Measuring Children’s Sodium and Potassium Intakes in NZ: A Pilot Study
Abstract
1. Introduction
2. Materials and Methods
3. Results
3.1. Recruitment and Retention
3.2. Demographics of Particpating Children
3.3. Completeness of Urine Samples
3.4. Sodium and Potassium Intake and Sodium:Potassium Molar Ratio
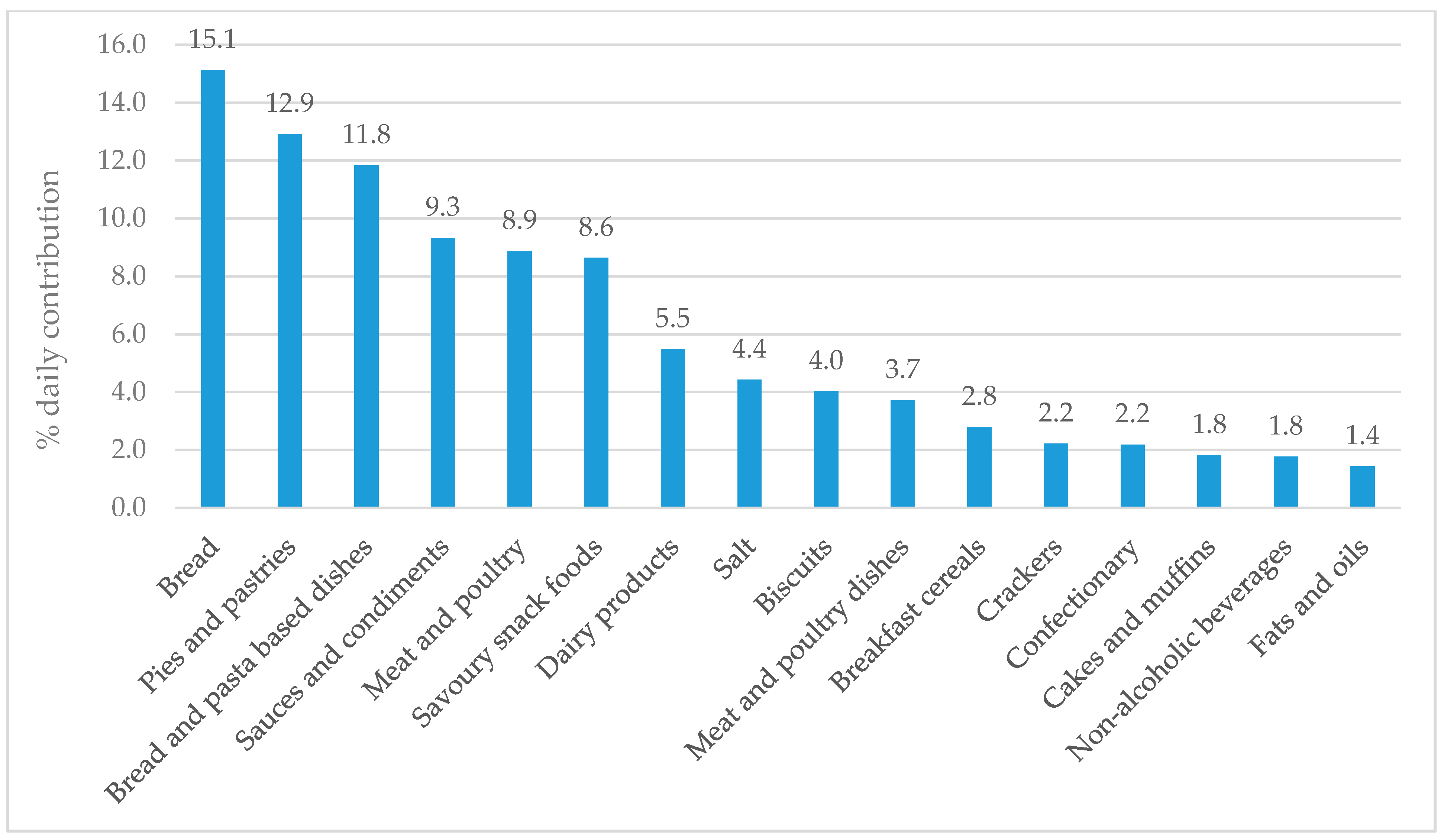
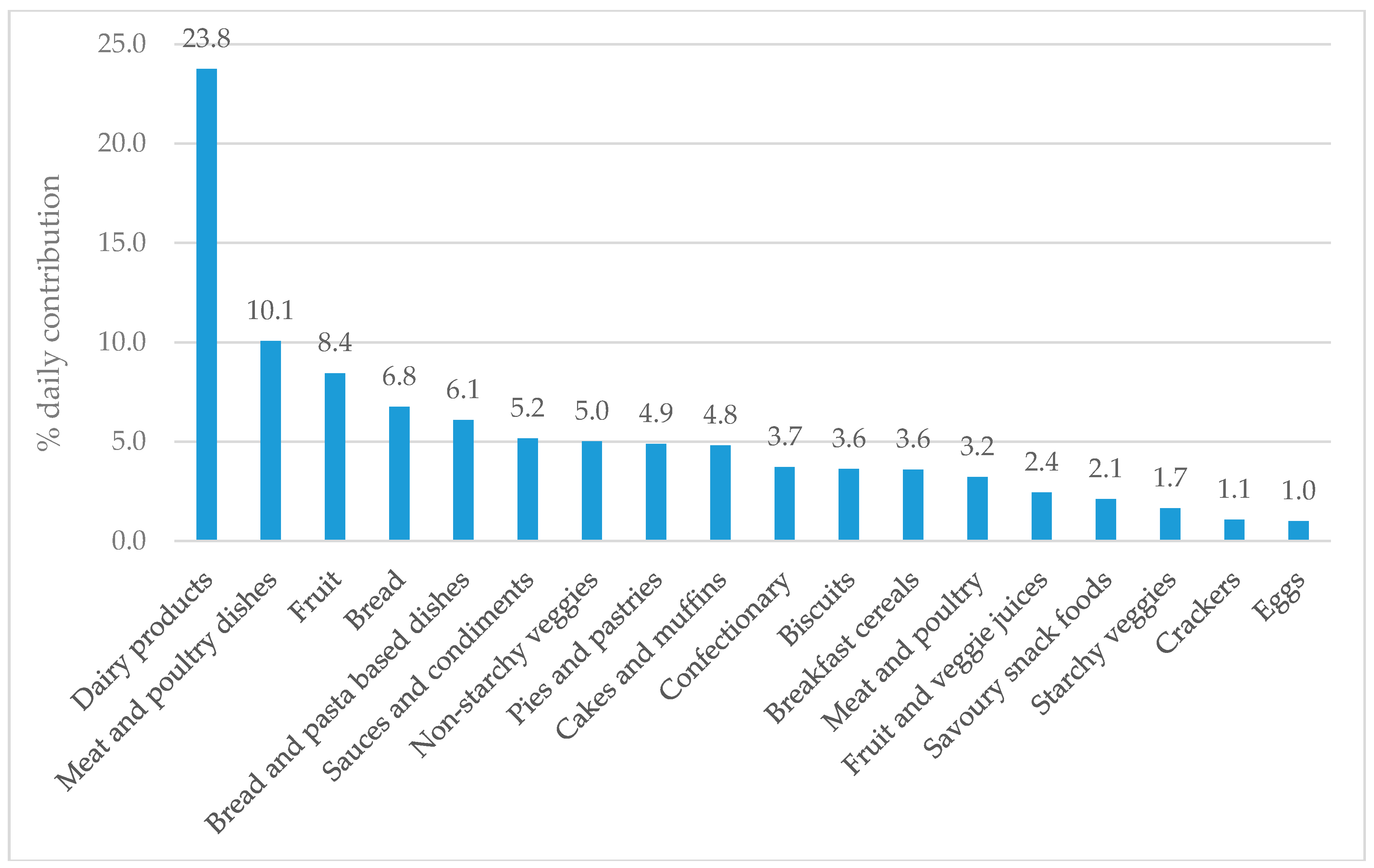
3.5. Major Food Sources of Sodium and Potassium
3.6. Use of Discretionary Salt
3.7. Blood Pressure
3.8. Feasiblity and Acceptibility of Methods for Collecting 24-h Urine Samples and Diet Recalls
3.8.1. 24-h Urine Samples
3.8.2. 24-h Diet Recalls
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Aburto, N.J.; Ziolkovska, A.; Hooper, L.; Elliott, P.; Cappuccio, F.P.; Meerpohl, J.J. Effect of lower sodium intake on health: Systematic review and meta-analyses. Br. Med. J. 2013, 346. [Google Scholar] [CrossRef] [PubMed]
- Aburto, N.J.; Hanson, S.; Gutierrez, H.R.; Hooper, L.; Elliott, P.; Cappuccio, F.P. Effect of increased potassium intake on cardiovascular risk factors and disease: Systematic review and meta-analyses. Br. Med. J. 2013, 346. [Google Scholar] [CrossRef] [PubMed]
- GBD 2013 Risk Factors Collaborators. Global, regional, and national comparative risk assessment of 79 behavioural, environmental and occupational, and metabolic risks or clusters of risks in 188 countries, 1990–2013: A systematic analysis for the Global Burden of Disease Study 2013. Lancet 2015, 386, 2287–2323. [Google Scholar] [CrossRef]
- Naghavi, M.; Abajobir, A.A.; Abbafati, C.; Abbas, K.M.; Abd-Allah, F.; Abera, S.F.; Aboyans, V.; Adetokunboh, O.; Afshin, A.; Agrawal, A.; et al. Global, regional, and national age-sex specific mortality for 264 causes of death, 1980–2016: A systematic analysis for the Global Burden of Disease Study 2016. Lancet 2017, 390, 1151–1210. [Google Scholar] [CrossRef]
