Dietary Cholesterol Contained in Whole Eggs Is Not Well Absorbed and Does Not Acutely Affect Plasma Total Cholesterol Concentration in Men and Women: Results from 2 Randomized Controlled Crossover Studies
Abstract
1. Introduction
2. Materials and Methods
2.1. Studies 1 and 2: Ethics and Protocol Registration
2.2. Study 1: Subjects
2.3. Study 1: Study Design
2.4. Study 1: Test Meal
2.5. Study 2: Subjects
2.6. Study 2: Study Design
2.7. Study 2: Test Meal
2.8. Studies 1 and 2: Sample Collection and Analyses
2.8.1. Baseline Blood Sample Collection and Lipid-Lipoprotein Analysis
2.8.2. Blood Sample Collection and Plasma and Triacylglycerol-Rich Lipoprotein Fraction Isolation
2.8.3. Total Cholesterol and Triacylglycerol Analyses
2.9. Studies 1 and 2: Power Calculation and Statistical Analysis
3. Results
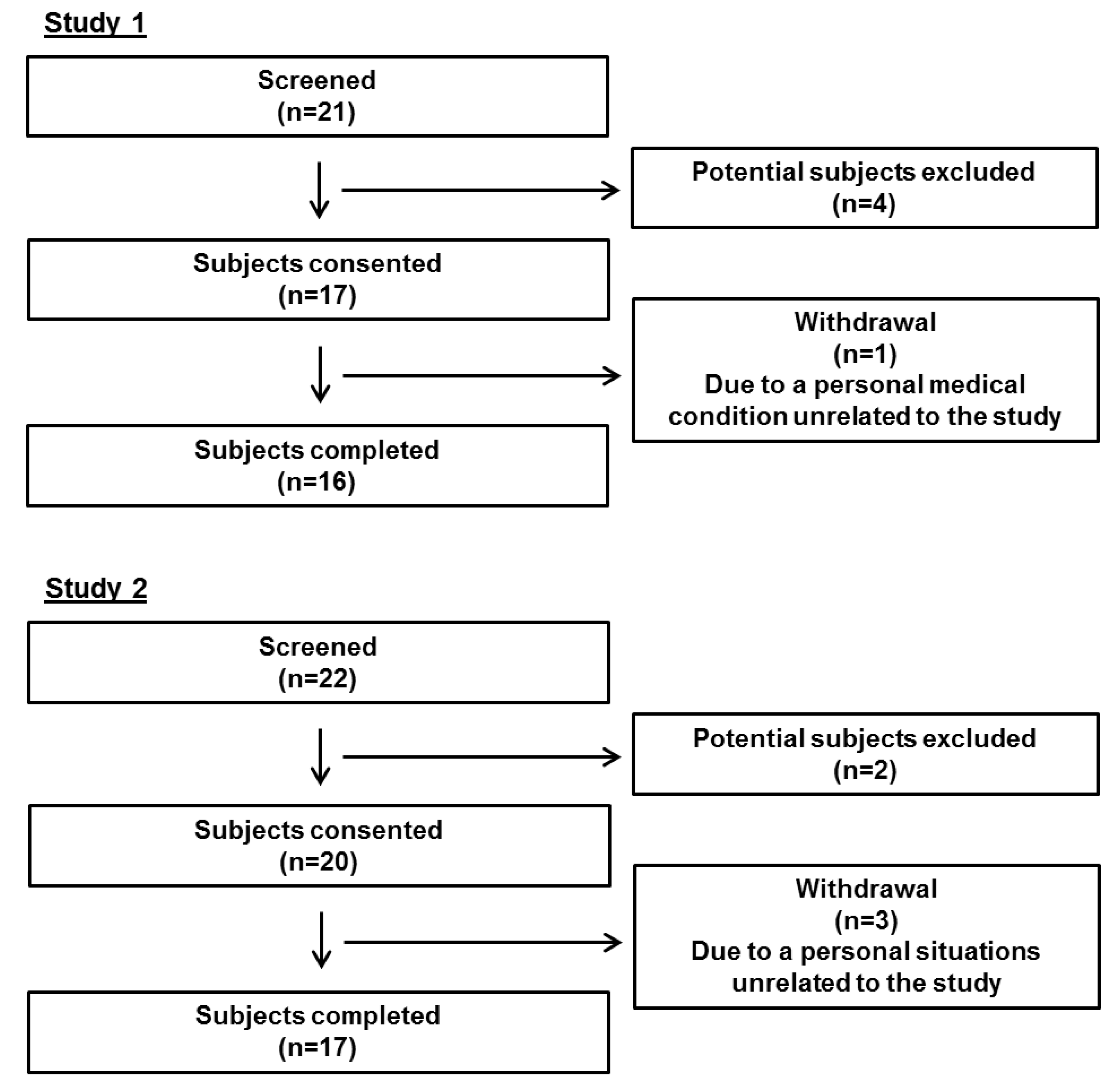
3.1. Subject Baseline Characteristics
3.1.1. Study 1
3.1.2. Study 2
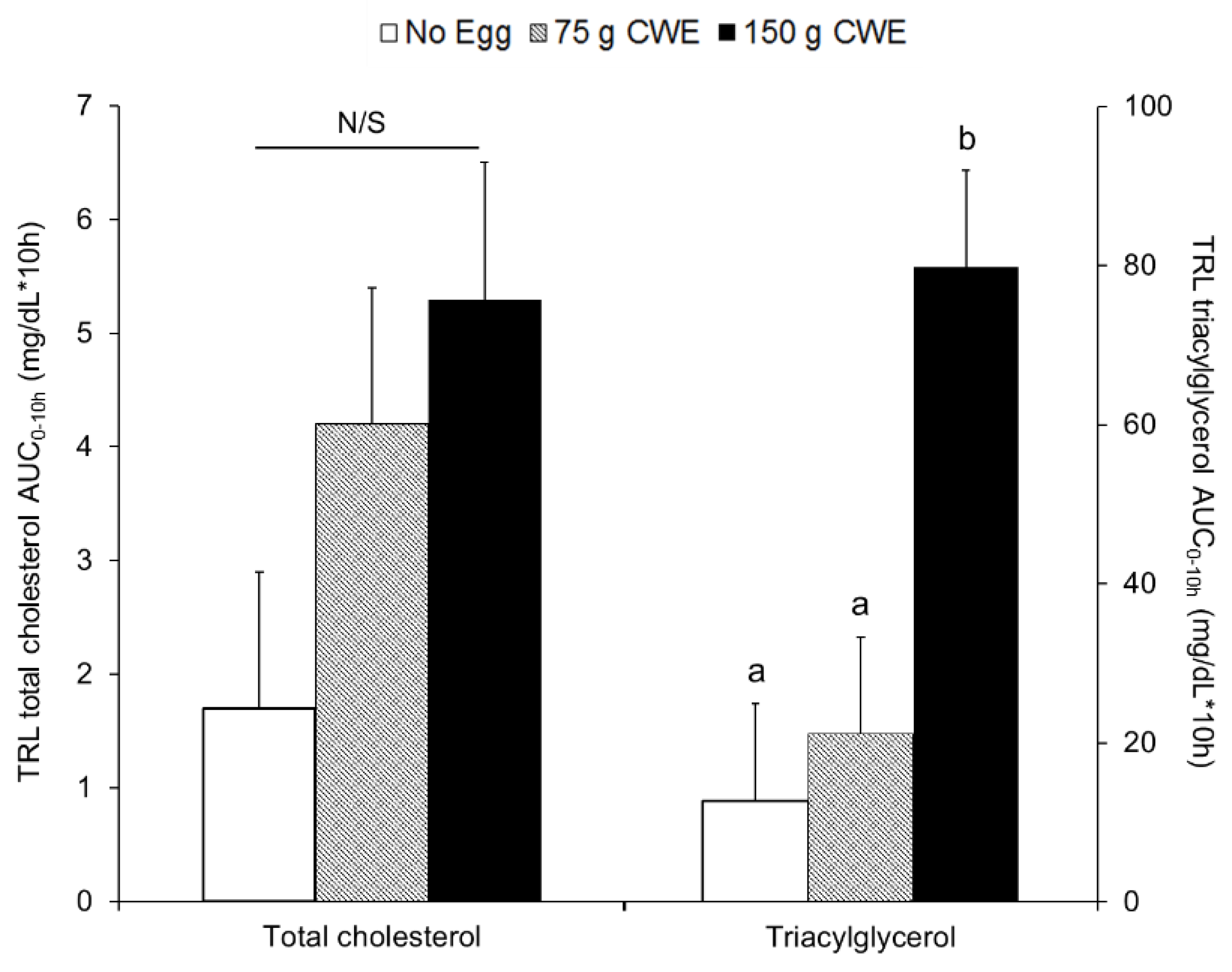
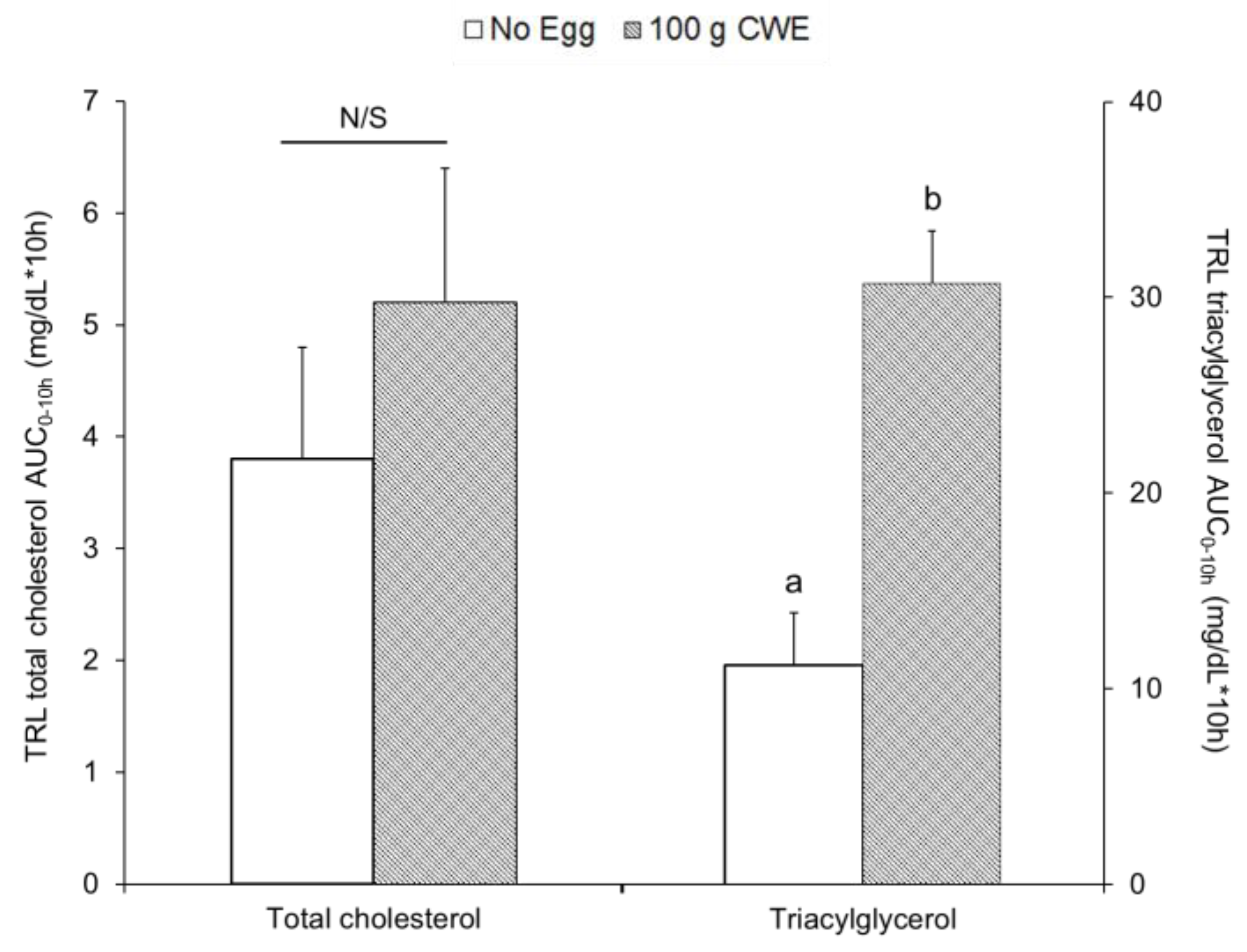
3.2. Total Cholesterol and Triacylglycerol Concentrations in TRL Fractions
3.2.1. Study 1
3.2.2. Study 2
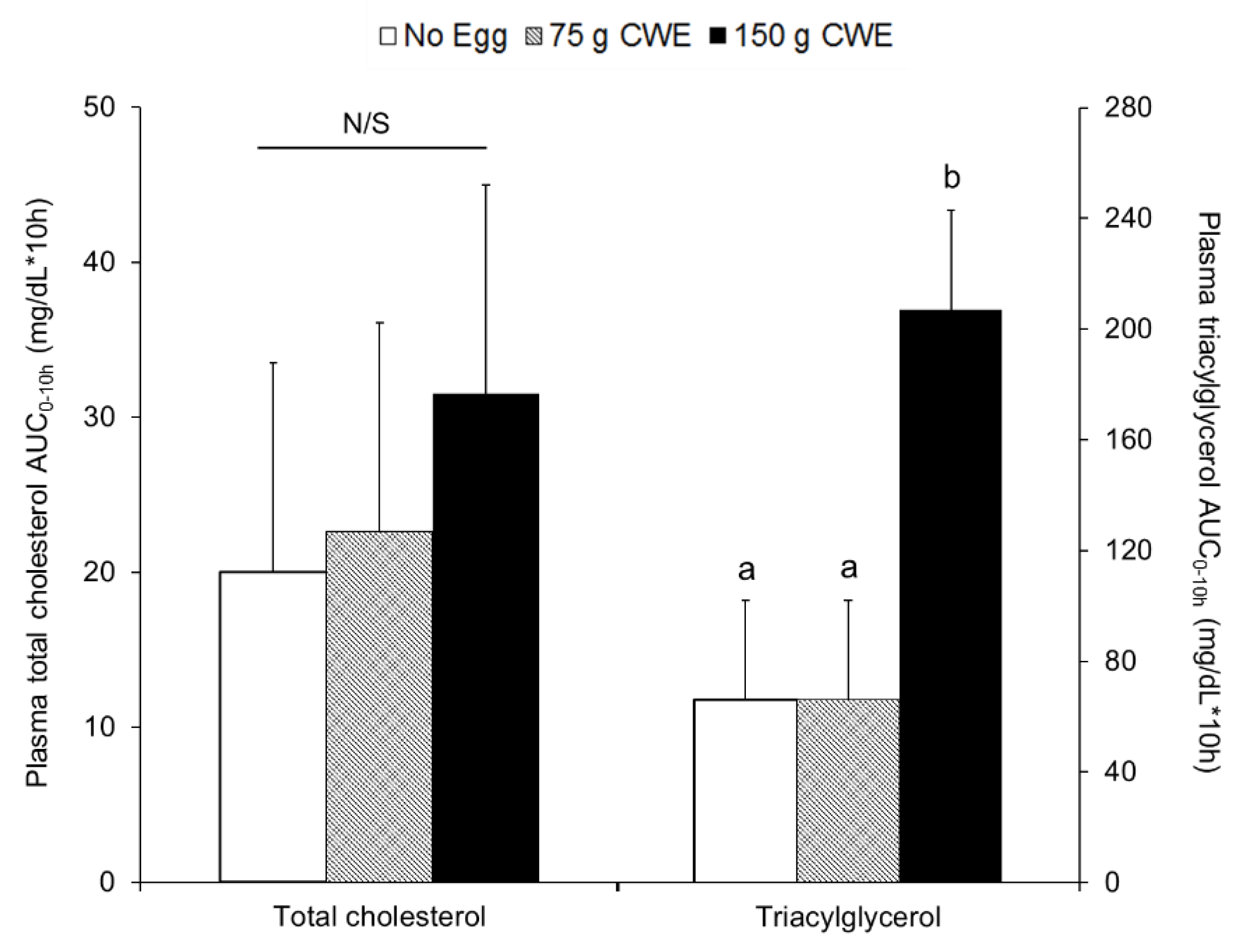
3.3. Total Cholesterol and Triacylglycerol Concentrations in Plasma
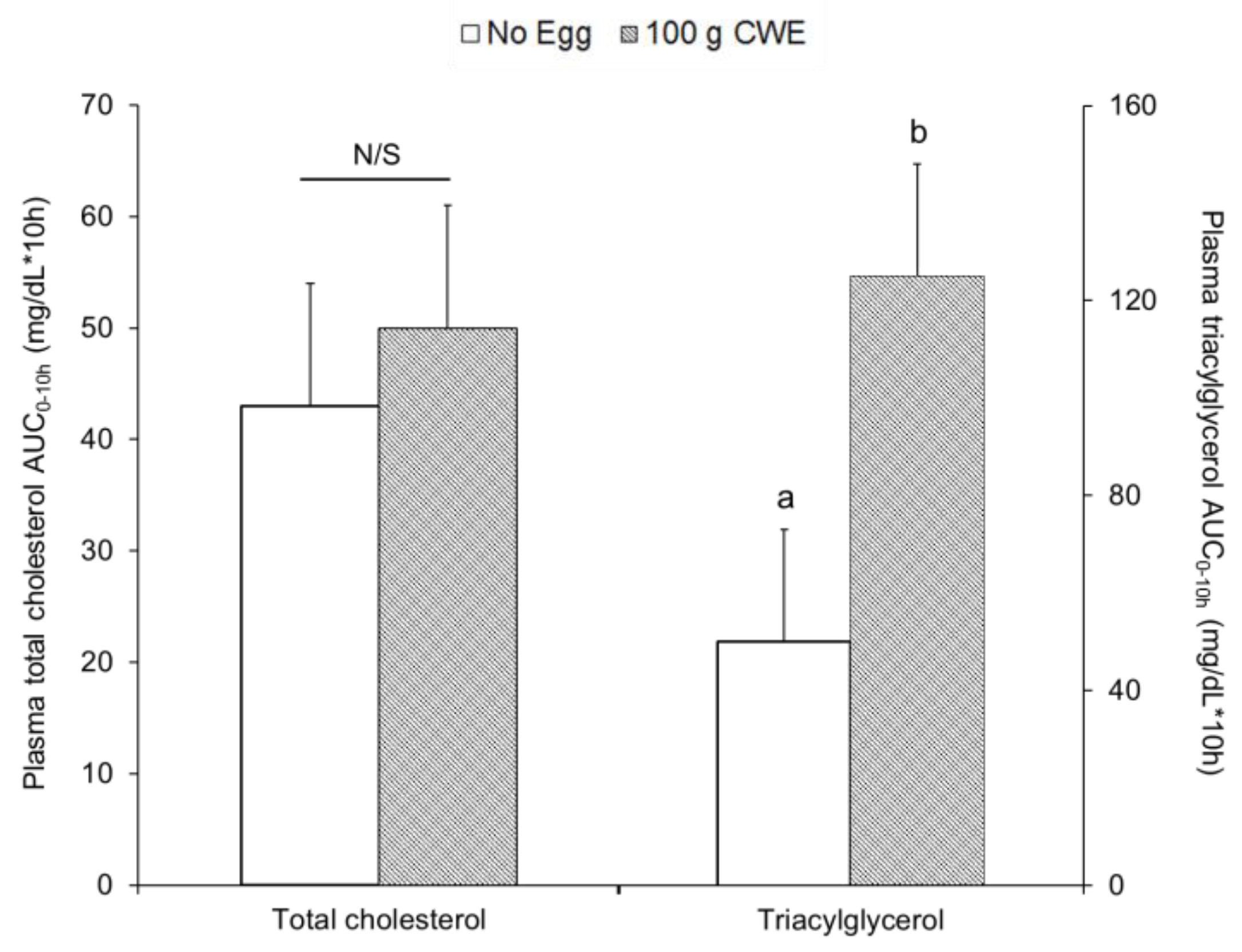
3.3.1. Study 1
3.3.2. Study 2
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Morgan, A.E.; Mooney, K.M.; Wilkinson, S.J.; Pickles, N.A.; Mc Auley, M.T. Cholesterol metabolism: A review of how ageing disrupts the biological mechanisms responsible for its regulation. Ageing Res. Rev. 2016, 27, 108–124. [Google Scholar] [CrossRef] [PubMed]
