Dietary Patterns and Cognitive Function among Older Community-Dwelling Adults
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Participants
2.2. Dietary Assessment
2.3. Alternate Mediterranean Diet Score
2.4. Alternate Healthy Eating Index—2010
2.5. Exploratory Factor Analysis of Dietary Nutrients
2.6. Cognitive Function
2.7. Covariate Assessment
2.8. Statistical Analysis
3. Results
3.1. Participant Characteristics
3.2. Dietary Pattern Scores
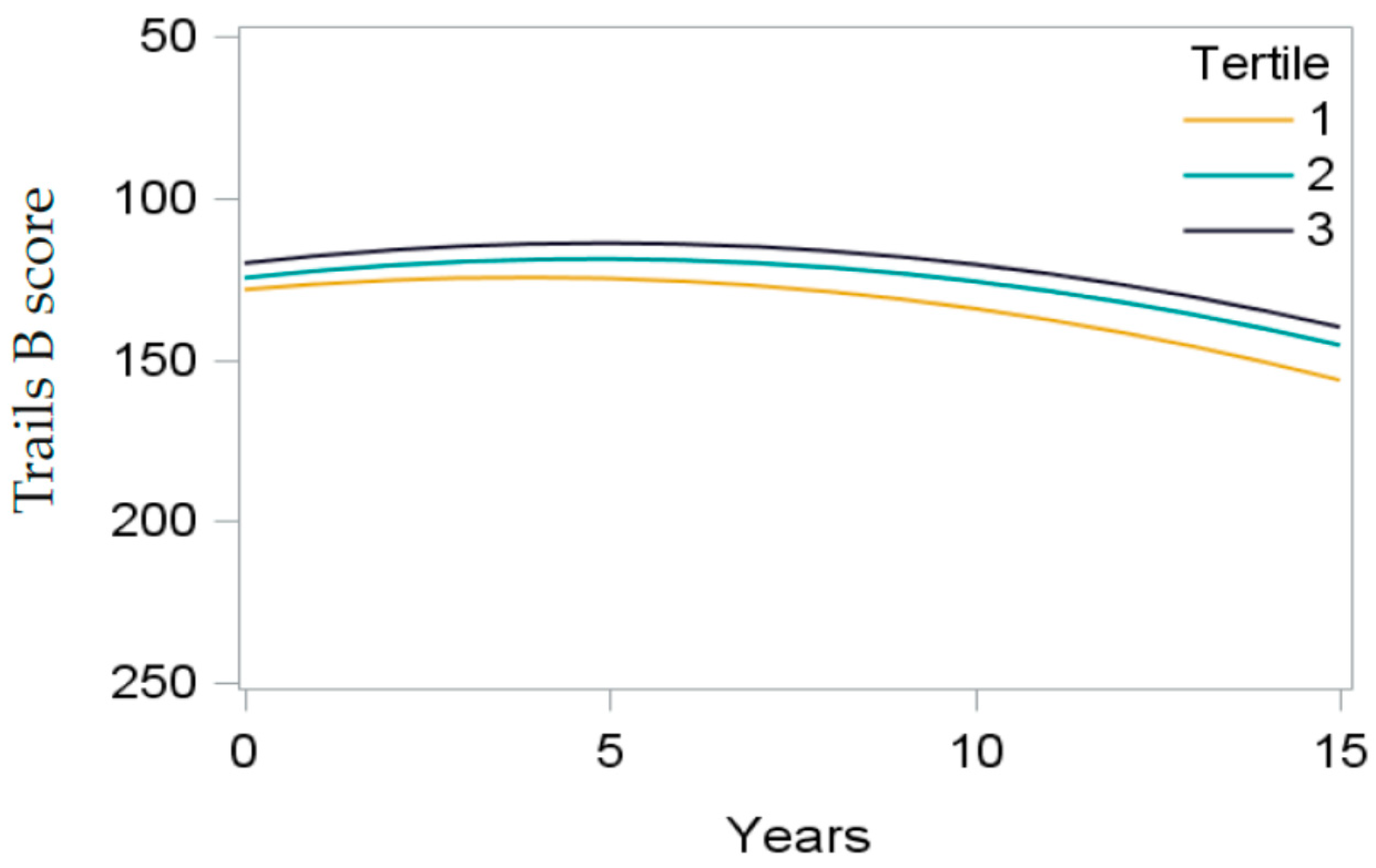
3.3. The aMed Score and Cognitive Function
3.4. The AHEI-2010 Score and Cognitive Function
3.5. Dietary Patterns from Exploratory Factor Analysis and Cognitive Function
3.6. Non-Participant Characteristics
3.7. Sensitivity Analyses
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Shah, H.; Albanese, E.; Duggan, C.; Rudan, I.; Langa, K.M.; Carrillo, M.C.; Chan, K.Y.; Joanette, Y.; Prince, M.; Rossor, M.; et al. Research priorities to reduce the global burden of dementia by 2025. Lancet Neurol. 2016, 15, 1285–1294. [Google Scholar] [CrossRef]
- Gauthier, S.; Albert, M.; Fox, N.; Goedert, M.; Kivipelto, M.; Mestre-Ferrandiz, J.; Middleton, L.T. Why has therapy development for dementia failed in the last two decades? Alzheimer’s Dement. 2016, 12, 60–64. [Google Scholar] [CrossRef] [PubMed]
- Rathod, R.; Kale, A.; Joshi, S. Novel insights into the effect of vitamin B12 and omega-3 fatty acids on brain function. J. Biomed. Sci. 2016, 23, 17. [Google Scholar] [CrossRef] [PubMed]
- Koh, F.; Charlton, K.; Walton, K.; McMahon, A.-T. Role of Dietary Protein and Thiamine Intakes on Cognitive Function in Healthy Older People: A Systematic Review. Nutrients 2015, 7, 2415–2439. [Google Scholar] [CrossRef] [PubMed]
- Solfrizzi, V.; Custodero, C.; Lozupone, M.; Imbimbo, B.P.; Valiani, V.; Agosti, P.; Schilardi, A.; D’Introno, A.; La Montagna, M.; Calvani, M.; et al. Relationships of Dietary Patterns, Foods, and Micro- and Macronutrients with Alzheimer’s Disease and Late-Life Cognitive Disorders: A Systematic Review. J. Alzheimer’s Dis. 2017, 59, 815–849. [Google Scholar] [CrossRef] [PubMed]
- van de Rest, O.; Berendsen, A.A.; Haveman-Nies, A.; de Groot, L.C. Dietary Patterns, Cognitive Decline, and Dementia: A Systematic Review. Adv. Nutr. 2015, 6, 154–168. [Google Scholar] [CrossRef] [PubMed]
- Trichopoulou, A.; Kyrozis, A.; Rossi, M.; Katsoulis, M.; Trichopoulos, D.; La Vecchia, C.; Lagiou, P. Mediterranean diet and cognitive decline over time in an elderly Mediterranean population. Eur. J. Nutr. 2015, 54, 1311–1321. [Google Scholar] [CrossRef] [PubMed]
- Qin, B.; Adair, L.S.; Plassman, B.L.; Batis, C.; Edwards, L.J.; Popkin, B.M.; Mendez, M.A. Dietary Patterns and Cognitive Decline among Chinese Older Adults. Epidemiology 2015, 26, 758–768. [Google Scholar] [CrossRef] [PubMed]
- Galbete, C.; Toledo, E.; Toledo, J.B.; Bes-Rastrollo, M.; Buil-Cosiales, P.; Marti, A.; Guillén-Grima, F.; Martínez-González, M.A. Mediterranean diet and cognitive function: The sun project. J. Nutr. Health Aging 2015, 19, 305–312. [Google Scholar] [CrossRef] [PubMed]
- Haring, B.; Wu, C.; Mossavar-Rahmani, Y.; Snetselaar, L.; Brunner, R.; Wallace, R.B.; Neuhouser, M.L.; Wassertheil-Smoller, S. No Association between Dietary Patterns and Risk for Cognitive Decline in Older Women with 9-Year Follow-Up: Data from the Women’s Health Initiative Memory Study. J. Acad. Nutr. Diet. 2016, 116, 921–930. [Google Scholar] [CrossRef] [PubMed]