- Ministry of Health. Health Loss in New Zealand: A Report from the New Zealand Burden of Diseases, Injuries and Risk Factors Study, 2006–2016; Ministry of Health: Wellington, New Zealand, 2013.
- Deckers, I.A.; van den Brandt, P.A.; van Engeland, M.; Soetekouw, P.M.; Baldewijns, M.M.; Goldbohm, R.A.; Schouten, L.J. Long-term dietary sodium, potassium and fluid intake; exploring potential novel risk factors for renal cell cancer in the Netherlands Cohort Study on diet and cancer. Br. J. Cancer 2014, 110, 797–801. [Google Scholar] [CrossRef] [PubMed]
- D’Elia, L.; Rossi, G.; Ippolito, R.; Cappuccio, F.P.; Strazzullo, P. Habitual salt intake and risk of gastric cancer: A meta-analysis of prospective studies. Clin. Nutr. 2012, 31, 489–498. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.; He, F.J.; MacGregor, G.A. High salt intake: Independent risk factor for obesity? Hypertens 2015, 66. [Google Scholar] [CrossRef] [PubMed]
- He, F.J.; Marrero, N.M.; MacGregor, G.A. Salt intake is related to soft drink consumption in children and adolescents: A link to obesity. Hypertension 2008, 51, 629–634. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Global Action Plan for the Prevention and Control of Noncommunicable Diseases 2013–2020; World Health Organization: Geneva, Switzerland, 2013. [Google Scholar]
- McLean, R.; Edmonds, J.; Williams, S.; Mann, J.; Skeaff, S.A. Balancing sodium and potassium: Estimates of intake in a New Zealand adult population sample. Nutrients 2015, 7, 8930–8938. [Google Scholar] [CrossRef] [PubMed]
- Stein, L.J.; Cowart, B.J.; Cowart, J.; Beauchamp, G.K. The development of salty taste acceptance is related to dietary experience in human infants: A prospective study. Am. J. Clin. Nutr. 2012, 94, 123–129. [Google Scholar] [CrossRef] [PubMed]
