Levodopa-Reduced Mucuna pruriens Seed Extract Shows Neuroprotective Effects against Parkinson’s Disease in Murine Microglia and Human Neuroblastoma Cells, Caenorhabditis elegans, and Drosophila melanogaster
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Preparation of Mucuna pruriens Seeds Extract (MPE)
2.3. Quantification of L-Dopa by Liquid Chromatography Electrospray Ionization Tandem Mass Spectrometry (LC-ESI-MS/MS)
2.4. Cell Culture
2.5. Cell Viability
2.6. Determination of Hydrogen Peroxide (H2O2)-Induced Reactive Oxygen Species (ROS) in Murine Microglia BV-2 Cells
2.7. Measurement of Lipopolysaccharide (LPS)-Induced Nitric Oxide Species (NOS) in Murine Microglia BV-2 Cells
2.8. Non-Contact Co-Culture Assay with BV-2 and SH-SY5Y Cells
2.9. 1-Methyl-4-Phenylpyridinium (MPP+) Induced Dopaminergic Neurotoxicity in C. elegans
2.10. D. melanogaster Strains and Maintenance
2.11. Negative Geotaxis (Climbing) Assay in D. melanogaster
2.12. Statistical Analyses
3. Results and Discussion
3.1. Preparation of Levodopa (L-Dopa)-Reduced Mucuna pruriens Extract (MPE)
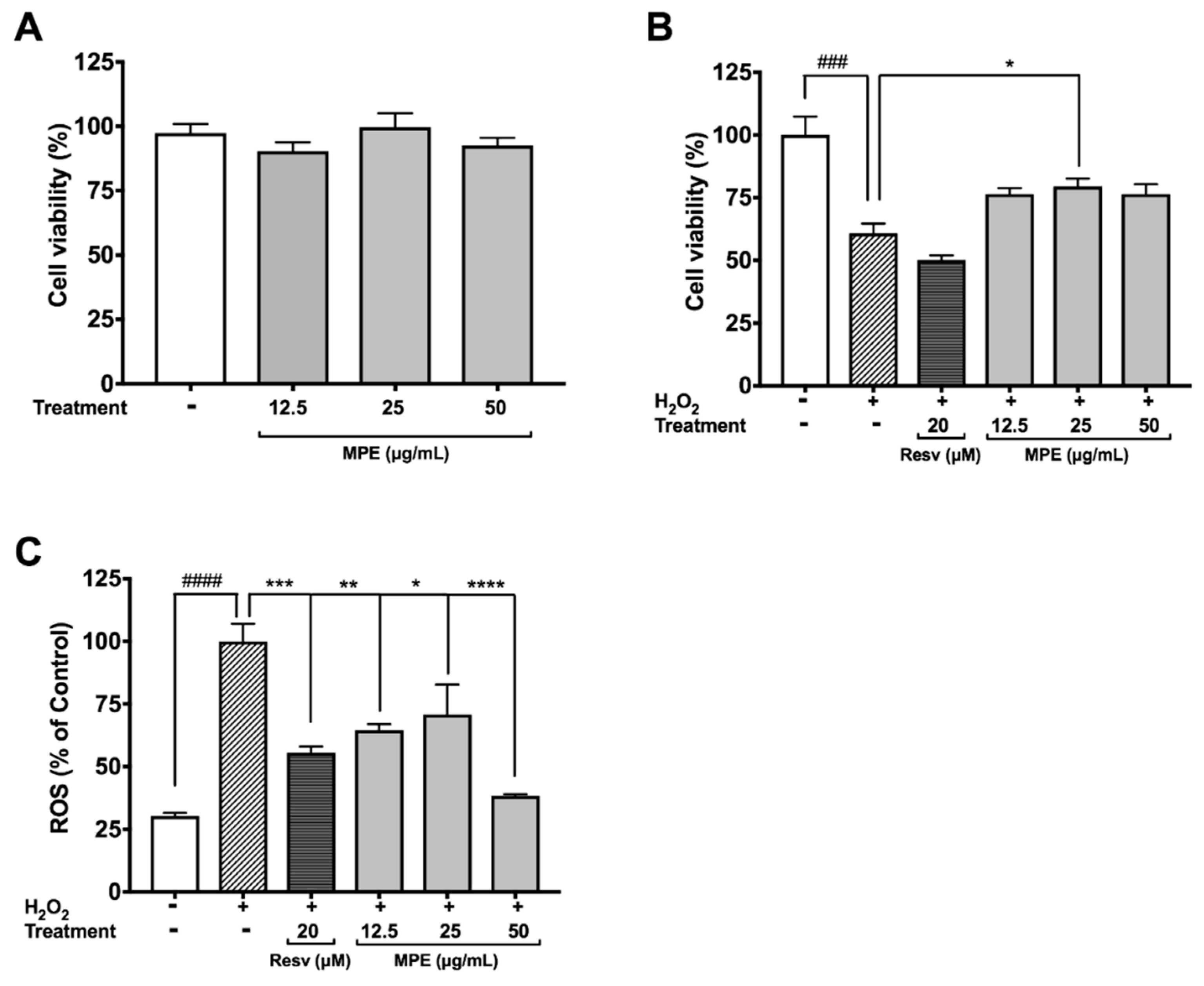
3.2. MPE Reduces Hydrogen Peroxide (H2O2)-Induced Toxicity and Reactive Oxygen Species (ROS) Production in Microglia BV-2 Cells
3.3. MPE Reduces Lipopolysaccharide (LPS)-Induced Nitric Oxide Species (NOS) Production in Microglia BV-2 Cells and Protects SH-SY5Y Cells in a Co-Culture Model
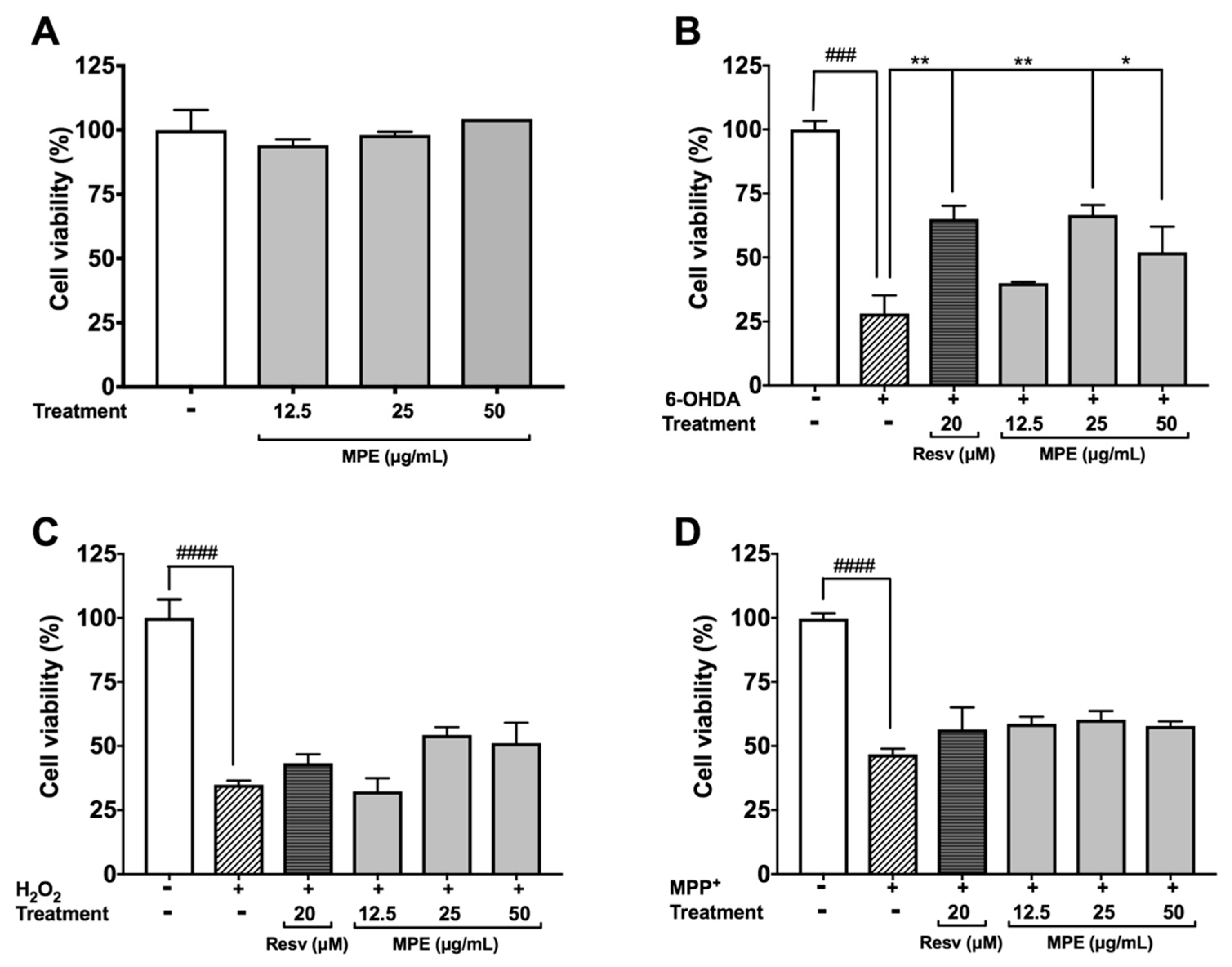
3.4. MPE Reduces Oxidative Stress Induced Cytotoxicity in SH-SY5Y Cells
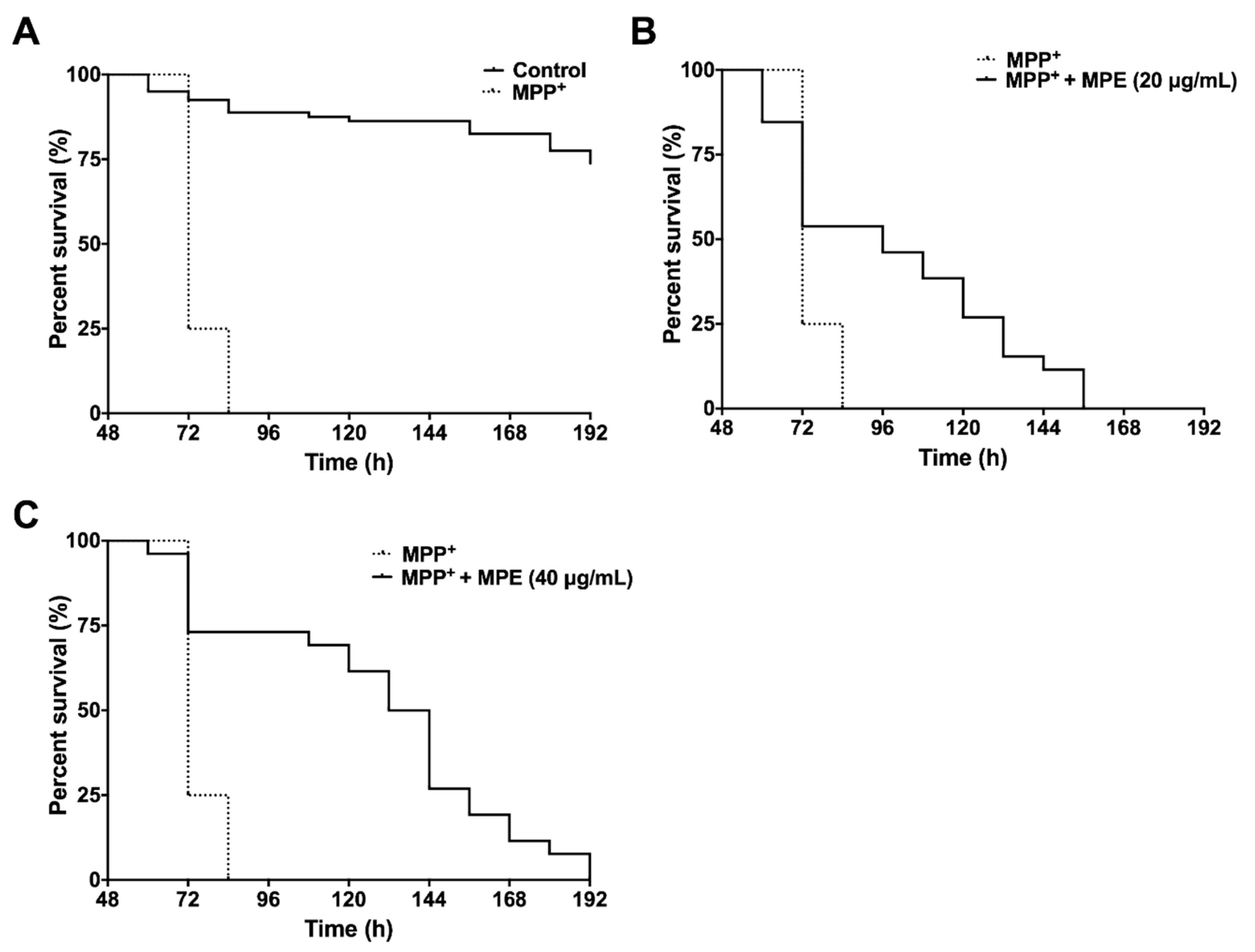
3.5. MPE Reduces Lethality of MPP+ Induced Dopaminergic Neurotoxicity in C. elegans
