 519–523. [Google Scholar] [CrossRef] [PubMed]
- Zuccotti, G.; Fabiano, V.; Dilillo, D.; Picca, M.; Cravidi, C.; Brambilla, P. Intakes of nutrients in Italian children with celiac disease and the role of commercially available gluten-free products. J. Hum. Nutr. Diet. 2013, 26, 436–444. [Google Scholar] [CrossRef] [PubMed]
- van Hees, N.J.M.; Giltay, E.J.; Tielemans, S.M.A.J.; Geleijnse, J.M.; Puvill, T.; Janssen, N.; van der Does, W. Essential Amino Acids in the Gluten-Free Diet and Serum in Relation to Depression in Patients with Celiac Disease. PLoS ONE 2015, 10, e0122619. [Google Scholar]
- Barone, M.; Della Valle, N.; Rosania, R.; Facciorusso, A.; Trotta, A.; Cantatore, F.P.; Falco, S.; Pignatiello, S.; Viggiani, M.T.; Amoruso, A.; et al. A comparison of the nutritional status between adult celiac patients on a long-term, strictly gluten-free diet and healthy subjects. Eur. J. Clin. Nutr. 2016, 70, 23–27. [Google Scholar] [CrossRef] [PubMed]
- Wild, D.; Robins, G.G.; Burley, V.J.; Howdle, P.D. Evidence of high sugar intake, and low fibre and mineral intake, in the gluten-free diet. Aliment. Pharmacol. Ther. 2010, 32, 573–581. [Google Scholar] [CrossRef]
- Thompson, T.; Dennis, M.; Higgins, L.A.; Lee, A.R.; Sharrett, M.K. Gluten-free diet survey: Are Americans with coeliac disease consuming recommended amounts of fibre, iron, calcium and grain foods? J. Hum. Nutr. Diet. 2005, 18, 163–169. [Google Scholar] [CrossRef]
- Thompson, T. Folate, iron, and dietary fiber contents of the gluten-free diet. J. Am. Diet. Assoc. 2000, 100, 1389–1396. [Google Scholar] [CrossRef]
- Penagini, F.; Dilillo, D.; Meneghin, F.; Mameli, C.; Fabiano, V.; Zuccotti, G.V. Gluten-free diet in children: An approach to a nutritionally adequate and balanced diet. Nutrients 2013, 5, 4553–4565. [Google Scholar] [CrossRef]
- Hallert, C.; Grant, C.; Grehn, S.; Grännö, C.; Hultén, S.; Midhagen, G.; Ström, M.; Svensson, H.; Valdimarsson, T. Evidence of poor vitamin status in coeliac patients on a gluten-free diet for 10 years. Aliment. Pharmacol. Ther. 2002, 16, 1333–1339. [Google Scholar] [CrossRef]
- Caruso, R.; Pallone, F.; Stasi, E.; Romeo, S.; Monteleone, G. Appropriate nutrient supplementation in celiac disease. Ann. Med. 2013, 45, 522–531. [Google Scholar] [CrossRef]
- Mager, D.R.; Qiao, J.; Turner, J. Vitamin D and K status influences bone mineral density and bone accrual in children and adolescents with celiac disease. Eur. J. Clin. Nutr. 2012, 66, 488–495. [Google Scholar] [CrossRef] [PubMed]
- Saturni, L.; Ferretti, G.; Bacchetti, T. The Gluten-Free Diet: Safety and Nutritional Quality. Nutrients 2010, 2, 16–34. [Google Scholar] [CrossRef] [PubMed]
- Kinsey, L.; Burden, S.T.; Bannerman, E. A dietary survey to determine if patients with coeliac disease are meeting current healthy eating guidelines and how their diet compares to that of the British general population. Eur. J. Clin. Nutr. 2008, 62, 1333–1342. [Google Scholar] [CrossRef] [PubMed]
- Rubio-Tapia, A.; Rahim, M.W.; See, J.A.; Lahr, B.D.; Wu, T.-T.; Murray, J.A. Mucosal recovery and mortality in adults with celiac disease after treatment with a gluten-free diet. Am. J. Gastroenterol. 2010, 105, 1412–1420. [Google Scholar] [CrossRef] [PubMed]
- Ciacci, C.; Cirillo, M.; Cavallaro, R.; Mazzacca, G. Long-term follow-up of celiac adults on gluten-free diet: Prevalence and correlates of intestinal damage. Digestion 2002, 66, 178–185. [Google Scholar] [CrossRef]
- Rosell, C.M. 27—Nutritionally enhanced wheat flours and breads A2—Cauvain, Stanley P. In Breadmaking, 2nd ed.; Woodhead Publishing Series in Food Science, Technology and Nutrition; Woodhead Publishing: Cambridge, UK, 2012; pp. 687–710. ISBN 978-0-85709-060-7. [Google Scholar]
- USDA Food Composition Databases. Available online: https://ndb.nal.usda.gov/ndb/ (accessed on 31 October 2018).