- Loughrey, D.G.; Lavecchia, S.; Brennan, S.; Lawlor, B.A.; Kelly, M.E. The Impact of the Mediterranean Diet on the Cognitive Functioning of Healthy Older Adults: A Systematic Review and Meta-Analysis. Adv. Nutr. 2017, 8, 571–586. [Google Scholar] [CrossRef] [PubMed]
- McCullough, M.L.; Feskanich, D.; Stampfer, M.J.; Giovannucci, E.L.; Rimm, E.B.; Hu, F.B.; Spiegelman, D.; Hunter, D.J.; Colditz, G.A.; Willett, W.C. Diet quality and major chronic disease risk in men and women: Moving toward improved dietary guidance. Am. J. Clin. Nutr. 2002, 76, 1261–1271. [Google Scholar] [CrossRef] [PubMed]
- Smyth, A.; Dehghan, M.; O’Donnell, M.; Anderson, C.; Teo, K.; Gao, P.; Sleight, P.; Dagenais, G.; Probstfield, J.L.; Mente, A.; et al. ONTARGET and TRANSCEND Investigators Healthy eating and reduced risk of cognitive decline: A cohort from 40 countries. Neurology 2015, 84, 2258–2265. [Google Scholar] [CrossRef] [PubMed]
- Ye, X.; Scott, T.; Gao, X.; Maras, J.E.; Bakun, P.J.; Tucker, K.L. Mediterranean Diet, Healthy Eating Index 2005, and Cognitive Function in Middle-Aged and Older Puerto Rican Adults. J. Acad. Nutr. Diet. 2013, 113, 276–281. [Google Scholar] [CrossRef] [PubMed]
- Tangney, C.C.; Kwasny, M.J.; Li, H.; Wilson, R.S.; Evans, D.A.; Morris, M.C. Adherence to a Mediterranean-type dietary pattern and cognitive decline in a community population. Am. J. Clin. Nutr. 2011, 93, 601–607. [Google Scholar] [CrossRef] [PubMed]
- Samieri, C.; Sun, Q.; Townsend, M.K.; Chiuve, S.E.; Okereke, O.I.; Willett, W.C.; Stampfer, M.; Grodstein, F. The relation of midlife diet to healthy aging: A cohort study. Ann. Intern. Med. 2013, 159, 584–591. [Google Scholar] [CrossRef] [PubMed]
- Reedy, J.; Wirfält, E.; Flood, A.; Mitrou, P.N.; Krebs-Smith, S.M.; Kipnis, V.; Midthune, D.; Leitzmann, M.; Hollenbeck, A.; Schatzkin, A.; et al. Comparing 3 dietary pattern methods-cluster analysis, factor analysis, and index analysis-with colorectal cancer risk. Am. J. Epidemiol. 2010, 171, 479–487. [Google Scholar] [CrossRef] [PubMed]
- Moskal, A.; Pisa, P.T.; Ferrari, P.; Byrnes, G.; Freisling, H.; Boutron-Ruault, M.-C.; Cadeau, C.; Nailler, L.; Wendt, A.; Kühn, T.; et al. Nutrient patterns and their food sources in an International Study Setting: Report from the EPIC study. PLoS ONE 2014, 9, e98647. [Google Scholar] [CrossRef] [PubMed]
- Trichopoulou, A.; Costacou, T.; Bamia, C.; Trichopoulos, D. Adherence to a Mediterranean diet and survival in a Greek population. N. Engl. J. Med. 2003, 348, 2599–2608. [Google Scholar] [CrossRef] [PubMed]
- Rimm, E.B.; Giovannucci, E.L.; Stampfer, M.J.; Colditz, G.A.; Litin, L.B.; Willett, W.C. Reproducibility and validity of an expanded self-administered semiquantitative food frequency questionnaire among male health professionals. Am. J. Epidemiol. 1992, 135, 1114–1126. [Google Scholar] [CrossRef] [PubMed]
- Litin, L.; Sacks, F. Trans-Fatty-Acid Content of Common Foods. N. Engl. J. Med. 1993, 329, 1969–1970. [Google Scholar] [CrossRef] [PubMed]
- Enig, M.G.; Pallansch, L.; Sampugna, J.; Keeney, M. Fatty acid composition of the fat in selected food items with emphasis on trans components. J. Am. Oil Chem. Soc. 1983, 60, 1788–1795. [Google Scholar] [CrossRef]