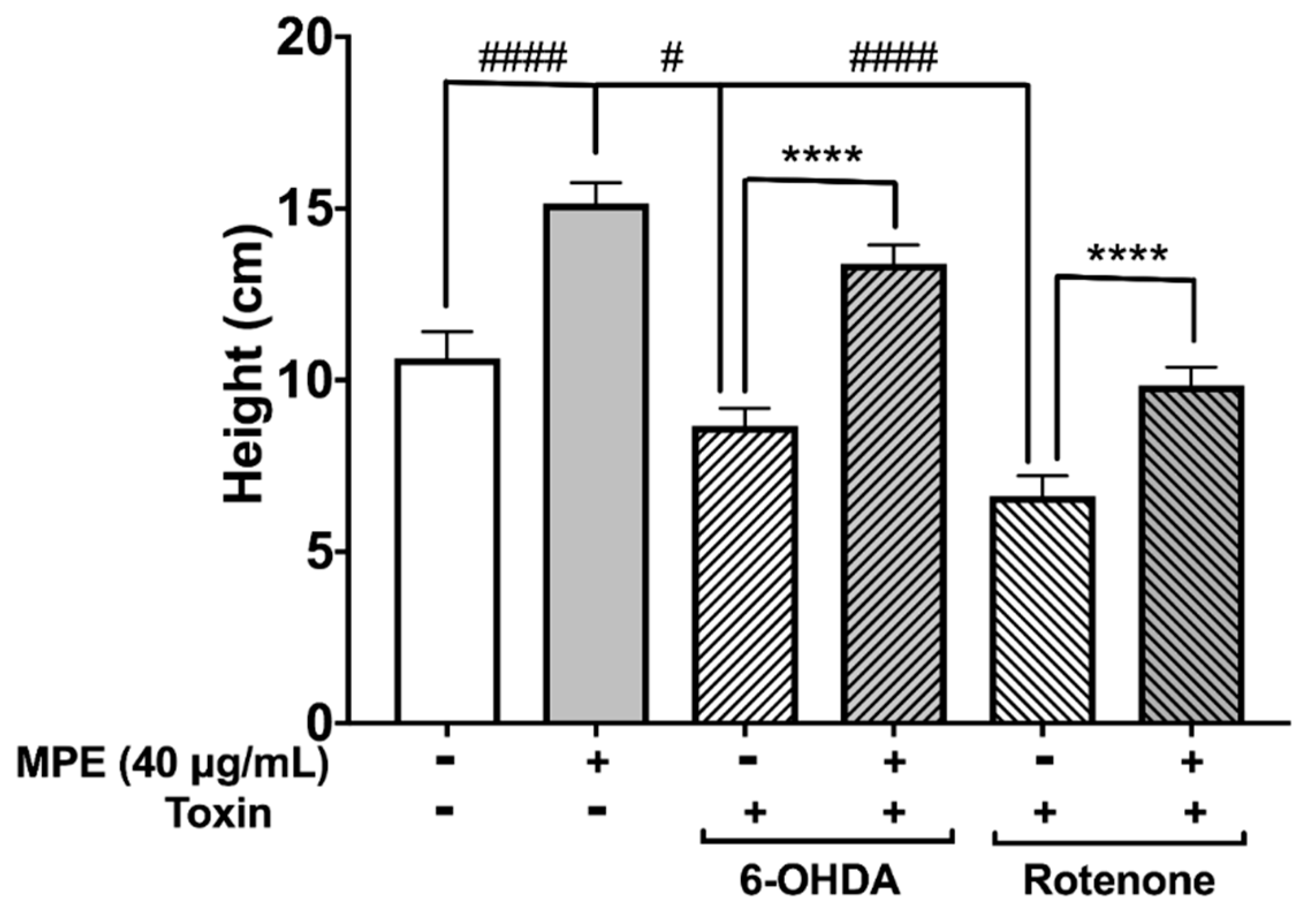
3.6. MPE Abrogates Chemically Induced Neurotoxicity in D. melanogaster
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Abushouk, A.I.; Negida, A.; Ahmed, H.; Abdel-Daim, M.M. Neuroprotective mechanisms of plant extracts against MPTP induced neurotoxicity: Future applications in Parkinson’s disease. Biomed. Pharmacother. 2017, 85, 635–645. [Google Scholar] [CrossRef] [PubMed]
- Dexter, D.T.; Jenner, P. Parkinson disease: From pathology to molecular disease mechanisms. Free Radic. Biol. Med. 2013, 62, 132–144. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.W.; Ko, H.S.; Dawson, V.L.; Dawson, T.M. Recent advances in our understanding of Parkinson’s disease. Drug Discov. Today Dis. Mech. 2005, 2, 427–433. [Google Scholar] [CrossRef]
- Tapias, V.; Cannon, J.R.; Greenamyre, J.T. Pomegranate juice exacerbates oxidative stress and nigrostriatal degeneration in Parkinson’s disease. Neurobiol. Aging 2014, 35, 161–169. [Google Scholar] [CrossRef] [PubMed]
- Lampariello, L.R.; Cortelazzo, A.; Guerranti, R.; Sticozzi, C.; Valacchi, G. The magic velvet bean of Mucuna pruriens. J. Tradit. Complement. Med. 2012, 2, 331–339. [Google Scholar] [CrossRef]
- Natarajan, K.; Narayanan, N.; Ravichandran, N. Review on “Mucuna”—The wonder plant. Int. J. Pharm. Sci. Rev. Res. 2012, 17, 86–93. [Google Scholar]
- Katzenschlager, R.; Evans, A.; Manson, A.; Patsalos, P.; Ratnaraj, N.; Watt, H.; Timmermann, L.; Van der Giessen, R.; Lees, A. Mucuna pruriens in Parkinson’s disease: A double blind clinical and pharmacological study. J. Neurol. Neurosurg. Psychiatry 2004, 75, 1672–1677. [Google Scholar] [CrossRef] [PubMed]