- Concha Collar Role of Bread on Nutrition and Health Worldwide. In Bread and Its Fortification; Food Biology Series; CRC Press: Boca Raton, FL, USA, 2015; pp. 26–52. ISBN 978-1-4987-0156-3.
- Hager, A.-S.; Wolter, A.; Jacob, F.; Zannini, E.; Arendt, E.K. Nutritional properties and ultra-structure of commercial gluten free flours from different botanical sources compared to wheat flours. J. Cereal Sci. 2012, 56, 239–247. [Google Scholar] [CrossRef]
- Melini, F.; Melini, V.; Luziatelli, F.; Ruzzi, M. Current and Forward-Looking Approaches to Technological and Nutritional Improvements of Gluten-Free Bread with Legume Flours: A Critical Review. Compr. Rev. Food Sci. Food Saf. 2017, 16, 1101–1122. [Google Scholar] [CrossRef]
- Lamacchia, C.; Camarca, A.; Picascia, S.; Di Luccia, A.; Gianfrani, C. Cereal-Based Gluten-Free Food: How to Reconcile Nutritional and Technological Properties of Wheat Proteins with Safety for Celiac Disease Patients. Nutrients 2014, 6, 575–590. [Google Scholar] [CrossRef]
- Bird, A.R.; Lopez-Rubio, A.; Shrestha, A.K.; Gidley, M.J. Resistant starch in vitro and in vivo: Factors determining yield, structure and physiological relevance. In Modern Biopolymer Science: Bridging the Divide between Fundamental Treatise and Industrial Application; Academic Press: New York, NY, USA, 2009; pp. 449–510. ISBN 978-0-12-374195-0. [Google Scholar]
- Holm, J.; Lundquist, I.; Björck, I.; Eliasson, A.C.; Asp, N.G. Degree of starch gelatinization, digestion rate of starch in vitro, and metabolic response in rats. Am. J. Clin. Nutr. 1988, 47, 1010–1016. [Google Scholar] [CrossRef]
- Berti, C.; Riso, P.; Monti, L.D.; Porrini, M. In vitro starch digestibility and in vivo glucose response of gluten-free foods and their gluten counterparts. Eur. J. Nutr. 2004, 43, 198–204. [Google Scholar] [CrossRef]
- Eliasson, A.; Larsson, K. Cereals in Breadmaking: A Molecular Colloidal Approach. Available online: https://www.crcpress.com/Cereals-in-Breadmaking-A-Molecular-Colloidal-Approach/liasson/p/book/9780824788162 (accessed on 13 November 2017).
