A Boost in Mitochondrial Activity Underpins the Cholesterol-Lowering Effect of Annurca Apple Polyphenols on Hepatic Cells
Abstract
1. Introduction
2. Materials and Methods
2.1. Reagents
2.2. Cell Culture and D2O Labeling
2.3. GC-MS
2.4. Mass Spectrometry-Based Metabolomic, Statistics, and Analysis.
2.5. Immunofluorescence
3. Results
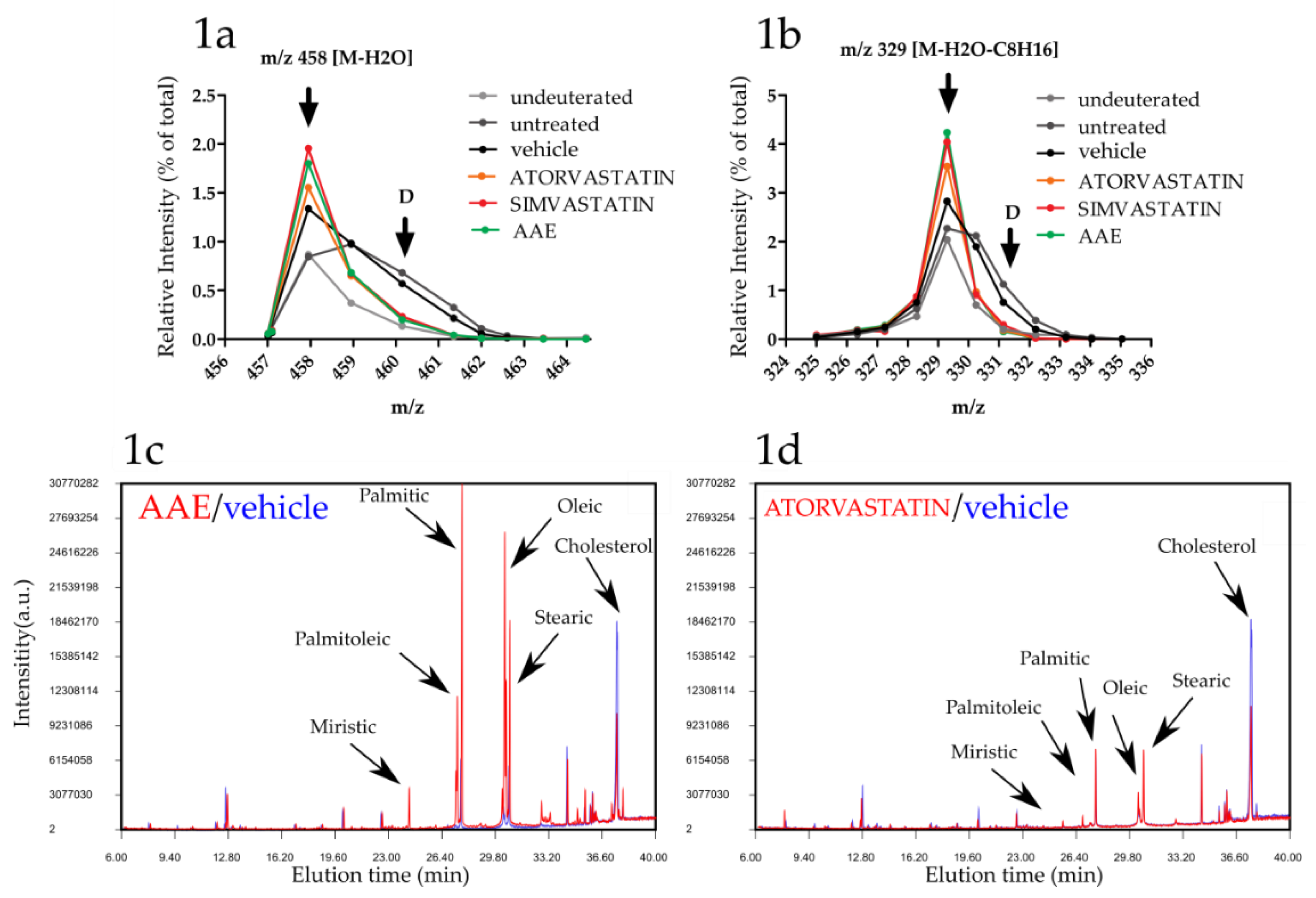
3.1. AAE Inhibits Cholesterogenesis in HuH7 Cells
3.2. AAE Induces Intracellular Accumulation of FAs
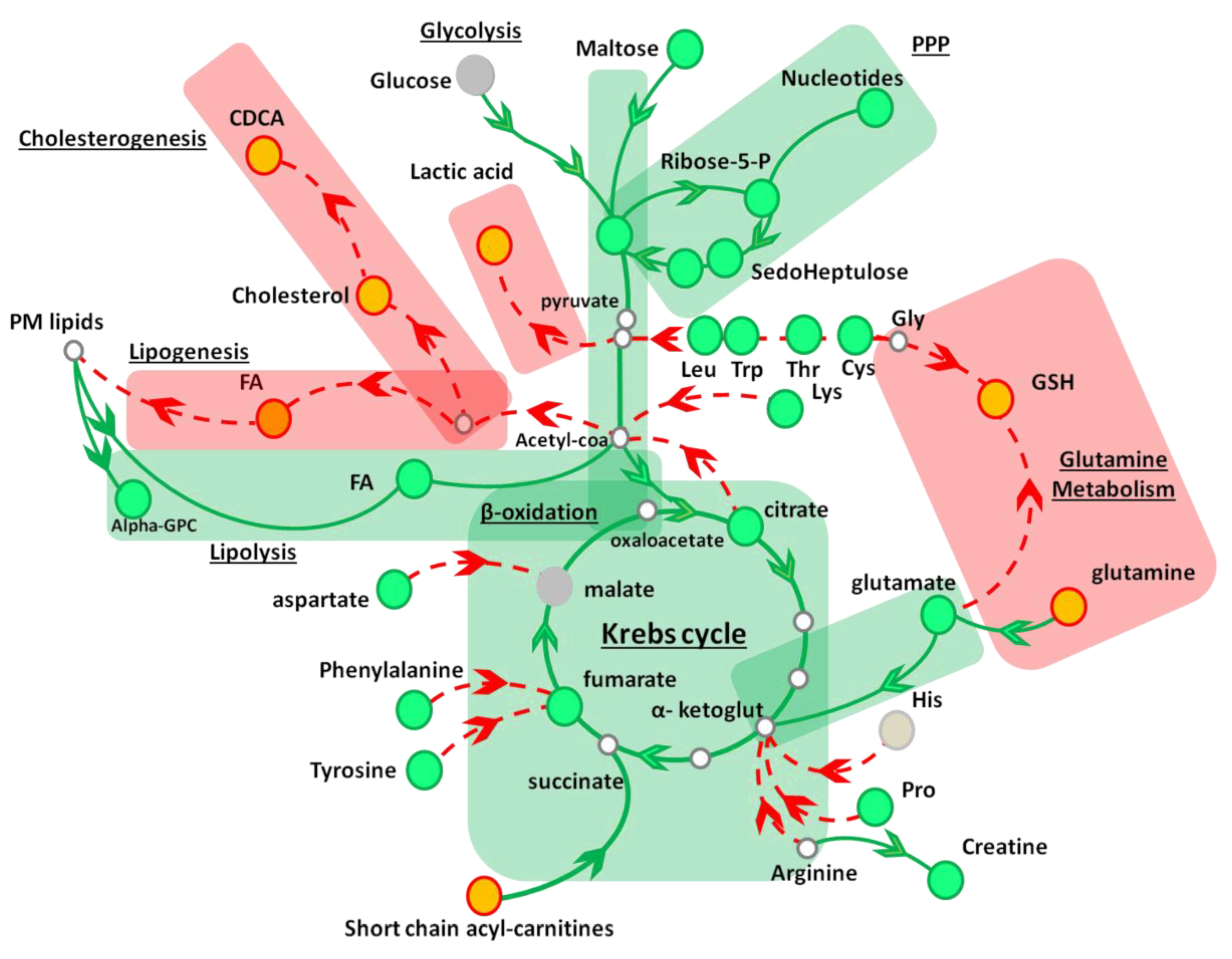
3.3. AAE Reprograms FA Metabolism and Diverts Acetyl-CoA to Krebs Cycle
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Townsend, N.; Nichols, M.; Scarborough, P.; Rayner, M. Cardiovascular disease in Europe--epidemiological update 2015. Eur Hear. J. 2015, 36, 2696–2705. [Google Scholar] [CrossRef] [PubMed]
- Koutsos, A.; Tuohy, K.M.; Lovegrove, J.A. Apples and cardiovascular health—Is the gut microbiota a core consideration? Nutrients 2015, 7, 3959–3998. [Google Scholar] [CrossRef]
- Catapano, A.L.; Graham, I.; De Backer, G.; Wiklund, O.; Chapman, M.J.; Drexel, H.; Hoes, A.W.; Jennings, C.S.; Landmesser, U.; Pedersen, T.R.; et al. 2016 ESC/EAS guidelines for the management of dyslipidaemias. Eur. Heart J. 2016, 37, 2999–3058. [Google Scholar] [CrossRef] [PubMed]
- Shivappa, N.; Godos, J.; Hébert, J.R.; Wirth, M.D.; Piuri, G.; Speciani, A.F.; Grosso, G. Dietary inflammatory index and cardiovascular risk and mortality—A meta-analysis. Nutrients 2018. [Google Scholar] [CrossRef] [PubMed]
- Briggs, A.D.M.; Mizdrak, A.; Scarborough, P. A statin a day keeps the doctor away: Comparative proverb assessment modelling study. BMJ 2013, 347, 1–6. [Google Scholar] [CrossRef]
- Taylor, B.A.; Lorson, L.; White, C.M.; Thompson, P.D. A randomized trial of coenzyme Q10 in patients with confirmed Statin Myopathy. Atherosclerosis 2015, 238, 329–335. [Google Scholar] [CrossRef] [PubMed]
- Bitzur, R.; Cohen, H.; Kamari, Y.; Harats, D. Intolerance to statins: Mechanisms and management. Diabetes Care 2013, 36. [Google Scholar] [CrossRef]
- Thompson, P.D.; Panza, G.; Zaleski, A.; Taylor, B. Statin-associated side effects. J. Am. Coll. Cardiol. 2016, 67, 2395–2410. [Google Scholar] [CrossRef]