- Nagashima, Y.; Kondo, T.; Sakata, M.; Koh, J.; Ito, H. Effects of soybean ingestion on pharmacokinetics of levodopa and motor symptoms of Parkinson’s disease—In relation to the effects of Mucuna pruriens. J. Neurol. Sci. 2016, 361, 229–234. [Google Scholar] [CrossRef] [PubMed]
- Cilia, R.; Laguna, J.; Cassani, E.; Cereda, E.; Pozzi, N.G.; Isaias, I.U.; Contin, M.; Barichella, M.; Pezzoli, G. Mucuna pruriens in Parkinson Disease A double-blind, randomized, controlled, crossover study. Neurology 2017, 89, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Manyam, B.V.; Dhanasekaran, M.; Hare, T.A. Neuroprotective effects of the antiparkinson drug Mucuna pruriens. Phyther. Res. 2004, 18, 706–712. [Google Scholar] [CrossRef] [PubMed]
- Kasture, S.; Pontis, S.; Pinna, A.; Schintu, N.; Spina, L.; Longoni, R.; Simola, N.; Ballero, M.; Morelli, M. Assessment of symptomatic and neuroprotective efficacy of Mucuna pruriens seed extract in rodent model of Parkinson’s disease. Neurotox. Res. 2009, 15, 111–122. [Google Scholar] [CrossRef] [PubMed]
- Lieu, C.A.; Kunselman, A.R.; Manyam, B.V.; Venkiteswaran, K.; Subramanian, T. A water extract of Mucuna pruriens provides long-term amelioration of parkinsonism with reduced risk for dyskinesias. Park. Relat. Disord. 2010, 16, 458–465. [Google Scholar] [CrossRef] [PubMed]
