Obesity in Inflammatory Bowel Disease: Gains in Adiposity despite High Prevalence of Myopenia and Osteopenia
Abstract
1. Introduction
- To evaluate body composition in patients with IBD, with serial prospective measurements over time;
- To explore the influence of clinical factors on body composition in patients with IBD;
- To explore whether standard anthropometric testing can detect aberrations in body composition.
2. Materials and Methods
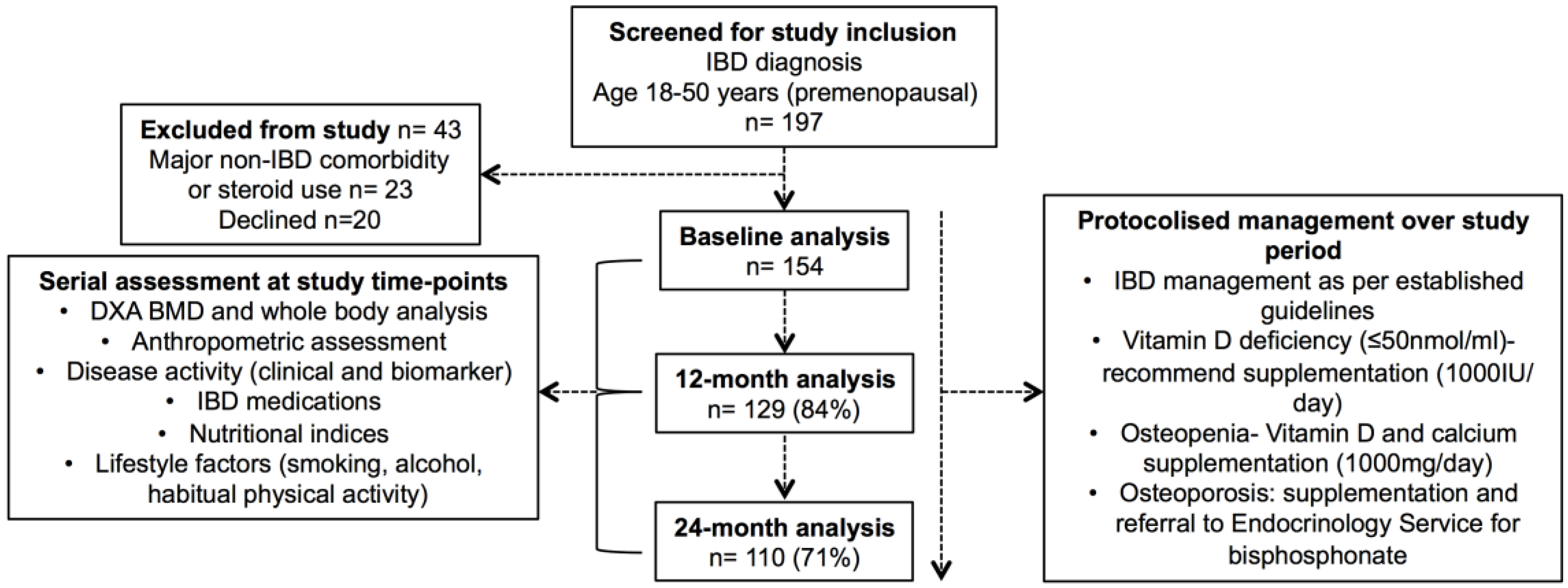
2.1. Subjects
2.2. Subject Data Collection
2.3. Body Composition, Anthropometric Assessment, and Nutritional Assessment
2.4. Management during Prospective Study Period
2.5. Ethical Considerations
2.6. Statistical Methods
3. Results
3.1. Subject Characteristics
3.2. Baseline Body Composition
3.3. Changes in Body Composition over 24 Months
3.4. Clinical Associations with Serial BMI Measurements
3.5. Clinical Associations with Serial FM and VAT Measurements
3.6. Clinical Associations with Serial ASMI Measurements
3.7. Clinical Associations with Serial BMD Measurements
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Bryant, R.V.; Trott, M.J.; Bartholomeusz, F.D.; Andrews, J.M. Systematic review: Body composition in adults with inflammatory bowel disease. Aliment. Pharmacol. Ther. 2013, 38, 213–225. [Google Scholar] [CrossRef] [PubMed]
- Kirchgesner, J.; Beaugerie, L.; Carrat, F.; Andersen, N.N.; Jess, T.; Schwarzinger, M. Increased risk of acute arterial events in young patients and severely active IBD: A nationwide French cohort study. Gut 2017. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.; Dulai, P.S.; Zarrinpar, A.; Ramamoorthy, S.; Sandborn, W.J. Obesity in IBD: Epidemiology, pathogenesis, disease course and treatment outcomes. Nat. Rev. Gastroenterol. Hepatol. 2017, 14, 110–121. [Google Scholar] [CrossRef] [PubMed]
- Winer, D.A.; Luck, H.; Tsai, S.; Winer, S. The intestinal immune system in obesity and insulin resistance. Cell Metab. 2016, 23, 413–426. [Google Scholar] [CrossRef] [PubMed]
- Karmiris, K.; Koutroubakis, I.E.; Xidakis, C.; Polychronaki, M.; Voudouri, T.; Kouroumalis, E.A. Circulating levels of leptin, adiponectin, resistin, and ghrelin in inflammatory bowel disease. Inflamm. Bowel Dis. 2006, 12, 100–105. [Google Scholar] [CrossRef] [PubMed]
- Fink, C.; Karagiannides, I.; Bakirtzi, K.; Pothoulakis, C. Adipose tissue and inflammatory bowel disease pathogenesis. Inflamm. Bowel Dis. 2012, 18, 1550–1557. [Google Scholar] [CrossRef] [PubMed]
- Flores, A.; Burstein, E.; Cipher, D.J.; Feagins, L.A. Obesity in inflammatory bowel disease: A marker of less severe disease. Dig. Dis. Sci. 2015, 60, 2436–2445. [Google Scholar] [CrossRef] [PubMed]
- Seminerio, J.L.; Koutroubakis, I.E.; Ramos-Rivers, C.; Hashash, J.G.; Dudekula, A.; Regueiro, M.; Baidoo, L.; Barrie, A.; Swoger, J.; Schwartz, M.; et al. Impact of obesity on the management and clinical course of patients with inflammatory bowel disease. Inflamm. Bowel Dis. 2015, 21, 2857–2863. [Google Scholar] [CrossRef] [PubMed]
- Hass, D.J.; Brensinger, C.M.; Lewis, J.D.; Lichtenstein, G.R. The impact of increased body mass index on the clinical course of crohn’s disease. Clin. Gastroenterol. Hepatol. Off. Clin. Pract. J. Am. Gastroenterol. Assoc. 2006, 4, 482–488. [Google Scholar] [CrossRef] [PubMed]
