White Sweet Potato as Meal Replacement for Overweight White-Collar Workers: A Randomized Controlled Trial
Abstract
1. Introduction
2. Participants and Methods
2.1. Study Participants
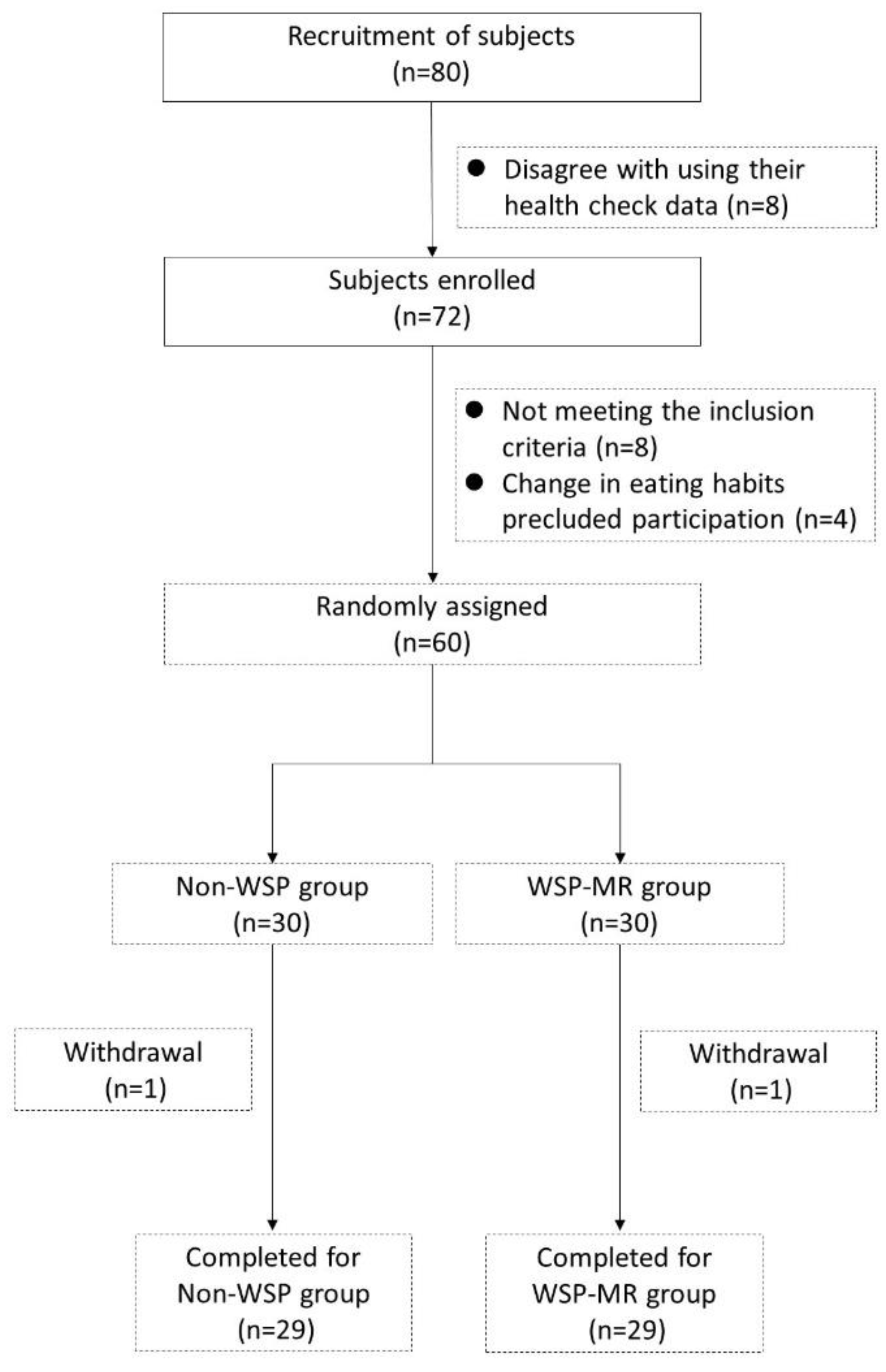
2.2. Trial Design
2.3. WSP-MR Formula
2.4. Biochemical Analyses
2.5. Statistical Analyses
3. Results
3.1. Participant Characteristics
3.2. Changes in Anthropometric Parameters and Clinical Characteristics
3.3. Changes in Biochemical Characteristics, Glycemic Markers, and Lipid Profiles
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Li, L.M.; Rao, K.Q.; Kong, L.Z.; Yao, C.H.; Xiang, H.D.; Zhai, F.Y.; Ma, G.S.; Yang, X.G.; Technical Working Group of China National Nutrition and Health Survey. A description on the Chinese national nutrition and health survey in 2002. Zhonghua Liu Xing Bing Xue Za Zhi 2005, 26, 478–484. [Google Scholar] [PubMed]
- Pan, W.H.; Wu, H.J.; Yeh, C.J.; Chuang, S.Y.; Chang, H.Y.; Yeh, N.H.; Hsieh, Y.T. Diet and health trends in Taiwan: Comparison of two nutrition and health surveys from 1993–1996 and 2005–2008. Asia Pac. J. Clin. Nutr. 2011, 20, 238–250. [Google Scholar] [PubMed]
- Gu, J.K.; Charles, L.E.; Bang, K.M.; Ma, C.C.; Andrew, M.E.; Violanti, J.M.; Burchfiel, C.M. Prevalence of obesity by occupation among US workers: The National Health Interview Survey 2004–2011. J. Occup. Environ. Med. 2014, 56, 516–528. [Google Scholar] [CrossRef] [PubMed]
- Lowe, M.R.; Butryn, M.L.; Zhang, F. Evaluation of meal replacements and a home food environment intervention for long-term weight loss: A randomized controlled trial. Am. J. Clin. Nutr. 2018, 107, 12–19. [Google Scholar] [CrossRef] [PubMed]
- Annunziato, R.A.; Timko, C.A.; Crerand, C.E.; Didie, E.R.; Bellace, D.L.; Phelan, S.; Kerzhnerman, I.; Lowe, M.R. A randomized trial examining differential meal replacement adherence in a weight loss maintenance program after one-year follow-up. Eat. Behav. 2009, 10, 176–183. [Google Scholar] [CrossRef] [PubMed]
- Vazquez, C.; Montagna, C.; Alcaraz, F.; Balsa, J.A.; Zamarron, I.; Arrieta, F.; Botella-Carretero, J.I. Meal replacement with a low-calorie diet formula in weight loss maintenance after weight loss induction with diet alone. Eur. J. Clin. Nutr. 2009, 63, 1226–1232. [Google Scholar] [CrossRef] [PubMed]
- Heymsfield, S.B.; van Mierlo, C.A.; van der Knaap, H.C.; Heo, M.; Frier, H.I. Weight management using a meal replacement strategy: Meta and pooling analysis from six studies. Int. J. Obes. 2003, 27, 537–549. [Google Scholar] [CrossRef] [PubMed]