- Orth, M.; Bellosta, S. Cholesterol: Its regulation and role in central nervous system disorders. Cholesterol 2012, 2012, 292598. [Google Scholar] [CrossRef] [PubMed]
- Dietschy, J.M.; Turley, S.D. Cholesterol metabolism in the brain. Curr. Opin. Lipidol. 2001, 12, 105–112. [Google Scholar] [CrossRef] [PubMed]
- Jesch, E.D.; Carr, T.P. Food ingredients that inhibit cholesterol absorption. Prev. Nutr. Food Sci. 2017, 22, 67–80. [Google Scholar] [PubMed]
- Berger, S.; Raman, G.; Vishwanathan, R.; Jacques, P.F.; Johnson, E.J. Dietary cholesterol and cardiovascular disease: A systematic review and meta-analysis. Am. J. Clin. Nutr. 2015, 102, 276–294. [Google Scholar] [CrossRef] [PubMed]
- Larsson, S.C.; Virtamo, J.; Wolk, A. Dietary fats and dietary cholesterol and risk of stroke in women. Atherosclerosis 2012, 221, 282–286. [Google Scholar] [CrossRef] [PubMed]
- Houston, D.K.; Ding, J.; Lee, J.S.; Garcia, M.; Kanaya, A.M.; Tylavsky, F.A.; Newman, A.B.; Visser, M.; Kritchevsky, S.B.; Health ABC Study. Dietary fat and cholesterol and risk of cardiovascular disease in older adults: The Health ABC Study. Nutr. Metab. Cardiovasc. Dis. 2011, 21, 430–437. [Google Scholar] [CrossRef] [PubMed]
- Lichtenstein, A.H.; Appel, L.J.; Brands, M.; Carnethon, M.; Daniels, S.; Franch, H.A.; Franklin, B.; Kris-Etherton, P.; Harris, W.S.; Howard, B.; et al. Diet and lifestyle recommendations revision 2006: A scientific statement from the American Heart Association Nutrition Committee. Circulation 2006, 114, 82–96. [Google Scholar] [CrossRef] [PubMed]
