Plasma 25-Hydroxy Vitamin D Is Not Associated with Either Cognitive Function or Academic Performance in Adolescents
Abstract
1. Introduction
2. Methods
2.1. Cognitive Function Tests and Students’ Academic Performance
2.2. Blood Collection and Biochemical Analyses
2.3. Statistical Methods
3. Results
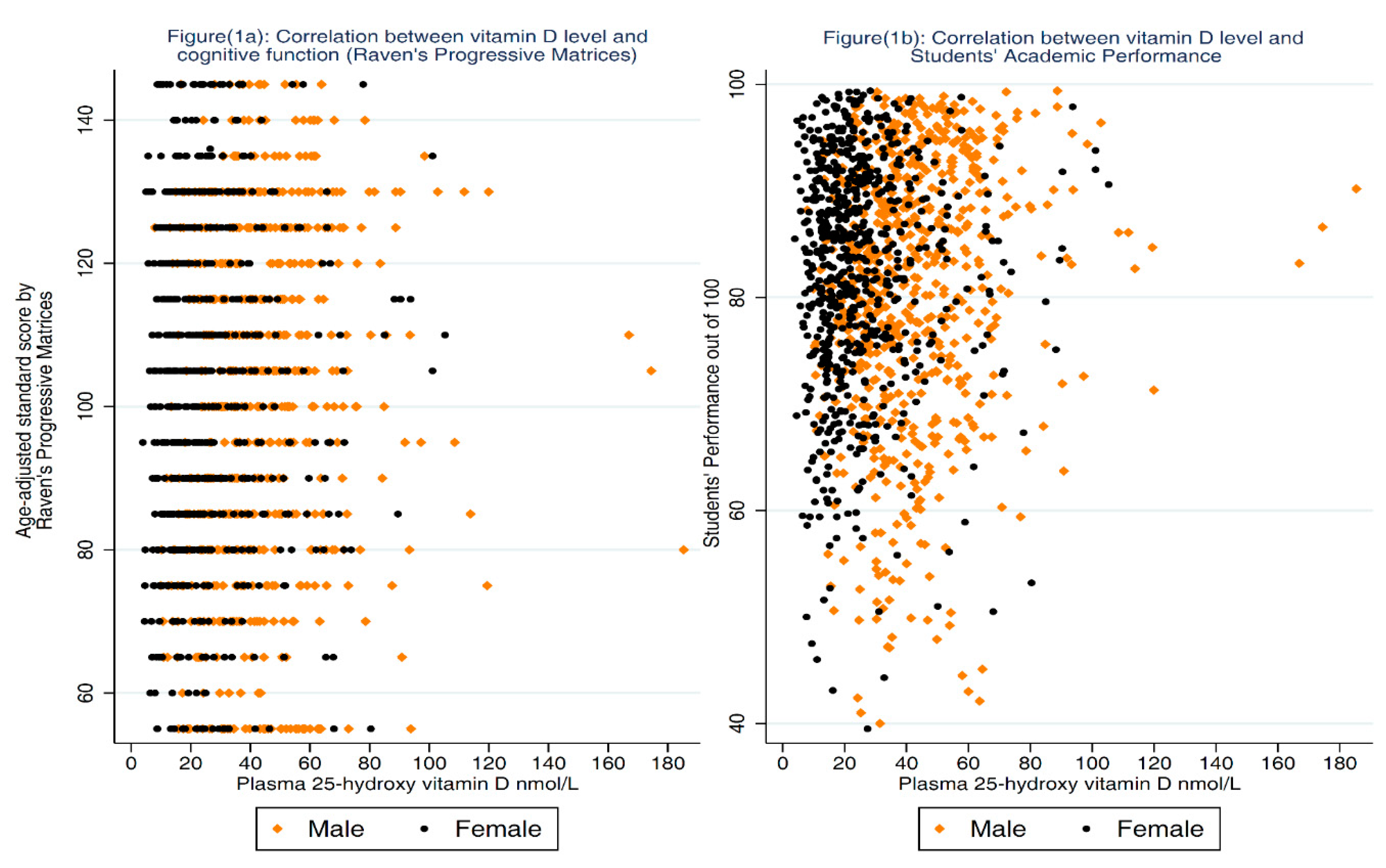
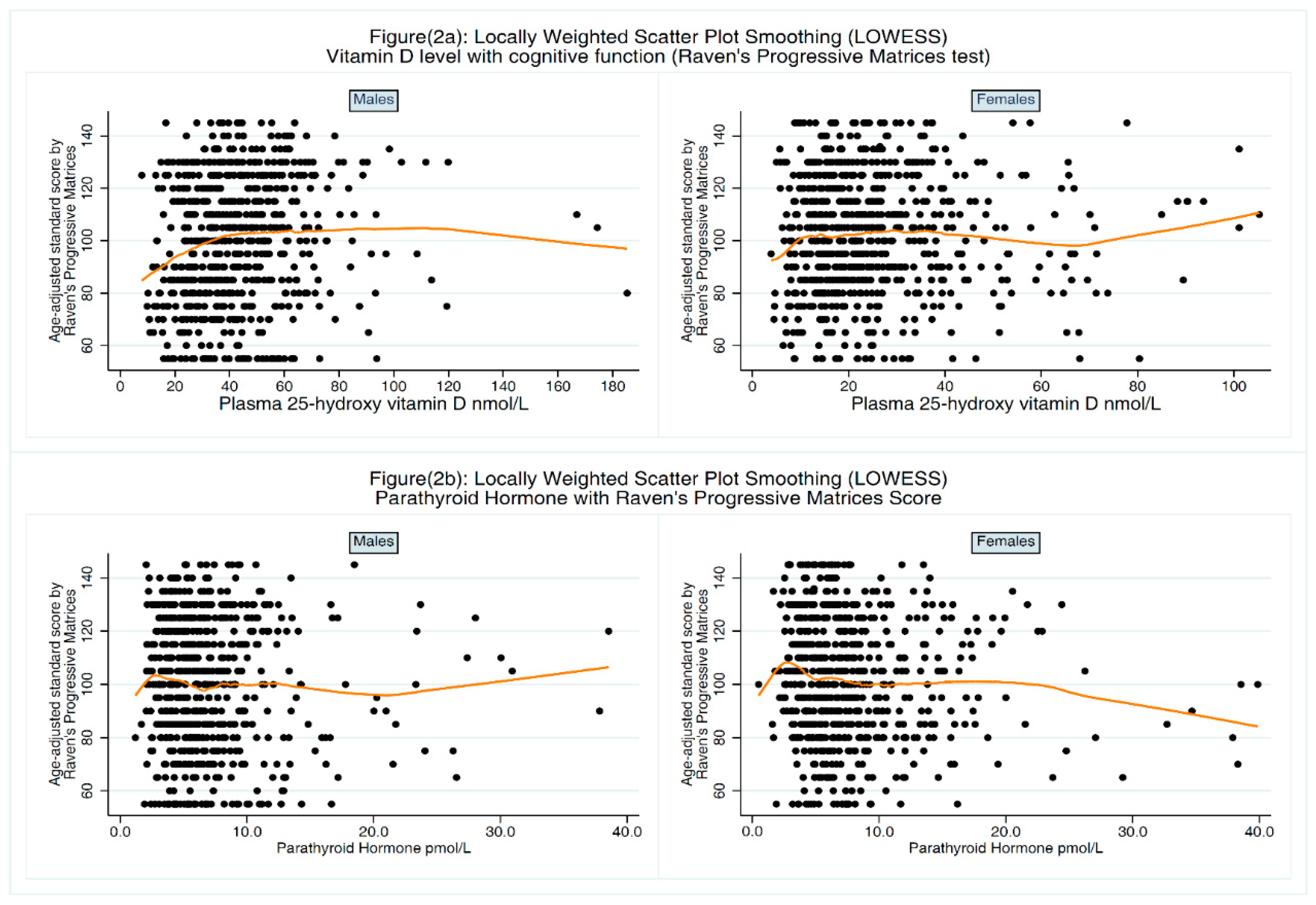
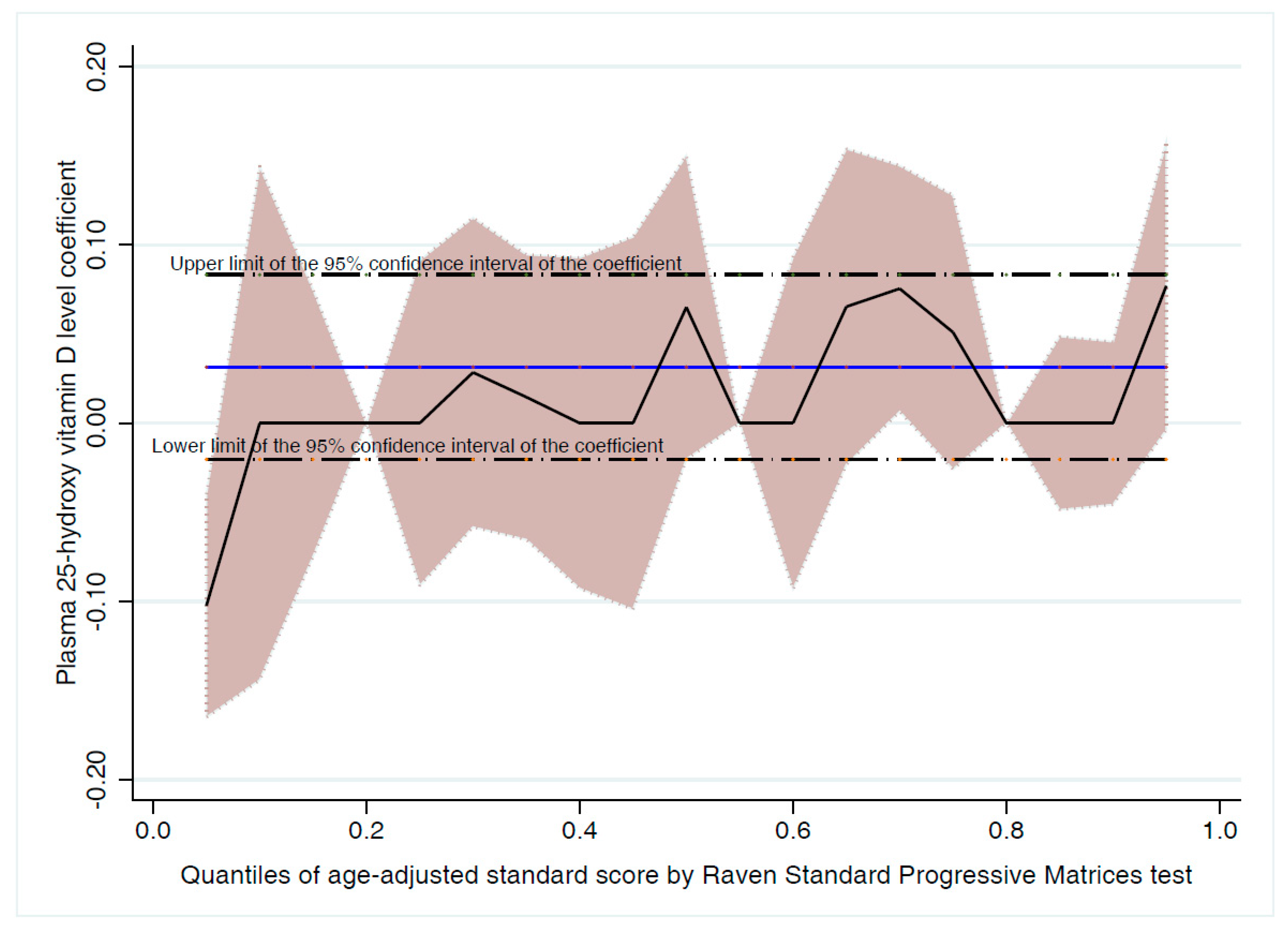
3.1. Association between 25-OH-D and Cognitive Function
3.2. Association between 25-OH-D and Academic Performance
4. Discussion
Strengths and Limitations
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviation
| 25-OH-D | 25-hydroxy vitamin D |
| ASC | age-adjusted standard score |
| CAP | College of American Pathologists |
| LC-MS/MS | liquid chromatography tandem mass spectrometry |
| PTH | parathyroid hormone |
| RCT | randomized control trials |
| SP | school performance |
| SPM | Standard Progressive Matrices |
| VD | vitamin D |
| VDR | vitamin D receptors |
References
- Van Schoor, N.; Lips, P. Global overview of vitamin D status. Endocrinol. Metab. Clin. N. Am. 2017, 46, 845–870. [Google Scholar] [CrossRef] [PubMed]
- Holick, M.F. The vitamin D deficiency pandemic: Approaches for diagnosis, treatment and prevention. Rev. Endocr. Metab. Disord. 2017, 18, 153–165. [Google Scholar] [CrossRef] [PubMed]
- Manson, J.E.; Brannon, P.M.; Rosen, C.J.; Taylor, C.L. Vitamin D deficiency—Is there really a pandemic? N. Engl. J. Med. 2016, 375, 1817–1820. [Google Scholar] [CrossRef] [PubMed]
- Shah, D.; Gupta, P. Vitamin D deficiency: Is the pandemic for real? Indian J. Community Med. 2015, 40, 215–217. [Google Scholar] [PubMed]
- Stokes, C.S.; Lammert, F. Vitamin D supplementation: Less controversy, more guidance needed. F1000Res 2016, 5. [Google Scholar] [CrossRef] [PubMed]
- Palacios, C.; Gonzalez, L. Is vitamin D deficiency a major global public health problem? J. Steroid Biochem. Mol. Boil. 2014, 144 Pt A, 138–145. [Google Scholar] [CrossRef]
- Holick, M.F. Vitamin D deficiency. N. Engl. J. Med. 2007, 357, 266–281. [Google Scholar] [CrossRef] [PubMed]
- Al-Taiar, A.; Rahman, A.; Al-Sabah, R.; Shaban, L.; Al-Harbi, A. Vitamin D status among adolescents in kuwait: A cross-sectional study. BMJ Open 2018, 8, e021401. [Google Scholar] [CrossRef] [PubMed]