- Leyvraz, M.; Chatelan, A.; da Costa, B.R.; Taffe, P.; Paradis, G.; Bovet, P.; Bochud, M.; Chiolero, A. Sodium intake and blood pressure in children and adolescents: A systematic review and meta-analysis of experimental and observational studies. Int. J. Epidemiol. 2018. [Google Scholar] [CrossRef] [PubMed]
- Tirosh, A.; Afek, A.; Rudich, A.; Percik, R.; Gordon, B.; Avalon, N.; Derazne, E.; Tzur, D.; Gershnabel, D.; Grossman, E.; et al. Progression of normotensive adolescents to hypertensive adults: A study of 26,980 teenagers. Hypertension 2010, 56, 203–209. [Google Scholar] [CrossRef] [PubMed]
- He, F.J.; MacGregor, G.A. Importance of salt in determining blood pressure in children: Meta-analysis of controlled trials. Hypertension 2006, 48, 861–869. [Google Scholar] [CrossRef] [PubMed]
- Buendia, J.R.; Bradlee, M.L.; Daniels, S.R.; Singer, M.R.; Moore, L.L. Longitudinal effects of dietary soduim and potassium on blood pressure in adolescent girls. JAMA Pediatr. 2015, 169, 560–568. [Google Scholar] [CrossRef] [PubMed]
- Geleijnse, J.M.; Grobbee, D.E.; Hofman, A. Sodium and potassium intake and blood pressure change in childhood. Br. Med. J. 1990, 300, 899–902. [Google Scholar] [CrossRef]
- Iwahori, T.; Miura, K.; Ueshima, H. Time to consider use of the sodium-to-potassium ratio for practical sodium reduction and potassium increase. Nutrients 2017, 9, 700. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Guideline: Sodium Intake for Adults and Children. Available online: http://www.who.int/nutrition/publications/guidelines/sodium_intake_printversion.pdf (accessed on 18 February 2013).
