Preconception Micronutrient Supplementation Reduced Circulating Branched Chain Amino Acids at 12 Weeks Gestation in an Open Trial of Guatemalan Women Who Are Overweight or Obese
Abstract
1. Introduction
2. Materials and Methods
2.1. Blood Collection and Amino Acid Profiling Using Dried Blood Spot Cards
2.2. Statistical Analysis
3. Results
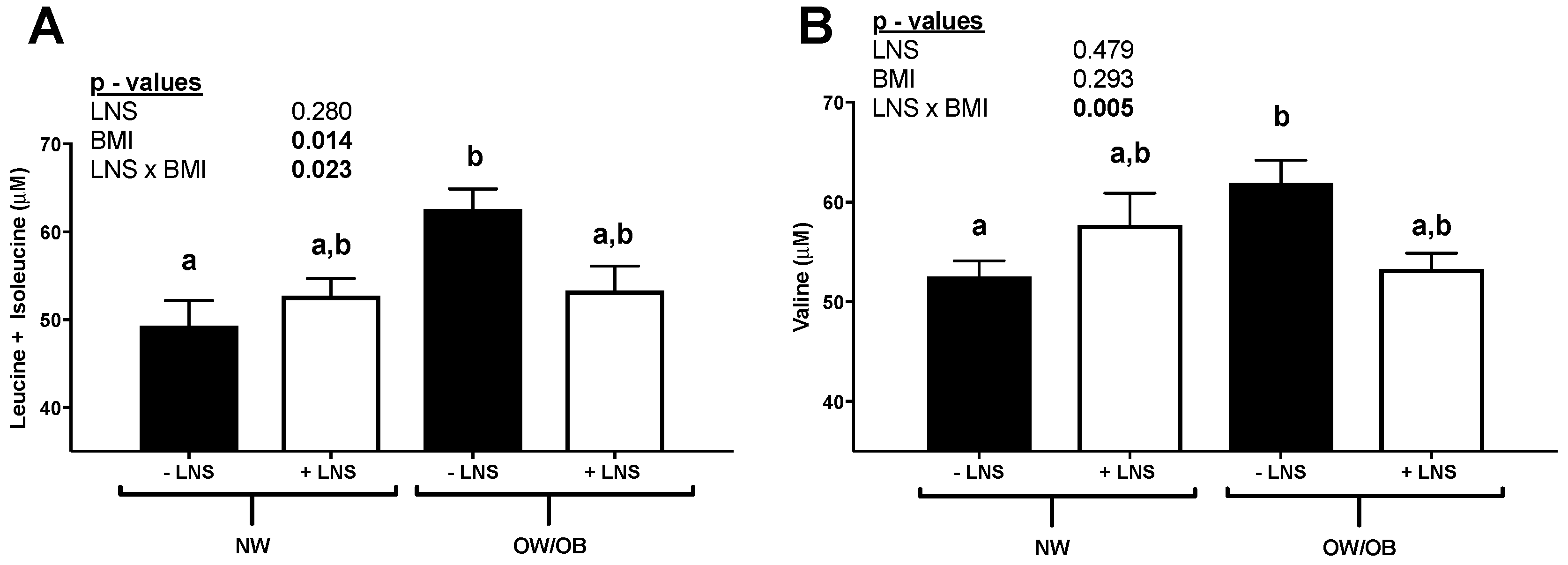
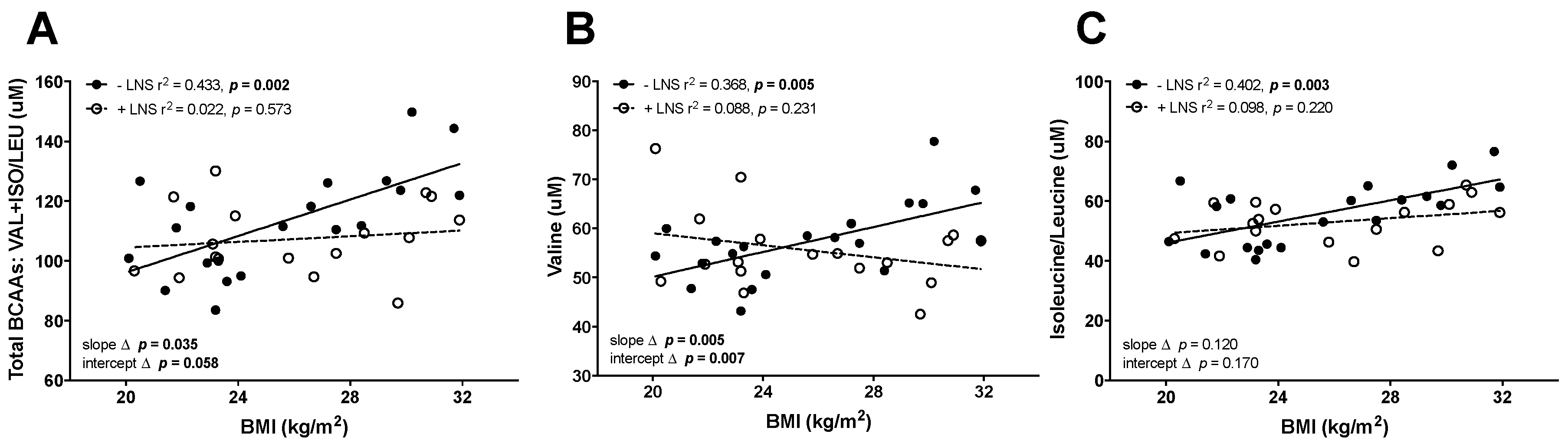
3.1. Elevated BCAAs in OW/OB Mothers and Maternal ppBMI Positively Correlated with the Leptin/Adiponectin Ratio at 12 Weeks Gestation in −LNS Groups
3.2. Maternal ppBMI Correlated with the Leptin/Adiponectin Ratio Was Not Correlated with BCAAs in +LNS Mothers
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Black, R.E.; Victora, C.G.; Walker, S.P.; Bhutta, Z.A.; Christian, P.; de Onis, M.; Ezzati, M.; Grantham-McGregor, S.; Katz, J.; Martorell, R.; et al. Maternal and child undernutrition and overweight in low-income and middle-income countries. Lancet 2013, 382, 427–451. [Google Scholar] [CrossRef]
- Victora, C.G.; Rivera, J.A. Optimal child growth and the double burden of malnutrition: research and programmatic implications. Am. J. Clin. Nutr. 2014, 100, 1611S–1612S. [Google Scholar] [CrossRef] [PubMed]
- Norris, S.A.; Osmond, C.; Gigante, D.; Kuzawa, C.W.; Ramakrishnan, L.; Lee, N.R.; Ramirez-Zea, M.; Richter, L.M.; Stein, A.D.; Tandon, N.; et al. Size at birth, weight gain in infancy and childhood, and adult diabetes risk in five low- or middle-income country birth cohorts. Diabetes Care 2012, 35, 72–79. [Google Scholar] [CrossRef] [PubMed]
- Ashwal, E.; Hadar, E.; Hod, M. Diabetes in low-resourced countries. Best Pract. Res. Clin. Obstet. Gynaecol. 2015, 29, 91–101. [Google Scholar] [CrossRef] [PubMed]
- Prentice, A.M. The double burden of malnutrition in countries passing through the economic transition. Ann. Nutr. Metab. 2018, 72 (Suppl. 3), 47–54. [Google Scholar] [CrossRef]
- Popkin, B.M.; Reardon, T. Obesity and the food system transformation in Latin America. Obes. Rev. 2018. [Google Scholar] [CrossRef] [PubMed]
- Black, R.E.; Allen, L.H.; Bhutta, Z.A.; Caulfield, L.E.; de Onis, M.; Ezzati, M.; Mathers, C.; Rivera, J.; Maternal and Child Undernutrition Study Group. Maternal and child undernutrition: global and regional exposures and health consequences. Lancet 2008, 371, 243–260. [Google Scholar] [CrossRef]
- Bhutta, Z.A.; Ahmed, T.; Black, R.E.; Cousens, S.; Dewey, K.; Giugliani, E.; Haider, B.A.; Kirkwood, B.; Morris, S.S.; Sachdev, H.P.; et al. What works? Interventions for maternal and child undernutrition and survival. Lancet 2008, 371, 417–440. [Google Scholar] [CrossRef]