- Bouillon, R. Extra-skeletal effects of vitamin D. Front. Horm. Res. 2018, 50, 72–88. [Google Scholar] [PubMed]
- Bjelakovic, G.; Gluud, L.L.; Nikolova, D.; Whitfield, K.; Wetterslev, J.; Simonetti, R.G.; Bjelakovic, M.; Gluud, C. Vitamin D supplementation for prevention of mortality in adults. Cochrane Database Syst. Rev. 2014, CD007470. [Google Scholar] [CrossRef] [PubMed]
- Straube, S.; Derry, S.; Straube, C.; Moore, R.A. Vitamin D for the treatment of chronic painful conditions in adults. Cochrane Database Syst. Rev. 2015, CD007771. [Google Scholar] [CrossRef] [PubMed]
- Beveridge, L.A.; Khan, F.; Struthers, A.D.; Armitage, J.; Barchetta, I.; Bressendorff, I.; Cavallo, M.G.; Clarke, R.; Dalan, R.; Dreyer, G.; et al. Effect of vitamin D supplementation on markers of vascular function: A systematic review and individual participant meta-analysis. J. Am. Heart Assoc. 2018, 7. [Google Scholar] [CrossRef] [PubMed]
- Das, R.R.; Singh, M.; Naik, S.S. Vitamin D as an adjunct to antibiotics for the treatment of acute childhood pneumonia. Cochrane Database Syst. Rev. 2018, 7, CD011597. [Google Scholar] [CrossRef] [PubMed]
- Lardner, A.L. Vitamin D and hippocampal development-the story so far. Front. Mol. Neurosci. 2015, 8, 58. [Google Scholar] [CrossRef] [PubMed]
- Eyles, D.W.; Smith, S.; Kinobe, R.; Hewison, M.; McGrath, J.J. Distribution of the vitamin D receptor and 1 alpha-hydroxylase in human brain. J. Chem. Neuroanat. 2005, 29, 21–30. [Google Scholar] [CrossRef] [PubMed]
- Prufer, K.; Veenstra, T.D.; Jirikowski, G.F.; Kumar, R. Distribution of 1,25-dihydroxyvitamin D3 receptor immunoreactivity in the rat brain and spinal cord. J. Chem. Neuroanat. 1999, 16, 135–145. [Google Scholar] [CrossRef]
- Miller, W.L.; Portale, A.A. Vitamin d 1 alpha-hydroxylase. Trends Endocrinol. Metab. TEM 2000, 11, 315–319. [Google Scholar] [CrossRef]
- Garcion, E.; Sindji, L.; Leblondel, G.; Brachet, P.; Darcy, F. 1,25-dihydroxyvitamin D3 regulates the synthesis of gamma-glutamyl transpeptidase and glutathione levels in rat primary astrocytes. J. Neurochem. 1999, 73, 859–866. [Google Scholar] [CrossRef] [PubMed]
- Cui, X.; Gooch, H.; Petty, A.; McGrath, J.J.; Eyles, D. Vitamin D and the brain: Genomic and non-genomic actions. Mol. Cell. Endocrinol. 2017, 453, 131–143. [Google Scholar] [CrossRef] [PubMed]