- Slover, H.T.; Thompson, R.H.; Davis, C.S.; Merola, G.V. Lipids in margarines and margarine-like foods. J. Am. Oil Chem. Soc. 1985, 62, 775–786. [Google Scholar] [CrossRef]
- Franz, M.; Sampson, L. Challenges in developing a whole grain database: Definitions, methods and quantification. J. Food Compos. Anal. 2006, 19, S38–S44. [Google Scholar] [CrossRef]
- Friday, J.E.; Bowman, S.A. MyPyramid Equivalents Database for USDA Survey Food Codes, 1994–2002, Version 1.0. U.S. Department of Agriculture, Agriculture Research Service, 2006. Available online: https://www.ars.usda.gov/research/publications/publication/?seqNo115=201058 (accessed on 23 August 2017).
- Fung, T.T.; Rexrode, K.M.; Mantzoros, C.S.; Manson, J.E.; Willett, W.C.; Hu, F.B. Mediterranean diet and incidence of and mortality from coronary heart disease and stroke in women. Circulation 2009, 119, 1093–1100. [Google Scholar] [CrossRef] [PubMed]
- Chiuve, S.E.; Fung, T.T.; Rimm, E.B.; Hu, F.B.; McCullough, M.L.; Wang, M.; Stampfer, M.J.; Willett, W.C. Alternative dietary indices both strongly predict risk of chronic disease. J. Nutr. 2012, 142, 1009–1018. [Google Scholar] [CrossRef] [PubMed]
- Willett, W.; Stampfer, M.J. Total energy intake: Implications for epidemiologic analyses. Am. J. Epidemiol. 1986, 124, 17–27. [Google Scholar] [CrossRef] [PubMed]
- Ri, K. Determining the dimensionality of data: A SAS macro for parallel analysis. In Proceedings of the Twenty Eighth Annual SAS Users Group International (SUGI 28), Seattle, WA, USA, 30 March 2003. [Google Scholar]
- Tombaugh, T.N.; McIntyre, N.J. The Mini-Mental State Examination: A comprehensive review. J. Am. Geriatr. Soc. 1992, 40, 922–935. [Google Scholar] [CrossRef] [PubMed]
- Reitan, R.M. Validity of the trail making test as an indicator of organic brain damage. Percept. Mot. Skills 1958, 8, 271–276. [Google Scholar] [CrossRef]
- Borkowski, J.G.; Benton, A.L.; Spreen, O. Word fluency and brain damage. Neuropsychologia 1967, 5, 135–140. [Google Scholar] [CrossRef]
- Buschke, H.; Fuld, P.A. Evaluating storage, retention, and retrieval in disordered memory and learning. Neurology 1974, 24, 1019–1025. [Google Scholar] [CrossRef] [PubMed]
- Reas, E.T.; Laughlin, G.A.; Bergstrom, J.; Kritz-Silverstein, D.; Barrett-Connor, E.; McEvoy, L.K. Effects of Sex and Education on Cognitive Change Over a 27-Year Period in Older Adults: The Rancho Bernardo Study. Am. J. Geriatr. Psychiatry 2017, 25, 889–899. [Google Scholar] [CrossRef] [PubMed]
- The Hypertension Detection and Follow-up Program. Prev. Med. 1976, 5, 207–215. [CrossRef]
- Alberti, K.G.; Zimmet, P.Z. Definition, diagnosis and classification of diabetes mellitus and its complications. Part 1: Diagnosis and classification of diabetes mellitus provisional report of a WHO consultation. Diabet. Med. 1998, 15, 539–553. [Google Scholar] [CrossRef]
- Storey, J.D. A direct approach to false discovery rates. J. R. Stat. Soc. Series. B Stat. Methodol. 2002, 64, 479–498. [Google Scholar] [CrossRef]
- Samieri, C.; Okereke, O.I.; Devore, E.E.; Grodstein, F. Long-term adherence to the Mediterranean diet is associated with overall cognitive status, but not cognitive decline, in women. J. Nutr. 2013, 143, 493–499. [Google Scholar] [CrossRef] [PubMed]