- Victora, C.G.; Adair, L.; Fall, C.; Hallal, P.C.; Martorell, R.; Richter, L.; Sachdev, H.S.; Maternal and Child Undernutrition Study Group. Maternal and child undernutrition: Consequences for adult health and human capital. Lancet 2008, 371, 340–357. [Google Scholar] [CrossRef]
- Dominguez-Salas, P.; Moore, S.E.; Baker, M.S.; Bergen, A.W.; Cox, S.E.; Dyer, R.A.; Fulford, A.J.; Guan, Y.; Laritsky, E.; Silver, M.J.; et al. Maternal nutrition at conception modulates DNA methylation of human metastable epialleles. Nat. Commun. 2014, 5, 3746. [Google Scholar] [CrossRef] [PubMed]
- Barker, D.J. Fetal origins of coronary heart disease. BMJ 1995, 311, 171–174. [Google Scholar] [CrossRef] [PubMed]
- Catalano, P.M. The impact of gestational diabetes and maternal obesity on the mother and her offspring. J. Dev. Orig. Health Dis. 2010, 1, 208–215. [Google Scholar] [CrossRef] [PubMed]
- Sharp, G.C.; Lawlor, D.A.; Richmond, R.C.; Fraser, A.; Simpkin, A.; Suderman, M.; Shihab, H.A.; Lyttleton, O.; McArdle, W.; Ring, S.M.; et al. Maternal pre-pregnancy BMI and gestational weight gain, offspring DNA methylation and later offspring adiposity: findings from the Avon Longitudinal Study of Parents and Children. Int. J. Epidemiol. 2015, 44, 1288–1304. [Google Scholar] [CrossRef] [PubMed]
- Berngard, S.C.; Berngard, J.B.; Krebs, N.F.; Garces, A.; Miller, L.V.; Westcott, J.; Wright, L.L.; Kindem, M.; Hambidge, K.M. Newborn length predicts early infant linear growth retardation and disproportionately high weight gain in a low-income population. Early Hum. Dev. 2013, 89, 967–972. [Google Scholar] [CrossRef] [PubMed]
- Mook-Kanamori, D.O.; Steegers, E.A.; Eilers, P.H.; Raat, H.; Hofman, A.; Jaddoe, V.W. Risk factors and outcomes associated with first-trimester fetal growth restriction. JAMA 2010, 303, 527–534. [Google Scholar] [CrossRef] [PubMed]
- Hambidge, K.M.; Mazariegos, M.; Kindem, M.; Wright, L.L.; Cristobal-Perez, C.; Juarez-Garcia, L.; Westcott, J.E.; Goco, N.; Krebs, N.F. Infant stunting is associated with short maternal stature. J. Pediatr. Gastroenterol. Nutr. 2012, 54, 117–119. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, T.; Hossain, M.; Sanin, K.I. Global burden of maternal and child undernutrition and micronutrient deficiencies. Ann. Nutr. Metab. 2012, 61 (Suppl. 1), 8–17. [Google Scholar] [CrossRef]
- Rao, K.R.; Padmavathi, I.J.; Raghunath, M. Maternal micronutrient restriction programs the body adiposity, adipocyte function and lipid metabolism in offspring: A review. Rev. Endocr. Metab. Disord. 2012, 13, 103–108. [Google Scholar] [CrossRef] [PubMed]
