Dietary Flavonoids and the Risk of Colorectal Cancer: An Updated Meta-Analysis of Epidemiological Studies
Abstract
1. Introduction
2. Methods
2.1. Search Strategy
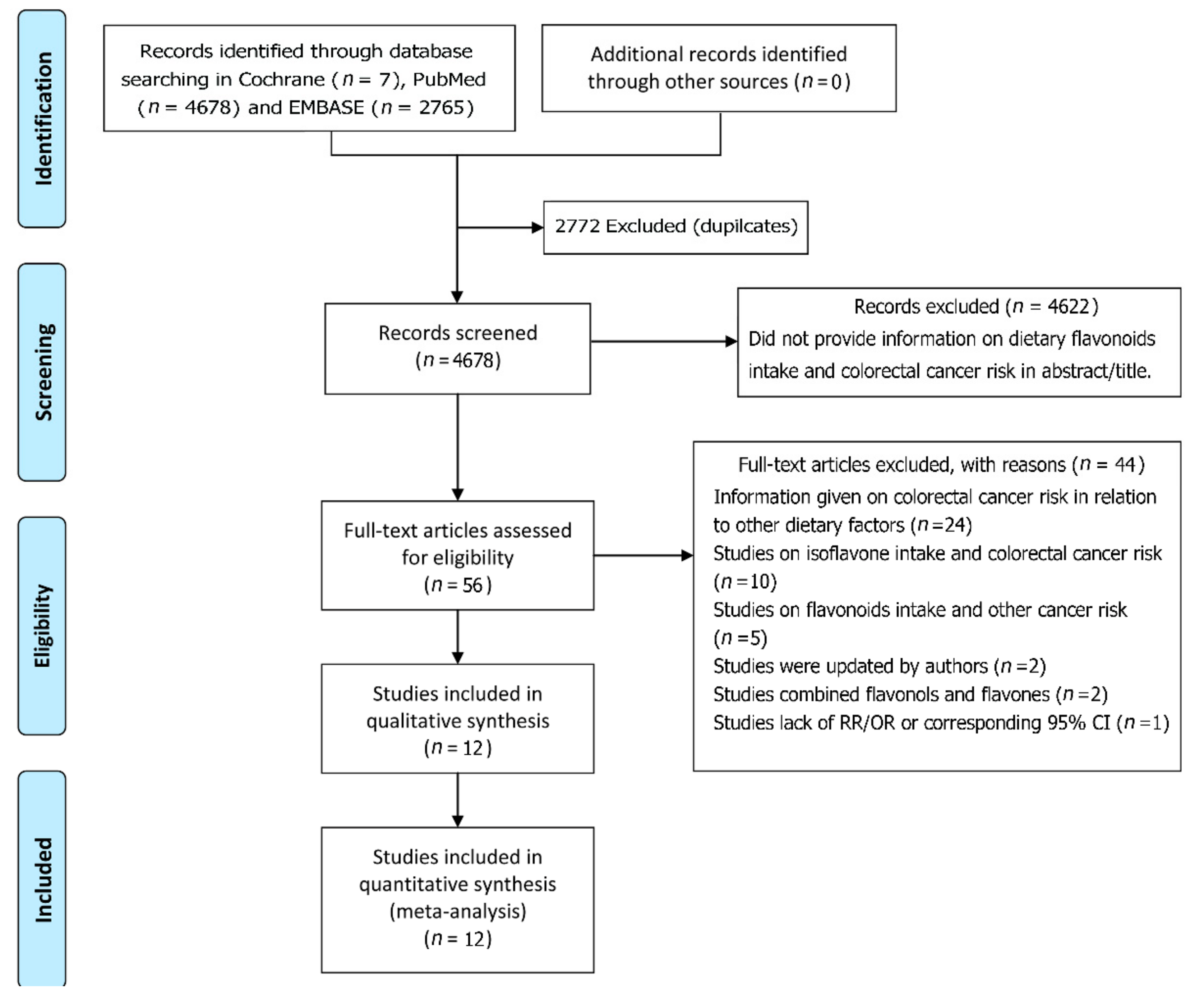
2.2. Study Selection
2.3. Data Extraction
2.4. Statistical Analysis
3. Results
3.1. Characteristics of the Included Studies
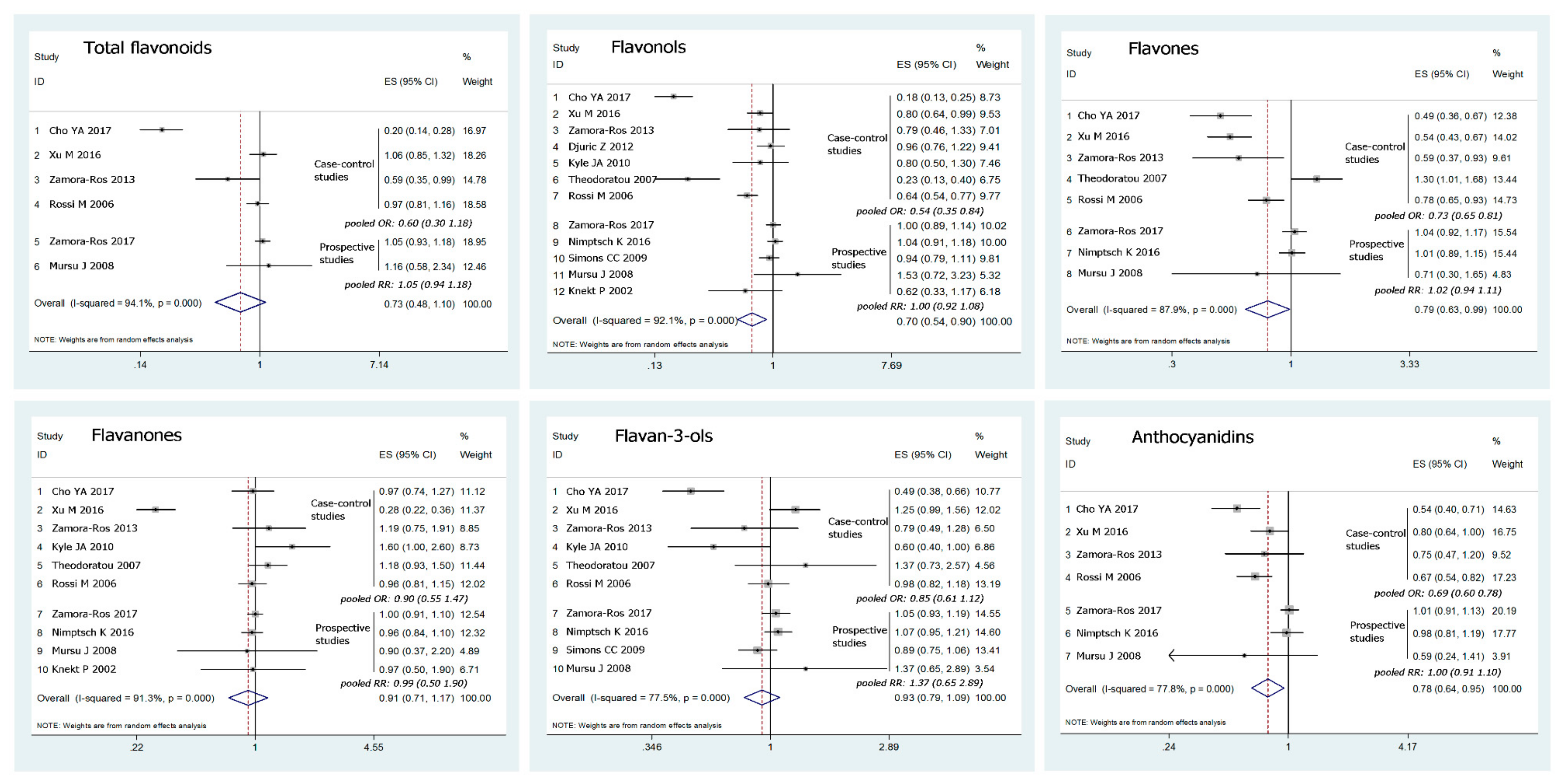
3.2. Dietary Flavonoid Intake and the Risk of Colorectal Cancer
3.3. Sensitivity Analysis
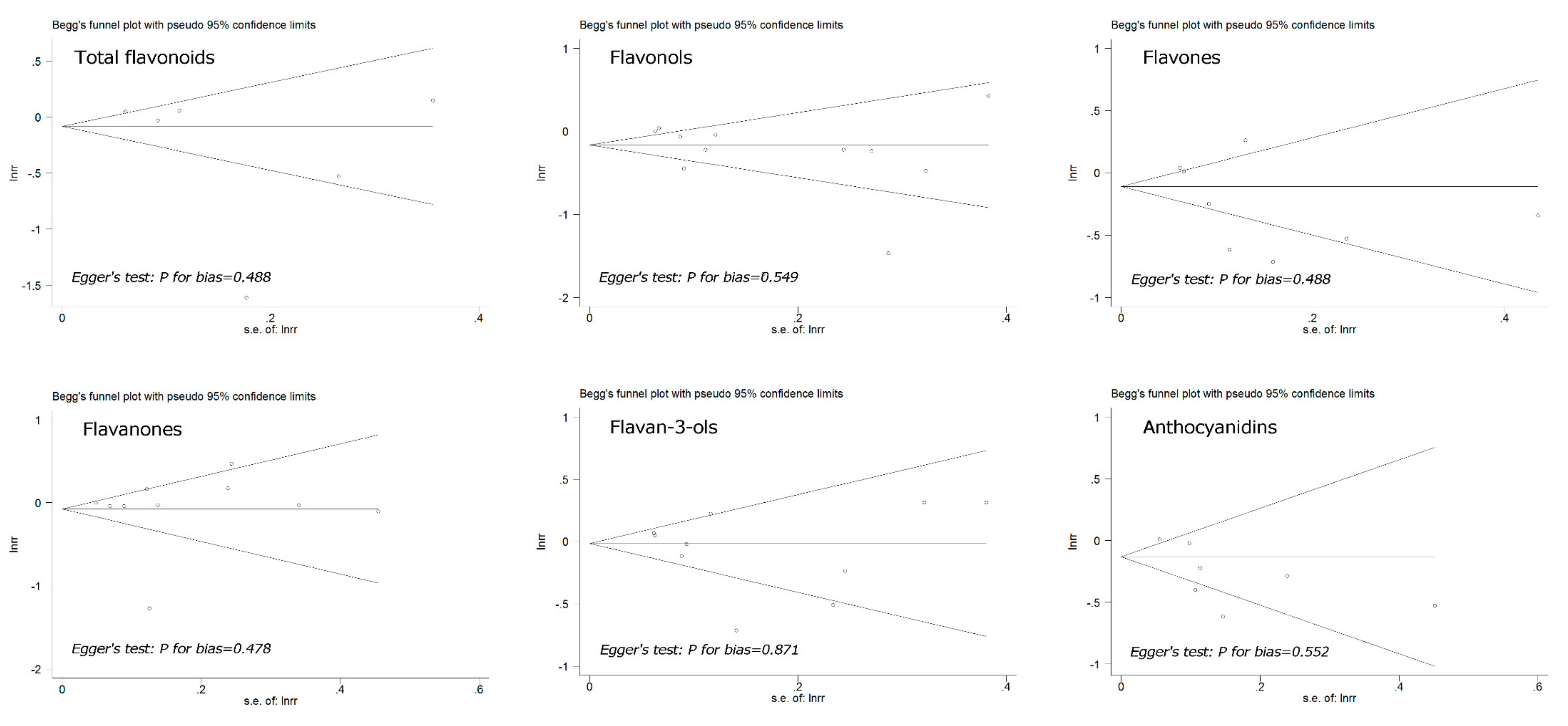
3.4. Publication Bias
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Cirmi, S.; Navarra, M.; Woodside, J.V.; Cantwell, M.M. Citrus fruits intake and oral cancer risk: A systematic review and meta-analysis. Pharmacol. Res. 2018, 133, 187–194. [Google Scholar] [CrossRef] [PubMed]
- Gu, M.J.; Huang, Q.C.; Bao, C.Z.; Li, Y.J.; Li, X.Q.; Ye, D.; Ye, Z.H.; Chen, K.; Wang, J.B. Attributable causes of colorectal cancer in China. BMC Cancer 2018, 18, 38. [Google Scholar] [CrossRef] [PubMed]
- Feng, Y.L.; Shu, L.; Zheng, P.F.; Zhang, X.Y.; Si, C.J.; Yu, X.L.; Gao, W.; Zhang, L. Dietary patterns and colorectal cancer risk: A meta-analysis. Eur. J. Cancer Prev. 2017, 26, 201–211. [Google Scholar] [CrossRef] [PubMed]
- Cirmi, S.; Ferlazzo, N.; Lombardo, G.E.; Maugeri, A.; Calapai, G.; Gangemi, S.; Navarra, M. Chemopreventive Agents and Inhibitors of Cancer Hallmarks: May Citrus Offer New Perspectives? Nutrients 2016, 8, 698. [Google Scholar] [CrossRef] [PubMed]
- Schwingshackl, L.; Schwedhelm, C.; Hoffmann, G.; Knüppel, S.; Laure Preterre, A.; Iqbal, K.; Bechthold, A.; De Henauw, S.; Michels, N.; Devleesschauwer, B.; et al. Food groups and risk of colorectal cancer. Int. J. Cancer 2018, 142, 1748–1758. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.; Je, Y. Flavonoid intake and mortality from cardiovascular disease and all causes: A meta-analysis of prospective cohort studies. Clin. Nutr. ESPEN 2017, 20, 68–77. [Google Scholar] [CrossRef] [PubMed]
- Petrick, J.L.; Steck, S.E.; Bradshaw, P.T.; Trivers, K.F.; Abrahamson, P.E.; Engel, L.S.; He, K.; Chow, W.H.; Mayne, S.T.; Risch, H.A.; et al. Dietary intake of flavonoids and oesophageal and gastric cancer: Incidence and survival in the United States of America (USA). Br. J. Cancer 2015, 112, 1291–300. [Google Scholar] [CrossRef] [PubMed]