- Grosso, G. Effects of polyphenol-rich foods on human health. Nutrients 2018. [Google Scholar] [CrossRef]
- Mink, P.J.; Scrafford, C.G.; Barraj, L.M.; Harnack, L.; Hong, C.-P.; Nettleton, J.A.; Jacobs, D.R. Flavonoid intake and cardiovascular disease mortality: A prospective study in postmenopausal women. Am. J. Clin. Nutr. 2007. [Google Scholar] [CrossRef]
- Hertog, M.G.L.; Feskens, E.J.M.; Kromhout, D.; Hertog, M.G.L.; Hollman, P.C.H.; Hertog, M.G.L.; Katan, M.B. Dietary antioxidant flavonoids and risk of coronary heart disease: The Zutphen Elderly Study. Lancet 1993. [Google Scholar] [CrossRef]
- Wang, X.; Ouyang, Y.Y.; Liu, J.; Zhao, G. Systematic review with meta-analysis Flavonoid intake and risk of CVD: A systematic review and meta-analysis of prospective cohort studies. Br. J. Nutr. 2014, 111, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Knekt, P.; Kumpulainen, J.; Järvinen, R.; Rissanen, H.; Heliövaara, M.; Reunanen, A.; Hakulinen, T.; Aromaa, A. Flavonoid intake and risk of chronic diseases. Am. J. Clin. Nutr. 2002. [Google Scholar] [CrossRef] [PubMed]
- Santini, A.; Tenore, G.C.; Novellino, E. Nutraceuticals: A paradigm of proactive medicine. Eur. J. Pharm. Sci. 2017, 96, 53–61. [Google Scholar] [CrossRef] [PubMed]
- Tang, G.Y.; Meng, X.; Li, Y.; Zhao, C.N.; Liu, Q.; Li, H.B. Effects of Vegetables on Cardiovascular Diseases and Related Mechanisms. Nutrients 2017. [Google Scholar] [CrossRef] [PubMed]
- Nagasako-Akazome, Y.; Kanda, T.; Ikeda, M.; Shimasaki, H. Serum Cholesterol-Lowering Effect of Apple Polyphenols in Healthy Subjects. J. Oleo Sci. 2005, 54, 143–151. [Google Scholar] [CrossRef]
- Nagasako-akazome, Y.; Kanda, T.; Ohtake, Y.; Shimasaki, H.; Kobayashi, T. Apple Polyphenols Influence Cholesterol Metabolism in Healthy Subjects with Relatively High Body Mass Index. J. Oleo Sci. 2007, 56, 417–428. [Google Scholar] [CrossRef]
- Osada, K.; Funayama, M.; Fuchi, S.; Sami, M.; Ohta, Y.; Kanda, T.; Ikeda, M. Effects of Dietary Procyanidins and Tea Polyphenols on Adipose Tissue Mass and Fatty Acid Metabolism in Rats on a High Fat Diet. J. Oleo Sci. 2006, 55, 79–89. [Google Scholar] [CrossRef]