- Butryn, M.L.; Webb, V.; Wadden, T.A. Behavioral treatment of obesity. Psychiatry Clin. N. Am. 2011, 34, 841–859. [Google Scholar] [CrossRef]
- Ozaki, S.; Oki, N.; Suzuki, S.; Kitamura, S. Structural characterization and hypoglycemic effects of arabinogalactan-protein from the tuberous cortex of the white-skinned sweet potato (Ipomoea batatas L.). J. Agric. Food Chem. 2010, 58, 11593–11599. [Google Scholar] [CrossRef] [PubMed]
- Ludvik, B.; Hanefeld, M.; Pacini, G. Improved metabolic control by Ipomoea batatas (Caiapo) is associated with increased adiponectin and decreased fibrinogen levels in type 2 diabetic subjects. Diabetes Obes. Metab. 2008, 10, 586–592. [Google Scholar] [CrossRef]
- Ludvik, B.; Waldhausl, W.; Prager, R.; Kautzky-Willer, A.; Pacini, G. Mode of action of ipomoea batatas (Caiapo) in type 2 diabetic patients. Metabolism 2003, 52, 875–880. [Google Scholar] [CrossRef]
- Ludvik, B.H.; Mahdjoobian, K.; Waldhaeusl, W.; Hofer, A.; Prager, R.; Kautzky-Willer, A.; Pacini, G. The effect of Ipomoea batatas (Caiapo) on glucose metabolism and serum cholesterol in patients with type 2 diabetes: A randomized study. Diabetes Care 2002, 25, 239–240. [Google Scholar] [CrossRef] [PubMed]
- Ju, R.; Zheng, S.; Luo, H.; Wang, C.; Duan, L.; Sheng, Y.; Zhao, C.; Xu, W.; Huang, K. Purple sweet potato attenuate weight gain in high fat diet induced obese mice. J. Food Sci. 2017, 82, 787–793. [Google Scholar] [CrossRef] [PubMed]
- Allison, D.B.; Gadbury, G.; Schwartz, L.G.; Murugesan, R.; Kraker, J.L.; Heshka, S.; Fontaine, K.R.; Heymsfield, S.B. A novel soy-based meal replacement formula for weight loss among obese individuals: A randomized controlled clinical trial. Eur. J. Clin. Nutr. 2003, 57, 514–522. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Hong, K.; Saltsman, P.; DeShields, S.; Bellman, M.; Thames, G.; Liu, Y.; Wang, H.J.; Elashoff, R.; Heber, D. Long-term efficacy of soy-based meal replacements vs an individualized diet plan in obese type II DM patients: Relative effects on weight loss, metabolic parameters, and C-reactive protein. Eur. J. Clin. Nutr. 2005, 59, 411–418. [Google Scholar] [CrossRef] [PubMed]
- Chronakis, I.S. On the molecular characteristics, compositional properties, and structural-functional mechanisms of maltodextrins: A review. Crit. Rev. Food Sci. Nutr. 1998, 38, 599–637. [Google Scholar] [CrossRef]