- Hardman, R.J.; Kennedy, G.; Macpherson, H.; Scholey, A.B.; Pipingas, A. Adherence to a Mediterranean-Style Diet and Effects on Cognition in Adults: A Qualitative Evaluation and Systematic Review of Longitudinal and Prospective Trials. Front. Nutr. 2016, 3. [Google Scholar] [CrossRef] [PubMed]
- Aridi, Y.; Walker, J.; Wright, O. The Association between the Mediterranean Dietary Pattern and Cognitive Health: A Systematic Review. Nutrients 2017, 9, 674. [Google Scholar] [CrossRef] [PubMed]
- Solfrizzi, V.; Capurso, C.; Panza, F. Adherence to a Mediterranean dietary pattern and risk of Alzheimer’s disease. Ann. Neurol. 2006, 60, 620. [Google Scholar] [CrossRef] [PubMed]
- Ministry of Health and Welfare Supreme Scientific Health Council of Greece. Dietary Guidelines for Adults in Greece. Available online: http://www.mednet.gr/archives/1999-5/pdf/516.pdf (accessed on 28 June 2017).
- Esposito, K.; Marfella, R.; Ciotola, M.; Di Palo, C.; Giugliano, F.; Giugliano, G.; D’Armiento, M.; D’Andrea, F.; Giugliano, D. Effect of a mediterranean-style diet on endothelial dysfunction and markers of vascular inflammation in the metabolic syndrome: A randomized trial. JAMA 2004, 292, 1440–1446. [Google Scholar] [CrossRef] [PubMed]
- Dai, J.; Jones, D.P.; Goldberg, J.; Ziegler, T.R.; Bostick, R.M.; Wilson, P.W.; Manatunga, A.K.; Shallenberger, L.; Jones, L.; Vaccarino, V. Association between adherence to the Mediterranean diet and oxidative stress. Am. J. Clin. Nutr. 2008, 88, 1364–1370. [Google Scholar] [PubMed]
- Widmer, R.J.; Flammer, A.J.; Lerman, L.O.; Lerman, A. The Mediterranean diet, its components, and cardiovascular disease. Am. J. Med. 2015, 128, 229–238. [Google Scholar] [CrossRef] [PubMed]
- Panagiotakos, D.B.; Pitsavos, C.; Stefanadis, C. Dietary patterns: A Mediterranean diet score and its relation to clinical and biological markers of cardiovascular disease risk. Nutr. Metab. Cardiovasc. Dis. 2006, 16, 559–568. [Google Scholar] [CrossRef] [PubMed]
- Rush, T.M.; Kritz-Silverstein, D.; Laughlin, G.A.; Fung, T.T.; Barrett-Connor, E.; McEvoy, L.K. Association between dietary sodium intake and cognitive function in older adults. J. Nutr. Health Aging 2017, 21, 276–283. [Google Scholar] [CrossRef] [PubMed]
- Thomas, J.; Thomas, C.J.; Radcliffe, J.; Itsiopoulos, C. Omega-3 Fatty Acids in Early Prevention of Inflammatory Neurodegenerative Disease: A Focus on Alzheimer’s Disease. Biomed Res. Int. 2015, 2015. [Google Scholar] [CrossRef] [PubMed]
- Kotani, S.; Sakaguchi, E.; Warashina, S.; Matsukawa, N.; Ishikura, Y.; Kiso, Y.; Sakakibara, M.; Yoshimoto, T.; Guo, J.; Yamashima, T. Dietary supplementation of arachidonic and docosahexaenoic acids improves cognitive dysfunction. Neurosci. Res. 2006, 56, 159–164. [Google Scholar] [CrossRef] [PubMed]
- Cardoso, C.; Afonso, C.; Bandarra, N.M. Dietary DHA and health: Cognitive function ageing. Nutr. Res. Rev. 2016, 29, 281–294. [Google Scholar] [CrossRef] [PubMed]
- Bazinet, R.P.; Layé, S. Polyunsaturated fatty acids and their metabolites in brain function and disease. Nat. Rev. Neurosci. 2014, 15, 771–785. [Google Scholar] [CrossRef] [PubMed]
- Shahidi, F.; Ambigaipalan, P. Omega-3 polyunsaturated fatty acids and their health benefits. Annu. Rev. Food Sci. Technol. 2018, 9, 345–381. [Google Scholar] [CrossRef] [PubMed]