- Opyd, P.M.; Jurgoński, A.; Juśkiewicz, J.; Milala, J.; Zduńczyk, Z.; Król, B. Nutritional and health-related effects of a diet containing apple seed meal in rats: The case of amygdalin. Nutrients 2017, 9, 1091. [Google Scholar] [CrossRef]
- Pirillo, A.; Catapano, A.L. Berberine, a plant alkaloid with lipid- and glucose-lowering properties: From in vitro evidence to clinical studies. Atherosclerosis 2015, 243, 449–461. [Google Scholar] [CrossRef]
- Zhao, C.; Meng, X.; Li, Y.; Li, S.; Liu, Q.; Tang, G.; Li, H. Fruits for Prevention and Treatment of Cardiovascular Diseases. 2017, 1–29. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Melnyk, J.P.; Tsao, R.; Marcone, M.F. How natural dietary antioxidants in fruits, vegetables and legumes promote vascular health. Food Res. Int. 2011, 44, 14–22. [Google Scholar] [CrossRef]
- Panche, A.N.; Diwan, A.D.; Chandra, S.R. Flavonoids: An overview. J. Nutr. Sci. 2016. [Google Scholar] [CrossRef] [PubMed]
- Serra, A.T.; Rocha, J.; Sepodes, B.; Matias, A.A.; Feliciano, R.P.; De Carvalho, A.; Bronze, M.R.; Duarte, C.M.M.; Figueira, M.E. Evaluation of cardiovascular protective effect of different apple varieties— Correlation of response with composition. Food Chem. 2012. [Google Scholar] [CrossRef] [PubMed]
- Tenore, G.C.; Campiglia, P.; Ritieni, A.; Novellino, E. In vitro bioaccessibility, bioavailability and plasma protein interaction of polyphenols from Annurca apple (M. pumila Miller cv Annurca). Food Chem. 2013, 141, 3519–3524. [Google Scholar] [CrossRef]
- Tenore, G.C.; Caruso, D.; Buonomo, G.; D’Urso, E.; D’Avino, M.; Campigli, P.; Marinelli, L.; Novellino, E. Annurca (Malus pumilamiller cv. Annurca) apple as a functional food for the contribution to a healthy balance of plasma cholesterol levels: Results of a randomized clinical trial. J. Sci. Food Agric. 2016, 97, 2107–2115. [Google Scholar] [CrossRef]
- Tenore, G.C.; Caruso, D.; Buonomo, G.; D’Avino, M.; Campiglia, P.; Marinelli, L.; Novellino, E. A Healthy Balance of Plasma Cholesterol by a Novel Annurca Apple-Based Nutraceutical Formulation: Results of a Randomized Trial. J. Med. Food 2017, 20, 288–300. [Google Scholar] [CrossRef]