- Di Somma, C.; Scarano, E.; Barrea, L.; Zhukouskaya, V.V.; Savastano, S.; Mele, C.; Scacchi, M.; Aimaretti, G.; Colao, A.; Marzullo, P. Vitamin D and neurological diseases: An endocrine view. Int. J. Mol. Sci. 2017, 18. [Google Scholar] [CrossRef] [PubMed]
- Annweiler, C.; Dursun, E.; Feron, F.; Gezen-Ak, D.; Kalueff, A.V.; Littlejohns, T.; Llewellyn, D.J.; Millet, P.; Scott, T.; Tucker, K.L.; et al. ‘Vitamin D and cognition in older adults’: Updated international recommendations. J. Intern. Med. 2015, 277, 45–57. [Google Scholar] [CrossRef] [PubMed]
- Goodwill, A.M.; Szoeke, C. A systematic review and meta-analysis of the effect of low vitamin D on cognition. J. Am. Geriatr. Soc. 2017, 65, 2161–2168. [Google Scholar] [CrossRef] [PubMed]
- Tolppanen, A.M.; Williams, D.; Lawlor, D.A. The association of circulating 25-hydroxyvitamin D and calcium with cognitive performance in adolescents: Cross-sectional study using data from the third national health and nutrition examination survey. Paediatr. Périnat. Epidemiol. 2011, 25, 67–74. [Google Scholar] [CrossRef] [PubMed]
- McGrath, J.; Scragg, R.; Chant, D.; Eyles, D.; Burne, T.; Obradovic, D. No association between serum 25-hydroxyvitamin D3 level and performance on psychometric tests in NHANES III. Neuroepidemiology 2007, 29, 49–54. [Google Scholar] [CrossRef] [PubMed]
- Cukic, I.; Brett, C.E.; Calvin, C.M.; Batty, G.D.; Deary, I.J. Childhood IQ and survival to 79: Follow-up of 94% of the Scottish mental survey 1947. Intelligence 2017, 63, 45–50. [Google Scholar] [CrossRef] [PubMed]
- Lawlor, D.A.; Batty, G.D.; Clark, H.; McIntyre, S.; Leon, D.A. Association of childhood intelligence with risk of coronary heart disease and stroke: Findings from the Aberdeen Children of the 1950s cohort study. Eur. J. Epidemiol. 2008, 23, 695–706. [Google Scholar] [CrossRef] [PubMed]
- Glanz, K.; Yaroch, A.L.; Dancel, M.; Saraiya, M.; Crane, L.A.; Buller, D.B.; Manne, S.; O’Riordan, D.L.; Heckman, C.J.; Hay, J.; et al. Measures of sun exposure and sun protection practices for behavioral and epidemiologic research. Arch. Dermatol. 2008, 144, 217–222. [Google Scholar] [CrossRef] [PubMed]
- Taylor, C.; Lamparello, B.; Kruczek, K.; Anderson, E.J.; Hubbard, J.; Misra, M. Validation of a food frequency questionnaire for determining calcium and vitamin D intake by adolescent girls with anorexia nervosa. J. Am. Diet. Assoc. 2009, 109, 479–485, 485.e1–485.e3. [Google Scholar] [CrossRef] [PubMed]