- Scaramuzza, A.E.; Mantegazza, C.; Bosetti, A.; Zuccotti, G.V. Type 1 diabetes and celiac disease: The effects of gluten free diet on metabolic control. World J. Diabetes 2013, 4, 130–134. [Google Scholar] [CrossRef] [PubMed]
- Brand-Miller, J.C.; Holt, S.H.; Pawlak, D.B.; McMillan, J. Glycemic index and obesity. Am. J. Clin. Nutr. 2002, 76, 281S–S285. [Google Scholar] [CrossRef] [PubMed]
- Ferrara, P.; Cicala, M.; Tiberi, E.; Spadaccio, C.; Marcella, L.; Gatto, A.; Calzolari, P.; Castellucci, G. High fat consumption in children with celiac disease. Acta Gastroenterol. Belg. 2009, 72, 296–300. [Google Scholar] [PubMed]
- do Nascimento, A.B.; Fiates, G.M.R.; dos Anjos, A.; Teixeira, E. Analysis of ingredient lists of commercially available gluten-free and gluten-containing food products using the text mining technique. Int. J. Food Sci. Nutr. 2013, 64, 217–222. [Google Scholar] [CrossRef] [PubMed]
- Kulai, T.; Rashid, M. Assessment of Nutritional Adequacy of Packaged Gluten-free Food Products. Can. J. Diet. Pract. Res. 2014, 75, 186–190. [Google Scholar] [CrossRef] [PubMed]
- Miranda, J.; Lasa, A.; Bustamante, M.A.; Churruca, I.; Simon, E. Nutritional differences between a gluten-free diet and a diet containing equivalent products with gluten. Plant Foods Hum. Nutr. 2014, 69, 182–187. [Google Scholar] [CrossRef]
- Wu, J.H.Y.; Neal, B.; Trevena, H.; Crino, M.; Stuart-Smith, W.; Faulkner-Hogg, K.; Louie, J.C.Y.; Dunford, E. Are gluten-free foods healthier than non-gluten-free foods? An evaluation of supermarket products in Australia. Br. J. Nutr. 2015, 114, 448–454. [Google Scholar] [CrossRef] [PubMed]
- Missbach, B.; Schwingshackl, L.; Billmann, A.; Mystek, A.; Hickelsberger, M.; Bauer, G.; König, J. Gluten-free food database: The nutritional quality and cost of packaged gluten-free foods. PeerJ 2015, 3, e337. [Google Scholar] [CrossRef] [PubMed]
- Mazzeo, T.; Cauzzi, S.; Brighenti, F.; Pellegrini, N. The development of a composition database of gluten-free products. Public Health Nutr. 2015, 18, 1353–1357. [Google Scholar] [CrossRef]
- Estévez, V.; Ayala, J.; Vespa, C.; Araya, M. The gluten-free basic food basket: A problem of availability, cost and nutritional composition. Eur. J. Clin. Nutr. 2016, 70, 1215–1217. [Google Scholar] [CrossRef]
- Fry, L.; Madden, A.M.; Fallaize, R. An investigation into the nutritional composition and cost of gluten-free versus regular food products in the UK. J. Hum. Nutr. Diet. 2018, 31, 108–120. [Google Scholar] [CrossRef] [PubMed]
- Allen, B.; Orfila, C. The Availability and Nutritional Adequacy of Gluten-Free Bread and Pasta. Nutrients 2018, 10, 1370. [Google Scholar] [CrossRef] [PubMed]
- Chumpitazi, B.P.; Lim, J.; McMeans, A.R.; Shulman, R.J.; Hamaker, B.R. Evaluation of FODMAP Carbohydrates Content in Selected Foods in the United States. J. Pediatr. 2018, 199, 252–255. [Google Scholar] [CrossRef] [PubMed]
- Cornicelli, M.; Saba, M.; Machello, N.; Silano, M.; Neuhold, S. Nutritional composition of gluten-free food versus regular food sold in the Italian market. Dig. Liver Dis. 2018, 50, 1305–1308. [Google Scholar] [CrossRef] [PubMed]
- Diamanti, A.; Capriati, T.; Basso, M.S.; Panetta, F.; Di Ciommo Laurora, V.M.; Bellucci, F.; Cristofori, F.; Francavilla, R. Celiac Disease and Overweight in Children: An Update. Nutrients 2014, 6, 207–220. [Google Scholar] [CrossRef] [PubMed]