- Tenore, G.C.; Carotenuto, A.; Caruso, D.; Buonomo, G.; D’Avino, M.; Brancaccio, D.; Ciampaglia, R.; Maisto, M.; Schisano, C.; Novellino, E. A nutraceutical formulation based on Annurca apple polyphenolic extract is effective on intestinal cholesterol absorption: A randomised, placebo-controlled, crossover study. PharmaNutrition 2018, 6, 85–94. [Google Scholar] [CrossRef]
- Tenore, G.C.; Calabrese, G.; Stiuso, P.; Ritieni, A.; Giannetti, D.; Novellino, E. Effects of Annurca apple polyphenols on lipid metabolism in HepG2 cell lines: A source of nutraceuticals potentially indicated for the metabolic syndrome. Food Res. Int. 2014, 63, 252–257. [Google Scholar] [CrossRef]
- Stirpe, M.; Palermo, V.; Bianchi, M.M.; Silvestri, R.; Falcone, C.; Tenore, G.; Novellino, E.; Mazzoni, C. Annurca apple (M. pumila Miller cv Annurca) extracts act against stress and ageing in S. cerevisiae yeast cells. BMC Complement. Altern. Med. 2017. [Google Scholar] [CrossRef]
- Sommella, E.; Ismail, O.H.; Pagano, F.; Pepe, G.; Ostacolo, C.; Mazzoccanti, G.; Russo, M.; Novellino, E.; Gasparrini, F.; Campiglia, P. Development of an improved online comprehensive hydrophilic interaction chromatography × reversed-phase ultra-high-pressure liquid chromatography platform for complex multiclass polyphenolic sample analysis. J. Sep. Sci. 2017, 40, 2188–2197. [Google Scholar] [CrossRef] [PubMed]
- Sommella, E.; Pepe, G.; Pagano, F.; Ostacolo, C.; Tenore, G.C.; Russo, M.T.; Novellino, E.; Manfra, M.; Campiglia, P. Detailed polyphenolic profiling of Annurca apple (M. pumila Miller cv Annurca) by a combination of RP-UHPLC and HILIC, both hyphenated to IT-TOF mass spectrometry. Food Res. Int. 2015, 76, 466–477. [Google Scholar] [CrossRef] [PubMed]
- Riccio, G.; Maisto, M.; Bottone, S.; Badolati, N.; Rossi, G.B.; Tenore, G.C.; Stornaiuolo, M.; Novellino, E. WNT inhibitory activity of malus pumila miller cv annurca and malus domestica cv limoncella apple extracts on human colon-rectal cells carrying familial adenomatous polyposis mutations. Nutrients 2017, 9. [Google Scholar] [CrossRef] [PubMed]
- Badolati, N.; Sommella, E.; Riccio, G.; Salviati, E.; Heintz, D.; Bottone, S.; Di Cicco, E.; Dentice, M.; Tenore, G.; Campiglia, P.; et al. Annurca Apple Polyphenols Ignite Keratin Production in Hair Follicles by Inhibiting the Pentose Phosphate Pathway and Amino Acid Oxidation. Nutrients 2018, 10. [Google Scholar] [CrossRef] [PubMed]