- Papandreou, D.; Rachaniotis, N.; Lari, M.; Al Mussabi, W. Validation of a food frequency questionnaire for vitamin D and calcium intake in healthy female college students. Food Nutr. Sci. 2014, 5, 2048–2052. [Google Scholar] [CrossRef]
- UEA. Youth Physical Activity Questionnaire. Available online: http://epi-meta.medschl.cam.ac.uk/includes/speedy/pdf/PhysicalActivity%20V4%20SPEEDY-3.pdf (accessed on 28 August 2015).
- Al-Hazzaa, H.M.; Al-Sobayel, H.I.; Musaiger, A.O. Convergent validity of the Arab Teens Lifestyle Study (ATLS) physical activity questionnaire. Int. J. Environ. Res. Public Health 2011, 8, 3810–3820. [Google Scholar] [CrossRef] [PubMed]
- Abdel-Khalek, A.M. Reliability and factorial validity of the standard progressive matrices among Kuwaiti children ages 8 to 15 years. Percept. Mot. Skills 2005, 101, 409–412. [Google Scholar] [CrossRef] [PubMed]
- Raven, J. Manual for Raven’s Progressive Matrices and Vocabullary Scale; Pearson Education: London, UK, 2000. [Google Scholar]
- Al-Harbi, A.N.; Khan, K.M.; Rahman, A. Developmental vitamin D deficiency affects spatial learning in wistar rats. J. Nutr. 2017, 147, 1795–1805. [Google Scholar] [CrossRef] [PubMed]
- Cleveland, W. Robust locally weighted fitting and smoothing scatterplots. J. Am. Stat. Assoc. 1979, 74, 829–836. [Google Scholar] [CrossRef]
- Holick, M.F.; Binkley, N.C.; Bischoff-Ferrari, H.A.; Gordon, C.M.; Hanley, D.A.; Heaney, R.P.; Murad, M.H.; Weaver, C.M. Evaluation, treatment, and prevention of vitamin D deficiency: An endocrine society clinical practice guideline. J. Clin. Endocrinol. Metab. 2011, 96, 1911–1930. [Google Scholar] [CrossRef] [PubMed]
- Society for Adolescent Health and Medicine. Recommended vitamin D intake and management of low vitamin D status in adolescents: A position statement of the society for adolescent health and medicine. J. Adolesc. Health 2013, 52, 801–803. [Google Scholar] [CrossRef] [PubMed]
- Nassar, M.F.; Amin, D.A.; Hamed, A.I.; Nassar, J.F.; Abou-Zeid, A.E.; Attaby, M.A. Vitamin D status and scholastic achievement in middle age childhood. J. Egypt. Soc. Parasitol. 2012, 42, 349–358. [Google Scholar] [CrossRef] [PubMed]