- World Health Organization. Guideline: Potassium Intake for Adults and Children; World Health Organization: Geneva, Switzerland, 2012; Available online: http://apps.who.int/iris/bitstream/handle/10665/77986/9789241504829_eng.pdf?sequence=1 (accessed on 15 May 2018).
- Ministry of Health. NZ Food NZ Children: Key Results of the 2002 National Children’s Nutrition Survey; Ministry of Health: Wellington, New Zealand, 2003.
- Grimes, C.; Riddell, L.J.; Campbell, K.J.; Beckford, K.; Baxter, J.R.; He, F.J.; Nowson, C.A. Dietary intake and sources of sodium and potassium amoung Australian schoolchildren: Results from the cross-sectional Salt and Other Nutrients in Children (SONIC) study. BMJ Open 2017, 7. [Google Scholar] [CrossRef] [PubMed]
- Bates, C.J.; Thurnham, D.I.; Bingham, S.A.; Margetts, B.M.; Nelson, M. Biochemical markers of nutrient intake. In Design Concepts in Nutritional Epidemiology, 2nd ed.; Margetts, B.M., Nelson, M., Eds.; Oxford University Press: Oxford, UK, 1997; Volume 2, pp. 170–240. [Google Scholar]
- Institute of Medicine. Dietary Reference Intakes for Water, Potassium, Sodium, Chloride, and Sulfate; The National Academies Press: Washington, DC, USA, 2004; Available online: http://www.nap.edu/catalog/10925/dietary-reference-intakes-for-water-potassium-sodium-chloride-and-sulfate (accessed on 5 October 2015).
- Marrero, N.M.; He, F.J.; Whincup, P.; Macgregor, G.A. Salt intake of children and adolescents in South London: Consumption levels and dietary sources. Hypertension 2014, 63, 1026–1032. [Google Scholar] [CrossRef] [PubMed]
- Libuda, L.; Kersting, M.; Alexy, U. Consumption of dietary salt measured by urinary sodium excretion and its association with body weight status in healthy children and adolescents. Public Health Nutr. 2012, 15, 433–441. [Google Scholar] [CrossRef] [PubMed]
- Welcome to Intake24. Available online: https://intake24.co.uk/ (accessed on 14 March 2018).