- Riccio, G.; Sommella, E.; Badolati, N.; Salviati, E.; Bottone, S.; Campiglia, P.; Dentice, M.; Tenore, G.; Stornaiuolo, M.; Novellino, E. Annurca Apple Polyphenols Protect Murine Hair Follicles from Taxane Induced Dystrophy and Hijacks Polyunsaturated Fatty Acid Metabolism toward β-Oxidation. Nutrients 2018, 10. [Google Scholar] [CrossRef] [PubMed]
- Esterman, A.L.; Cohen, B.I.; Javitt, N.B. Cholesterol metabolism: Use of D2O for determination of synthesis rate in cell culture. J. Lipid Res. 1985, 26, 950–954. [Google Scholar] [PubMed]
- Castro-Perez, J.; Previs, S.F.; McLaren, D.G.; Shah, V.; Herath, K.; Bhat, G.; Johns, D.G.; Wang, S.-P.; Mitnaul, L.; Jensen, K.; et al. In vivo D2O labeling to quantify static and dynamic changes in cholesterol and cholesterol esters by high resolution LC/MS. J. Lipid Res. 2011, 52, 159–169. [Google Scholar] [CrossRef]
- Johnson, S.; Rabinovitch, P. Ex vivo imaging of excised tissue using vital dyes and confocal microscopy. Curr. Protoc. Cytom. 2012. [Google Scholar] [CrossRef]
- Riccio, G.; Bottone, S.; La Regina, G.; Badolati, N.; Passacantilli, S.; Rossi, G.B.; Accardo, A.; Dentice, M.; Silvestri, R.; Novellino, E.; et al. A Negative Allosteric Modulator of WNT Receptor Frizzled 4 Switches into an Allosteric Agonist. Biochemistry 2018, 57, 839–851. [Google Scholar] [CrossRef]
- Ser, Z.; Liu, X.; Tang, N.N.; Locasale, J.W. Extraction parameters for metabolomics from cultured cells. Anal. Biochem. 2015. [Google Scholar] [CrossRef]
- Sommella, E.; Conte, G.M.; Salviati, E.; Pepe, G.; Bertamino, A.; Ostacolo, C.; Sansone, F.; Prete, F.D.; Aquino, R.P.; Campiglia, P. Fast profiling of natural pigments in different spirulina (arthrospira platensis) dietary supplements by DI-FT-ICR and evaluation of their antioxidant potential by pre-column DPPH-UHPLC assay. Molecules 2018, 23. [Google Scholar] [CrossRef] [PubMed]
- Tenore, G.C.; Campiglia, P.; Stiuso, P.; Ritieni, A.; Novellino, E. Nutraceutical potential of polyphenolic fractions from Annurca apple (M. pumila Miller cv Annurca). Food Chem. 2013, 140, 614–622. [Google Scholar] [CrossRef] [PubMed]
- Dhaliya, S.A.; Surya, A.S.; Dawn, V.T.; Betty, C.; Arun, K.; Sunil, C. A Review of Hyperlipidemia and Medicinal Plants. Int.J.A.PS.BMS 2013, 2, 219–237. [Google Scholar]