- Ivey, K.L.; Jensen, M.K.; Hodgson, J.M.; Eliassen, A.H.; Cassidy, A.; Rimm, E.B. Association of flavonoid-rich foods and flavonoids with risk of all-cause mortality. Br. J. Nutr. 2017, 117, 1470–1477. [Google Scholar] [CrossRef] [PubMed]
- Grosso, G.; Godos, J.; Lamuela-Raventos, R.; Ray, S.; Micek, A.; Pajak, A.; Sciacca, S.; D’Orazio, N.; Del Rio, D.; Galvano, F. A comprehensive meta-analysis on dietary flavonoid and lignan intake and cancer risk: Level of evidence and limitations. Mol. Nutr. Food. Res. 2017, 61. [Google Scholar] [CrossRef] [PubMed]
- Sebastian, R.S.; Wilkinson Enns, C.; Goldman, J.D.; Moshfegh, A.J. Dietary Flavonoid Intake Is Inversely Associated with Cardiovascular Disease Risk as Assessed by Body Mass Index and Waist Circumference among Adults in the United States. Nutrients 2017, 9, 827. [Google Scholar] [CrossRef] [PubMed]
- Amawi, H.; Ashby, C.R.; Tiwari, A.K. Cancer chemoprevention through dietary flavonoids: What’s limiting? Chin. J. Cancer. 2017, 36, 50. [Google Scholar] [CrossRef] [PubMed]
- Xie, Y.; Huang, S.; Su, Y. Dietary Flavonols Intake and Risk of Esophageal and Gastric Cancer: A Meta-Analysis of Epidemiological Studies. Nutrients 2016, 8, 91. [Google Scholar] [CrossRef] [PubMed]
- Hua, X.; Yu, L.; You, R.; Yang, Y.; Liao, J.; Chen, D.; Yu, L. Association among Dietary Flavonoids; Flavonoid Subclasses and Ovarian Cancer Risk: A Meta-Analysis. PLoS ONE 2016, 11, e0151134. [Google Scholar] [CrossRef] [PubMed]
- Cho, Y.A.; Lee, J.; Oh, J.H.; Chang, H.J.; Sohn, D.K.; Shin, A.; Kim, J. Dietary Flavonoids; CYP1A1 Genetic Variants; and the Risk of Colorectal Cancer in a Korean population. Sci. Rep. 2017, 7, 128. [Google Scholar] [CrossRef] [PubMed]
- Xu, M.; Chen, Y.M.; Huang, J.; Fang, Y.J.; Huang, W.Q.; Yan, B.; Lu, M.S.; Pan, Z.Z.; Zhang, C.X. Flavonoid intake from vegetables and fruits is inversely associated with colorectal cancer risk: A case-control study in China. Br. J. Nutr. 2016, 116, 1275–1287. [Google Scholar] [CrossRef] [PubMed]
- Zamora-Ros, R.; Not, C.; Guinó, E.; Luján-Barroso, L.; García, R.M.; Biondo, S.; Salazar, R.; Moreno, V. Association between habitual dietary flavonoid and lignan intake and colorectal cancer in a Spanish case-control study (the Bellvitge Colorectal Cancer Study). Cancer Causes Control 2013, 24, 549–557. [Google Scholar] [CrossRef] [PubMed]