- Stewart, C.P.; Christian, P.; Schulze, K.J.; Leclerq, S.C.; West, K.P., Jr.; Khatry, S.K. Antenatal micronutrient supplementation reduces metabolic syndrome in 6- to 8-year-old children in rural Nepal. J. Nutr. 2009, 139, 1575–1581. [Google Scholar] [CrossRef] [PubMed]
- Carlin, J.; George, R.; Reyes, T.M. Methyl donor supplementation blocks the adverse effects of maternal high fat diet on offspring physiology. PLoS ONE 2013, 8, e63549. [Google Scholar] [CrossRef] [PubMed]
- Dahlhoff, C.; Worsch, S.; Sailer, M.; Hummel, B.A.; Fiamoncini, J.; Uebel, K.; Obeid, R.; Scherling, C.; Geisel, J.; Bader, B.L.; et al. Methyl-donor supplementation in obese mice prevents the progression of NAFLD, activates AMPK and decreases acyl-carnitine levels. Mol. Metab. 2014, 3, 565–580. [Google Scholar] [CrossRef] [PubMed]
- Waterland, R.A.; Travisano, M.; Tahiliani, K.G.; Rached, M.T.; Mirza, S. Methyl donor supplementation prevents transgenerational amplification of obesity. Int. J. Obes. (Lond.) 2008, 32, 1373–1379. [Google Scholar] [CrossRef] [PubMed]
- Newgard, C.B.; An, J.; Bain, J.R.; Muehlbauer, M.J.; Stevens, R.D.; Lien, L.F.; Haqq, A.M.; Shah, S.H.; Arlotto, M.; Slentz, C.A.; et al. A branched-chain amino acid-related metabolic signature that differentiates obese and lean humans and contributes to insulin resistance. Cell Metab. 2009, 9, 311–326. [Google Scholar] [CrossRef] [PubMed]
- Newgard, C.B. Interplay between lipids and branched-chain amino acids in development of insulin resistance. Cell Metab. 2012, 15, 606–614. [Google Scholar] [CrossRef] [PubMed]
- Lynch, C.J.; Adams, S.H. Branched-chain amino acids in metabolic signalling and insulin resistance. Nat. Rev. Endocrinol. 2014, 10, 723–736. [Google Scholar] [CrossRef] [PubMed]
- Jacob, S.; Nodzenski, M.; Reisetter, A.C.; Bain, J.R.; Muehlbauer, M.J.; Stevens, R.D.; Ilkayeva, O.R.; Lowe, L.P.; Metzger, B.E.; Newgard, C.B.; et al. Targeted metabolomics demonstrates distinct and overlapping maternal metabolites associated with bmi, glucose, and insulin sensitivity during pregnancy across four ancestry groups. Diabetes Care 2017, 40, 911–919. [Google Scholar] [CrossRef] [PubMed]
- Moretti, F.; Birarelli, M.; Carducci, C.; Pontecorvi, A.; Antonozzi, I. Simultaneous high-performance liquid chromatographic determination of amino acids in a dried blood spot as a neonatal screening test. J. Chromatogr. 1990, 511, 131–136. [Google Scholar] [CrossRef]
- Wagner, M.; Tonoli, D.; Varesio, E.; Hopfgartner, G. The use of mass spectrometry to analyze dried blood spots. Mass Spectrom. Rev. 2016, 35, 361–438. [Google Scholar] [CrossRef] [PubMed]
- Hambidge, K.M.; Krebs, N.F.; Westcott, J.E.; Garces, A.; Goudar, S.S.; Kodkany, B.S.; Pasha, O.; Tshefu, A.; Bose, C.L.; Figueroa, L.; et al. Preconception maternal nutrition: A multi-site randomized controlled trial. BMC Pregnancy Childbirth 2014, 14, 111. [Google Scholar] [CrossRef] [PubMed]