- Baierle, M.; Vencato, P.; Oldenburg, L.; Bordignon, S.; Zibetti, M.; Trentini, C.; Duarte, M.; Veit, J.; Somacal, S.; Emanuelli, T.; et al. Fatty Acid Status and Its Relationship to Cognitive Decline and Homocysteine Levels in the Elderly. Nutrients 2014, 6, 3624–3640. [Google Scholar] [CrossRef] [PubMed]
- Laitinen, M.H.; Ngandu, T.; Rovio, S.; Helkala, E.-L.; Uusitalo, U.; Viitanen, M.; Nissinen, A.; Tuomilehto, J.; Soininen, H.; Kivipelto, M. Fat intake at midlife and risk of dementia and Alzheimer’s disease: A population-based study. Dement. Geriatr. Cogn. Disord. 2006, 22, 99–107. [Google Scholar] [CrossRef] [PubMed]
- Zamroziewicz, M.K.; Paul, E.J.; Zwilling, C.E.; Barbey, A.K. Predictors of Memory in Healthy Aging: Polyunsaturated Fatty Acid Balance and Fornix White Matter Integrity. Aging Dis. 2017, 8, 372–383. [Google Scholar] [CrossRef] [PubMed]
- Zamroziewicz, M.K.; Paul, E.J.; Zwilling, C.E.; Barbey, A.K. Determinants of fluid intelligence in healthy aging: Omega-3 polyunsaturated fatty acid status and frontoparietal cortex structure. Nutr. Neurosci. 2017, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Gu, Y.; Vorburger, R.S.; Gazes, Y.; Habeck, C.G.; Stern, Y.; Luchsinger, J.A.; Manly, J.J.; Schupf, N.; Mayeux, R.; Brickman, A.M. White matter integrity as a mediator in the relationship between dietary nutrients and cognition in the elderly. Ann. Neurol. 2016, 79, 1014–1025. [Google Scholar] [CrossRef] [PubMed]
- Raederstorff, D.; Wyss, A.; Calder, P.C.; Weber, P.; Eggersdorfer, M. Vitamin E function and requirements in relation to PUFA. Br. J. Nutr. 2015, 114, 1113–1122. [Google Scholar] [CrossRef] [PubMed]
- Fata, G.; Weber, P.; Mohajeri, M. Effects of Vitamin E on Cognitive Performance during Ageing and in Alzheimer’s Disease. Nutrients 2014, 6, 5453–5472. [Google Scholar] [CrossRef] [PubMed]
- Perkins, A.J.; Hendrie, H.C.; Callahan, C.M.; Gao, S.; Unverzagt, F.W.; Xu, Y.; Hall, K.S.; Hui, S.L. Association of antioxidants with memory in a multiethnic elderly sample using the Third National Health and Nutrition Examination Survey. Am. J. Epidemiol. 1999, 150, 37–44. [Google Scholar] [CrossRef] [PubMed]
- Dong, Y.; Chen, X.; Liu, Y.; Shu, Y.; Chen, T.; Xu, L.; Li, M.; Guan, X. Do low-serum vitamin E levels increase the risk of Alzheimer disease in older people? Evidence from a meta-analysis of case-control studies. Int. J. Geriatr. Psychiatry 2018, 33, e257–e263. [Google Scholar] [CrossRef] [PubMed]
- Kryscio, R.J.; Abner, E.L.; Caban-Holt, A.; Lovell, M.; Goodman, P.; Darke, A.K.; Yee, M.; Crowley, J.; Schmitt, F.A. Association of Antioxidant Supplement Use and Dementia in the Prevention of Alzheimer’s Disease by Vitamin E and Selenium Trial (PREADViSE). JAMA Neurol. 2017, 74, 567. [Google Scholar] [CrossRef] [PubMed]
- Farina, N.; Llewellyn, D.; Isaac, M.G.E.K.N.; Tabet, N. Vitamin E for Alzheimer’s dementia and mild cognitive impairment. Cochrane Database Syst. Rev. 2017, 4, CD002854. [Google Scholar] [CrossRef]
- Ye, X.; Gao, X.; Scott, T.; Tucker, K.L. Habitual sugar intake and cognitive function among middle-aged and older Puerto Ricans without diabetes. Br. J. Nutr. 2011, 106, 1423–1432. [Google Scholar] [CrossRef] [PubMed]
- Simeon, V.; Chiodini, P.; Mattiello, A.; Sieri, S.; Panico, C.; Brighenti, F.; Krogh, V.; Panico, S. Dietary glycemic load and risk of cognitive impairment in women: Findings from the EPIC-Naples cohort. Eur. J. Epidemiol. 2015, 30, 425–433. [Google Scholar] [CrossRef] [PubMed]