- Djuric, Z.; Severson, R.K.; Kato, I. Association of dietary quercetin with reduced risk of proximal colon cancer. Nutr. Cancer 2012, 64, 351–360. [Google Scholar] [CrossRef] [PubMed]
- Kyle, J.A.; Sharp, L.; Little, J.; Duthie, G.G.; McNeill, G. Dietary flavonoid intake and colorectal cancer: A case-control study. Br. J. Nutr. 2010, 103, 429–436. [Google Scholar] [CrossRef] [PubMed]
- Theodoratou, E.; Kyle, J.; Cetnarskyj, R.; Farrington, S.M.; Tenesa, A.; Barnetson, R.; Porteous, M.; Dunlop, M.; Campbell, H. Dietary flavonoids and the risk of colorectal cancer. Cancer Epidemiol. Prev. Biomark. 2007, 16, 684–693. [Google Scholar] [CrossRef] [PubMed]
- Rossi, M.; Negri, E.; Talamini, R.; Bosetti, C.; Parpinel, M.; Gnagnarella, P.; Franceschi, S.; Dal Maso, L.; Montella, M.; Giacosa, A.; et al. Flavonoids and colorectal cancer in Italy. Cancer Epidemiol. Prev. Biomark. 2006, 15, 1555–1558. [Google Scholar] [CrossRef] [PubMed]
- Zamora-Ros, R.; Barupal, D.K.; Rothwell, J.A.; Jenab, M.; Fedirko, V.; Romieu, I.; Aleksandrova, K.; Overvad, K.; Kyrø, C.; Tjønneland, A.; et al. Dietary flavonoid intake and colorectal cancer risk in the European prospective investigation into cancer and nutrition (EPIC) cohort. Int. J. Cancer 2017, 140, 1836–1844. [Google Scholar] [CrossRef] [PubMed]
- Nimptsch, K.; Zhang, X.; Cassidy, A.; Song, M.; O’Reilly, É.J.; Lin, J.H.; Pischon, T.; Rimm, E.B.; Willett, W.C.; Fuchs, C.S.; et al. Habitual intake of flavonoid subclasses and risk of colorectal cancer in 2 large prospective cohorts. Am. J. Clin. Nutr. 2016, 103, 184–191. [Google Scholar] [CrossRef] [PubMed]
- Simons, C.C.; Hughes, L.A.; Arts, I.C.; Goldbohm, R.A.; van den Brandt, P.A.; Weijenberg, M.P. Dietary flavonol.; flavone and catechin intake and risk of colorectal cancer in the Netherlands Cohort Study. Int. J. Cancer 2009, 125, 2945–2952. [Google Scholar] [CrossRef] [PubMed]
- Mursu, J.; Nurmi, T.; Tuomainen, T.P.; Salonen, J.T.; Pukkala, E.; Voutilainen, S. Intake of flavonoids and risk of cancer in Finnish men: The Kuopio Ischaemic Heart Disease Risk Factor Study. Int. J. Cancer 2008, 123, 660–663. [Google Scholar] [CrossRef] [PubMed]