- U.S. Department of Agriculture and U.S. Department of Health and Human Services. Dietary Guidelines for Americans 2010, 7th ed.; US Government Printing Office: Washington, DC, USA, 2010.
- Scientific Report of the 2015 Dietary Guidelines Advisory Committee 2015. Available online: https://health.gov/dietaryguidelines/2015-scientific-report/pdfs/scientific-report-of-the-2015-dietary-guidelines-advisory-committee.pdf (accessed on 1 December 2016).
- Eckel, R.H.; Jakicic, J.M.; Ard, J.D.; de Jesus, J.M.; Houston Miller, N.; Hubbard, V.S.; Lee, I.M.; Lichtenstein, A.H.; Loria, C.M.; Millen, B.E.; et al. 2013 AHA/ACC guideline on lifestyle management to reduce cardiovascular risk: A report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines. J. Am. Coll. Cardiol. 2014, 63, 2960–2984. [Google Scholar] [CrossRef] [PubMed]
- Song, W.O.; Kerver, J.M. Nutritional contribution of eggs to American diets. J. Am. Coll. Nutr. 2000, 19, 556S–562S. [Google Scholar] [CrossRef] [PubMed]
- Hu, F.B.; Stampfer, M.J.; Rimm, E.B.; Manson, J.E.; Ascherio, A.; Colditz, G.A.; Rosner, B.A.; Spiegelman, D.; Speizer, F.E.; Sacks, F.M.; et al. A prospective study of egg consumption and risk of cardiovascular disease in men and women. JAMA 1999, 281, 1387–1394. [Google Scholar] [CrossRef] [PubMed]
- American Egg Board. Available online: http://www.aeb.org/food-manufacturers/egg-nutrition-and-trends/nutrient-composition (accessed on 15 January 2018).