- Agrawal, A.D. Pharmacological activities of flavonoids: A review. Int. J. Pharm. Sci. Nanotechnol. 2011, 4, 1394–1398. [Google Scholar] [CrossRef]
- Amani, R. Flavonoid-rich beverage effects on lipid profile and blood pressure in diabetic patients. World J. Diabetes 2014, 5, 962–968. [Google Scholar] [CrossRef] [PubMed]
- Chavez-Santoscoy, R.A.; Gutierrez-Uribe, J.A.; Granados, O.; Torre-Villalvazo, I.; Serna-Saldivar, S.O.; Torres, N.; Palacios-González, B.; Tovar, A.R. Flavonoids and saponins extracted from black bean (Phaseolus vulgaris L.) seed coats modulate lipid metabolism and biliary cholesterol secretion in C57BL/6 mice. Br. J. Nutr. 2014, 112, 886–899. [Google Scholar] [CrossRef] [PubMed]
- Shao, D.; Lian, Z.; Di, Y.; Zhang, L.; shahid riaz Rajoka, M.; Zhang, Y.; Kong, J.; Jiang, C.; Shi, J. Dietary compounds have potential in controlling atherosclerosis by modulating macrophage cholesterol metabolism and inflammation via miRNA. npj Sci. Food 2018. [Google Scholar] [CrossRef]
- Want, E.J.; Masson, P.; Michopoulos, F.; Wilson, I.D.; Theodoridis, G.; Plumb, R.S.; Shockcor, J.; Loftus, N.; Holmes, E.; Nicholson, J.K. Global metabolic profiling of animal and human tissues via UPLC MS. Nat. Protoc. 2013, 8, 17–32. [Google Scholar] [CrossRef]
- Sunagawa, T.; Ohta, Y.; Sami, M.; Kanda, T.; Osada, K. Hypocholesterolemic Effect of Dietary Apple Polyphenol Is Associated with Alterations in Hepatic Gene Expression Related to Cholesterol Metabolism in Rats. Int. J. Life Sci. Med. Res. 2016. [Google Scholar] [CrossRef]
- Masuda, I.; Koike, M.; Nakashima, S.; Mizutani, Y.; Ozawa, Y.; Watanabe, K.; Sawada, Y.; Sugiyama, H.; Sugimoto, A.; Nojiri, H.; et al. Apple procyanidins promote mitochondrial biogenesis and proteoglycan biosynthesis in chondrocytes. Sci. Rep. 2018, 8, 1–13. [Google Scholar] [CrossRef]
- Abraham Domínguez-Avila, J.; González-Aguilar, G.A.; Alvarez-Parrilla, E.; de la Rosa, L.A. Modulation of PPAR expression and activity in response to polyphenolic compounds in high fat diets. Int. J. Mol. Sci. 2016. [CrossRef]
- Ali, F.; Ismail, A.; Esa, N.M.; Pei, C.P. Transcriptomics expression analysis to unveil the molecular mechanisms underlying the cocoa polyphenol treatment in diet-induced obesity rats. Genomics 2015, 105, 23–30. [Google Scholar] [CrossRef] [PubMed]