- Knekt, P.; Kumpulainen, J.; Järvinen, R.; Rissanen, H.; Heliövaara, M.; Reunanen, A.; Hakulinen, T.; Aromaa, A. Flavonoid intake and risk of chronic diseases. Am. J. Clin. Nutr. 2002, 76, 560–568. [Google Scholar] [CrossRef] [PubMed]
- Kruger, C.; Zhou, Y. Red meat and colon cancer: A review of mechanistic evidence for heme in the context of risk assessment methodology. Food. Chem. Toxicol. 2018, 118, 131–153. [Google Scholar] [CrossRef] [PubMed]
- Hughes, L.A.E.; Simons, C.C.J.M.; van den Brandt, P.A.; van Engeland, M.; Weijenberg, M.P. Lifestyle, Diet, and Colorectal Cancer Risk According to (Epi)genetic Instability: Current Evidence and Future Directions of Molecular Pathological Epidemiology. Curr. Colorectal Cancer Rep. 2017, 13, 455–469. [Google Scholar] [CrossRef] [PubMed]
- He, X.; Sun, L.M. Dietary intake of flavonoid subclasses and risk of colorectal cancer: Evidence from population studies. Oncotarget 2016, 7, 26617–26627. [Google Scholar] [CrossRef] [PubMed]
- Madunić, J.; Madunić, I.V.; Gajski, G.; Popić, J.; Garaj-Vrhovac, V. Apigenin: A dietary flavonoid with diverse anticancer properties. Cancer Lett. 2018, 413, 11–22. [Google Scholar] [CrossRef] [PubMed]
- Koosha, S.; Alshawsh, M.A.; Looi, C.Y.; Seyedan, A.; Mohamed, Z. An Association Map on the Effect of Flavonoids on the Signaling Pathways in Colorectal Cancer. Int. J. Med. Sci. 2016, 13, 374–385. [Google Scholar] [CrossRef] [PubMed]
- George, V.C.; Dellaire, G.; Rupasinghe, H.P.V. Plant flavonoids in cancer chemoprevention: Role in genome stability. J. Nutr. Biochem. 2017, 45, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Zhong, C.; Wall, N.R.; Zu, Y.; Sui, G. Therapeutic Application of Natural Medicine Monomers in Cancer Treatment. Curr. Med. Chem. 2017, 24, 3681–3697. [Google Scholar] [CrossRef] [PubMed]
- Hoensch, H.; Groh, B.; Edler, L.; Kirch, W. Prospective cohort comparison of flavonoid treatment in patients with resected colorectal cancer to prevent recurrence. World J. Gastroenterol. 2008, 14, 2187–2193. [Google Scholar] [CrossRef] [PubMed]