- Arimond, M.; Zeilani, M.; Jungjohann, S.; Brown, K.H.; Ashorn, P.; Allen, L.H.; Dewey, K.G. Considerations in developing lipid-based nutrient supplements for prevention of undernutrition: Experience from the International Lipid-Based Nutrient Supplements (iLiNS) project. Matern. Child Nutr. 2015, 11 (Suppl. 4), 31–61. [Google Scholar] [CrossRef] [PubMed]
- Ashorn, P.; Alho, L.; Ashorn, U.; Cheung, Y.B.; Dewey, K.G.; Harjunmaa, U.; Lartey, A.; Nkhoma, M.; Phiri, N.; Phuka, J.; et al. The impact of lipid-based nutrient supplement provision to pregnant women on newborn size in rural Malawi: A randomized controlled trial. Am. J. Clin. Nutr. 2015, 101, 387–397. [Google Scholar] [CrossRef] [PubMed]
- Chace, D.H.; DiPerna, J.C.; Mitchell, B.L.; Sgroi, B.; Hofman, L.F.; Naylor, E.W. Electrospray tandem mass spectrometry for analysis of acylcarnitines in dried postmortem blood specimens collected at autopsy from infants with unexplained cause of death. Clin. Chem. 2001, 47, 1166–1182. [Google Scholar] [PubMed]
- Chace, D.H.; Kalas, T.A.; Naylor, E.W. Use of tandem mass spectrometry for multianalyte screening of dried blood specimens from newborns. Clin. Chem. 2003, 49, 1797–1817. [Google Scholar] [CrossRef] [PubMed]
- Chace, D.H.; Adam, B.W.; Smith, S.J.; Alexander, J.R.; Hillman, S.L.; Hannon, W.H. Validation of accuracy-based amino acid reference materials in dried-blood spots by tandem mass spectrometry for newborn screening assays. Clin. Chem. 1999, 45, 1269–1277. [Google Scholar] [PubMed]
- Hochberg, Y.; Benjamini, Y. More powerful procedures for multiple significance testing. Stat. Med. 1990, 9, 811–818. [Google Scholar] [CrossRef] [PubMed]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2017. [Google Scholar]
- Fitch, W.L.; King, J.C. Plasma amino acid, glucose, and insulin responses to moderate-protein and high-protein test meals in pregnant, nonpregnant, and gestational diabetic women. Am. J. Clin. Nutr. 1987, 46, 243–249. [Google Scholar] [CrossRef] [PubMed]
- Ghadimi, H.; Pecora, P. Free amino acids of cord plasma as compared with maternal plasma during pregnancy. Pediatrics 1964, 33, 500–506. [Google Scholar] [PubMed]
- Metzger, B.E.; Phelps, R.L.; Freinkel, N.; Navickas, I.A. Effects of gestational diabetes on diurnal profiles of plasma glucose, lipids, and individual amino acids. Diabetes Care 1980, 3, 402–409. [Google Scholar] [CrossRef] [PubMed]
- Potter, J.M.; Green, A.; Cullen, D.R.; Milner, R.D. Amino acid profiles in early diabetic and non-diabetic pregnancy. Diabetes Res. Clin. Pract. 1986, 2, 123–126. [Google Scholar] [CrossRef]