- Norsa, L.; Shamir, R.; Zevit, N.; Verduci, E.; Hartman, C.; Ghisleni, D.; Riva, E.; Giovannini, M. Cardiovascular disease risk factor profiles in children with celiac disease on gluten-free diets. World J. Gastroenterol. 2013, 19, 5658–5664. [Google Scholar] [CrossRef] [PubMed]
- Aurangzeb, B.; Leach, S.T.; Lemberg, D.A.; Day, A.S. Nutritional status of children with coeliac disease. Acta Paediatr. 2010, 99, 1020–1025. [Google Scholar] [CrossRef]
- Venkatasubramani, N.; Telega, G.; Werlin, S.L. Obesity in pediatric celiac disease. J. Pediatr. Gastroenterol. Nutr. 2010, 51, 295–297. [Google Scholar] [CrossRef] [PubMed]
- Balamtekin, N.; Uslu, N.; Baysoy, G.; Usta, Y.; Demir, H.; Saltık-Temizel, İ.N.; Özen, H.; Gürakan, F.; Yüce, A. The presentation of celiac disease in 220 Turkish children. Turk. J. Pediatr. 2010, 52, 6. [Google Scholar]
- Valletta, E.; Fornaro, M.; Cipolli, M.; Conte, S.; Bissolo, F.; Danchielli, C. Celiac disease and obesity: Need for nutritional follow-up after diagnosis. Eur. J. Clin. Nutr. 2010, 64, 1371–1372. [Google Scholar] [CrossRef] [PubMed]
- Brambilla, P.; Picca, M.; Dilillo, D.; Meneghin, F.; Cravidi, C.; Tischer, M.C.; Vivaldo, T.; Bedogni, G.; Zuccotti, G.V. Changes of body mass index in celiac children on a gluten-free diet. Nutr. Metab. Cardiovasc. Dis. 2013, 23, 177–182. [Google Scholar] [CrossRef]
- Reilly, N.R.; Aguilar, K.; Hassid, B.G.; Cheng, J.; Defelice, A.R.; Kazlow, P.; Bhagat, G.; Green, P.H. Celiac disease in normal-weight and overweight children: Clinical features and growth outcomes following a gluten-free diet. J. Pediatr. Gastroenterol. Nutr. 2011, 53, 528–531. [Google Scholar] [CrossRef] [PubMed]
- Forchielli, M.L.; Fernicola, P.; Diani, L.; Scrivo, B.; Salfi, N.C.; Pessina, A.C.; Lima, M.; Conti, V.; Pession, A. Gluten-Free Diet and Lipid Profile in Children With Celiac Disease: Comparison With General Population Standards. J. Pediatr. Gastroenterol. Nutr. 2015, 61, 224–229. [Google Scholar] [CrossRef] [PubMed]
- Więch, P.; Chmiel, Z.; Bazaliński, D.; Sałacińska, I.; Bartosiewicz, A.; Mazur, A.; Korczowski, B.; Binkowska-Bury, M.; Dąbrowski, M. The Relationship between Body Composition and a Gluten Free Diet in Children with Celiac Disease. Nutrients 2018, 10, 1817. [Google Scholar] [CrossRef] [PubMed]
- Kabbani, T.A.; Goldberg, A.; Kelly, C.P.; Pallav, K.; Tariq, S.; Peer, A.; Hansen, J.; Dennis, M.; Leffler, D.A. Body mass index and the risk of obesity in coeliac disease treated with the gluten-free diet. Aliment. Pharmacol. Ther. 2012, 35, 723–729. [Google Scholar] [CrossRef] [PubMed]
- Ukkola, A.; Mäki, M.; Kurppa, K.; Collin, P.; Huhtala, H.; Kekkonen, L.; Kaukinen, K. Changes in body mass index on a gluten-free diet in coeliac disease: A nationwide study. Eur. J. Intern. Med. 2012, 23, 384–388. [Google Scholar] [CrossRef] [PubMed]
- Levran, N.; Wilschanski, M.; Livovsky, J.; Shachar, E.; Moskovitz, M.; Assaf-Jabrin, L.; Shteyer, E. Obesogenic habits among children and their families in response to initiation of gluten-free diet. Eur. J. Pediatr. 2018, 177, 859–866. [Google Scholar] [CrossRef]
- Semeraro, L.A.; Barwick, K.W.; Gryboski, J.D. Obesity in celiac sprue. J. Clin. Gastroenterol. 1986, 8, 177–180. [Google Scholar] [CrossRef]
- Papastamataki, M.; Papassotiriou, I.; Bartzeliotou, A.; Vazeou, A.; Roma, E.; Chrousos, G.P.; Kanaka-Gantenbein, C. Incretins, amylin and other gut-brain axis hormones in children with coeliac disease. Eur. J. Clin. Investig. 2014, 44, 74–82. [Google Scholar] [CrossRef]