- Poddighe, S.; De Rose, F.; Marotta, R.; Ruffilli, R.; Fanti, M.; Secci, P.P.; Mostallino, M.C.; Setzu, M.D.; Zuncheddu, M.A.; Collu, I.; et al. Mucuna pruriens (Velvet bean) rescues motor, olfactory, mitochondrial and synaptic impairment in PINK1B9 Drosophila melanogaster genetic model of Parkinson’s disease. PLoS ONE 2014, 9, e110802. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Ma, H.; Dasilva, N.A.; Rose, K.N.; Johnson, S.L.; Zhang, L.; Wan, C.; Dain, J.A.; Seeram, N.P. Development of a neuroprotective potential algorithm for medicinal plants. Neurochem. Int. 2016, 100, 164–177. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Seeram, N.P. Liquid chromatography coupled with time-of-flight tandem mass spectrometry for comprehensive phenolic characterization of pomegranate fruit and flower extracts used as ingredients in botanical dietary supplements. J. Sep. Sci. 2018. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Siard, M.; Adams, A.; Keowen, M.L.; Miller, T.K.; Garza, F.; Andrews, F.M.; Seeram, N.P. Simultaneous quantification of free curcuminoids and their metabolites in equine plasma by LC-ESI–MS/MS. J. Pharm. Biomed. Anal. 2018, 154, 31–39. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Seeram, N.P. Ultra-fast liquid chromatography coupled with electrospray ionization time-of-flight mass spectrometry for the rapid phenolic profiling of red maple (Acer rubrum) leaves. J. Sep. Sci. 2018, 41, 2331–2346. [Google Scholar] [CrossRef] [PubMed]
- Eisenhofer, G.; Brown, S.; Peitzsch, M.; Pelzel, D.; Lattke, P.; Glöckner, S.; Stell, A.; Prejbisz, A.; Fassnacht, M.; Beuschlein, F.; et al. Levodopa therapy in Parkinson’s disease: Influence on liquid chromatographic tandem mass spectrometric-based measurements of plasma and urinary normetanephrine, metanephrine and methoxytyramine. Ann. Clin. Biochem. 2014, 51, 38–46. [Google Scholar] [CrossRef] [PubMed]