- Chan, S.S.; Luben, R.; Olsen, A.; Tjonneland, A.; Kaaks, R.; Teucher, B.; Lindgren, S.; Grip, O.; Key, T.; Crowe, F.L.; et al. Body mass index and the risk for crohn’s disease and ulcerative colitis: Data from a european prospective cohort study (the IBD in EPIC study). Am. J. Gastroenterol. 2013, 108, 575–582. [Google Scholar] [CrossRef] [PubMed]
- Buning, C.; von Kraft, C.; Hermsdorf, M.; Gentz, E.; Wirth, E.K.; Valentini, L.; Haas, V. Visceral adipose tissue in patients with crohn’s disease correlates with disease activity, inflammatory markers, and outcome. Inflamm. Bowel Dis. 2015, 21, 2590–2597. [Google Scholar] [CrossRef] [PubMed]
- Erhayiem, B.; Dhingsa, R.; Hawkey, C.J.; Subramanian, V. Ratio of visceral to subcutaneous fat area is a biomarker of complicated crohn’s disease. Clin. Gastroenterol. Hepatol. Off. Clin. Pract. J. Am. Gastroenterol. Assoc. 2011, 9, 684–687.e681. [Google Scholar] [CrossRef] [PubMed]
- Holt, D.Q.; Moore, G.T.; Strauss, B.J.; Hamilton, A.L.; De Cruz, P.; Kamm, M.A. Visceral adiposity predicts post-operative crohn’s disease recurrence. Aliment. Pharmacol. Ther. 2017, 45, 1255–1264. [Google Scholar] [CrossRef] [PubMed]
- Liu, G.; Wu, X.; Li, Y.; Rui, Y.; Stocchi, L.; Remzi, F.H.; Shen, B. Postoperative excessive gain in visceral adipose tissue as well as body mass index are associated with adverse outcomes of an ileal pouch. Gastroenterol. Rep. 2016, 5, 29–35. [Google Scholar] [CrossRef] [PubMed]
- Van Langenberg, D.R.; Gatta, P.D.; Hill, B.; Zacharewicz, E.; Gibson, P.R.; Russell, A.P. Delving into disability in crohn’s disease: Dysregulation of molecular pathways may explain skeletal muscle loss in crohn’s disease. J. Crohn Colitis 2013, 8, 626–634. [Google Scholar] [CrossRef] [PubMed]
- Schneider, S.M.; Al-Jaouni, R.; Filippi, J.; Wiroth, J.B.; Zeanandin, G.; Arab, K.; Hebuterne, X. Sarcopenia is prevalent in patients with crohn’s disease in clinical remission. Inflamm. Bowel Dis. 2008, 14, 1562–1568. [Google Scholar] [CrossRef] [PubMed]
- Bryant, R.V.; Ooi, S.; Schultz, C.G.; Goess, C.; Grafton, R.; Hughes, J.; Lim, A.; Bartholomeusz, F.D.; Andrews, J.M. Low muscle mass and sarcopenia: Common and predictive of osteopenia in inflammatory bowel disease. Aliment. Pharmacol. Ther. 2015, 41, 895–906. [Google Scholar] [CrossRef] [PubMed]
- Adams, D.W.; Gurwara, S.; Silver, H.J.; Horst, S.N.; Beaulieu, D.B.; Schwartz, D.A.; Seidner, D.L. Sarcopenia is common in overweight patients with inflammatory bowel disease and may predict need for surgery. Inflamm. Bowel Dis. 2017, 23, 1182–1186. [Google Scholar] [CrossRef] [PubMed]
- Pedersen, M.; Cromwell, J.; Nau, P. Sarcopenia is a predictor of surgical morbidity in inflammatory bowel disease. Inflamm. Bowel Dis. 2017, 23, 1867–1872. [Google Scholar] [CrossRef] [PubMed]
- Bamba, S.; Sasaki, M.; Takaoka, A.; Takahashi, K.; Imaeda, H.; Nishida, A.; Inatomi, O.; Sugimoto, M.; Andoh, A. Sarcopenia is a predictive factor for intestinal resection in admitted patients with crohn’s disease. PLoS ONE 2017, 12, e0180036. [Google Scholar] [CrossRef] [PubMed]
- Harbord, M.; Annese, V.; Vavricka, S.R.; Allez, M.; Barreiro-de Acosta, M.; Boberg, K.M.; Burisch, J.; De Vos, M.; De Vries, A.M.; Dick, A.D.; et al. The first european evidence-based consensus on extra-intestinal manifestations in inflammatory bowel disease. J. Crohn Colitis 2016, 10, 239–254. [Google Scholar] [CrossRef] [PubMed]