- Hofman, D.L.; van Buul, V.J.; Brouns, F.J. Nutrition, health, and regulatory aspects of digestible maltodextrins. Crit. Rev. Food Sci. Nutr. 2016, 56, 2091–2100. [Google Scholar] [CrossRef]
- Cheang, K.-U.; Chen, C.-M.; Chen, C.-Y.O.; Liang, F.-Y.; Shih, C.-K.; Li, S.-C. Effects of glucomannan noodle on diabetes risk factors in patients with metabolic syndrome: A double-blinded, randomized crossover controlled trial. J. Food Nutr. Res. 2017, 5, 622–628. [Google Scholar] [CrossRef]
- Ketel, I.J.; Volman, M.N.; Seidell, J.C.; Stehouwer, C.D.; Twisk, J.W.; Lambalk, C.B. Superiority of skinfold measurements and waist over waist-to-hip ratio for determination of body fat distribution in a population-based cohort of Caucasian Dutch adults. Eur. J. Endocrinol. 2007, 156, 655–661. [Google Scholar] [CrossRef] [PubMed]
- Bosy-Westphal, A.; Geisler, C.; Onur, S.; Korth, O.; Selberg, O.; Schrezenmeir, J.; Muller, M.J. Value of body fat mass vs anthropometric obesity indices in the assessment of metabolic risk factors. Int. J. Obes. (Lond.) 2006, 30, 475–483. [Google Scholar] [CrossRef]
- Metzner, C.E.; Folberth-Vogele, A.; Bitterlich, N.; Lemperle, M.; Schafer, S.; Alteheld, B.; Stehle, P.; Siener, R. Effect of a conventional energy-restricted modified diet with or without meal replacement on weight loss and cardiometabolic risk profile in overweight women. Nutr. Metab. (Lond) 2011, 8, 64. [Google Scholar] [CrossRef]
- Gulati, S.; Misra, A.; Tiwari, R.; Sharma, M.; Pandey, R.M.; Yadav, C.P. Effect of high-protein meal replacement on weight and cardiometabolic profile in overweight/obese Asian Indians in North India. Br. J. Nutr. 2017, 117, 1531–1540. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Liu, Y.; Yang, Q.; Li, X.; Yang, J.; Wang, J.; Shi, L.; Chen, Y.; Zhu, S. The effect of protein-enriched meal replacement on waist circumference reduction among overweight and obese Chinese with hyperlipidemia. J. Am. Coll. Nutr. 2016, 35, 236–244. [Google Scholar] [CrossRef] [PubMed]
- Malik, V.S.; Hu, F.B. Popular weight-loss diets: From evidence to practice. Nat. Clin. Pract. Cardiovasc. Med. 2007, 4, 34. [Google Scholar] [CrossRef] [PubMed]
- Brand-Miller, J.C.; Holt, S.H.A.; Pawlak, D.B.; McMillan, J. Glycemic index and obesity. Am. J. Clin. Nutr. 2002, 76, 281S–285S. [Google Scholar] [CrossRef] [PubMed]