- Vilhena, R.O.; Pontes, F.L.; Marson, B.M.; Ribeiro, R.P.; de Carvalho, K.A.T.; Cardoso, M.A.; Pontarolo, R. A new HILIC-MS/MS method for the simultaneous analysis of carbidopa, levodopa, and its metabolites in human plasma. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2014, 967, 41–49. [Google Scholar] [CrossRef] [PubMed]
- Ma, H.; Johnson, S.L.; Liu, W.; Dasilva, N.A.; Meschwitz, S.; Dain, J.A.; Seeram, N.P. Evaluation of polyphenol anthocyanin-enriched extracts of blackberry, black raspberry, blueberry, cranberry, red raspberry, and strawberry for free radical scavenging, reactive carbonyl species trapping, anti-glycation, anti-β-amyloid aggregation, and microglial neuroprotective effects. Int. J. Mol. Sci. 2018, 19, 416. [Google Scholar] [CrossRef]
- Zhao, L.; Chen, J.; Su, J.; Li, L.; Hu, S.; Li, B.; Zhang, X.; Xu, Z.; Chen, T. In vitro antioxidant and antiproliferative activities of 5-hydroxymethylfurfural. J. Agric. Food Chem. 2013, 61, 10604–10611. [Google Scholar] [CrossRef] [PubMed]
- Ma, H.; DaSilva, N.A.; Liu, W.; Nahar, P.P.; Wei, Z.; Liu, Y.; Pham, P.T.; Crews, R.; Vattem, D.A.; Slitt, A.L.; et al. Effects of a standardized phenolic-enriched maple syrup extract on β-amyloid aggregation, neuroinflammation in microglial and neuronal cells, and β-amyloid induced neurotoxicity in Caenorhabditis elegans. Neurochem. Res. 2016, 41, 2836–2841. [Google Scholar] [CrossRef] [PubMed]