- Grimes, C.; Baxter, J.R.; Campbell, K.J.; Riddell, L.J.; Rigo, M.; Liem, D.G.; Keast, R.S.; He, F.J.; Nowson, C.A. Cross-sectional study of 24-hour urinary electrolyte excretion and associated health outcomes in a convenience sample of Australian primary schoolchildren: The salt and other nutrients in children (SONIC) study protocol. J. Med. Int. Res. Protoc. 2015, 4, e7. [Google Scholar] [CrossRef] [PubMed]
- Grimes, C.A.; Riddell, L.J.; Campbell, K.J.; Nowson, C.A. Dietary salt intake assessed by 24 h urinary sodium excretion in Australian schoolchildren aged 5–13 years. Public Health Nutr. 2012, 16, 1789–1795. [Google Scholar] [CrossRef] [PubMed]
- Education Counts. Data Services. Directories. New Zealand Schools. Available online: https://www.educationcounts.govt.nz/data-services/directories/list-of-nz-schools (accessed on 18 May 2018).
- REDCap. REDCap: Research Electronic Data Capture. Available online: https://www.project-redcap.org/ (accessed on 21 May 2018).
- Pan American Health Organization, World Health Organization. Protocol for Population Level Sodium Determination in 24-Hour Urine Samples. Available online: https://www.paho.org/hq/dmdocuments/2013/24h-urine-Protocol-eng.pdf (accessed on 18 June 2018).
- Jaffe, M. Ueber den Niederschlag, welchen Pikrinsäure in normalem Harn erzeugt und über eine neue Reaction des Kreatinins. Z. Physiol. Chem. 1886, 10, 391–400. [Google Scholar]
- Raper, N.P.B.; Ingwersen, L.; Steinfeldt, L.; Anand, J. An overview of USDA’s dietary intake data system. J. Food Comp. Anal. 2004, 17, 545–555. [Google Scholar] [CrossRef]
- The Concise New Zealand Food Composition Tables. Available online: https://www.foodcomposition.co.nz/about/ (accessed on 27 July 2018).
- Bradley, J.; Simpson, E.; Poliakov, I.; Matthews, J.N.S.; Olivier, P.; Adamson, A.J.; Foster, E. Comparison of Intake24 (an online 24-h dietary recall tool) with interviewer-led 24-h recall in 11–24 year-olds. Nutrients 2016, 8, 358. [Google Scholar] [CrossRef] [PubMed]
- Eyles, H.; Neal, B.; Jiang, Y.; Ni Mhhurchu, C. Estimating population food and nutrient exposure: Comparison of store survey data with household panel food purchases. Br. J. Nutr. 2016. [Google Scholar] [CrossRef] [PubMed]
- Flynn, J.T.; Kaelber, D.C.; Baker-Smith, C.M.; Blowey, D.; Carroll, A.E.; Daniels, S.R.; de Ferranti, S.D.; Dionne, J.M.; Falkner, B.; Flinn, S.K.; et al. Subcommittee on screening and management of high blood pressure in children. Clinical Practice Guideline for Screening and Management of High Blood Pressure in Children and Adolescents. Pediatrics 2017, 140, e20171904. [Google Scholar] [CrossRef] [PubMed]
- Statistics New Zealand. Household Expenditure Statistics: Year Ended June 2016. Available online: http://www.stats.govt.nz/browse_for_stats/people_and_communities/Households/HouseholdExpenditureStatistics_HOTPYeJun16.aspx (accessed on 25 September 2017).
- Nutrient Reference Values for Australia and New Zealand: Sodium. Available online: https://www.nrv.gov.au/nutrients/sodium (accessed on 16 July 2018).
- Nutrient Reference Values for Australia and New Zealand: Potassium. Available online: https://www.nrv.gov.au/nutrients/potassium (accessed on 16 July 2018).
- Durie, M.H. A Maori perspective of health. Soc. Sci. Med. 1985, 20, 483–486. [Google Scholar] [CrossRef]
- Ministry of Health. Annual Update of Key Results 2016/17: New Zealand Health Survey; Ministry of Health: Wellington, New Zealand, 2017. Available online: https://www.health.govt.nz/publication/annual-update-key-results-2016-17-new-zealand-health-survey (accessed on 16 March 2018).