- Ballester-Plane, J.; Laporta-Hoyos, O.; Macaya, A.; Poo, P.; Melendez-Plumed, M.; Vazquez, E.; Delgado, I.; Zubiaurre-Elorza, L.; Narberhaus, A.; Toro-Tamargo, E.; et al. Measuring intellectual ability in cerebral palsy: The comparison of three tests and their neuroimaging correlates. Res. Dev. Disabil. 2016, 56, 83–98. [Google Scholar] [CrossRef] [PubMed]
- Tolppanen, A.M.; Sayers, A.; Fraser, W.D.; Lawlor, D.A. Association of serum 25-hydroxyvitamin D3 and D2 with academic performance in childhood: Findings from a prospective birth cohort. J. Epidemiol. Community Health 2012, 66, 1137–1142. [Google Scholar] [CrossRef] [PubMed]
- Usdin, T.B.; Gruber, C.; Bonner, T.I. Identification and functional expression of a receptor selectively recognizing parathyroid hormone, the PTH2 receptor. J. Boil. Chem. 1995, 270, 15455–15458. [Google Scholar] [CrossRef]
- Joborn, C.; Hetta, J.; Niklasson, F.; Rastad, J.; Wide, L.; Agren, H.; Akerstrom, G.; Ljunghall, S. Cerebrospinal fluid calcium, parathyroid hormone, and monoamine and purine metabolites and the blood-brain barrier function in primary hyperparathyroidism. Psychoneuroendocrinology 1991, 16, 311–322. [Google Scholar] [CrossRef]
- Henderson, V.H.; Velleman, F.P. Building multiple regression models interactively. Biometrics 1981, 37, 391–411. [Google Scholar] [CrossRef]
- Jorde, R.; Waterloo, K.; Saleh, F.; Haug, E.; Svartberg, J. Neuropsychological function in relation to serum parathyroid hormone and serum 25-hydroxyvitamin D levels. The tromsø study. J. Neurol. 2006, 253, 464–470. [Google Scholar] [CrossRef] [PubMed]
- Bjorkman, M.P.; Sorva, A.J.; Tilvis, R.S. Does elevated parathyroid hormone concentration predict cognitive decline in older people? Aging Clin. Exp. Res. 2010, 22, 164–169. [Google Scholar] [CrossRef] [PubMed]
- Feart, C.; Helmer, C.; Merle, B.; Herrmann, F.R.; Annweiler, C.; Dartigues, J.F.; Delcourt, C.; Samieri, C. Associations of lower vitamin D concentrations with cognitive decline and long-term risk of dementia and alzheimer’s disease in older adults. Alzheimers Dement. 2017, 13, 1207–1216. [Google Scholar] [CrossRef] [PubMed]
- Matchar, D.B.; Chei, C.L.; Yin, Z.X.; Koh, V.; Chakraborty, B.; Shi, X.M.; Zeng, Y. Vitamin D levels and the risk of cognitive decline in Chinese elderly people: The Chinese longitudinal healthy longevity survey. J. Gerontol. Ser. A Boil. Sci. Med Sci. 2016, 71, 1363–1368. [Google Scholar] [CrossRef] [PubMed]