- Ormerod, K.G.; LePine, O.K.; Abbineni, P.S.; Bridgeman, J.M.; Coorssen, J.R.; Mercier, A.J.; Tattersall, G.J. Drosophila development, physiology, behavior, and lifespan are influenced by altered dietary composition. Fly (Austin) 2017, 11, 153–170. [Google Scholar] [CrossRef] [PubMed]
- Nichols, C.D.; Becnel, J.; Pandey, U.B. Methods to assay Drosophila behavior. J. Vis. Exp. 2012, 3–7. [Google Scholar] [CrossRef]
- Kasture, S.; Mohan, M.; Kasture, V. Mucuna pruriens seeds in treatment of Parkinson’s disease: Pharmacological review. Orient. Pharm. Exp. Med. 2013, 13, 165–174. [Google Scholar] [CrossRef]
- Sridhar, K.R.; Bhat, R. Agrobotanical, nutritional and bioactive potential of unconventional legume—Mucuna. Livest. Res. Rural Dev. 2007, 19, 1–31. Available online: http://www.lrrd.org/lrrd19/9/srid19126.htm (accessed on 3 August 2018).
- Damodaran, M.; Ramaswamy, R. Isolation of I-3:4-dihydroxyphenylalanine from the seeds of Mucuna pruriens. Biochem. J. 1937, 31, 2149–2152. [Google Scholar] [CrossRef] [PubMed]
- Misra, L.; Wagner, H. Lipid derivatives from Mucuna pruriens seeds. Indian J. Chem. 2006, 45, 801–804. [Google Scholar]
- Janardhanan, K.; Gurumoorthi, P.; Pugalenthi, M. Nutritional Potential of Five Accessions of a South Indian Tribal Pulse, Mucuna pruriens var utilis I. The effect of processing methods on the content of L-Dopa, phytic acid, and oligosaccharides. Trop. Subtrop. Agroecosyst. 2003, 1, 141–152. [Google Scholar] [CrossRef]
- Lieu, C.A.; Venkiteswaran, K.; Gilmour, T.P.; Rao, A.N.; Petticoffer, A.C.; Gilbert, E.V.; Deogaonkar, M.; Manyam, B.V.; Subramanian, T. The antiparkinsonian and antidyskinetic mechanisms of Mucuna pruriens in the MPTP-treated nonhuman primate. Evid. Based Complement. Altern. Med. 2012, 2012. [Google Scholar] [CrossRef] [PubMed]