- Bovell-Benjamin, A.C. Sweet potato: A review of its past, present, and future role in human nutrition. In Advances in Food and Nutrition Research; Toldra, F., Ed.; Academic Press: Cambridge, MA, USA, 2007; Volume 52, pp. 1–59. [Google Scholar]
- Leader, N.J.; Ryan, L.; Molyneaux, L.; Yue, D.K. How best to use partial meal replacement in managing overweight or obese patients with poorly controlled type 2 diabetes. Obesity (Silver Spring) 2013, 21, 251–253. [Google Scholar] [CrossRef]
- Larsen, T.M.; Dalskov, S.M.; van Baak, M.; Jebb, S.A.; Papadaki, A.; Pfeiffer, A.F.; Martinez, J.A.; Handjieva-Darlenska, T.; Kunešová, M.; Pihlsgård, M.; et al. Diets with high or low protein content and glycemic index for weight-loss maintenance. N. Engl. J. Med. 2010, 363, 2102–2113. [Google Scholar] [CrossRef]
- Nicklas, J.M.; Sacks, F.M.; Smith, S.R.; LeBoff, M.S.; Rood, J.C.; Bray, G.A.; Ridker, P.M. Effect of dietary composition of weight loss diets on high-sensitivity c-reactive protein: The Randomized POUNDS LOST trial. Obesity (Silver Spring) 2013, 21, 681–689. [Google Scholar] [CrossRef]
- Howarth, N.C.; Saltzman, E.; Roberts, S.B. Dietary fiber and weight regulation. Nutr. Rev. 2001, 59, 129–139. [Google Scholar] [CrossRef]
- Wu, S.J.; Pan, W.H.; Yeh, N.H.; Chang, H.Y. Trends in nutrient and dietary intake among adults and the elderly: From NAHSIT 1993–1996 to 2005–2008. Asia Pac. J. Clin. Nutr. 2011, 20, 251–265. [Google Scholar]
- Zhang, L.; Li, H.T.; Shen, L.; Fang, Q.C.; Qian, L.L.; Jia, W.P. Effect of dietary resistant starch on prevention and treatment of obesity-related diseases and its possible mechanisms. Biomed. Environ. Sci. 2015, 28, 291–297. [Google Scholar] [CrossRef] [PubMed]
- Nichenametla, S.N.; Weidauer, L.A.; Wey, H.E.; Beare, T.M.; Specker, B.L.; Dey, M. Resistant starch type 4-enriched diet lowered blood cholesterols and improved body composition in a double blind controlled cross-over intervention. Mol. Nutr. Food Res. 2014, 58, 1365–1369. [Google Scholar] [CrossRef] [PubMed]
- Musilová, J.; Bystrická, J.; Árvay, J.; Harangózo, L. Polyphenols and phenolic acids in sweet potato (Ipomoea batatas L.) roots. Potravinárstvo 2017, 11, 82–87. [Google Scholar] [CrossRef]
- Yoshino, J.; Conte, C.; Fontana, L.; Mittendorfer, B.; Imai, S.; Schechtman, K.B.; Gu, C.; Kunz, I.; Rossi Fanelli, F.; Patterson, B.W.; et al. Resveratrol supplementation does not improve metabolic function in nonobese women with normal glucose tolerance. Cell Metab. 2012, 16, 658–664. [Google Scholar] [CrossRef] [PubMed]