- Santamaria, C.; Bi, W.G.; Leduc, L.; Tabatabaei, N.; Jantchou, P.; Luo, Z.C.; Audibert, F.; Nuyt, A.M.; Wei, S.Q. Prenatal vitamin D status and offspring’s growth, adiposity and metabolic health: A systematic review and meta-analysis. Br. J. Nutr. 2018, 119, 310–319. [Google Scholar] [CrossRef] [PubMed]
- Pet, M.A.; Brouwer-Brolsma, E.M. The impact of maternal vitamin D status on offspring brain development and function: A systematic review. Adv. Nutr. 2016, 7, 665–678. [Google Scholar] [CrossRef] [PubMed]
- Rose, G. Sick individuals and sick populations. Int. J. Epidemiol. 2001, 30, 427–432. [Google Scholar] [CrossRef] [PubMed]
- Vogeser, M. Quantification of circulating 25-hydroxyvitamin D by liquid chromatography-tandem mass spectrometry. J. Steroid Biochem. Mol. Boil. 2010, 121, 565–573. [Google Scholar] [CrossRef] [PubMed]
- Sadat-Ali, M.; Al-Elq, A.H.; Al-Shaikh, I.H.; Al-Turki, H.A.; Al-Ali, A.K.; Al-Othman, A.A. Assessment of low vitamin D among Saudi Arabians. Did we overshoot the runway? Saudi Med. J. 2014, 35, 1243–1249. [Google Scholar] [PubMed]
- Lai, J.K.; Lucas, R.M.; Banks, E.; Ponsonby, A.L. Variability in vitamin D assays impairs clinical assessment of vitamin D status. Intern. Med. J. 2012, 42, 43–50. [Google Scholar] [CrossRef] [PubMed]
| Characteristics | ||
|---|---|---|
| Age in years, Mean (SD) years | 12.4 | (0.9) |
| n | (%) | |
| Gender | ||
| Male | 674 | (49.2) |
| Nationality | ||
| Kuwaiti | 1047 | (76.4) |
| Non-Kuwait | 323 | (23.6) |
| Father’s Education 1 | ||
| No formal education | 15 | (1.11) |
| Primary/Intermediate | 215 | (16.1) |
| Secondary (high school) | 330 | (24.7) |
| Diploma | 251 | (18.8) |
| University & above | 526 | (39.3) |
| Mother’s Education 2 | ||
| No formal education | 31 | (2.3) |
| Primary/Intermediate | 145 | (10.7) |
| Secondary (high school) | 294 | (21.9) |
| Diploma | 293 | (21.7) |
| University & above | 587 | (43.5) |
| Father’s Income 3 (Kuwaiti Dinars) | ||
| Less than 500 | 89 | (6.7) |
| 500 to 1000 | 291 | (22.0) |
| 1001 to 1500 | 414 | (31.3) |
| 1501 to 2000 | 213 | (16.1) |
| More than 2000 | 164 | (12.4) |
| Do not wish to tell | 153 | (11.6) |
| Mother’s Employment Status 4 | ||
| Housewife | 466 | (34.7) |
| Paid employment | 664 | (49.5) |
| Others | 212 | (15.8) |
| Housing 5 | ||