- Tortora, R.; Capone, P.; Stefano, G.D.; Imperatore, N.; Gerbino, N.; Donetto, S.; Monaco, V.; Caporaso, N.; Rispo, A. Metabolic syndrome in patients with coeliac disease on a gluten-free diet. Aliment. Pharmacol. Ther. 2015, 41, 352–359. [Google Scholar] [CrossRef]
- Ciccone, A.; Gabrieli, D.; Cardinale, R.; Di Ruscio, M.; Vernia, F.; Stefanelli, G.; Necozione, S.; Melideo, D.; Viscido, A.; Frieri, G.; et al. Metabolic Alterations in Celiac Disease Occurring after Following a Gluten-Free Diet. Digestion 2018, 14, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Kabbani, T.A.; Kelly, C.P.; Betensky, R.A.; Hansen, J.; Pallav, K.; Villafuerte-Gálvez, J.A.; Vanga, R.; Mukherjee, R.; Novero, A.; Dennis, M.; et al. Patients with celiac disease have a lower prevalence of non-insulin-dependent diabetes mellitus and metabolic syndrome. Gastroenterology 2013, 144, 912–917. [Google Scholar] [CrossRef]
- Zifman, E.; Waisbourd-Zinman, O.; Marderfeld, L.; Zevit, N.; Guz-Mark, A.; Silbermintz, A.; Assa, A.; Mozer-Glassberg, Y.; Biran, N.; Reznik, D.; et al. The Effect of Gluten-Free Diet on Cardiovascular Risk Factors in Newly Diagnosed Pediatric Celiac Disease Patients. J. Pediatr. Gastroenterol. Nutr. 2018. [Google Scholar] [CrossRef] [PubMed]
- Potter, M.D.E.; Brienesse, S.C.; Walker, M.M.; Boyle, A.; Talley, N.J. Effect of the gluten-free diet on cardiovascular risk factors in patients with coeliac disease: A systematic review. J. Gastroenterol. Hepatol. 2018, 33, 781–791. [Google Scholar] [CrossRef] [PubMed]
- Emilsson, L.; Semrad, C.E. Obesity, Metabolic Syndrome, and Cardiac Risk Factors: Going Gluten-Free, for Better or Worse? Dig. Dis. Sci. 2017, 62, 2215–2216. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.-S.; Demyen, M.F.; Mathew, J.; Kothari, N.; Feurdean, M.; Ahlawat, S.K. Obesity, Metabolic Syndrome, and Cardiovascular Risk in Gluten-Free Followers Without Celiac Disease in the United States: Results from the National Health and Nutrition Examination Survey 2009-2014. Dig. Dis. Sci. 2017, 62, 2440–2448. [Google Scholar] [CrossRef] [PubMed]
- Ehteshami, M.; Shakerhosseini, R.; Sedaghat, F.; Hedayati, M.; Eini-Zinab, H.; Hekmatdoost, A. The Effect of Gluten Free Diet on Components of Metabolic Syndrome: A Randomized Clinical Trial. Asian Pac. J. Cancer Prev. 2018, 19, 2979. [Google Scholar] [PubMed]
- Flint, H.J.; Scott, K.P.; Louis, P.; Duncan, S.H. The role of the gut microbiota in nutrition and health. Nat. Rev. Gastroenterol. Hepatol. 2012, 9, 577–589. [Google Scholar] [CrossRef]
- Collado, M.C.; Donat, E.; Ribes-Koninckx, C.; Calabuig, M.; Sanz, Y. Specific duodenal and faecal bacterial groups associated with paediatric coeliac disease. J. Clin. Pathol. 2009, 62, 264–269. [Google Scholar] [CrossRef]
- Collado, M.C.; Donat, E.; Ribes-Koninckx, C.; Calabuig, M.; Sanz, Y. Imbalances in faecal and duodenal Bifidobacterium species composition in active and non-active coeliac disease. BMC Microbiol. 2008, 8, 232. [Google Scholar] [CrossRef]
- Di Cagno, R.; De Angelis, M.; De Pasquale, I.; Ndagijimana, M.; Vernocchi, P.; Ricciuti, P.; Gagliardi, F.; Laghi, L.; Crecchio, C.; Guerzoni, M.E.; et al. Duodenal and faecal microbiota of celiac children: Molecular, phenotype and metabolome characterization. BMC Microbiol. 2011, 11, 219. [Google Scholar] [CrossRef] [PubMed]