- Singh, A.P.; Sarkar, S.; Tripathi, M.; Rajender, S. Mucuna pruriens and its major constituent L-DOPA recover spermatogenic loss by combating ROS, loss of mitochondrial membrane potential and apoptosis. PLoS ONE 2013, 8, e54655. [Google Scholar] [CrossRef] [PubMed]
- Yuste, J.E.; Tarragon, E.; Campuzano, C.M.; Ros-Bernal, F. Implications of glial nitric oxide in neurodegenerative diseases. Front. Cell. Neurosci. 2015, 9, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Yadav, S.K.; Prakash, J.; Chouhan, S.; Westfall, S.; Verma, M.; Singh, T.D.; Singh, S.P. Comparison of the neuroprotective potential of Mucuna pruriens seed extract with estrogen in 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP)-induced PD mice model. Neurochem. Int. 2014, 65, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Shimohama, S.; Sawada, H.; Kitamura, Y.; Taniguchi, T. Disease model: Parkinson’s disease. Trends Mol. Med. 2003, 9, 360–365. [Google Scholar] [CrossRef]
- Dauer, W.; Przedborski, S. Parkinson’s Disease: Mechanisms and Models. Neuron 2003, 39, 889–909. [Google Scholar] [CrossRef]
- Muñoz-Soriano, V.; Paricio, N. Drosophila models of Parkinson’s disease: Discovering relevant pathways and novel therapeutic strategies. Parkinson’s Dis. 2011, 2011, 520640. [Google Scholar] [CrossRef] [PubMed]

| Extract | Yield (%; w/w) | L-Dopa Content (%; w/w) |
|---|---|---|
| methanol | 100 | 28.0 |
| hexanes | 3.1 | 0.54 |
| ethyl acetate | 4.7 | 0.03 |
| butanol | 45.3 | 10.05 |
| water | 46.9 | 21.39 |
| Survival (h) | MPP+ | MPP+ + MPE (20 μg/mL) | MPP+ + MPE (40 μg/mL) |