- Targownik, L.E.; Bernstein, C.N.; Nugent, Z.; Johansson, H.; Oden, A.; McCloskey, E.; Kanis, J.A.; Leslie, W.D. Inflammatory bowel disease and the risk of fracture after controlling for frax. J. Bone Miner. Res. 2013, 28, 1007–1013. [Google Scholar] [CrossRef] [PubMed]
- Targownik, L.E.; Bernstein, C.N.; Nugent, Z.; Leslie, W.D. Inflammatory bowel disease has a small effect on bone mineral density and risk for osteoporosis. Clin. Gastroenterol. Hepatol. Off. Clin. Pract. J. Am. Gastroenterol. Assoc. 2013, 11, 278–285. [Google Scholar] [CrossRef] [PubMed]
- Bernstein, C.N.; Blanchard, J.F.; Leslie, W.; Wajda, A.; Yu, B.N. The incidence of fracture among patients with inflammatory bowel disease. A population-based cohort study. Ann. Intern. Med. 2000, 133, 795–799. [Google Scholar] [CrossRef] [PubMed]
- Casals-Seoane, F.; Chaparro, M.; Mate, J.; Gisbert, J.P. Clinical course of bone metabolism disorders in patients with inflammatory bowel disease: A 5-year prospective study. Inflamm. Bowel Dis. 2016, 22, 1929–1936. [Google Scholar] [CrossRef] [PubMed]
- Targownik, L.E.; Leslie, W.D.; Carr, R.; Clara, I.; Miller, N.; Rogala, L.; Graff, L.A.; Walker, J.R.; Bernstein, C.N. Longitudinal change in bone mineral density in a population-based cohort of patients with inflammatory bowel disease. Calcif. Tissue Int. 2012, 91, 356–363. [Google Scholar] [CrossRef] [PubMed]
- Tosato, M.; Marzetti, E.; Cesari, M.; Savera, G.; Miller, R.R.; Bernabei, R.; Landi, F.; Calvani, R. Measurement of muscle mass in sarcopenia: From imaging to biochemical markers. Aging Clin. Exp. Res. 2017, 29, 19–27. [Google Scholar] [CrossRef] [PubMed]
- Satsangi, J.; Silverberg, M.S.; Vermeire, S.; Colombel, J.F. The montreal classification of inflammatory bowel disease: Controversies, consensus, and implications. Gut 2006, 55, 749–753. [Google Scholar] [CrossRef] [PubMed]
- Ainsworth, B.E.; Macera, C.A.; Jones, D.A.; Reis, J.P.; Addy, C.L.; Bowles, H.R.; Kohl, H.W. Comparison of the 2001 BRFSS and the IPAQ physical activity questionnaires. Med. Sci. Sports Exerc. 2006, 38, 1584–1592. [Google Scholar] [CrossRef] [PubMed]
- Hans, D.B.; Shepherd, J.A.; Schwartz, E.N.; Reid, D.M.; Blake, G.M.; Fordham, J.N.; Fuerst, T.; Hadji, P.; Itabashi, A.; Krieg, M.A.; et al. Peripheral dual-energy X-ray absorptiometry in the management of osteoporosis: The 2007 ISCD official positions. J. Clin. Densitom. Off. J. Int. Soc. Clin. Densitom. 2008, 11, 188–206. [Google Scholar] [CrossRef] [PubMed]
- Cruz-Jentoft, A.J.; Baeyens, J.P.; Bauer, J.M.; Boirie, Y.; Cederholm, T.; Landi, F.; Martin, F.C.; Michel, J.P.; Rolland, Y.; Schneider, S.M.; et al. Sarcopenia: European consensus on definition and diagnosis: Report of the european working group on sarcopenia in older people. Age Ageing 2010, 39, 412–423. [Google Scholar] [CrossRef] [PubMed]
- Kelly, T.L.; Wilson, K.E.; Heymsfield, S.B. Dual energy X-ray absorptiometry body composition reference values from NHANES. PLoS ONE 2009, 4, e7038. [Google Scholar] [CrossRef] [PubMed]
- Kaul, S.; Rothney, M.P.; Peters, D.M.; Wacker, W.K.; Davis, C.E.; Shapiro, M.D.; Ergun, D.L. Dual-energy X-ray absorptiometry for quantification of visceral fat. Obesity 2012, 20, 1313–1318. [Google Scholar] [CrossRef] [PubMed]