| Rented flat | 499 | (36.9) |
| Rented house | 159 | (11.8) |
| Owned flat | 55 | (4.1) |
| Owned house | 638 | (47.2) |
| Vitamin D Status | Model 1 | Model 2 | Model 3 | Model 4 | Model 5 |
|---|---|---|---|---|---|
| β [95%CI] | β [95%CI] | β [95%CI] | β [95%CI] | β [95%CI] | |
| 25-OH-D levels nmol/L | 0.03 | 0.01 | 0.00 | 0.00 | −0.01 |
| [−0.02, 0.08] | [−0.04, 0.07] | [−0.05, 0.06] | [−0.05, 0.06] | [−0.07, 0.05] | |
| p-value | 0.23 | 0.70 | 0.95 | 0.85 | 0.73 |
| Q1 (25-OH-D < 19.2 nmol/L) (n = 337) | [Reference] | [Reference] | [Reference] | [Reference] | [Reference] |
| Q2 (25-OH-D ≥ 19.2 to <29.7 nmol/L) (n = 346) | 0.86 | 1.12 | −0.39 | 1.15 | 0.82 |
| [−2.65, 4.38] | [−2.32, 4.58] | [−4.28, 3.49] | [−2.29, 4.59] | [−2.82, 4.46] | |
| Q3 (25-OH-D from 29.7 to <44.1 nmol/L) (n = 344) | 2.67 | 2.29 | −0.26 | 2.69 | 0.84 |
| [−0.86, 6.19] | [−1.43, 6.02] | [−6.25−5.72] | [−1.03, 6.41] | [−3.24, 4.92] | |
| Q4 (25-OH-D ≥ 44.1 nmol/L) (n = 343) | 2.91 | 0.95 | −0.70 | 0.50 | −0.42 |
| [−0.62, 6.43] | [−3.00, 4.90] | [−7.92, 6.53] | [−3.47, 4.47] | [−4.82, 4.01] | |
| p-value | 0.30 | 0.67 | 1.00 | 0.45 | 0.88 |
| Severe deficiency (25-OH-D < 25 nmol/L) (n = 544) | [Reference] | [Reference] | [Reference] | [Reference] | [Reference] |
| Deficiency (25-OH-D ≥ 25 to <50 nmol/L) (n = 572) | 2.28 | 2.52 | 2.70 | 2.71 | 1.36 |
| [−0.46, 5.04] | [−0.39, 5.44] | [0.22, 5.63] | [−0.22, 5.63] | [−1.86, 4.59] | |
| Insufficiency (25-OH-D ≥ 50 to <75 nmol/L) (n = 205) | 1.40 | −0.66 | −0.93 | −0.94 | −1.53 |
| [−2.36, 5.16] | [−4.66, 3.34] | [−4.94, 3.09] | [−4.97, 3.08] | [−6.01, 2.94] | |
| Sufficiency (25-OH-D ≥ 75 nmol/L) (n = 49) | 5.46 | 4.98 | 2.79 | 3.02 | 1.47 |
| [−1.39, 12.31] | [−1.96, 11.92] | [−4.16, 9.74] | [−3.93, 9.98] | [−5.96, 8.91] | |
| p-value | 0.24 | 0.11 | 0.11 | 0.10 | 0.48 |
| Vitamin D Status | Model 1 | Model 2 | Model 3 | Model 4 | Model 5 |
|---|---|---|---|---|---|
| β [95%CI] | β [95%CI] | β [95%CI] | β [95%CI] | β [95%CI] | |
| 25-OH-D levels nmol/L | 0.02 | −0.00 | −0.00 | −0.01 | −0.01 |
| [−0.01, 0.06] | [−0.03, 0.03] | [−0.04, 0.03] | [−0.04, 0.03] | [−0.04, 0.03] | |
| p-value | 0.22 | 0.94 | 0.77 | 0.67 | 0.69 |
| Q1 (25-OH-D < 19.2 nmol/L) (n = 337) | [Reference] | [Reference] | [Reference] | [Reference] | [Reference] |
| Q2 (25-OH-D ≥19.2 to <29.7 nmol/L) (n = 346) | 1.10 | 1.61 | 2.01 | 1.66 | 1.74 |