|---|---|---|---|
| Median | 72 | 96 * | 138 *** |
| Maximum | 72 | 132 * | 162 *** |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Johnson, S.L.; Park, H.Y.; DaSilva, N.A.; Vattem, D.A.; Ma, H.; Seeram, N.P. Levodopa-Reduced Mucuna pruriens Seed Extract Shows Neuroprotective Effects against Parkinson’s Disease in Murine Microglia and Human Neuroblastoma Cells, Caenorhabditis elegans, and Drosophila melanogaster. Nutrients 2018, 10, 1139. https://doi.org/10.3390/nu10091139
Johnson SL, Park HY, DaSilva NA, Vattem DA, Ma H, Seeram NP. Levodopa-Reduced Mucuna pruriens Seed Extract Shows Neuroprotective Effects against Parkinson’s Disease in Murine Microglia and Human Neuroblastoma Cells, Caenorhabditis elegans, and Drosophila melanogaster. Nutrients. 2018; 10(9):1139. https://doi.org/10.3390/nu10091139
Chicago/Turabian StyleJohnson, Shelby L., Hyun Y. Park, Nicholas A. DaSilva, Dhiraj A. Vattem, Hang Ma, and Navindra P. Seeram. 2018. "Levodopa-Reduced Mucuna pruriens Seed Extract Shows Neuroprotective Effects against Parkinson’s Disease in Murine Microglia and Human Neuroblastoma Cells, Caenorhabditis elegans, and Drosophila melanogaster" Nutrients 10, no. 9: 1139. https://doi.org/10.3390/nu10091139
APA StyleJohnson, S. L., Park, H. Y., DaSilva, N. A., Vattem, D. A., Ma, H., & Seeram, N. P. (2018). Levodopa-Reduced Mucuna pruriens Seed Extract Shows Neuroprotective Effects against Parkinson’s Disease in Murine Microglia and Human Neuroblastoma Cells, Caenorhabditis elegans, and Drosophila melanogaster. Nutrients, 10(9), 1139. https://doi.org/10.3390/nu10091139