- Young, H.; Benton, D. The glycemic load of meals, cognition and mood in middle and older aged adults with differences in glucose tolerance: A randomized trial. ESPEN. J. 2014, 9, e147–e154. [Google Scholar] [CrossRef]
- Benton, D.; Ruffin, M.-P.; Lassel, T.; Nabb, S.; Messaoudi, M.; Vinoy, S.; Desor, D.; Lang, V. The delivery rate of dietary carbohydrates affects cognitive performance in both rats and humans. Psychopharmacology 2003, 166, 86–90. [Google Scholar] [CrossRef] [PubMed]
- Seneff, S.; Wainwright, G.; Mascitelli, L. Is the metabolic syndrome caused by a high fructose, and relatively low fat, low cholesterol diet? Arch. Med. Sci. 2011, 1, 8–20. [Google Scholar] [CrossRef] [PubMed]
- Yaffe, K.; Blackwell, T.; Kanaya, A.M.; Davidowitz, N.; Barrett-Connor, E.; Krueger, K. Diabetes, impaired fasting glucose, and development of cognitive impairment in older women. Neurology 2004, 63, 658–663. [Google Scholar] [CrossRef] [PubMed]
- Laterra, J.; Keep, R.; Betz, A.L.; Goldstein, G.W. Blood-brain-cerebrospinal fluid barriers. In Basic Neurochemistry: Molecular, Cellular, and Medical Aspects, 5th ed.; Siegel, G.J., Ed.; Raven Press: New York, NY, USA, 1994; pp. 681–698. [Google Scholar]
- Roberts, R.O.; Roberts, L.A.; Geda, Y.E.; Cha, R.H.; Pankratz, V.S.; O’Connor, H.M.; Knopman, D.S.; Petersen, R.C. Relative intake of macronutrients impacts risk of mild cognitive impairment or dementia. J. Alzheimer’s Dis. 2012, 32, 329–339. [Google Scholar] [CrossRef] [PubMed]
- van de Rest, O.; van der Zwaluw, N.L.; de Groot, L.C.P.G.M. Literature review on the role of dietary protein and amino acids in cognitive functioning and cognitive decline. Amino Acids 2013, 45, 1035–1045. [Google Scholar] [CrossRef] [PubMed]
- Steinbach, S.; Hundt, W.; Vaitl, A.; Heinrich, P.; Förster, S.; Bürger, K.; Zahnert, T. Taste in mild cognitive impairment and Alzheimer’s disease. J. Neurol. 2010, 257, 238–246. [Google Scholar] [CrossRef] [PubMed]
- Schiffman, S.S. Perception of taste and smell in elderly persons. Crit. Rev. Food Sci. Nutr. 1993, 33, 17–26. [Google Scholar] [CrossRef] [PubMed]
- Doty, R.L. Olfactory capacities in aging and Alzheimer’s disease. Psychophysical and anatomic considerations. Ann. N. Y. Acad. Sci. 1991, 640, 20–27. [Google Scholar] [CrossRef] [PubMed]
- Mungas, D.; Cooper, J.K.; Weiler, P.G.; Gietzen, D.; Franzi, C.; Bernick, C. Dietary Preference for Sweet Foods in Patients with Dementia. J. Am. Geriatr. Soc. 1990, 38, 999–1007. [Google Scholar] [CrossRef] [PubMed]
- Sakai, M.; Kazui, H.; Shigenobu, K.; Komori, K.; Ikeda, M.; Nishikawa, T. Gustatory Dysfunction as an Early Symptom of Semantic Dementia. Dement. Geriatr. Cogn. Dis. Extra 2017, 7, 395–405. [Google Scholar] [CrossRef] [PubMed]
- Feskanich, D.; Rimm, E.B.; Giovannucci, E.L.; Colditz, G.A.; Stampfer, M.J.; Litin, L.B.; Willett, W.C. Reproducibility and validity of food intake measurements from a semiquantitative food frequency questionnaire. J. Am. Diet. Assoc. 1993, 93, 790–796. [Google Scholar] [CrossRef]
- Lopez, L.B.; Kritz-Silverstein, D.; Barrett Connor, E. High dietary and plasma levels of the omega-3 fatty acid docosahexaenoic acid are associated with decreased dementia risk: The Rancho Bernardo study. J. Nutr. Health Aging 2011, 15, 25–31. [Google Scholar] [CrossRef] [PubMed]