- Ellis, T.P.; Wright, A.G.; Clifton, P.M.; Ilag, L.L. Postprandial insulin and glucose levels are reduced in healthy subjects when a standardised breakfast meal is supplemented with a filtered sugarcane molasses concentrate. Eur. J. Nutr. 2016, 55, 2365–2376. [Google Scholar] [CrossRef] [PubMed]
| Nutrient | WSP | WSP-MR |
|---|---|---|
| Calories (kcal) | 380.8 | 390.7 |
| Protein (g) | 2.9 | 22.4 |
| Total fat (g) | 0 | 10.9 |
| Saturated Fat | 0 | 2.5 |
| MUFA | 0 | 4.9 |
| PUFA | 0 | 2.9 |
| Carbohydrates (g) | 92.3 | 58.5 |
| Dietary fiber | 10.1 | 15.5 |
| Sugars | 31.1 | 13.4 |
| Water (g) | 9.6 | 3.8 |
| Ash (g) | 3.0 | 4.5 |
| Minerals | ||
| Sodium (mg) | 176 | 410.6 |
| Manganese (mg) | 3.34 | 1 |
| Potassium (mg) | 11,700 | 469 |
| Calcium (mg) | 1080 | 610.3 |
| Magnesium (mg) | 675 | 223.5 |
| Iron (mg) | 18.3 | 6.4 |
| Zinc (mg) | 7.98 | 7.3 |
| Phosphate (mg) | 2660 | 317.5 |
| Chloride (mg) | 2500 | 356.1 |
| Copper (ug) | 5.38 | 364 |
| Characteristic | Non-WSP | WSP-MR | p |
|---|---|---|---|
| Age (year-old) | 37.33 ± 5.50 | 38.76 ± 6.24 | 0.371 |
| Male/female (n) | 13/14 | 14/15 | 0.992 |
| Height (cm) | 163.91 ± 9.17 | 164.58 ± 7.81 | 0.767 |
| Body weight (kg) | 66.77 ± 8.4 | 68.01 ± 6.88 | 0.546 |
| Body mass index (kg/m2) | 24.68 ± 0.90 | 24.99 ± 1.05 | 0.245 |
| Body fat (%) | 29.12 ± 5.39 | 29.42 ± 6.37 | 0.851 |
| WC (cm) | 84.49 ± 7.67 | 85.87 ± 4.78 | 0.427 |
| HC (cm) | 99.24 ± 10.51 | 101.87 ± 3.39 | 0.206 |
| W-H ratio (%) | 86.52 ± 16.24 | 84.33 ± 4.49 | 0.487 |
| Basal metabolic rate (kcal) | 1507.16 ± 203.52 | 1508.40 ± 176.10 | 0.981 |
| Systolic blood pressure (mmHg) | 116.59 ± 12.24 | 112.45 ± 15.01 | 0.265 |
| Diastolic blood pressure (mmHg) | 69.41 ± 9.60 | 68.31 ± 15.63 | 0.751 |
| FBG (mg/dL) | 86.81 ± 4.69 | 85.28 ± 7.33 | 0.357 |
| Insulin (uU/mL) | 9.41 ± 4.86 | 8.63 ± 3.53 | 0.493 |
| TC (mg/dL) | 195.48 ± 26.68 | 188.24 ± 25.35 | 0.302 |
| TG (mg/dL) | 113.93 ± 41.91 | 111.31 ± 53.19 | 0.840 |
| HDL-C (mg/dL) | 50.44 ± 10.34 | 49.07 ± 7.77 | 0.574 |
| LDL-C (mg/dL) | 128.78 ± 24.08 | 123.14 ± 22.95 | 0.374 |
| Uric acid (mg/dL) | 5.33 ± 1.41 | 5.19 ± 1.28 | 0.691 |
| AST (U/L) | 29.96 ± 6.42 | 23.17 ± 23.84 | 0.800 |
| ALT (U/L) | 22.26 ± 9.53 | 29.17 ± 49.63 | 0.480 |
| Non-WSP | WSP-MR | |||||
|---|---|---|---|---|---|---|
| Week 0 | Week 8 | Changes | Week 0 | Week 8 | Changes | |
| Body weight (kg) | 66.77 ± 8.40 | 64.66 ± 8.39 * | −2.11 ± 1.70 | 68.01 ± 6.88 | 64.31 ± 6.21 * | −3.70 ± 2.24 # |