- Kozłowska, A.; Szostak-Wegierek, D. Flavonoids—Food sources and health benefits. Rocz. Państwowego Zakładu Hig. 2014, 65, 79–85. [Google Scholar]
- Jung, M.; Triebel, S.; Anke, T.; Richling, E.; Erkel, G. Influence of apple polyphenols on inflammatory gene expression. Mol. Nutr. Food Res. 2009, 53, 1263–1280. [Google Scholar] [CrossRef] [PubMed]
- Wang, N.; Liu, W.; Zhang, T.; Jiang, S.; Xu, H.; Wang, Y.; Zhang, Z.; Wang, C.; Chen, X. Transcriptomic Analysis of Red-Fleshed Apples Reveals the Novel Role of MdWRKY11 in Flavonoid and Anthocyanin Biosynthesis. J. Agric. Food Chem. 2018, 66, 7076–7086. [Google Scholar] [CrossRef] [PubMed]
- Ohta, Y.; Sami, M.; Kanda, T.; Saito, K.; Osada, K.; Kato, H. Gene expression analysis of the anti-obesity effect by apple polyphenols in rats fed a high fat diet or a normal diet. J. Oleo Sci. 2006, 55, 305–314. [Google Scholar] [CrossRef]
- Shabrova, E.V.; Tarnopolsky, O.; Singh, A.P.; Plutzky, J.; Vorsa, N.; Quadro, L. Insights into the molecular mechanisms of the anti-atherogenic actions of flavonoids in normal and obese mice. PLoS ONE 2011, 6, e24634. [Google Scholar] [CrossRef] [PubMed]
- Kopustinskiene, D.M.; Savickas, A.; Vetchý, D.; Masteikova, R.; Kasauskas, A.; Bernatoniene, J. Direct effects of (-)-Epicatechin and procyanidin B2 on the respiration of rat heart mitochondria. Biomed. Res. Int. 2015. [Google Scholar] [CrossRef]
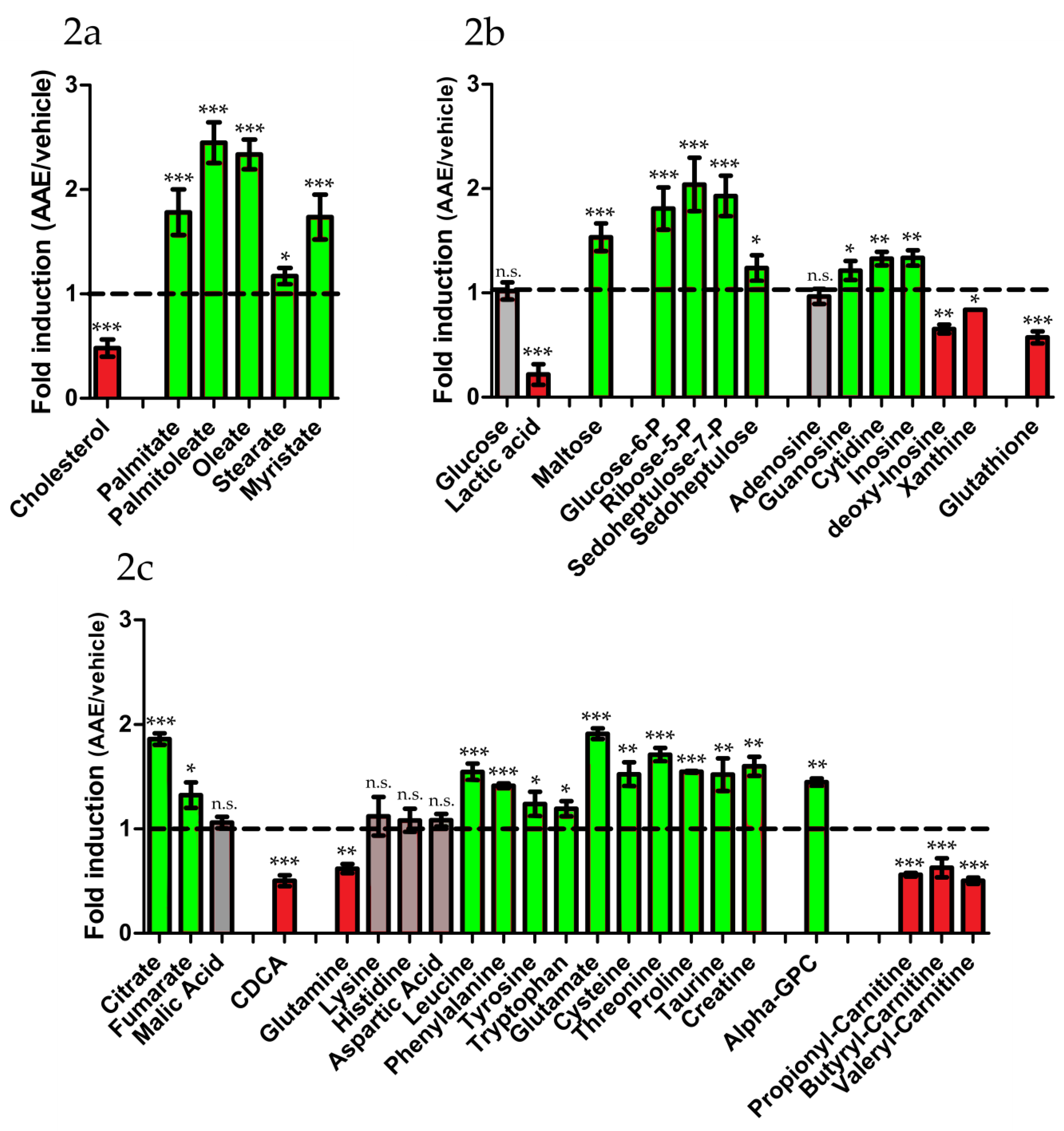
| Metabolic Pathway | Metabolite | Fold change 1 | Metabolic Pathway | Metabolite | Fold Change 1 |
|---|---|---|---|---|---|
| Glycolysis | PPP | ||||
| Glucose | 1.02 ± 0.08 | Ribose 5-P | 2.04 ± 0.26 | ||
| Glucose 6-P | 1.81 ± 0.2 | Sedoheptulose | 1.24 ± 0.12 | ||
| Lactate | 0.22 ± 0.10 | Sedoheptulose-7P | 1.93 ± 0.20 | ||
| Glycogenolysis | Nucleotides | ||||
| Maltose | 1.56 ± 0.13 | Xanthine | 0.84 ± 0.01 | ||
| Amino acids | Adenosine | 0,96 ± 0.07 | |||
| Proline | 1.54 ± 0.04 | Cytidine | 1.33 ± 0.07 | ||
| Threonine | 1.71 ± 0.07 | Guanosine | 1.21 ± 0.09 | ||
| Glutamine | 0.62 ± 0.04 | Inosine | 1.34 ± 0.07 | ||
| Lysine | 0.97 ± 0.12 | Deoxy-inosine | 0.65 ± 0.04 | ||