- Oliveira, A.C.; Padrao, P.; Moreira, A.; Pinto, M.; Neto, M.; Santos, T.; Madureira, J.; de oliveira Fernandes, E.; Graca, P.; Breda, J.; et al. Potassium urinary excretion and dietary intake: A cross-sectional analysis in 8–10 year-old children. BMC Pediatr. 2015, 15. [Google Scholar] [CrossRef] [PubMed]
- Aparicio, A.; Rodriguez-Rodriguez, E.; Cuadrado-Soto, E.; Navia, B.; Lopez-Sobaler, A.M.; Ortega, R.M. Estimation of salt intake assessed by urinary excretion of sodium over 24 h in Spanish subjects aged 7–11 years. Eur. J. Nutr. 2017, 56, 171–178. [Google Scholar] [CrossRef] [PubMed]
- Wall, C.R.; Murphy, R.; Waldie, K.E.; Mitchell, E.A.; Wati, P.; Thompson, J.M.D.; The ABC Study Group. Dietary intakes in children born small for gestational age and appropriate for gestational age: A longitudinal study. Mater. Child Nutr. 2016, 13. [Google Scholar] [CrossRef]
- Ministry of Health. Food and Nutrition Guidelines for Healthy Children and Young People (Aged 2–18 Years): A Background Paper; Ministry of Health: Wellington, New Zealand, 2015. Available online: https://www.health.govt.nz/publication/food-and-nutrition-guidelines-healthy-children-and-young-people-aged-2-18-years-background-paper (accessed on 27 July 2018).
- Australian Bureau of Statistics. Australian Health Survey: Consumption of Food Groups from the Australian Dietary Guidelines; Australian Bureau of Statistics: Canberra, Australia, 2016. Available online: http://www.ausstats.abs.gov.au/ausstats/subscriber.nsf/0/77E3E65E4D967249CA2581F40016ABD6/$File/4364.0.55.012%20-%20consumption%20of%20food%20groups%20from%20australian%20dietary%20guidelines.pdf (accessed on 23 August 2018).
- Campanozzi, A.; Avallone, S.; Barbato, A.; Iacone, R.; Russo, O.; De Filippo, G.; D’Angelo, G.; Pensabene, L.; Malamisura, B.; Cecere, G.; et al. High sodium and low potassium intake among Italian children: Relationship with age, body mass and blood pressure. PLoS ONE 2015, 10, e0121183. [Google Scholar] [CrossRef] [PubMed]
- Ministry of Health, University of Auckland. Nutrition and the Burden of Disease: New Zealand 1997–2011; Ministry of Health: Wellington, New Zealand, 2003. Available online: http://www.moh.govt.nz/notebook/nbbooks.nsf/0/A8D85BC5BAD17610CC256D970072A0AA/$file/nutritionandtheburdenofdisease.pdf (accessed on 2 August 2018).
- Eyles, H.; Shields, E.; Webster, J.; Ni Mhhurchu, C. Achieving the WHO sodium target: Estimation of reductions required in the sodium conent of packaged foods and other sources of dietary sodium. Am. J. Clin. Nutr. 2016. [Google Scholar] [CrossRef] [PubMed]
- Ministry of Health, University of Otago. A Focus on Nutrition: Key Findings of the 2008/09 New Zealand Adult Nutrition Survey; Ministry of Health: Wellington, New Zealand, 2011. Available online: http://www.health.govt.nz/publication/focus-nutrition-key-findings-2008-09-nz-adult-nutrition-survey (accessed on 11 November 2015).