| Body fat (%) | 29.12 ± 5.39 | 28.17 ± 5.31 * | −0.95 ± 2.21 | 29.42 ± 6.37 | 27.16 ± 6.07 * | −2.26 ± 2.09 # |
| BMI (kg/m2) | 24.68 ± 0.90 | 23.92 ± 0.96 * | −0.76 ± 0.68 | 24.99 ± 1.05 | 23.65 ± 1.22 * | −1.33 ± 0.82 # |
| WC (cm) | 84.49 ± 7.67 | 82.93 ± 6.11 | −1.57 ± 4.42 | 85.87 ± 4.78 | 83.78 ± 5.65 * | −2.10 ± 4.46 |
| HC (cm) | 99.24 ± 10.51 | 99.22 ± 3.52 | −0.02 ± 8.91 | 101.87 ± 3.39 | 98.98 ± 3.31 * | −2.89 ± 2.88 |
| W-H ratio (%) | 86.52 ± 16.24 | 83.54 ± 4.80 | −2.99 ± 15.99 | 84.33 ± 4.49 | 84.64 ± 5.14 | 0.32 ± 3.88 |
| WrC (cm) | 15.80 ± 1.01 | 15.59 ± 0.88 * | −0.21 ± 0.44 | 16.00 ± 1.04 | 15.58 ± 1.04 * | −0.43 ± 0.71 |
| ThC (cm) | 59.46 ± 2.35 | 57.29 ± 2.35 * | −2.18 ± 2.82 | 59.90 ± 3.11 | 57.15 ± 2.52 * | −2.76 ± 3.32 |
| CaC (cm) | 37.37 ± 1.84 | 37.00 ± 1.93 * | −0.37 ± 0.82 | 37.97 ± 1.69 | 37.33 ± 1.58 * | −0.64 ± 0.88 |
| MAC (cm) | 29.69 ± 2.31 | 28.84 ± 2.15 * | −0.85 ± 0.74 | 29.96 ± 1.93 | 28.47 ± 1.82 * | −1.49 ± 0.96 # |
| MAMC (cm) | 22.61 ± 2.61 | 23.36 ± 3.10 * | 0.74 ± 1.89 | 22.76 ± 2.79 | 22.94 ± 2.50 | 0.18 ± 2.02 |
| TrS (mm) | 22.54 ± 4.79 | 17.47 ± 4.62 * | −5.07 ± 5.58 | 22.92 ± 5.84 | 17.60 ± 4.28 * | −5.31 ± 5.65 |
| Non-WSP | WSP-MR | |||||
|---|---|---|---|---|---|---|
| Week 0 | Week 8 | Changes | Week 0 | Week 8 | Changes | |
| Albumin (g/dL) | 4.83 ± 0.19 | 4.60 ± 0.24 * | −0.23 ± 0.19 | 4.51 ± 0.21 | 4.55 ± 0.24 * | 0.04 ± 0.15 # |
| Total protein (g/dL) | 7.41 ± 0.31 | 7.29 ± 0.25 * | −0.13 ± 0.25 | 7.38 ± 0.40 | 7.33 ± 0.37 * | −0.05 ± 0.23 |
| Sodium (meq/L) | 137.30 ± 2.15 | 139.96 ± 1.23 * | 2.66 ± 2.31 | 137.67 ± 1.24 | 139.60 ± 1.37 * | 1.93 ± 1.21 |
| Potassium (meq/L) | 4.09 ± 0.30 | 4.18 ± 0.30 * | 0.09 ± 0.29 | 4.16 ± 0.31 | 4.22 ± 0.29 | 0.06 ± 0.30 |
| Calcium (meq/L) | 10.02 ± 0.22 | 9.33 ± 0.24 * | −0.69 ± 0.25 | 9.67 ± 0.29 | 9.31 ± 0.31 * | −0.36 ± 0.20 # |
| Phosphorus (meq/L) | 3.21 ± 0.46 | 3.17 ± 0.42 * | −0.04 ± 0.37 | 3.14 ± 0.44 | 3.30 ± 0.33 * | 0.16 ± 0.35 # |
| Magnesium (meq/L) | 2.05 ± 0.12 | 2.11 ± 0.13 * | 0.06 ± 0.11 | 2.02 ± 0.11 | 2.14 ± 0.13 * | 0.11 ± 0.11 |
| AST (U/L) | 21.96 ± 6.42 | 19.59 ± 6.40 | −2.37 ± 6.23 | 23.17 ± 23.84 | 17.48 ± 3.86 | −5.69 ± 22.14 |
| ALT (U/L) | 22.26 ± 9.53 | 21.78 ± 18.11 | −0.48 ± 16.18 | 29.17 ± 49.63 | 16.31 ± 7.90 | −12.86 ± 43.81 |
| γ-GTP (U/L) | 19.33 ± 10.04 | 16.44 ± 7.88 * | −2.89 ± 4.37 | 25.17 ± 35.13 | 13.72 ± 8.18 | −11.45 ± 30.89 |