- Shin, J.Y.; Xun, P.; Nakamura, Y.; He, K. Egg consumption in relation to risk of cardiovascular disease and diabetes: A systematic review and meta-analysis. Am. J. Clin. Nutr. 2013, 98, 146–159. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Zhou, C.; Zhou, X.; Li, L. Egg consumption and risk of cardiovascular diseases and diabetes: A meta-analysis. Atherosclerosis 2013, 229, 524–530. [Google Scholar] [CrossRef] [PubMed]
- Rong, Y.; Chen, L.; Zhu, T.; Song, Y.; Yu, M.; Shan, Z.; Sands, A.; Hu, F.B.; Liu, L. Egg consumption and risk of coronary heart disease and stroke: Dose-response meta-analysis of prospective cohort studies. BMJ 2013, 346, e8539. [Google Scholar] [CrossRef] [PubMed]
- DiMarco, D.M.; Norris, G.H.; Millar, C.L.; Blesso, C.N.; Fernandez, M.L. Intake of up to 3 eggs per day is associated with changes in HDL function and increased plasma antioxidants in healthy, young adults. J. Nutr. 2017, 147, 323–329. [Google Scholar] [PubMed]
- Dubois, C.; Armand, M.; Mekki, N.; Portugal, H.; Pauli, A.M.; Bernard, P.M.; Lafont, H.; Lairon, D. Effects of increasing amounts of dietary cholesterol on postprandial lipemia and lipoproteins in human subjects. J. Lipid. Res. 1994, 35, 1993–2007. [Google Scholar] [PubMed]
- Havel, R.J. Postprandial lipid metabolism: An overview. Proc. Nutr. Soc. 1997, 56, 659–666. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.E.; Ferruzzi, M.G.; Campbell, W.W. Egg Consumption Increases Vitamin E Absorption from Co-Consumed Raw Mixed Vegetables in Healthy Young Men. J. Nutr. 2016, 146, 2199–2205. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.E.; Gordon, S.L.; Ferruzzi, M.G.; Campbell, W.W. Effects of egg consumption on carotenoid absorption from co-consumed, raw vegetables. Am. J. Clin. Nutr. 2015, 102, 75–83. [Google Scholar] [CrossRef] [PubMed]
- Mattsson, L.A.; Silfverstolpe, G.; Samsioe, G. Lipid composition of serum lipoproteins in relation to gonadal hormones during the normal menstrual cycle. Eur. J. Obstet. Gynecol. Reprod. Biol. 1984, 17, 327–335. [Google Scholar] [CrossRef]
- Muesing, R.A.; Forman, M.R.; Graubard, B.I.; Beecher, G.R.; Lanza, E.; McAdam, P.A.; Campbell, W.S.; Olson, B.R. Cyclic changes in lipoprotein and apolipoprotein levels during the menstrual cycle in healthy premenopausal women on a controlled diet. J. Clin. Endocrinol. Metab. 1996, 81, 3599–3603. [Google Scholar]
- Friedewald, W.T.; Levy, R.I.; Fredrickson, D.S. Estimation of the concentration of low-density lipoprotein cholesterol in plasma, without use of the preparative ultracentrifuge. Clin. Chem. 1972, 18, 499–502. [Google Scholar] [PubMed]
- Clayton, Z.S.; Scholar, K.R.; Shelechi, M.; Hernandez, L.M.; Barber, A.M.; Petrisko, Y.J.; Hooshmand, S.; Kern, M. Influence of resistance training combined with daily consumption of an egg-based or bagel-based breakfast on risk factors for chronic diseases in healthy untrained individuals. J. Am. Coll. Nutr. 2015, 34, 113–119. [Google Scholar] [CrossRef] [PubMed]
- Katz, D.L.; Evans, M.A.; Nawaz, H.; Njike, V.Y.; Chan, W.; Comerford, B.P.; Hoxley, M.L. Egg consumption and endothelial function: A randomized controlled crossover trial. Int. J. Cardiol. 2005, 99, 65–70. [Google Scholar] [CrossRef] [PubMed]
- Mutungi, G.; Ratliff, J.; Puglisi, M.; Torres-Gonzalez, M.; Vaishnav, U.; Leite, J.O.; Quann, E.; Volek, J.S.; Fernandez, M.L. Dietary cholesterol from eggs increases plasma HDL cholesterol in overweight men consuming a carbohydrate-restricted diet. J. Nutr. 2008, 138, 272–276. [Google Scholar] [CrossRef] [PubMed]
- Fernandez, M.L. Rethinking dietary cholesterol. Curr. Opin. Clin. Nutr. Metab. Care 2012, 15, 117–121. [Google Scholar] [CrossRef] [PubMed]
- McNamara, D.J. The Fifty Year Rehabilitation of the Egg. Nutrients 2015, 7, 8716–8722. [Google Scholar] [CrossRef] [PubMed]