| Demographic (Mean, SD) | Girls (n = 13) | Boys (n = 14) | Total (n = 27) |
|---|---|---|---|
| Age (years) 1 | 9.1 (8, 11) | 8.6 (8, 10) | 8.9 (8, 11) |
| Weight (kg) | 46.9 (10.3) | 38.8 (11.9) | 42.7 (11.7) |
| Height (cm) | 143.9 (7.8) | 138.1 (6.4) | 140.9 (7.5) |
| Waist circumference (cm) | 75.7 (9.8) | 68.1 (12.0) | 71.8 (11.5) |
| Ethnicity 2 (n) | |||
| European | 4 | 6 | 10 |
| Māori 3 | 3 | 7 | 10 |
| Samoan | 6 | 4 | 10 |
| Cook Island Māori | 3 | 0 | 3 |
| Tongan | 7 | 8 | 15 |
| Other ethnicity 4 | 1 | 5 | 6 |
| Household income (n) | |||
| <$NZ 70,000 | 11 | 8 | 19 |
| >70,001 | 2 | 3 | 5 |
| Declined to answer | 0 | 3 | 3 |
| Median (Range) | Girls | Boys | Total |
|---|---|---|---|
| 24-h urine (all samples; n) | 13 | 14 | 27 |
| Sodium (mg/day) | 1494 (870–2992) | 2504 (831,4786) | 1934 (831–4786) |
| Salt equivalent (g/day) | 3.7 (2.2–7.5) | 6.3 (2.1–12.0) | 4.8 (2.1–12.0) |
| Potassium (mg/day) | 1738 (434–2244) | 1589 (495–2981) | 1696 (434–2981) |
| Sodium: potassium (molar ratio) | 1.7 (1.1–6.2) | 2.9 (1.4–7.2) | 2.5 (1.1–7.2) |
| Volume output (mL/day) | 526 (127–897) | 644 (202–1167) | 762 (45–1167) |
| 24-h urine (complete only; n) | 9 | 10 | 19 |
| Sodium (mg/day) | 1623 (1087–2992) | 2407 (1381–4786) | 2191 (1087–4786) |
| Salt equivalent (g/day) | 4.1 (2.7–7.5) | 6.0 (3.5–12.0) | 5.5 (2.7–12.0) |
| Potassium (mg/day) | 1845 (1504–2244) | 1659 (800–2981) | 1776 (800–2981) |
| Sodium: potassium (molar ratio) | 1.6 (1.1–2.6) | 2.9 (1.4–4.8) | 2.0 (1.1–4.8) |
| Volume output (mL/day) | 767 (468–897) | 703 (450–1167) | 762 (450–1167) |
| Theme | Explanation | Supporting Quotes |
|---|---|---|
| Feasibility | ||
| Participant satisfaction | Most children said they would take part in the study again. However, there were mixed feelings regarding how satisfied children felt taking part. | “It was weird to collect.. there was nothing I didn’t like. I would take part again” (Child) “Easy to do, but I didn’t really like it. Would do it again” (Child) |
| Autonomy/supervision | Most children were able to self-manage collection of their own urine. Some parents chose to start their child on a weekend day so they could monitor the collection process. The age group was deemed appropriate with children having a sense of responsibility and autonomy over their own collection. | “It was all straight forward and easy to manage. The children did it themselves, didn’t need to do much. Classroom interference was barely noticeable” (Teacher) “Better to do it on the weekend at home. Able to watch and make sure it was collected. Tricky to do at school” (Parent/caregiver) |
| Student learning experience | Children said given the opportunity most children would take part in the study again because it was fun, interesting, and they were able to learn new things. The study support topics hauora (well-being) and whanaungatanga (relationship, sense of family connection) taught earlier in the school year and developed their interests further. The school and participating students were appreciative of being given the opportunity to take part. Some children commented that taking part meant they were helping with something bigger, demonstrating the vision, values and competencies of the NZ school curriculum. | “The students were really excited to participate in the science study. We do a lot of science in the classroom so they were eager to get involved” (Teacher) “Would do it again because it’s about how much salt we’re eating. It was weird because we had to catch our pee and hold the jug close to us” (Child) “Best part was doing the blood pressure and stuff” |
| Ease of collection | Overall, the children found it easy to collect their urine. | “Collected it all. Just a few drops spilled. Easy to do” (Child) “Collected full time. No spills. No problem collecting” (Child) |
| Acceptability | ||
| Communication | Text reminders were the preferred method of contact. The email with the on-line survey had a low initial response rate despite several reminder emails. | “Survey with consent form. Text reminder helped—prefer text instead of email” (Parent/caregiver) “Text reminders were helpful. Would have forgotten otherwise” (Parent/caregiver) |