| Total bilirubin (mg/dL) | 0.72 ± 0.31 | 0.81 ± 0.31 | 0.09 ± 0.26 | 0.77 ± 0.34 | 0.78 ± 0.37 | 0.01 ± 0.29 |
| Creatinine (mg/dL) | 0.76 ± 0.14 | 0.68 ± 0.14 * | −0.09 ± 0.05 | 0.76 ± 0.18 | 0.68 ± 0.15 * | −0.09 ± 0.06 |
| BUN (mg/dL) | 13.26 ± 2.38 | 11.67 ± 2.60 * | −1.59 ± 2.22 | 11.79 ± 2.93 | 11.93 ± 2.94 | 0.14 ± 3.07 # |
| Uric acid (mg/dL) | 5.33 ± 1.41 | 5.14 ± 1.21 * | −0.19 ± 0.63 | 5.19 ± 1.28 | 5.16 ± 1.11 | −0.03 ± 0.66 |
| Hs-CRP (mg/dL) | 0.21 ± 0.18 | 0.13 ± 0.05 * | −0.09 ± 0.17 | 0.27 ± 0.34 | 0.38 ± 0.88 | 0.11 ± 0.98 |
| Non-WSP | WSP-MR | |||||
|---|---|---|---|---|---|---|
| Week 0 | Week 8 | Changes | Week 0 | Week 8 | Changes | |
| FBG (mg/dL) | 86.81 ± 4.69 | 80.30 ± 6.33 * | −6.52 ± 5.60 | 85.28 ± 7.33 | 78.90 ± 6.14 * | −6.38 ± 5.30 |
| HbA1c (%) | 5.14 ± 0.37 | 5.12 ± 0.44 | −0.02 ± 0.17 | 5.38 ± 0.35 | 5.20 ± 0.25 * | −0.19 ± 0.20 # |
| Insulin (uU/mL) | 9.41 ± 4.86 | 8.04 ± 4.09 | −1.37 ± 3.70 | 8.63 ± 3.53 | 5.71 ± 2.98 * | −2.92 ± 3.79 |
| TC (mg/dL) | 195.48 ± 26.68 | 181.89 ± 25.16 * | −13.59 ± 12.91 | 188.24 ± 25.35 | 177.41 ± 25.31* | −10.83 ± 18.83 |
| TG (mg/dL) | 113.93 ± 41.91 | 87.63 ± 40.23 * | −26.30 ± 32.11 | 111.31 ± 53.19 | 78.14 ± 25.49 * | −33.17 ± 47.23 |
| HDL-C (mg/dL) | 50.44 ± 10.34 | 51.78 ± 10.48 | 1.33 ± 3.81 | 49.07 ± 7.77 | 51.52 ± 8.50 * | 2.45 ± 4.45 |
| LDL-C (mg/dL) | 128.78 ± 24.08 | 116.44 ± 23.43 * | −12.33 ±13.08 | 123.14 ± 22.95 | 113.07 ± 22.58 * | −10.07 ± 17.51 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shih, C.-K.; Chen, C.-M.; Hsiao, T.-J.; Liu, C.-W.; Li, S.-C. White Sweet Potato as Meal Replacement for Overweight White-Collar Workers: A Randomized Controlled Trial. Nutrients 2019, 11, 165. https://doi.org/10.3390/nu11010165
Shih C-K, Chen C-M, Hsiao T-J, Liu C-W, Li S-C. White Sweet Potato as Meal Replacement for Overweight White-Collar Workers: A Randomized Controlled Trial. Nutrients. 2019; 11(1):165. https://doi.org/10.3390/nu11010165
Chicago/Turabian StyleShih, Chun-Kuang, Chiao-Ming Chen, Tun-Jen Hsiao, Ching-Wen Liu, and Sing-Chung Li. 2019. "White Sweet Potato as Meal Replacement for Overweight White-Collar Workers: A Randomized Controlled Trial" Nutrients 11, no. 1: 165. https://doi.org/10.3390/nu11010165
APA StyleShih, C.-K., Chen, C.-M., Hsiao, T.-J., Liu, C.-W., & Li, S.-C. (2019). White Sweet Potato as Meal Replacement for Overweight White-Collar Workers: A Randomized Controlled Trial. Nutrients, 11(1), 165. https://doi.org/10.3390/nu11010165