- Sanz, Y. Effects of a gluten-free diet on gut microbiota and immune function in healthy adult humans. Gut Microbes 2010, 1, 135–137. [Google Scholar] [CrossRef] [PubMed]
- De Palma, G.; Nadal, I.; Collado, M.C.; Sanz, Y. Effects of a gluten-free diet on gut microbiota and immune function in healthy adult human subjects. Br. J. Nutr. 2009, 102, 1154–1160. [Google Scholar] [CrossRef] [PubMed]
- Richards, A.L.; Burns, M.B.; Alazizi, A.; Barreiro, L.B.; Pique-Regi, R.; Blekhman, R.; Luca, F. Genetic and Transcriptional Analysis of Human Host Response to Healthy Gut Microbiota. mSystems 2016, 1, e00067-16. [Google Scholar] [CrossRef] [PubMed]
- Melini, F.; Melini, V. Immunological Methods in Gluten Risk Analysis: A Snapshot. Safety 2018, 4, 56. [Google Scholar] [CrossRef]
| PubMed | No. of Documents Found | Scopus | No. of Documents Found |
|---|---|---|---|
| Search | Search | ||
| (((coeliac disease OR celiac disease)) AND gluten-free diet) AND nutrition | 316 | (TITLE-ABS-KEY (coeliac AND disease OR celiac AND disease) AND TITLE-ABS-KEY (gluten-free AND diet) AND TITLE-ABS-KEY (nutrition)) | 220 |
| ((gluten-free food OR gluten-free food product)) AND ((nutritional profile OR nutritional composition)) | 61 | (TITLE-ABS-KEY (gluten-free AND food OR gluten-free AND food AND product) AND TITLE-ABS-KEY (nutritional AND profile OR nutritional AND composition)) | 78 |
| (coeliac disease OR celiac disease) AND (nutritional inadequacies) | 3 | (TITLE-ABS-KEY (“coeliac disease OR celiac disease”) AND TITLE-ABS-KEY (“nutritional inadequacies”)) | 53 |
| (((“coeliac disease” OR “celiac disease”)) AND gluten-free diet) AND deficiency | 112 | TITLE-ABS-KEY (coeliac AND disease OR celiac AND disease) AND TITLE-ABS-KEY (gluten-free AND diet) AND TITLE-ABS-KEY (deficiency) | 328 |
| ((“coeliac disease” OR “celiac disease”)) AND malnutrition | 129 | TITLE-ABS-KEY (coeliac AND disease OR celiac AND disease) AND TITLE-ABS-KEY (malnutrition) | 157 |
| (((coeliac disease OR celiac disease)) AND gluten-free diet) AND fat intake | 14 | (TITLE-ABS-KEY (“gluten-free diet”) AND TITLE-ABS-KEY (“fat intake”)) | 28 |
| (((coeliac disease OR celiac disease)) AND gluten-free diet) AND carbohydrate intake | 19 | (TITLE-ABS-KEY (“gluten-free diet”) AND TITLE-ABS-KEY (“carbohydrate intake”)) | 12 |
| (((coeliac disease OR celiac disease)) AND gluten-free diet) AND protein intake | 45 | (TITLE-ABS-KEY (“gluten-free diet”) AND TITLE-ABS-KEY (“protein intake”)) | 44 |
| ((“coeliac disease” OR “celiac disease”)) AND micronutrients | 104 | TITLE-ABS-KEY(coeliac disease OR celiac disease) AND TITLE-ABS-KEY(micronutrients) | 61 |
| ((coeliac disease OR celiac disease)) AND vitamin | 195 | TITLE-ABS-KEY(coeliac disease OR celiac disease) AND TITLE-ABS-KEY(vitamins) | 397 |
| ((“coeliac disease” OR “celiac disease”)) AND minerals | 22 | TITLE-ABS-KEY(coeliac disease OR celiac disease) AND TITLE-ABS-KEY(minerals) | 169 |
| (((coeliac disease OR celiac disease)) AND gluten-free diet) AND obesity | 22 | (TITLE-ABS-KEY (gluten-free diet) AND TITLE-ABS-KEY (obesity)) | 99 |
| (((coeliac disease OR celiac disease)) AND gluten-free diet) AND microbiota | 60 | (TITLE-ABS-KEY (gluten-free diet) AND TITLE-ABS-KEY (microbiota)) | 107 |