- Niederreiter, L.; Adolph, T.E.; Tilg, H. Food, microbiome and colorectal cancer. Dig. Liver Dis. 2018, 50, 647–652. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Yu, J. The association of diet.; gut microbiota and colorectal cancer: What we eat may imply what we get. Protein Cell 2018. [Google Scholar] [CrossRef] [PubMed]
- Thomas, S.; Izard, J.; Walsh, E.; Batich, K.; Chongsathidkiet, P.; Clarke, G.; Sela, D.A.; Muller, A.J.; Mullin, J.M.; Albert, K.; et al. The Host Microbiome Regulates and Maintains Human Health: A Primer and Perspective for Non-Microbiologists. Cancer Res. 2017, 77, 1783–1812. [Google Scholar] [CrossRef] [PubMed]
- Houghton, D.; Stewart, C.J.; Day, C.P.; Trenell, M. Gut Microbiota and Lifestyle Interventions in NAFLD. Int. J. Mol. Sci. 2016, 17, 447. [Google Scholar] [CrossRef] [PubMed]
- Cope, E.K.; Goldberg, A.N.; Pletcher, S.D.; Lynch, S.V. Compositionally and functionally distinct sinus microbiota in chronic rhinosinusitis patients have immunological and clinically divergent consequences. Microbiome 2017, 5, 53. [Google Scholar] [CrossRef] [PubMed]
- Barko, P.C.; McMichael, M.A.; Swanson, K.S.; Williams, D.A. The Gastrointestinal Microbiome: A Review. J. Vet. Intern. Med. 2018, 32, 9–25. [Google Scholar] [CrossRef] [PubMed]
- Murota, K.; Nakamura, Y.; Uehara, M. Flavonoid metabolism: The interaction of metabolites and gut microbiota. Biosci. Biotechnol. Biochem. 2018, 82, 600–610. [Google Scholar] [CrossRef] [PubMed]
- Marranzano, M.; Ray, S.; Godos, J.; Galvano, F. Association between dietary flavonoids intake and obesity in a cohort of adults living in the Mediterranean area. Int. J. Food. Sci. Nutr. 2018, 26, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Oteiza, P.I.; Fraga, C.G.; Mills, D.A.; Taft, D.H. Flavonoids and the gastrointestinal tract: Local and systemic effects. Mol. Aspects Med. 2018, 61, 41–49. [Google Scholar] [CrossRef] [PubMed]
| Author, Year Region, Design, Cases/Controls | Flavonoids Exposure (mg/day), RR or OR and 95% CI | Adjustments | |||
|---|---|---|---|---|---|
| Cho YA 2017 Korean Case-control923/1846 [14] | Total flavonoids, MI (86.3) | Flavanones, MI (3.7) | Age, sex, BMI, education, total caloric intake, FHCC, and regular exercise. | ||
| Q1 (<67.7) | 1.00 | Q1 (<1.16) | 1.00 | ||
| Q2 (67.7–98.3) | 0.91 (0.72–1.14) | Q2 (1.16–3.54) | 1.16 (0.90–1.50) | ||