- Persson, B.; Pschera, H.; Lunell, N.O.; Barley, J.; Gumaa, K.A. Amino acid concentrations in maternal plasma and amniotic fluid in relation to fetal insulin secretion during the last trimester of pregnancy in gestational and type I diabetic women and women with small-for-gestational-age infants. Am. J. Perinatol. 1986, 3, 98–103. [Google Scholar] [CrossRef] [PubMed]
- Zimmer, D.M.; Golichowski, A.M.; Karn, C.A.; Brechtel, G.; Baron, A.D.; Denne, S.C. Glucose and amino acid turnover in untreated gestational diabetes. Diabetes Care 1996, 19, 591–596. [Google Scholar] [CrossRef] [PubMed]
- Bentley-Lewis, R.; Huynh, J.; Xiong, G.; Lee, H.; Wenger, J.; Clish, C.; Nathan, D.; Thadhani, R.; Gerszten, R. Metabolomic profiling in the prediction of gestational diabetes mellitus. Diabetologia 2015, 58, 1329–1332. [Google Scholar] [CrossRef] [PubMed]
- Kalkhoff, R.K.; Kandaraki, E.; Morrow, P.G.; Mitchell, T.H.; Kelber, S.; Borkowf, H.I. Relationship between neonatal birth weight and maternal plasma amino acid profiles in lean and obese nondiabetic women and in type I diabetic pregnant women. Metabolism 1988, 37, 234–239. [Google Scholar] [CrossRef]
- Park, S.; Park, J.Y.; Lee, J.H.; Kim, S.H. Plasma levels of lysine, tyrosine, and valine during pregnancy are independent risk factors of insulin resistance and gestational diabetes. Metab. Syndr. Relat. Disord. 2015, 13, 64–70. [Google Scholar] [CrossRef] [PubMed]
- Sandler, V.; Reisetter, A.C.; Bain, J.R.; Muehlbauer, M.J.; Nodzenski, M.; Stevens, R.D.; Ilkayeva, O.; Lowe, L.P.; Metzger, B.E.; Newgard, C.B.; et al. Associations of maternal BMI and insulin resistance with the maternal metabolome and newborn outcomes. Diabetologia 2017, 60, 518–530. [Google Scholar] [CrossRef] [PubMed]
- Lowe, W.L., Jr.; Bain, J.R.; Nodzenski, M.; Reisetter, A.C.; Muehlbauer, M.J.; Stevens, R.D.; Ilkayeva, O.R.; Lowe, L.P.; Metzger, B.E.; Newgard, C.B.; et al. Maternal BMI and glycemia impact the fetal metabolome. Diabetes Care 2017, 40, 902–910. [Google Scholar] [CrossRef] [PubMed]
- Perng, W.; Gillman, M.W.; Fleisch, A.F.; Michalek, R.D.; Watkins, S.M.; Isganaitis, E.; Patti, M.E.; Oken, E. Metabolomic profiles and childhood obesity. Obesity 2014, 22, 2570–2578. [Google Scholar] [CrossRef] [PubMed]
- She, P.; Van, H.C.; Reid, T.; Hutson, S.M.; Cooney, R.N.; Lynch, C.J. Obesity-related elevations in plasma leucine are associated with alterations in enzymes involved in branched-chain amino acid metabolism. Am. J. Physiol. Endocrinol. Metab. 2007, 293, E1552–E1563. [Google Scholar] [CrossRef] [PubMed]