- Harris, P.A.; Taylor, R.; Thielke, R.; Payne, J.; Gonzalez, N.; Conde, J.G. Research electronic data capture (REDCap)—A metadata-driven methodology and workflow process for providing translational research informatics support. J. Biomed. Inf. 2009, 42, 377–381. [Google Scholar] [CrossRef] [PubMed]
| Fortified Cereals Factor | Fruit/Vegetable Factor | Animal Fat/Vit B12 Factor | Dairy Factor | Plant PUFA/Vit E Factor | Sugar/Low Protein Factor | |
|---|---|---|---|---|---|---|
| Vit B1 (Thiamine) | 0.86 | – | – | – | – | – |
| Iron | 0.84 | – | – | – | – | – |
| Vit B6 | 0.73 | 0.44 | – | – | – | – |
| Folate | 0.69 | 0.50 | – | – | – | – |
| Zinc | 0.66 | – | 0.42 | – | – | −0.22 |
| Crude fiber | 0.34 | 0.78 | – | – | – | – |
| Beta-carotene | – | 0.78 | – | – | – | – |
| Vit C | – | 0.73 | – | – | – | 0.26 |
| Dietary Cholesterol | – | – | 0.91 | – | – | |
| Arachidonic acid 20:4 (ω-6) | – | – | 0.84 | – | – | −0.20 |
| Vit B12 | 0.31 | – | 0.66 | 0.28 | – | – |
| Saturated Fat | – | −0.46 | 0.57 | – | 0.42 | – |
| Lactose | – | – | – | 0.94 | – | – |
| Calcium | – | – | – | 0.92 | – | – |
| Vit D | – | – | – | 0.81 | – | – |
| Linoleic acid 18:2 (ω-6) | – | – | – | – | 0.90 | – |
| Alpha-linolenic acid 18:3 (ω-3) | – | – | – | – | 0.87 | – |
| Vit E | 0.49 | – | – | – | 0.57 | – |
| Sucrose | – | – | – | – | – | 0.85 |
| Fructose | – | 0.53 | – | – | −0.21 | 0.60 |
| Protein | – | 0.53 | 0.25 | – | −0.65 | |
| Variance explained (%) | 16.7 | 13.8 | 13.3 | 12.8 | 10.5 | 8.6 |
| Female | Mean Age | Some College | Current Smoker | Exercise ≥3x/Wk | Alcohol Daily | Fair/Poor Health a | Energy Intake | BMI | ||
|---|---|---|---|---|---|---|---|---|---|---|
| n | % | Years (SD) | % | % | % | % | % | kcal/day | kg/m2 | |
| aMed diet score | ||||||||||
| tertile 1 | 525 | 59.4 | 72.7 (9.2) | 65.1 | 12.2 | 62.0 | 37.2 | 5.0 | 1448 (473) | 25.5 (4.0) |
| tertile 2 | 576 | 57.3 | 73.5 (9.6) | 71.9 | 8.7 | 71.7 | 35.4 | 3.3 | 1667 (504) | 25.2 (4.0) |
| tertile 3 | 398 | 58.3 | 73.6 (8.6) | 71.6 | 5.3 | 80.1 | 38.9 | 3.5 | 1868 (523) | 24.8 (3.4) |
| p-value | 0.77 | 0.22 | 0.03 | <0.001 | <0.001 | 0.53 | 0.33 | <0.001 | 0.02 | |
| AHEI-2010 score | ||||||||||
| tertile 1 | 498 | 57.4 | 73.1 (9.4) | 65.1 | 13.3 | 61.2 | 35.9 | 5.2 | 1674 (524) | 25.3 (4.0) |
| tertile 2 | 507 | 59.0 | 73.6 (9.0) | 71.2 | 9.3 | 73.6 | 37.4 | 3.2 | 1613 (519) | 25.3 (4.1) |
| tertile 3 | 494 | 58.5 | 73.0 (9.1) | 72.1 | 4.5 | 76.8 | 37.7 | 3.4 | 1645 (530) | 24.9 (3.4) |
| p-value | 0.88 | 0.51 | 0.03 | <0.001 | <0.001 | 0.84 | 0.19 | 0.19 | 0.18 | |
| Plant PUFA/vit E factor | ||||||||||
| tertile 1 | 496 | 58.3 | 73.6 (8.9) | 67.7 | 7.9 | 68.8 | 30.8 | 3.8 | 1580 (622) | 25.2 (3.9) |
| tertile 2 | 500 | 58.4 | 72.9 (9.4) | 69.4 | 9.8 | 73.6 | 39.7 | 4.2 | 1701 (503) | 25.4 (4.1) |
| tertile 3 | 503 | 58.3 | 73.2 (9.2) | 71.2 | 9.3 | 69.2 | 40.4 | 3.8 | 1650 (425) | 24.9 (3.6) |
| p-value | 0.99 | 0.50 | 0.5 | 0.54 | 0.17 | <0.001 | 0.93 | <0.001 | 0.15 | |
| Sugar/low protein factor | ||||||||||
| tertile 1 | 498 | 58.2 | 70.8 (8.5) | 74.3 | 10.8 | 71.9 | 43.6 | 3.0 | 1618 (580) | 25.3 (3.8) |
| tertile 2 | 498 | 58.2 | 73.5 (9.2) | 69.5 | 9 | 69.9 | 33.1 | 3.8 | 1617 (515) | 25.4 (4.0) |
| tertile 3 | 503 | 58.4 | 75.4 (9.3) | 64.6 | 7.2 | 69.9 | 34.3 | 5.0 | 1696 (471) | 24.9 (3.7) |