| Q3 (98.3–141.7) | 0.66 (0.52–0.85) | Q3 (3.54–8.99) | 1.37 (1.07–1.76) | ||
| Q4 (≥141.7) | 0.20 (0.14–0.28) | Q4 (≥8.99) | 0.97 (0.74–1.27) | ||
| Flavonols, MI (16.4) | Flavan-3-ols, MI (10.2) | ||||
| Q1 (<13.0) | 1.00 | Q1 (<5.62) | 1.00 | ||
| Q2 (13.0–19.2) | 1.05 (0.84–1.32) | Q2 (5.62–13.19) | 0.90 (0.71–1.14) | ||
| Q3 (19.2–27.4) | 0.50 (0.39–0.65) | Q3 (13.19–30.38) | 0.67 (0.52–0.87) | ||
| Q4 (≥27.4) | 0.18 (0.13–0.25) | Q4 (≥30.38) | 0.49 (0.38–0.66) | ||
| Flavones, MI (1.0) | Anthocyanidins, MI (17.0) | ||||
| Q1 (<0.75) | 1.00 | Q1 (<11.4) | 1.00 | ||
| Q2 (0.75–1.12) | 1.50 (1.17–1.92) | Q2 (11.4–18.9) | 1.22 (0.96–1.56) | ||
| Q3 (1.12–1.70) | 1.34 (1.04–1.73) | Q3 (18.9–29.7) | 0.99 (0.77–1.26) | ||
| Q4 (≥1.70) | 0.49 (0.36–0.67) | Q4 (≥29.7) | 0.54 (0.40–0.71) | ||
| Xu M 2016 China Case-control 1632/1632 [15] | Total flavonoids, MI (248.5) | Flavanones, MI (3.5) | Age, sex, marital status, education, income, occupation, family history of cancer, smoking status, passive smoking, alcohol, activity, BMI, and intakes of red and processed meat, poultry and fish, total dairy products and eggs. | ||
| Q1 | 1.00 | Q1 | 1.00 | ||
| Q2 | 1.11 (0.90–1.38) | Q2 | 0.76 (0.62–0.92) | ||
| Q3 | 1.04 (0.84–1.31) | Q3 | 0.61 (0.50–0.76) | ||
| Q4 | 1.06 (0.85–1.32) | Q4 | 0.28 (0.22–0.36) | ||
| Flavonols, MI (34.5) | Flavan-3-ols, MI (26.6) | ||||
| Q1 | 1.00 | Q1 | 1.00 | ||
| Q2 | 0.83 (0.66–1.03) | Q2 | 1.18 (0.94–1.47) | ||
| Q3 | 0.81 (0.65–1.00) | Q3 | 1.18 (0.95–1.48) | ||
| Q4 | 0.80 (0.64–0.99) | Q4 | 1.25 (0.99–1.56) | ||
| Flavones, MI (2.6) | Anthocyanidins, MI (18.9) | ||||
| Q1 | 1.00 | Q1 | 1.00 | ||
| Q2 | 0.55 (0.44–0.68) | Q2 | 0.91 (0.73–1.12) | ||
| Q3 | 0.54 (0.44–0.67) | Q3 | 0.93 (0.75–1.15) | ||
| Q4 | 0.54 (0.43–0.67) | Q4 | 0.80 (0.64–1.00) | ||
| Zamora-Ros R Spain 2013 Case-control 424/401 [16] | Total flavonoids, MI (198.0) | Flavanones, MI (17.1) | Sex, age, BMI, FHCC, energy intake, alcohol and fiber intake, red and processed meat intake, tobacco, PA, aspirin, NSAID. | ||
| Q1 (<68.9) | 1.00 | Q1 (<3.7) | 1.00 | ||
| Q2 (68.9–108.9) | 0.99 (0.66–1.49) | Q2 (3.7–9.1) | 1.46 (0.97–2.19) | ||
| Q3 (108.9–167.9) | 0.88 (0.57–1.37) | Q3 (9.1–17.7) | 1.09 (0.71–1.66) | ||