| [−1.54, 3.74] | [−0.34, 3.57] | [0.01, 4.01] | [−0.42, 3.75] | [−0.33, 3.80] | |
| Q3 (25-OH-D from 29.7 to <44.1 nmol/L) (n = 344) | 0.40 | 0.70 | 0.98 | 0.90 | 0.94 |
| [−2.15, 2.95] | [−1.58, 2.99] | [−1.26, 3.21] | [−1.39, 3.20] | [−1.41, 3.30] | |
| Q4 (25-OH-D ≥ 44.1 nmol/L) (n = 343) | 2.60 | 0.82 | 1.00 | 1.40 | 1.67 |
| [0.06, 5.25] | [−1.51, 3.14] | [−1.38, 3.38] | [−1.03, 3.83] | [−0.86, 4.19] | |
| p-value | 0.17 | 0.43 | 0.27 | 0.45 | 0.36 |
| Severe deficiency (25-OH-D < 25 nmol/L) (n = 544) | [Reference] | [Reference] | [Reference] | [Reference] | [Reference] |
| Deficiency (25-OH-D ≥ 25 to <50 nmol/L) (n = 572) | −0.30 | −1.36 | −0.84 | −0.99 | −0.94 |
| [−2.35, 1.75] | [−3.04, 0.32] | [−2.65, 0.96] | [−2.73, 0.73] | [−2.88, 0.99] | |
| Insufficiency (25-OH-D ≥ 50 to <75 nmol/L) (n = 205) | 2.30 | −0.34 | 0.31 | 0.28 | 0.38 |
| [−0.33, 4.93] | [−2.46, 1.77] | [−2.51, 1.88] | [−1.89, 2.47] | [−1.97, 2.73] | |
| Sufficiency (25-OH-D ≥ 75 nmol/L) (n = 49) | 3.10 | −1.71 | −1.27 | −2.12 | −1.99 |
| [−0.83, 7.03] | [−5.15, 1.73] | [−5.39, 2.84] | [−5.99, 1.74] | [−5.86, 1.89] | |
| p-value | 0.08 | 0.38 | 0.78 | 0.35 | 0.45 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rahman, A.; Al-Taiar, A.; Shaban, L.; Al-Sabah, R.; Al-Harbi, A.; Mojiminiyi, O. Plasma 25-Hydroxy Vitamin D Is Not Associated with Either Cognitive Function or Academic Performance in Adolescents. Nutrients 2018, 10, 1197. https://doi.org/10.3390/nu10091197
Rahman A, Al-Taiar A, Shaban L, Al-Sabah R, Al-Harbi A, Mojiminiyi O. Plasma 25-Hydroxy Vitamin D Is Not Associated with Either Cognitive Function or Academic Performance in Adolescents. Nutrients. 2018; 10(9):1197. https://doi.org/10.3390/nu10091197
Chicago/Turabian StyleRahman, Abdur, Abdullah Al-Taiar, Lemia Shaban, Reem Al-Sabah, Anwar Al-Harbi, and Olusegun Mojiminiyi. 2018. "Plasma 25-Hydroxy Vitamin D Is Not Associated with Either Cognitive Function or Academic Performance in Adolescents" Nutrients 10, no. 9: 1197. https://doi.org/10.3390/nu10091197
APA StyleRahman, A., Al-Taiar, A., Shaban, L., Al-Sabah, R., Al-Harbi, A., & Mojiminiyi, O. (2018). Plasma 25-Hydroxy Vitamin D Is Not Associated with Either Cognitive Function or Academic Performance in Adolescents. Nutrients, 10(9), 1197. https://doi.org/10.3390/nu10091197