| (((coeliac disease OR celiac disease)) AND gluten-free diet) AND metabolic syndrome | 17 | (TITLE-ABS-KEY (gluten-free diet) AND TITLE-ABS-KEY (metabolic syndrome)) | 24 |
| (((coeliac disease OR celiac disease)) AND gluten-free diet) AND (glycaemic index OR glycemic index) | 12 | (TITLE-ABS-KEY (gluten-free diet) AND TITLE-ABS-KEY (glycaemic index OR glycemic index)) | 29 |
| ((coeliac disease OR celiac disease)) AND (co-morbidity OR co-morbidities) | 136 | (TITLE-ABS-KEY (coeliac disease OR celiac disease) AND TITLE-ABS-KEY (co-morbidity OR co-morbidities)) | 33 |
| ((coeliac disease OR celiac disease)) AND (gluten-free diet compliance) | 161 | (TITLE-ABS-KEY (coeliac disease OR celiac disease) AND TITLE-ABS-KEY (“gluten-free diet compliance”)) | 4 |
| ((coeliac disease OR celiac disease)) AND ((cardiovascular diseases OR cardio-vascular diseases)) | 382 | (TITLE-ABS-KEY(coeliac disease OR celiac disease) AND TITLE-ABS-KEY(“cardiovascular diseases” OR “cardio-vascular diseases”)) | 37 |
| ((coeliac disease OR celiac disease)) AND (weight gain) | 40 | (TITLE-ABS-KEY (coeliac disease OR celiac disease) AND TITLE-ABS-KEY (“weight gain”)) | 38 |
| TOTAL | 1895 | 1918 |
| Reference | Publication Year | Geographical Area | No. Products | Food Category | Nutritional Profile of GF Food Products | |
|---|---|---|---|---|---|---|
| Nascimento et al. [64] | 2013 | Brazil | 168 (GF) | 162 (GC) | cookies | Higher energy profile Higher protein, saturated fat and sodium content |
| bread and pasta | Lower protein and DF content | |||||
| snacks | Lower energy profile Lower total fat, saturated fat and sodium content Higher protein and DF | |||||
| breakfast cereals | Lower energy profile Higher sodium content | |||||
| Kulai and Rashid [65] | 2014 | Canada | 71 (GF) | 60 (GC) | GF food products | Energy profile comparable to GC food products; |
| bread | Higher fat content, two-fold No differences in saturated fat content Lower protein content | |||||
| pasta | Higher mean carbohydrate content Low in dietary fibre, iron, folates | |||||
| breakfast cereals and cake mixes | No significant difference | |||||
| Miranda et al. [66] | 2014 | Spain | 206 (GF) | 289 (GC) | bread | Higher fat content, especially saturated fat Lower protein content More salt Less fibre |
| pasta | Nutrient profile similar to bread | |||||
| bakery products | Lower energy, protein and carbohydrate content Higher sodium and cholesterol content | |||||
| Differences among brands | ||||||
| Wu et al. [67] | 2015 | Australia | Similar nutritional profile between GF and GC food products | |||
| pasta, bread, breakfast cereals | Lower protein content Similar total energy, sodium, saturated fats, total sugars | |||||
| bread | High mean DF content | |||||
| cereal bars, cake mixes, sweet biscuits | High content of sugar, saturated fats and salt | |||||
| cake mixes, cakes | Low saturated fat levels High total sugar level Total energy similar to GC | |||||
| Missbach et al. [68] | 2015 | Austria | 63 (GF) | 126 (GC) | - | Energy content, carbohydrates, total fats, saturated fatty acids, fibre and sugar did not differ between GF and GC products Protein content more than two-folds lower in 57% of all GF food categories Sodium content lower in GF products Potassium content was overall significantly lower in GF food products |
| pasta | Zinc content significantly lower in GF pasta products | |||||
| Mazzeo et al. [69] | 2015 | Italy | 60 (GF) | sweet products | High fat and sugar content | |