| Q5 (>167.9) | 0.59 (0.35–0.99) | Q5 (>17.7) | 1.19 (0.75–1.91) | ||
| Flavonols, MI (14.5) | Flavan-3-ols, MI (15.2) | ||||
| Q1 (<5.1) | 1.00 | Q1 (<4.9) | 1.00 | ||
| Q2 (5.1–8.3) | 0.98 (0.65–1.47) | Q2 (4.9–8.1) | 0.93 (0.61–1.40) | ||
| Q3 (8.4–11.5) | 0.78 (0.50–1.23) | Q3 (8.2–12.9) | 1.05 (0.69–1.61) | ||
| Q5 (>11.5) | 0.79 (0.46–1.33) | Q5 (>12.9) | 0.79 (0.49–1.28) | ||
| Flavones, MI (2.2) | Anthocyanidins, MI (11.4) | ||||
| Q1 (<0.7) | 1.00 | Q1 (<3.3) | 1.00 | ||
| Q2 (0.7–1.2) | 0.76 (0.51–1.15) | Q2 (3.3–6.5) | 0.74 (0.49–1.12) | ||
| Q3 (1.3–2.1) | 0.79 (0.52–1.21) | Q3 (6.6–10.6) | 0.75 (0.50–1.14) | ||
| Q5 (>2.1) | 0.59 (0.37–0.93) | Q5 (>10.6) | 0.75 (0.47–1.20) | ||
| Djuric Z USA 2012 Case-control 1163/1501 [17] | Flavonol (quercetin), MI (8.35) | Age, gender, PA, BMI, FHCC, education, NSAID, total fat, fiber, carotenoids, folate. | |||
| Q1 (<4.3) | 1.00 | ||||
| Q2 (4.3–6.3) | 0.80 (0.64–1.01) | ||||
| Q3 (6.3–10.3) | 1.06 (0.84–1.33) | ||||
| Q5 (>10.3) | 0.96 (0.76–1.22) | ||||
| Kyle JA UK 2010 Case-control 264/408 [18] | Flavonols, MI (30.1) | Flavanones, MI (15.6) | Energy, age at diagnosis, family history, NSAID, aspirin, Mn, riboflavin, vitamin C, folate. | ||
| Q1 (<19.3) | 1.00 | Q1 (<2.7) | 1.00 | ||
| Q2 (19.3–30.4) | 1.0 (0.6–1.7) | Q2 (2.7–13.4) | 1.5 (0.9–2.5) | ||
| Q3 (30.4–40.4) | 1.3 (0.8–2.1) | Q3 (13.4–32.2) | 1.4 (0.9–2.4) | ||
| Q5 (>40.4) | 0.8 (0.5–1.3) | Q5 (>32.2) | 1.6 (1.0–2.6) | ||
| Flavan-3-ols, MI (127.8) | |||||
| Q1 (<67.1) | 1.00 | ||||
| Q2 (67.1–119.2) | 0.7 (0.4–1.1) | ||||
| Q3 (119.2–188.8) | 1.3 (0.8–2.2) | ||||
| Q5 (>188.8) | 0.6 (0.4–1.0) | ||||
| Theodoratou E Scotland 2007 Case-control 1456/1456 [19] | Flavonols, MI (27.4) | Flavanones, MI (20.4) | Total energy, FHCC, fiber intake, alcohol, NSAID intake, smoking, BMI, and PA, fruit, vegetable intake. | ||
| Q1 (<16.0) | 1.00 | Q1 (<16.7) | 1.00 | ||
| Q2 (16.1–27.4) | 0.57 (0.43–0.76) | Q2 (16.7–32.7) | 1.43 (1.15–1.80) | ||
| Q3 (27.5–36.8) | 0.41 (0.27–0.63) | Q3 (32.7–45.2) | 1.35 (1.08–1.70) | ||
| Q4 (>36.8) | 0.23 (0.13–0.40) | Q5 (>45.2) | 1.18 (0.93–1.50) | ||
| Flavones, MI (1.0) | Flavan-3-ols, MI (115.4) | ||||
| Q1 (<0.5) | 1.00 | Q1 (<42.6) | 1.00 | ||
| Q2 (0.5–1.1) | 1.05 (0.85–1.31) | Q2 (42.6–115.3) | 1.10 (0.81–1.49) | ||