- Lackey, D.E.; Lynch, C.J.; Olson, K.C.; Mostaedi, R.; Ali, M.; Smith, W.H.; Karpe, F.; Humphreys, S.; Bedinger, D.H.; Dunn, T.N.; et al. Regulation of adipose branched-chain amino acid catabolism enzyme expression and cross-adipose amino acid flux in human obesity. Am. J. Physiol. Endocrinol. Metab. 2013, 304, E1175–E1187. [Google Scholar] [CrossRef] [PubMed]
- Goffredo, M.; Santoro, N.; Trico, D.; Giannini, C.; D’Adamo, E.; Zhao, H.; Peng, G.; Yu, X.; Lam, T.T.; Pierpont, B.; et al. A branched-chain amino acid-related metabolic signature characterizes obese adolescents with non-alcoholic fatty liver disease. Nutrients 2017, 9, E642. [Google Scholar] [CrossRef] [PubMed]
- McCormack, S.E.; Shaham, O.; McCarthy, M.A.; Deik, A.A.; Wang, T.J.; Gerszten, R.E.; Clish, C.B.; Mootha, V.K.; Grinspoon, S.K.; Fleischman, A. Circulating branched-chain amino acid concentrations are associated with obesity and future insulin resistance in children and adolescents. Pediatr. Obes. 2013, 8, 52–61. [Google Scholar] [CrossRef] [PubMed]
- Connelly, M.A.; Wolak-Dinsmore, J.; Dullaart, R.P.F. Branched chain amino acids are associated with insulin resistance independent of leptin and adiponectin in subjects with varying degrees of glucose tolerance. Metab. Syndr. Relat. Disord. 2017, 15, 183–186. [Google Scholar] [CrossRef] [PubMed]
- Adams, S.H. Emerging perspectives on essential amino acid metabolism in obesity and the insulin-resistant state. Adv. Nutr. 2011, 2, 445–456. [Google Scholar] [CrossRef] [PubMed]
- Butte, N.F.; Liu, Y.; Zakeri, I.F.; Mohney, R.P.; Mehta, N.; Voruganti, V.S.; Goring, H.; Cole, S.A.; Comuzzie, A.G. Global metabolomic profiling targeting childhood obesity in the Hispanic population. Am. J. Clin. Nutr. 2015, 102, 256–267. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Han, Q.; Liu, Y.; Sun, C.; Gang, X.; Wang, G. The relationship between branched-chain amino acid related metabolomic signature and insulin resistance: A systematic review. J. Diabetes Res. 2016, 2016, 2794591. [Google Scholar] [CrossRef] [PubMed]
- Trico, D.; Prinsen, H.; Giannini, C.; de Graff, R.; Juchem, C.; Li, F.; Caprio, S.; Santoro, N.; Herzog, R.I. Elevated alpha-hydroxybutyrate and BCAA levels predict deterioration of glycemic control in adolescents. J. Clin. Endocrinol. Metab. 2017, 102, 2473–2481. [Google Scholar] [CrossRef] [PubMed]
- Shah, S.H.; Crosslin, D.R.; Haynes, C.S.; Nelson, S.; Turer, C.B.; Stevens, R.D.; Muehlbauer, M.J.; Wenner, B.R.; Bain, J.R.; Laferrere, B.; et al. Branched-chain amino acid levels are associated with improvement in insulin resistance with weight loss. Diabetologia 2012, 55, 321–330. [Google Scholar] [CrossRef] [PubMed]