- Lairon, D.; Lopez-Miranda, J.; Williams, C. Methodology for studying postprandial lipid metabolism. Eur. J. Clin. Nutr. 2007, 61, 1145–1161. [Google Scholar] [CrossRef] [PubMed]
- Cohn, J.S.; Kamili, A.; Wat, E.; Chung, R.W.; Tandy, S. Dietary phospholipids and intestinal cholesterol absorption. Nutrients 2010, 2, 116–127. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Y.; Noh, S.K.; Koo, S.I. Egg phosphatidylcholine decreases the lymphatic absorption of cholesterol in rats. J. Nutr. 2001, 131, 2358–2363. [Google Scholar] [CrossRef] [PubMed]
- Noh, S.K.; Koo, S.I. Egg sphingomyelin lowers the lymphatic absorption of cholesterol and alpha-tocopherol in rats. J. Nutr. 2003, 133, 3571–3576. [Google Scholar] [CrossRef] [PubMed]
- Matsuoka, R.; Kimura, M.; Muto, A.; Masuda, Y.; Sato, M.; Imaizumi, K. Mechanism for the cholesterol-lowering action of egg white protein in rats. Biosci. Biotechnol. Biochem. 2008, 72, 1506–1512. [Google Scholar] [CrossRef] [PubMed]
- Ginsberg, H.N.; Karmally, W.; Siddiqui, M.; Holleran, S.; Tall, A.R.; Rumsey, S.C.; Deckelbaum, R.J.; Blaner, W.S.; Ramakrishnan, R. A dose-response study of the effects of dietary cholesterol on fasting and postprandial lipid and lipoprotein metabolism in healthy young men. Arterioscler. Thromb. 1994, 14, 576–586. [Google Scholar] [CrossRef] [PubMed]
- Yang, F.; Ma, M.; Xu, J.; Yu, X.; Qiu, N. An egg-enriched diet attenuates plasma lipids and mediates cholesterol metabolism of high-cholesterol fed rats. Lipids 2012, 47, 269–277. [Google Scholar] [CrossRef] [PubMed]
- Yamaoto, S.; Kina, T.; Yamagata, N.; Kokubu, T.; Shinjo, S.; Asato, L. Favorable effects of egg white protein on lipid metabolism in rats and mice. Nutr. Res. 1993, 3, 1453–1457. [Google Scholar] [CrossRef]
- Asato, L.; Wang, M.F.; Chan, Y.C.; Yeh, S.H.; Chung, H.M.; Chung, S.Y.; Chida, S.; Uezato, T.; Suzuki, I.; Yamagata, N.; et al. Effect of egg white on serum cholesterol concentration in young women. J. Nutr. Sci. Vitaminol. 1996, 42, 87–96. [Google Scholar] [CrossRef] [PubMed]
- Goltz, S.R.; Campbell, W.W.; Chitchumroonchokchai, C.; Failla, M.L.; Ferruzzi, M.G. Meal triacylglycerol profile modulates postprandial absorption of carotenoids in humans. Mol. Nutr. Food Res. 2012, 56, 866–877. [Google Scholar] [CrossRef] [PubMed]
- Borel, P.; Preveraud, D.; Desmarchelier, C. Bioavailability of vitamin E in humans: An update. Nutr. Rev. 2013, 71, 319–331. [Google Scholar] [CrossRef] [PubMed]
- Jeanes, Y.M.; Hall, W.L.; Ellard, S.; Lee, E.; Lodge, J.K. The absorption of vitamin E is influenced by the amount of fat in a meal and the food matrix. Br. J. Nutr. 2004, 92, 575–579. [Google Scholar] [CrossRef] [PubMed]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, J.E.; Campbell, W.W. Dietary Cholesterol Contained in Whole Eggs Is Not Well Absorbed and Does Not Acutely Affect Plasma Total Cholesterol Concentration in Men and Women: Results from 2 Randomized Controlled Crossover Studies. Nutrients 2018, 10, 1272. https://doi.org/10.3390/nu10091272
Kim JE, Campbell WW. Dietary Cholesterol Contained in Whole Eggs Is Not Well Absorbed and Does Not Acutely Affect Plasma Total Cholesterol Concentration in Men and Women: Results from 2 Randomized Controlled Crossover Studies. Nutrients. 2018; 10(9):1272. https://doi.org/10.3390/nu10091272
Chicago/Turabian StyleKim, Jung Eun, and Wayne W. Campbell. 2018. "Dietary Cholesterol Contained in Whole Eggs Is Not Well Absorbed and Does Not Acutely Affect Plasma Total Cholesterol Concentration in Men and Women: Results from 2 Randomized Controlled Crossover Studies" Nutrients 10, no. 9: 1272. https://doi.org/10.3390/nu10091272
APA StyleKim, J. E., & Campbell, W. W. (2018). Dietary Cholesterol Contained in Whole Eggs Is Not Well Absorbed and Does Not Acutely Affect Plasma Total Cholesterol Concentration in Men and Women: Results from 2 Randomized Controlled Crossover Studies. Nutrients, 10(9), 1272. https://doi.org/10.3390/nu10091272