- Clinical Guidelines on the Identification; Evaluation; Treatment of Overweight and Obesity in Adults: Executive Summary. Expert panel on the identification, evaluation, and treatment of overweight in adults. Am. J. Clin. Nutr. 1998, 68, 899–917. [CrossRef] [PubMed]
- Australian Bureau of Statistics. National Health Survey: First Results, 2014–2015, 8 December 2015 ed.; Australian Bureau of Statistics: Canberra, Australia, 2015.
- World Health Organization. Assessment of fracture risk and its application to screening for postmenopausal osteoporosis. Report of a WHO study group. World Health Organ. Tech. Rep. Ser. 1994, 843, 1–129. [Google Scholar]
- Dignass, A.; Lindsay, J.O.; Sturm, A.; Windsor, A.; Colombel, J.F.; Allez, M.; D’Haens, G.; D’Hoore, A.; Mantzaris, G.; Novacek, G.; et al. Second european evidence-based consensus on the diagnosis and management of ulcerative colitis part 2: Current management. J. Crohn Colitis 2012, 6, 991–1030. [Google Scholar] [CrossRef] [PubMed]
- Dignass, A.; Van Assche, G.; Lindsay, J.O.; Lemann, M.; Soderholm, J.; Colombel, J.F.; Danese, S.; D’Hoore, A.; Gassull, M.; Gomollon, F.; et al. The second european evidence-based consensus on the diagnosis and management of crohn’s disease: Current management. J. Crohn Colitis 2010, 4, 28–62. [Google Scholar] [CrossRef] [PubMed]
- Britton, K.A.; Massaro, J.M.; Murabito, J.M.; Kreger, B.E.; Hoffmann, U.; Fox, C.S. Body fat distribution, incident cardiovascular disease, cancer, and all-cause mortality. J. Am. Coll. Cardiol. 2013, 62, 921–925. [Google Scholar] [CrossRef] [PubMed]
- Preis, S.R.; Massaro, J.M.; Robins, S.J.; Hoffmann, U.; Vasan, R.S.; Irlbeck, T.; Meigs, J.B.; Sutherland, P.; D’Agostino, R.B., Sr.; O’Donnell, C.J.; et al. Abdominal subcutaneous and visceral adipose tissue and insulin resistance in the framingham heart study. Obesity 2010, 18, 2191–2198. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.; Singh, H.; Loftus, E.V., Jr.; Pardi, D.S. Risk of cerebrovascular accidents and ischemic heart disease in patients with inflammatory bowel disease: A systematic review and meta-analysis. Clin. Gastroenterol. Hepatol. Off. Clin. Pract. J. Am. Gastroenterol. Assoc. 2014, 12, 382–393. [Google Scholar] [CrossRef] [PubMed]
- Cheng, S.; Massaro, J.M.; Fox, C.S.; Larson, M.G.; Keyes, M.J.; McCabe, E.L.; Robins, S.J.; O’Donnell, C.J.; Hoffmann, U.; Jacques, P.F.; et al. Adiposity, cardiometabolic risk, and vitamin d status: The framingham heart study. Diabetes 2010, 59, 242–248. [Google Scholar] [CrossRef] [PubMed]
- Pedersen, H.K.; Gudmundsdottir, V.; Nielsen, H.B.; Hyotylainen, T.; Nielsen, T.; Jensen, B.A.; Forslund, K.; Hildebrand, F.; Prifti, E.; Falony, G.; et al. Human gut microbes impact host serum metabolome and insulin sensitivity. Nature 2016, 535, 376–381. [Google Scholar] [CrossRef] [PubMed]
- Qin, J.; Li, Y.; Cai, Z.; Li, S.; Zhu, J.; Zhang, F.; Liang, S.; Zhang, W.; Guan, Y.; Shen, D.; et al. A metagenome-wide association study of gut microbiota in type 2 diabetes. Nature 2012, 490, 55–60. [Google Scholar] [CrossRef] [PubMed]
- Le Chatelier, E.; Nielsen, T.; Qin, J.; Prifti, E.; Hildebrand, F.; Falony, G.; Almeida, M.; Arumugam, M.; Batto, J.M.; Kennedy, S.; et al. Richness of human gut microbiome correlates with metabolic markers. Nature 2013, 500, 541–546. [Google Scholar] [CrossRef] [PubMed]