- Tan, H.C.; Khoo, C.M.; Tan, M.Z.; Kovalik, J.P.; Ng, A.C.; Eng, A.K.; Lai, O.F.; Ching, J.H.; Tham, K.W.; Pasupathy, S. The effects of sleeve gastrectomy and gastric bypass on branched-chain amino acid metabolism 1 year after bariatric surgery. Obes. Surg. 2016, 26, 1830–1835. [Google Scholar] [CrossRef] [PubMed]
- Lips, M.A.; Van Klinken, J.B.; van Harmelen, V.; Dharuri, H.K.; Ac’t Hoen, P.; Laros, J.F.; van Ommen, G.J.; Janssen, I.M.; Van Ramshorst, B.; Van Wagensveld, B.A.; et al. Roux-en-Y gastric bypass surgery, but not calorie restriction, reduces plasma branched-chain amino acids in obese women independent of weight loss or the presence of type 2 diabetes. Diabetes Care 2014, 37, 3150–3156. [Google Scholar] [CrossRef] [PubMed]
- Magkos, F.; Bradley, D.; Schweitzer, G.G.; Finck, B.N.; Eagon, J.C.; Ilkayeva, O.; Newgard, C.B.; Klein, S. Effect of Roux-en-Y gastric bypass and laparoscopic adjustable gastric banding on branched-chain amino acid metabolism. Diabetes 2013, 62, 2757–2761. [Google Scholar] [CrossRef] [PubMed]
- Barcelo, A.; Bauca, J.M.; Pena-Zarza, J.A.; Morell-Garcia, D.; Yanez, A.; Perez, G.; Pierola, J.; Toledo, N.; de la Pena, M. Circulating branched-chain amino acids in children with obstructive sleep apnea. Pediatr. Pulmonol. 2017, 52, 1085–1091. [Google Scholar] [CrossRef] [PubMed]
- Barcelo, A.; Morell-Garcia, D.; Salord, N.; Esquinas, C.; Perez, G.; Perez, A.; Monasterio, C.; Gasa, M.; Fortuna, A.M.; Montserrat, J.M.; et al. A randomized controlled trial: branched-chain amino acid levels and glucose metabolism in patients with obesity and sleep apnea. J. Sleep Res. 2017, 26, 773–781. [Google Scholar] [CrossRef] [PubMed]
- Reece, E.A.; Coustan, D.R.; Sherwin, R.S.; Tuck, S.; Bates, S.; O’Connor, T.; Tamborlane, W.V. Does intensive glycemic control in diabetic pregnancies result in normalization of other metabolic fuels? Am. J. Obstet. Gynecol. 1991, 165, 126–130. [Google Scholar] [CrossRef]
| BMI (<25) = NW | BMI (≥25) = OW/OB | p-Value | |||||
|---|---|---|---|---|---|---|---|
| Maternal Characteristics | −LNS (n = 10) | +LNS (n = 9) | −LNS (n = 10) | +LNS (n = 10) | LNS | BMI | LNS × BMI |
| Maternal Characteristics: Pre-Pregnancy | |||||||
| Age (years) | 23.8 ± 1.4 | 23.8 ± 1.6 | 24.7 ± 1.3 | 25.7 ± 1.5 | 0.728 | 0.333 | 0.728 |
| Parity | 1.6 ± 0.2 | 1.4 ± 0.3 | 1.6 ± 0.3 | 1.5 ± 0.3 | 0.605 | 0.863 | 0.863 |
| Ht (cm) | 146 ± 1 | 145 ± 2 | 145 ± 1 | 142 ± 2 | 0.305 | 0.295 | 0.559 |
| BW (kg) | 47.6 ± 1.1 | 47.1 ± 1.5 | 60.8 ± 1.6 | 58.6 ± 1.6 | 0.384 | <0.001 | 0.571 |
| BMI (kg/m2) | 22.3 ± 0.4 | 22.3 ± 0.4 | 28.8 ± 0.7 | 28.9 ± 0.6 | 0.935 | <0.001 | 0.892 |
| Compliance (%) | 77.4 ± 5.6 | 72.8 ± 4.7 | 0.533 * | ||||
| SES | 3.6 ± 0.3 | 3.9 ± 0.4 | 4.0 ± 0.2 | 3.2 ± 0.5 | 0.466 | 0.679 | 0.125 |