| Instructions | Overall, families and teachers were positive about the study, felt well-informed, and understood the requirements. However, there was disconnect observed between these comments and the correct return of the record sheet (for urine collection timeframe and spillages) and collection bottle. Only six participants initially returned the record sheet, and 10 returned the collection bottle with all of the correct documentation. | “Instructions were all good. Instructions needed more pictures” (Parent/caregiver) “Instructions easy to relay to the children. Didn’t read the children’s instruction sheet Mum showed us how to do it” (Child) |
| Equipment | The jug and funnel were considered key components of the collection kit, but some children would have liked a larger jug to catch their urine and avoid chance of contact. Few children used the reminder door hanger and/or safety pin. Many kits were returned with the instructions and record sheet unopened in the resealable bag. | “Equipment was good. Funnel and jug made it easy to do” (Child) “Big jug so it doesn’t splatter when it gets filled to the top” (Child) “He wasn’t interested in the door hanger. Kept the bottle in the bathroom as a visual reminder” (Parent) |
| Cultural aspects | Most families were accepting of the methods required and few reported any cultural concerns pertaining to study methods. One parent shared a concern around storage of urine containers in a bathroom—because other small children were in the house the containers were stored above the child’s head which was considered tapu (sacred, restricted) 1. Many children were shy of talking about their experiences (whakamā) and appeared embarrassed to talk about going to the toilet or collecting their urine. Comments expressed some discomfort/anxiety around touching their urine and parts of their body that are not typically spoken about. | “No cultural issues, no I didn’t have concerns about cultural appropriateness” (Parent/caregiver) “Giving each student the blue bag with all the utensils was awesome. They were able to carry it home/back to school with ease, and although everyone else knew what was inside it there was no mocking or anything at all (Parent/caregiver) “..placed the bottle on the window sill, which is above where you would stand, so tapu, be good to acknowledge this in instructions” (Parent) |
| Country | Year | Age (Years) | n | Salt (g) | Potassium (mg) | ||||
|---|---|---|---|---|---|---|---|---|---|
| Girls | Boys | Total | Girls | Boys | Total | ||||
| Australia [22] | 2017 | 9–12 | 383 | 6.1 | 7.0 | 6.6 | 1838 | 2111 | 1994 |
| Britain [25] | 2014 | 8–9 | 111 | 4.8 | 4.7 | 3.8 | NR | NR | NR |
| Germany [26] | 2011 | 7–10 | 83 | NR | NR | 4.6 | NR | NR | NR |
| Italy [49] | 2015 | 8–10 (G) & 9–11 (B) | 408 | 5.6 | 7.8 | NR | 1290 | 1642 | NR |
| Portugal [44] | 2015 | 8–10 | 163 | 6.0 | 7.3 | NR | 1682 | 1701 | NR |
| Spain [45] | 2017 | 7–11 | 205 | 7.0 | 8.2 | 7.8 | 1826 | 2021 | 1932 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Eyles, H.; Bhana, N.; Lee, S.E.; Grimes, C.; McLean, R.; Nowson, C.; Wall, C. Measuring Children’s Sodium and Potassium Intakes in NZ: A Pilot Study. Nutrients 2018, 10, 1198. https://doi.org/10.3390/nu10091198
Eyles H, Bhana N, Lee SE, Grimes C, McLean R, Nowson C, Wall C. Measuring Children’s Sodium and Potassium Intakes in NZ: A Pilot Study. Nutrients. 2018; 10(9):1198. https://doi.org/10.3390/nu10091198
Chicago/Turabian StyleEyles, Helen, Neela Bhana, Sang Eun Lee, Carley Grimes, Rachael McLean, Caryl Nowson, and Clare Wall. 2018. "Measuring Children’s Sodium and Potassium Intakes in NZ: A Pilot Study" Nutrients 10, no. 9: 1198. https://doi.org/10.3390/nu10091198
APA StyleEyles, H., Bhana, N., Lee, S. E., Grimes, C., McLean, R., Nowson, C., & Wall, C. (2018). Measuring Children’s Sodium and Potassium Intakes in NZ: A Pilot Study. Nutrients, 10(9), 1198. https://doi.org/10.3390/nu10091198