- Zeevi, D.; Korem, T.; Zmora, N.; Israeli, D.; Rothschild, D.; Weinberger, A.; Ben-Yacov, O.; Lador, D.; Avnit-Sagi, T.; Lotan-Pompan, M.; et al. Personalized nutrition by prediction of glycemic responses. Cell 2015, 163, 1079–1094. [Google Scholar] [CrossRef] [PubMed]
- Subramaniam, K.; Fallon, K.; Ruut, T.; Lane, D.; McKay, R.; Shadbolt, B.; Ang, S.; Cook, M.; Platten, J.; Pavli, P.; et al. Infliximab reverses inflammatory muscle wasting (sarcopenia) in crohn’s disease. Aliment. Pharmacol. Ther. 2015, 41, 419–428. [Google Scholar] [CrossRef] [PubMed]
- Ding, N.S.; Malietzis, G.; Lung, P.F.C.; Penez, L.; Yip, W.M.; Gabe, S.; Jenkins, J.T.; Hart, A. The body composition profile is associated with response to anti-TNF therapy in crohn’s disease and may offer an alternative dosing paradigm. Aliment. Pharmacol. Ther. 2017, 46, 883–891. [Google Scholar] [CrossRef] [PubMed]
- Holt, D.Q.; Varma, P.; Strauss, B.J.G.; Rajadurai, A.S.; Moore, G.T. Low muscle mass at initiation of anti-TNF therapy for inflammatory bowel disease is associated with early treatment failure: A retrospective analysis. Eur. J. Clin. Nutr. 2017, 71, 773–777. [Google Scholar] [CrossRef] [PubMed]
- Krajcovicova, A.; Hlavaty, T.; Killinger, Z.; Miznerova, E.; Toth, J.; Letkovsky, J.; Nevidanska, M.; Cierny, D.; Koller, T.; Zelinkova, Z.; et al. Combination therapy with an immunomodulator and anti-TNFα agent improves bone mineral density in ibd patients. J. Crohn Colitis 2014, 8, 1693–1701. [Google Scholar] [CrossRef] [PubMed]
- Peyrin-Biroulet, L.; Van Assche, G.; Gomez-Ulloa, D.; Garcia-Alvarez, L.; Lara, N.; Black, C.M.; Kachroo, S. Systematic review of tumor necrosis factor antagonists in extraintestinal manifestations in inflammatory bowel disease. Clin. Gastroenterol. Hepatol. Off. Clin. Pract. J. Am. Gastroenterol. Assoc. 2017, 15, 25–36. [Google Scholar] [CrossRef] [PubMed]
| Baseline | 12 Months | 24 Months | Δ p Value | |||
|---|---|---|---|---|---|---|
| Demographics | Patients (n, %) | 129 (84%) | 129 (84%) | 110 (71%) | - | |
| Male (n, %) | 75 (58%) | 75 (58%) | 62 (58%) | - | ||
| Age (years) (median, IQR) | 31 (25–40) | 32 (26–41) | 33 (27–42) | - | ||
| IBD phenotype | Crohn’s disease (n, %) | 95 (74%) | 92 (71%) | 79 (72%) | ||
| Ulcerative colitis (n, %) | 34 (26%) | 37 (29%) | 28 (25%) | |||
| IBD-related abdominal surgery (n, %) | 39 (30%) | 7 (5%) | 12 (11%) | - | ||
| IBD-related hospitalisation (n, %) | - | 22 (17%) | 12 (11%) | - | ||
| IBD Disease Activity | IBD clinical disease activity score | - | ||||
| Crohn’s Disease CDAI Mean ± SD | 90 ± 99 | 71 ± 67 | 65 ± 58 | |||
| Median, IQR | 60 (26–126) | 62 (26–110) | 49 (20–90) | |||
| Ulcerative colitis partial Mayo mean ± SD | 1.5 ± 2.2 | 1.4 ± 2.2 | 1.5 ± 1.9 | |||
| Median, IQR | 0.5 (0–2) | 0 (0–2) | 0 (0–4) | |||
| C-reactive protein (mg/L) Mean ± SD | 0.40 | |||||
| 8.7 ± 21 | 5.2 ± 11 | 5.3 ± 8 | ||||
| Median, IQR | 1.7 (0.5–7.7) | 1.9 (0.5–5.8) | 1.6 (0.4–6.3) | |||
| Faecal calprotectin (μg/g) | 0.49 | |||||
| Mean ± SD Median, IQR | 247.5 ± 305 | 155.9 ± 206 | 179.4 ± 227 | |||
| 84 (20–450) | 50 (20–205) | 93 (20–245) | ||||
| Composite disease activity assessment § (n active disease, %) | - | |||||
| 65 (50%) | 59 (46%) | 57 (52% | ||||
| IBD Therapy | Oral corticosteroids ^ Current (n, %) | 38 (29%) | 11 (9%) | 7 (6%) | <0.0001 * | |
| Duration usage (mths) Mean ± SD | 28 ± 57 | |||||
| Median, IQR | 6 (1-24) | |||||
| Biologic therapy overall (n, %) | 53 (41%) | 63 (49%) | 59 (54%) | 0.0004 * | ||