| Histidine | 1.08 ± 0.11 | GSH | 0.57 ± 0.02 | ||
| Cysteine | 1.52 ± 0.11 | β-oxidation | |||
| Tryptophan | 1.19 ± 0.07 | Propionyl-carn | 0.54 ± 0.05 | ||
| Taurine | 1.52 ± 0.16 | Butyryl-carn | 0.56 ± 0.02 | ||
| Creatine | 1.60 ± 0.09 | Valeryl-carn | 0.50 ± 0.03 | ||
| Glutamic Acid | 1.91 ± 0.05 | ||||
| Leucine | 1.54 ± 0.08 | Krebs cycle | |||
| Tyrosine | 1.24 ± 0.12 | Citrate | 1.86 ± 0.06 | ||
| Phenylalanine | 1.41 ± 0.02 | Fumarate | 1.32 ± 0.12 | ||
| Aspartic Acid | 1.08 ± 0.06 | Malate | 1.06 ± 0.05 | ||
| Lipids | Bile acids | ||||
| Cholesterol | 0.48 ± 0.08 | CDCA | 0.50 ± 0.05 | ||
| Palmitic Acid | 1.78 ± 0.22 | ||||
| Palmitoleic Acid | 2.45 ± 0.20 | ||||
| Stearic Acid | 1.17 ± 0.22 | ||||
| Oleic Acid | 2.34 ± 0.14 | ||||
| Myristic acid | 1.73± 0.14 | ||||
| α - GPC | 1.44 ± 0.04 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sommella, E.; Badolati, N.; Riccio, G.; Salviati, E.; Bottone, S.; Dentice, M.; Campiglia, P.; Tenore, G.C.; Stornaiuolo, M.; Novellino, E. A Boost in Mitochondrial Activity Underpins the Cholesterol-Lowering Effect of Annurca Apple Polyphenols on Hepatic Cells. Nutrients 2019, 11, 163. https://doi.org/10.3390/nu11010163
Sommella E, Badolati N, Riccio G, Salviati E, Bottone S, Dentice M, Campiglia P, Tenore GC, Stornaiuolo M, Novellino E. A Boost in Mitochondrial Activity Underpins the Cholesterol-Lowering Effect of Annurca Apple Polyphenols on Hepatic Cells. Nutrients. 2019; 11(1):163. https://doi.org/10.3390/nu11010163
Chicago/Turabian StyleSommella, Eduardo, Nadia Badolati, Gennaro Riccio, Emanuela Salviati, Sara Bottone, Monica Dentice, Pietro Campiglia, Gian Carlo Tenore, Mariano Stornaiuolo, and Ettore Novellino. 2019. "A Boost in Mitochondrial Activity Underpins the Cholesterol-Lowering Effect of Annurca Apple Polyphenols on Hepatic Cells" Nutrients 11, no. 1: 163. https://doi.org/10.3390/nu11010163
APA StyleSommella, E., Badolati, N., Riccio, G., Salviati, E., Bottone, S., Dentice, M., Campiglia, P., Tenore, G. C., Stornaiuolo, M., & Novellino, E. (2019). A Boost in Mitochondrial Activity Underpins the Cholesterol-Lowering Effect of Annurca Apple Polyphenols on Hepatic Cells. Nutrients, 11(1), 163. https://doi.org/10.3390/nu11010163