| p-value | 0.99 | <0.001 | <0.001 | 0.13 | 0.73 | <0.001 | 0.28 | 0.02 | 0.06 | |
| MMSE | Trails B | Verbal Fluency | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Base Model a | Full Model b | Base Model a | Full Model b | Base Model a | Full Model b | |||||||
| Beta | 95% CI | Beta | 95% CI | Beta | 95% CI | Beta | 95% CI | Beta | 95% CI | Beta | 95% CI | |
| aMed score | ||||||||||||
| tertile 2 | 0.22 | (0.03, 0.41) | 0.19 | (−0.006, 0.38) | −6.10 | (−7.85, −0.89) | −4.92 | (−10.08, 0.25) | 0.31 | (−0.12, 0.75) | 0.22 | (−0.18, 0.62) |
| tertile 3 | 0.39 | (0.17, 0.60) | 0.33 | (0.11, 0.55) | −5.28 | (−11.11, 0.55) | −2.77 | (−8.63, 3.01) | 0.56 | (0.06, 1.04) | 0.51 | (0.06, 0.97) |
| p-value | 0.002 * | 0.01 * | 0.05 | 0.18 | 0.08 | 0.14 | ||||||
| p-trend | 0.0003 * | 0.002 * | 0.06 | 0.32 | 0.02 * | 0.03 | ||||||
| AHEI-2010 score | ||||||||||||
| tertile 2 | 0.22 | (0.0, 0.40) | 0.18 | (−0.02, 0.37) | −4.33 | (−9.70, 1.04) | −2.35 | (−7.72, 3.02) | 0.64 | (0.23, 1.05) | 0.46 | (−0.01, 0.92) |
| tertile 3 | 0.18 | (0.003, 0.37) | 0.11 | (−0.09, 0.31) | −3.06 | (−8.45, 2.33) | −0.48 | (−5.91, 4.95) | 0.55 | (0.13, 0.96) | 0.40 | (−0.05, 0.86) |
| p-value | 0.04 * | 0.22 | 0.27 | 0.66 | 0.01 * | 0.10 | ||||||
| p-trend | 0.13 | 0.17 | 0.80 | 0.85 | 0.01 * | 0.09 | ||||||
| Plant PUFA/vit E | ||||||||||||
| tertile 2 | 0.007 | (−0.20, 0.20) | −0.02 | (−0.22, 0.18) | −3.90 | (−9.31, 1.57) | −3.22 | (−8.6, 2.6) | 0.45 | (−0.01, 0.90) | 0.43 | (−0.02, 0.88) |
| tertile 3 | 0.23 | (0.02, 0.42) | 0.22 | (0.02, 0.42) | −8.29 | (−13.70, −2.89) | −7.85 | (−13.2, −2.47) | 0.31 | (−0.15, 0.76) | 0.29 | (−0.16, 0.75) |
| p-value | 0.04 | 0.03 | 0.01 * | 0.02 * | 0.15 | 0.17 | ||||||
| p-trend | 0.01 * | 0.01 * | 0.003 * | 0.005 * | 0.17 | 0.19 | ||||||
| Sugar/low protein | ||||||||||||
| tertile 2 | −0.04 | (−0.24, 0.16) | −0.03 | (−0.23, 0.17) | 2.13 | (−3.25, 7.51) | 1.34 | (−4.02, 6.71) | 0.04 | (−0.41, 0.49) | 0.05 | (−0.40, 0.50) |
| tertile 3 | −0.30 | (−0.50, −0.10) | −0.29 | (−0.49, −0.09) | 6.94 | (1.47, 12.41) | 6.22 | (0.76, 11.68) | −0.60 | (−1.05, −0.13) | −0.57 | (−1.03, −0.11) |
| p-value | 0.007 * | 0.008 * | 0.04 | 0.06 | 0.01 * | 0.01 * | ||||||
| p-trend | 0.007 * | 0.01 * | 0.01 * | 0.03 | 0.03 | 0.04 | ||||||
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Richard, E.L.; Laughlin, G.A.; Kritz-Silverstein, D.; Reas, E.T.; Barrett-Connor, E.; McEvoy, L.K. Dietary Patterns and Cognitive Function among Older Community-Dwelling Adults. Nutrients 2018, 10, 1088. https://doi.org/10.3390/nu10081088
Richard EL, Laughlin GA, Kritz-Silverstein D, Reas ET, Barrett-Connor E, McEvoy LK. Dietary Patterns and Cognitive Function among Older Community-Dwelling Adults. Nutrients. 2018; 10(8):1088. https://doi.org/10.3390/nu10081088
Chicago/Turabian StyleRichard, Erin L., Gail A. Laughlin, Donna Kritz-Silverstein, Emilie T. Reas, Elizabeth Barrett-Connor, and Linda K. McEvoy. 2018. "Dietary Patterns and Cognitive Function among Older Community-Dwelling Adults" Nutrients 10, no. 8: 1088. https://doi.org/10.3390/nu10081088
APA StyleRichard, E. L., Laughlin, G. A., Kritz-Silverstein, D., Reas, E. T., Barrett-Connor, E., & McEvoy, L. K. (2018). Dietary Patterns and Cognitive Function among Older Community-Dwelling Adults. Nutrients, 10(8), 1088. https://doi.org/10.3390/nu10081088