| Infliximab (n, %) | 35 (27%) | 41 (32%) | 35 (32%) | |||
| Adalimumab (n, %) | 16 (12%) | 19 (15%) | 22 (20%) | |||
| Vedolizumab (n, %) | 2 (2%) | 3 (2%) | 2 (2%) | |||
| 5-aminosalicylic acid therapy (n, %) | 55 (43%) | 58 (45%) | 53 (48%) | 0.18 | ||
| Immunomodulator therapy Overall (n, %) | 73 (57%) | 78 (60%) | 67 (61%) | 0.12 | ||
| Azathioprine (n, %) | 46 (36%) | 47 (36%) | 41 (37%) | |||
| Mercaptopurine (n, %) | 6 (5%) | 6 (5%) | 5 (5%) | |||
| Methotrexate (n, %) | 3 (2%) | 5 (3%) | 4 (4%) | |||
| Thiopurine/allopurinol (n, %) | 18 (14%) | 20 (16%) | 17 (15%) | |||
| Exercise (IPAQ) ¶ | Continuous | Mean ± SD | 4385 ± 5908 | 5570 ± 8952 | 4935 ± 6879 | 0.36 |
| Median, IQR | 2284 (693–5690) | 2106 (862–5745) | 2445 (942–5558) | |||
| Categorical | Inactive | 50 (39%) | 47 (36%) | 37 (34%) | ||
| Minimally active | 30 (23%) | 24 (19%) | 23 (21%) | |||
| Active | 28 (22%) | 41 (32%) | 35 (32%) | |||
| Nutritional assessment | Albumin (g/dL) | Mean ± SD | 40 ± 4.6 | 40 ± 3.8 | 40 ± 3.5 | 0.64 |
| Median, IQR | 40 (37–43) | 40 (37–42) | 40 (38–42) | |||
| Haemoglobin (g/L) | Mean ± SD | 141 ± 15 | 141 ± 14 | 143 ± 13 | 0.07 | |
| Median, IQR | 141 (132–150) | 141 (130–149) | 144 (136–152) | |||
| Ferritin (ng/mL) | Mean ± SD | 87 ± 85 | 93 ± 122 | 115 ± 186 | 0.09 | |
| Median, IQR | 62 (34–106) | 63 (35–104) | 71 (42–123) | |||
| Calcium Mean ± SD | 2.35 ± 0.11 | 2.34 ± 0.1 | 2.34 ± 0.09 | 0.16 | ||
| Median, IQR | 2.35 (2.28–2.42) | 2.36 (2.29–2.4) | 2.34 (2.30–2.4) | |||
| Vitamin D (nmol/mL) # | Mean ± SD | 64 ± 28 | 65 ± 25 | 70 ± 25 | 0.01 * | |
| Median, IQR | 63 (41–84) | 65 (48–80) | 67 (53–85) | |||
| Low Vitamin D level | 52 (40%) | 40 (31%) | 38 (35%) | |||
| Vitamin D supplementation | 50 (39%) | 40 (31%) | 37 (34%) | |||
| Bisphosphonate therapy | 2 (1%) | 4 (3%) | 4 (4%) | |||
| Body Composition | Baseline (n = 129) | Year 1 (n = 129) | Year 2 (n = 110) | p Value | ||
|---|---|---|---|---|---|---|
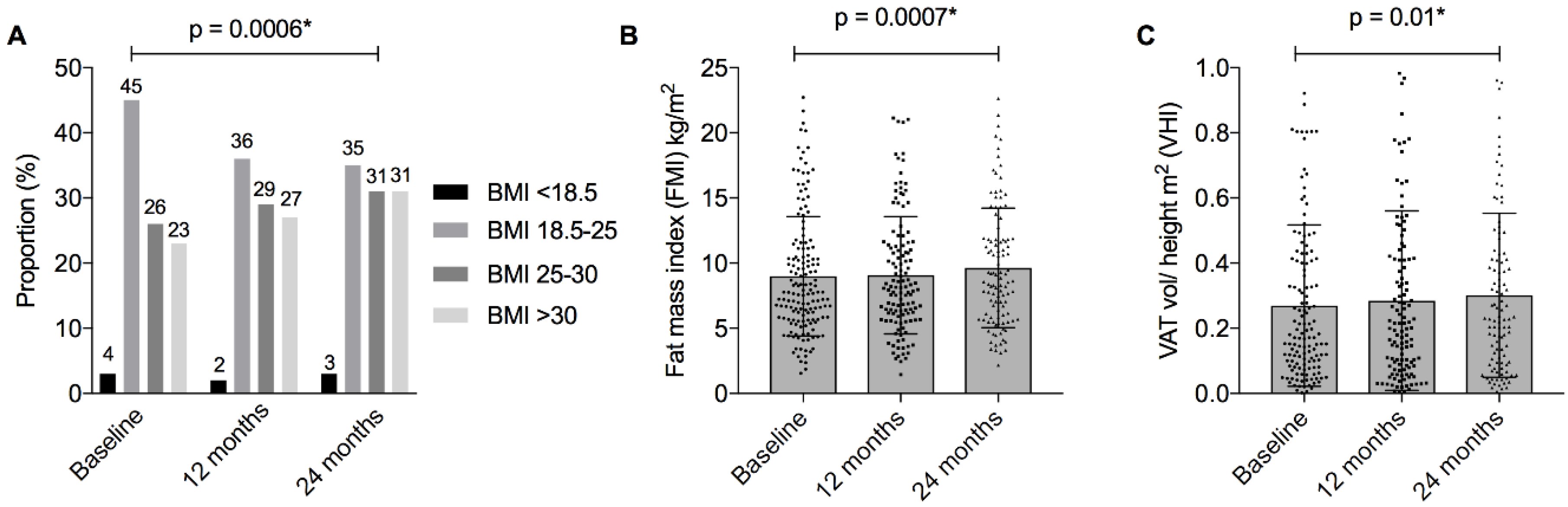
| Anthropometric assessment | Body mass index (BMI) | Mean ± SD (kg/m2) | 26.5 ± 5.1 | 27.4 ± 5.8 | 27.7 ± 5.6 | 0.0006 * |
| Δ ± SD | - | 0.52 ± 2.51 | 0.84 ± 2.66 | |||
| Median (IQR) | 25.1 (22.9–29.7) | 26.5 (23.2–30.7) | 26.8 (23.8–30.8) | |||
| BMI categorical ¶ | Underweight < 18.5 | 4 (3%) | 3 (2%) | 3 (3%) | 0.0006 * | |
| Normal 18.5–25 | 58 (45%) | 47 (36%) | 38 (35%) | |||
| Overweight 25–30 | 33 (26%) | 38 (29%) | 34 (31%) | |||
| Obese ≥ 30 | 30 (23%) | 35 (27%) | 34 (31%) | |||
| Waist circumference (cm) | Mean ± SD | 90.8 ± 15.9 | 92.4 ± 14.5 | 94.1 ± 14.4 | 0.003 * | |
| Δ ± SD | - | 0.73 ± 10.32 | 2.26 ± 9.18 | |||
| Median (IQR) | 88 (80–101) | 91 (81–102) | 93 (83–103) | |||
| Waist:hip ratio | Mean ± SD | 0.88 ± 0.11 | 0.88 ± 0.10 | 0.89 ± 0.10 | 0.31 | |
| Δ ± SD | - | −0.004 ± 0.11 | 0.01 ± 0.12 | |||
| Median (IQR) | 0.86 (0.81–0.95) | 0.87 (0.82–0.92) | 0.89 (0.83–0.94) | |||
| Grip strength (PSI) | Mean ± SD | 40.2 ± 12.0 | 39.5 ± 12.2 | 39.8 ± 12.4 | 0.68 | |
| Δ ± SD | - | −0.48 ± 5.56 | −0.30 ± 5.01 | |||
| Median (IQR) | 37.8 (30.0–50.0) | 38.0 (29.0–49.3) | 37.4 (31.1–49.1) | |||
| Grip strength z-score | Mean ± SD | −0.63 ± 1.30 | −0.9 ± 1.49 | −0.67 ± 1.23 | 0.46 | |
| Δ ± SD | - | −0.27 ± 1.55 | −0.01 ± 1.27 | |||
| Median (IQR) | −0.48 (−1.29–0.15) | −0.75 (−1.41–0.12) | −0.6 (−1.32–0.15) | |||