| Maternal Characteristics: 12 Weeks Gestation | |||||||
| Age (years) | 24.0 ± 1.4 | 24.4 ± 1.8 | 25.1 ± 1.3 | 25.9 ± 1.4 | 0.680 | 0.399 | 0.906 |
| BW (kg) | 47.1 ± 1.1 | 47.9 ± 1.6 | 59.8 ± 1.2 | 58.1 ± 1.8 | 0.764 | <0.001 | 0.407 |
| BMI | 22.1 ± 0.4 | 22.5 ± 0.5 | 28.4 ± 0.6 | 28.7 ± 0.8 | 0.536 | <0.001 | 0.921 |
| (−)LNS | (+)LNS | |||
|---|---|---|---|---|
| Category | Pathway | Variable | vs. ppBMI r-Value (p-Value) | vs. ppBMI r-Value (p-Value) |
| Serum | NA | Total Adiponectin (µg/mL) | −0.323 (0.164) | −0.312 (0.194) |
| Serum | NA | HMW Adiponectin (µg/mL) | −0.107 (0.662) | −0.39 (0.099) |
| Serum | NA | %HMW/Total Ratio | 0.383 (0.095) | −0.372 (0.117) |
| Serum | NA | Leptin (ng/mL) | 0.143 (0.547) | 0.492 (0.033) |
| Serum | NA | Insulin (µIU/mL) | 0.194 (0.413) | 0.266 (0.271) |
| Serum | NA | Leptin/Total Adipo Ratio | 0.466 (0.044) | 0.512 (0.025) |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Borengasser, S.J.; Baker, P.R., II; Kerns, M.E.; Miller, L.V.; Palacios, A.P.; Kemp, J.F.; Westcott, J.E.; Morrison, S.D.; Hernandez, T.L.; Garces, A.; et al. Preconception Micronutrient Supplementation Reduced Circulating Branched Chain Amino Acids at 12 Weeks Gestation in an Open Trial of Guatemalan Women Who Are Overweight or Obese. Nutrients 2018, 10, 1282. https://doi.org/10.3390/nu10091282
Borengasser SJ, Baker PR II, Kerns ME, Miller LV, Palacios AP, Kemp JF, Westcott JE, Morrison SD, Hernandez TL, Garces A, et al. Preconception Micronutrient Supplementation Reduced Circulating Branched Chain Amino Acids at 12 Weeks Gestation in an Open Trial of Guatemalan Women Who Are Overweight or Obese. Nutrients. 2018; 10(9):1282. https://doi.org/10.3390/nu10091282
Chicago/Turabian StyleBorengasser, Sarah J., Peter R. Baker, II, Mattie E. Kerns, Leland V. Miller, Alexandra P. Palacios, Jennifer F. Kemp, Jamie E. Westcott, Seth D. Morrison, Teri L. Hernandez, Ana Garces, and et al. 2018. "Preconception Micronutrient Supplementation Reduced Circulating Branched Chain Amino Acids at 12 Weeks Gestation in an Open Trial of Guatemalan Women Who Are Overweight or Obese" Nutrients 10, no. 9: 1282. https://doi.org/10.3390/nu10091282
APA StyleBorengasser, S. J., Baker, P. R., II, Kerns, M. E., Miller, L. V., Palacios, A. P., Kemp, J. F., Westcott, J. E., Morrison, S. D., Hernandez, T. L., Garces, A., Figueroa, L., Friedman, J. E., Hambidge, K. M., & Krebs, N. F. (2018). Preconception Micronutrient Supplementation Reduced Circulating Branched Chain Amino Acids at 12 Weeks Gestation in an Open Trial of Guatemalan Women Who Are Overweight or Obese. Nutrients, 10(9), 1282. https://doi.org/10.3390/nu10091282