| brioches | High content of salt | |||||
| bread, pizza, snack, flours | High available carbohydrate and sugar content | |||||
| Estévez et al. [70] | 2016 | Chile | 19 (GF) | 34 (GC) | bread | Low protein content High DF content |
| Fat content similar to GC products. | ||||||
| Fry et al. [71] | 2018 | United Kingdom | 679 (GF) | 1045 (GC) | - | Lack of a pattern in the comparison of overall nutritional quality of GF dietary foods |
| biscuits, crackers, white and brown bread, breakfast cereals, white and wholegrain flour, pizza bases, wholegrain and white pasta | Low protein content | |||||
| commercial white bread, breakfast cereals, wholegrain pasta | High and medium fat and saturated fats | |||||
| prescribed breakfast cereals, crackers, biscuits | High and medium salt content | |||||
| brown and white bread, white and wholegrain flour, pizza bases, crackers, biscuits | Higher sugar content in prescribed GF food products than commercial | |||||
| wholegrain flours, white pasta | Very high salt content | |||||
| white and brown bread | High DF content | |||||
| breakfast cereals, white and wholegrain pasta | Low DF content | |||||
| Allen and Orfila [72] | 2018 | United Kingdom | 49 (GF) | 61 (GC) | - | Average total energy, saturated fat, and salt values were similar between GF and GC products |
| brown bread, pasta | Lower levels of sugar | |||||
| white, brown, seeded bread | Higher fat content | |||||
| white, brown, seeded bread, pasta | Lower carbohydrate intake per portion | |||||
| white, brown, seeded bread, pasta | Lower protein content | |||||
| white, brown, seeded bread | Higher DF content | |||||
| GF pasta | Significantly lower DF content | |||||
| bread | Only 28% of GF breads were fortified with calcium carbonate and iron Only 5% of the total GF breads were fortified with all four fortification minerals, in addition to folic acid and riboflavin. | |||||
| Chumpitazi et al. [73] | 2018 | USA | 3 (GF) | - | excessive FODMAP content | |
| Cornicelli et al. [74] | 2018 | Italy | 235 (GF) | 349 (GC) | - | Overall, energy content was not different to that of regular equivalents. Two exceptions: Lower content in bread and higher in pasta |
| all GF food categories, but especially bread and rusks | Lower protein content | |||||
| - | Fat content was not different. GF biscuits and pasta have the highest content of saturated fats; | |||||
| GF biscuits, rusks, pasta and bread substitutes | Higher content of carbohydrates | |||||
| GF biscuits, bread substitutes, pasta | Lower DF content | |||||
| GF bread | Higher DF content | |||||
| GF pasta and rusks | Higher salt content | |||||
| GF biscuits | Lower salt content |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Melini, V.; Melini, F. Gluten-Free Diet: Gaps and Needs for a Healthier Diet. Nutrients 2019, 11, 170. https://doi.org/10.3390/nu11010170
Melini V, Melini F. Gluten-Free Diet: Gaps and Needs for a Healthier Diet. Nutrients. 2019; 11(1):170. https://doi.org/10.3390/nu11010170
Chicago/Turabian StyleMelini, Valentina, and Francesca Melini. 2019. "Gluten-Free Diet: Gaps and Needs for a Healthier Diet" Nutrients 11, no. 1: 170. https://doi.org/10.3390/nu11010170
APA StyleMelini, V., & Melini, F. (2019). Gluten-Free Diet: Gaps and Needs for a Healthier Diet. Nutrients, 11(1), 170. https://doi.org/10.3390/nu11010170