| Fat mass index (FMI) (kg/height m2) | FMI (kg/m2) | Mean ± SD | 8.83 ± 4.57 | 9.07 ± 4.50 | 9.64 ± 4.59 | 0.0007 * |
| Δ ± SD | - | 0.26 ± 1.54 | 0.70 ± 2.05 | |||
| Median (IQR) | 7.67 (5.67–10.60) | 8.09 (6.07–11.32) | 8.61 (5.82–11.81) | |||
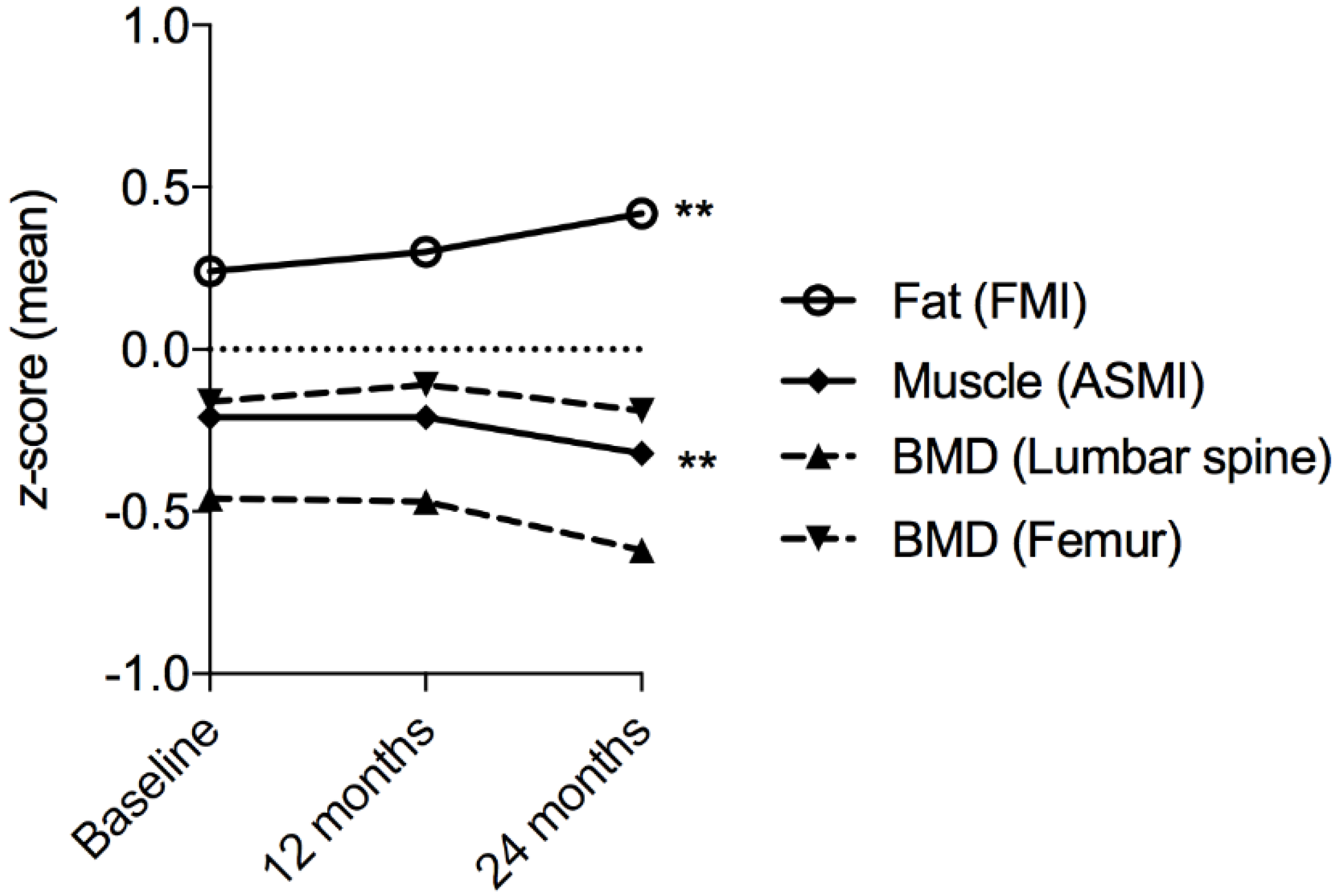
| FMI z-score | Mean ± SD | 0.24 ± 1.26 | 0.30 ± 1.26 | 0.42 ± 1.24 | 0.006 * | |
| Δ ± SD | - | 0.06 ± 0.48 | 0.17 ± 0.61 | |||
| Median (IQR) | −0.13 (−0.59–0.93) | 0.08 (−0.57–0.89) | 0.25 (−0.47–1.15) | |||
| Visceral adipose tissue (VAT) | VAT volume (cm3) | Mean ± SD | 834.2 ± 821.7 | 903.9 ± 901.6 | 949.9 ± 826.7 | 0.01 * |
| Δ ± SD | - | 67.8 ± 331.8 | 102.1 ± 376.6 | |||
| Median (IQR) | 493.7 (216.9, 1175.9) | 600.5 (248.8, 1291) | 702.1 (266.6–1354) | |||
| VAT (grams) ^ | Mean ± SD | 786.9 ± 775.2 | 852.76 ± 850.53 | 896.09 ± 779.94 | 0.01 * | |
| Δ ± SD | - | 64.0 ± 313.0 | 96.3 ± 355.3 | |||
| Median (IQR) | 465.74 (204.59–1109.38) | 566.51 (241.63–1209.85) | 662.35 (256.23–1274.44) | |||
| VAT:SAT ^ | Mean ± SD | 0.64 ± 0.68 | 0.64 ± 0.55 | 0.63 ± 0.49 | 0.36 | |
| Δ ± SD | - | −0.005 ± 0.50 | 0.026 ± 0.585 | |||
| Median (IQR) | 0.45 (0.24–0.8) | 0.46 (0.31–0.83) | 0.49 (0.28–0.89) | |||
| VHI ^ | Mean ± SD | 0.26 ± 0.25 | 0.28 ± 0.28 | 0.3 ± 0.25 | 0.01 * | |
| Δ ± SD | - | 0.022 ± 0.10 | 0.127 ± 0.604 | |||
| Median (IQR) | 0.15 (0.07–0.35) | 0.2 (0.08–0.41) | 0.23 (0.1–0.41) | |||
| Bone mineral density (BMD) | WHO t-score femur | Mean ± SD | −0.18 ± 1.02 | −0.1 ± 1.03 | −0.13 ± 1.03 | 0.001 * |
| Δ ± SD | - | 0.075 ± 0.191 | 0.081 ±0.266 | |||
| Median (IQR) | −0.21 (−0.96–0.52) | −0.20 (−0.99–0.55) | −0.17 (−0.97–0.69) | |||
| WHO t-score spine | Mean ± SD | −0.35 ± 1.1 | −0.29 ± 1.11 | −0.38 ± 1.17 | 0.47 | |
| Δ ± SD | - | 0.033 ± 0.34 | 0.027 ± 0.357 | |||
| Median (IQR) | −0.48 (−1.06–0.4) | −0.37 (−1.05–0.39) | −0.36 (−1.21–0.33) | |||
| WHO z-score femur | Mean ± SD | −0.16 ± 1.06 | −0.11 ± 1.03 | −0.19 ± 0.93 | 0.86 | |
| Δ ± SD | - | 0.049 ± 0.31 | 0.007 ± 0.52 | |||
| Median (IQR) | −0.24 (−0.93–0.46) | −0.14 (−0.90–0.51) | −0.12 (−0.91–0.48) | |||
| WHO z-score spine | Mean ± SD | −0.46 ± 1.08 | −0.47 ± 1.09 | −0.62 ± 1.14 | 0.51 | |
| Δ ± SD | - | 0.004 ± 0.385 | −0.025 ± 0.41 | |||
| Median (IQR) | −0.52 (−1.19–0.29) | −0.52 (−1.22–0.23) | −0.58 (−1.4–0.18) | |||
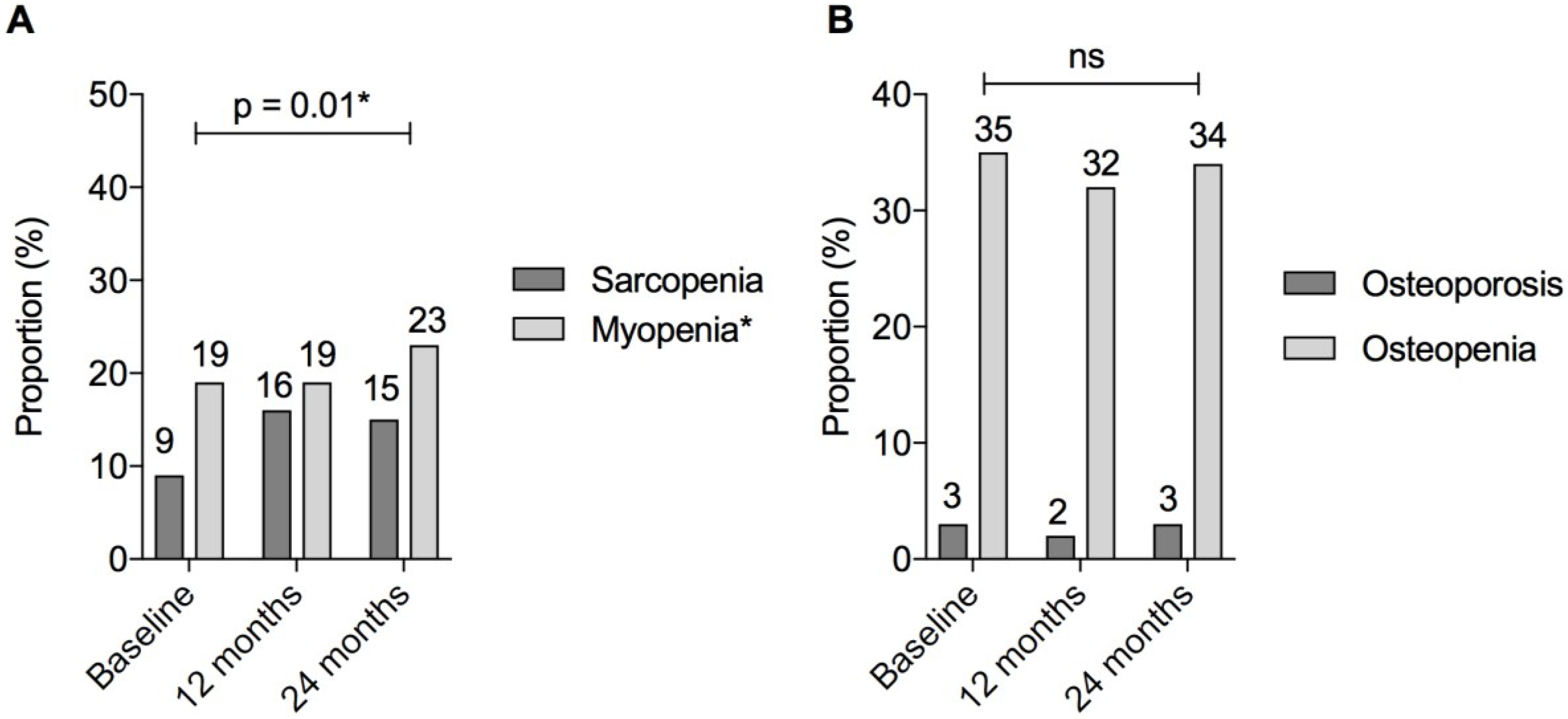
| Bone status # | Osteopenia (n, %) | 45 (35%) | 41 (32%) | 37 (34%) | 0.23 | |
| Osteoporosis (n, %) | 3 (2%) | 2 (2%) | 3 (3%) | |||
| Overall osteopenia/porosis (n, %) | 48 (37%) | 43 (34%) | 40 (37%) | |||
| Appendicular skeletal muscle index (ASMI) (kg/height m2) | ASMI (kg/m2) | Mean ± SD | 7.76 ± 1.40 | 7.80 ± 1.45 | 7.68 ± 1.40 | 0.01 * |
| Δ ± SD | - | 0.014 ± 0.49 | −0.127 ± 0.55 | |||
| Median (IQR) | 7.60 (6.57–8.48) | 7.84 (6.74–8.62) | 7.64 (6.66–8.45) | |||
| ASMI z-score | Mean ± SD | −0.21 ± 0.96 | −0.21 ± 0.98 | −0.32 ± 0.99 | 0.002 * | |
| Δ ± SD | - | −0.006 ± 0.392 | −0.0129 ± 0.444 | |||
| Median (IQR) | −0.31 (−0.95–0.17) | −0.3 (−0.88–0.24) | −0.45 (−0.93–0.29) | |||
| Myopenia (Low ASMI §) | 24 (19%) | 25 (19%) | 25 (23%) | 0.01 * | ||
| Functional sarcopenia (Low ASMI AND low grip strength §) | 12 (9%) | 21 (16%) | 17 (15%) | 0.05 | ||
| Variable | Univariable | Full Multivariable Model | |||
|---|---|---|---|---|---|
| Est. (95%CI) | p Value | Est. (95%CI) | p Value | ||
| Time | Repeated measures over 24 m | 0.43 [0.21, 0.65] | <0.0001 | 0.08 [−0.08, 0.24] | 0.28 |
| Demographics | Age at study entry | 0.24 [0.15, 0.33] | <0.0001 | 0.021 [−0.004, 0.046] | 0.08 |
| Gender (Male vs. female) | 0.0 [−2.0, 2.0] | 0.99 | 0.38 [−0.18, 0.95] | 0.17 | |
| IBD-related factors | IBD phenotype (Ulcerative colitis vs. Crohn’s disease) | −0.7 [−2.9, 1.5] | 0.52 | −0.04 [−0.46, 0.38] | 0.86 |
| IBD disease duration | 0.014 [0.005, 0.022] | 0.002 | 0.0003 [−0.0020, 0.0026] | 0.78 | |
| Faecal calprotectin (μg/g) | −0.0015 [−0.0025, 0.004] | 0.007 | 0.00021 [−0.00044, 0.00086] | 0.48 | |
| C-reactive protein (mg/L) | −0.011 [−0.027, 0.006] | 0.19 | 0.001 [−0.009, 0.011] | 0.85 | |
| Current corticosteroid use | −0.4 [−1.0, 0.2] | 0.22 | −0.10 [−0.49, 0.29] | 0.55 | |
| Biologic therapy | 0.25 [−0.31, 0.81] | 0.38 | 0.04 [−0.18, 0.25] | 0.69 | |
| Immunomodulator therapy | 0.42 [−0.03, 0.88] | 0.06 | 0.11 [−0.02, 0.23] | 0.08 | |
| Lifestyle and nutritional factors | Smoking status Current vs. never Ex vs. never | 0.8 [−1.9, 3.4] 2.0 [−0.4, 4.4] | 0.23 | −0.24 [−0.75, 0.28] 0.34 [−0.11, 0.78] | 0.07 |
| Excess alcohol intake ^ | 0.2 [−4.9, 5.3] | 0.95 | 0.8 [−0.1, 1.8] | 0.06 | |
| Vitamin D level (nmol/mL) | 0.006 [−0.005, 0.017] | 0.25 | 0.006 [0.000, 0.011] | 0.03 | |
| Habitual exercise (IPAQ score, continuous) § | 0.002 [−0.036, 0.041] | 0.91 | 0.009 [−0.011, 0.029] | 0.36 | |
| Albumin (g/dL) | 0.06 [−0.02, 0.13] | 0.16 | 0.008 [−0.034, 0.049] | 0.76 | |
| Body composition factors | Fat mass index (FMI) | 1.1 [1.0, 1.2] | <0.0001 | 1.0 [1.0, 1.1] | <0.0001 |
| Appendicular skeletal muscle index (ASMI) | 2.1 [1.8, 2.5] | <0.0001 | 1.3 [1.1, 1.5] | <0.0001 | |
| Functional sarcopenia ¶ | −1.5 [−2.5, −0.4] | 0.0006 | 0.11 [−0.41, 0.62] | 0.65 | |
| Grip strength (pounds per square inch, PSI) | 0.024 [−0.024, 0.072] | 0.32 | 0.028 [0.008, 0.049] | 0.006 | |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bryant, R.V.; Schultz, C.G.; Ooi, S.; Goess, C.; Costello, S.P.; Vincent, A.D.; Schoeman, S.N.; Lim, A.; Bartholomeusz, F.D.; Travis, S.P.L.; et al. Obesity in Inflammatory Bowel Disease: Gains in Adiposity despite High Prevalence of Myopenia and Osteopenia. Nutrients 2018, 10, 1192. https://doi.org/10.3390/nu10091192
Bryant RV, Schultz CG, Ooi S, Goess C, Costello SP, Vincent AD, Schoeman SN, Lim A, Bartholomeusz FD, Travis SPL, et al. Obesity in Inflammatory Bowel Disease: Gains in Adiposity despite High Prevalence of Myopenia and Osteopenia. Nutrients. 2018; 10(9):1192. https://doi.org/10.3390/nu10091192
Chicago/Turabian StyleBryant, Robert Venning, Christopher G. Schultz, Soong Ooi, Charlotte Goess, Samuel Paul Costello, Andrew D. Vincent, Scott N. Schoeman, Amanda Lim, Francis Dylan Bartholomeusz, Simon P.L. Travis, and et al. 2018. "Obesity in Inflammatory Bowel Disease: Gains in Adiposity despite High Prevalence of Myopenia and Osteopenia" Nutrients 10, no. 9: 1192. https://doi.org/10.3390/nu10091192
APA StyleBryant, R. V., Schultz, C. G., Ooi, S., Goess, C., Costello, S. P., Vincent, A. D., Schoeman, S. N., Lim, A., Bartholomeusz, F. D., Travis, S. P. L., & Andrews, J. M. (2018). Obesity in Inflammatory Bowel Disease: Gains in Adiposity despite High Prevalence of Myopenia and Osteopenia. Nutrients, 10(9), 1192. https://doi.org/10.3390/nu10091192