| Q3 (1.1–1.9) | 1.01 (0.81–1.26) | Q3 (115.3–162.1) | 1.56 (0.98–2.50) | ||
| Q4 (>1.9) | 1.30 (1.01–1.68) | Q5 (>162.1) | 1.37 (0.73–2.57) | ||
| Rossi M Italy 2006 Case-control 1953/4154 [20] | Total flavonoids, MI (137.8) | Flavanones, MI (38.3) | Age, sex, study center, FHCC, education, alcohol consumption, BMI, occupational PA, and energy intake. | ||
| Q1 (<75.3) | 1.00 | Q1 (<12.5) | 1.00 | ||
| Q2 (75.4–108.5) | 0.90 (0.75–1.08) | Q2 (12.6–28.7) | 0.88 (0.74–1.05) | ||
| Q3 (108.6–141.6) | 0.79 (0.66–0.94) | Q3 (28.8–35.5) | 0.89 (0.75–1.07) | ||
| Q4 (141.7–191.1) | 0.81 (0.67–0.97) | Q4 (35.6–67.0) | 0.80 (0.67–0.96) | ||
| Q5 (>191.1) | 0.97 (0.81–1.16) | Q5 (>67.0) | 0.96 (0.81–1.15) | ||
| Flavonols, MI (21.6) | Flavan-3-ols, MI (54.0) | ||||
| Q1 (<13.2) | 1.00 | Q1 (<20.8) | 1.00 | ||
| Q2 (13.3–17.3) | 0.80 (0.67–0.95) | Q2 (20.9–34.4) | 0.75 (0.63–0.91) | ||
| Q3 (17.4–22.0) | 0.77 (0.64–0.91) | Q3 (34.5–51.7) | 0.75 (0.62–0.90) | ||
| Q4 (22.1–28.5) | 0.74 (0.62–0.88) | Q4 (51.8–88.5) | 0.79 (0.65–0.95) | ||
| Q5 (>28.6) | 0.64 (0.54–0.77) | Q5 (>88.5) | 0.98 (0.82–1.18) | ||
| Flavones, MI (0.5) | Anthocyanidins, MI (20.0) | ||||
| Q1 (<0.3) | 1.00 | Q1 (<5.3) | 1.00 | ||
| Q2 (0.3–0.4) | 0.82 (0.69–0.98) | Q2 (5.4–11.5) | 0.81 (0.68–0.96) | ||
| Q3 (0.4–0.5) | 0.72 (0.61–0.86) | Q3 (11.6–19.4) | 0.78 (0.65–0.93) | ||
| Q4 (0.5–0.7) | 0.76 (0.64–0.91) | Q4 (19.5–31.7) | 0.64 (0.53–0.77) | ||
| Q5 (>0.7) | 0.78 (0.65–0.93) | Q5 (>31.7) | 0.67 (0.54–0.82) | ||
| Zamora-Ros R Europe 2017 Prospective cohort 4517(477,312) [21] | Total flavonoids, MI (418) | Flavanones, MI (40) | Sex, age, center, smoking, PA, education, BMI, total energy, alcohol, red and processed meat, fibre and calcium intakes, menopausal status, hormone replacement therapy use, contraceptive use. | ||
| Q1(<223) | 1.00 | Q1 (<8.2) | 1.00 | ||
| Q2 (223–346) | 1.09 (0.99–1.20) | Q2 (8.2–18.1) | 0.96 (0.88–1.05) | ||
| Q3 (347–507) | 1.10 (0.99–1.22) | Q3 (18.2–33.3) | 0.99 (0.91–1.09) | ||
| Q4 (508–771) | 1.07 (0.96–1.20) | Q4 (33.4–65.9) | 0.95 (0.86–1.05) | ||
| Q5 (>771) | 1.05 (0.93–1.18) | Q5 (>65.9) | 1.00 (0.91–1.10) | ||
| Flavonols, MI (28) | Flavan-3-ols, MI (325) | ||||
| Q1 (<13.9) | 1.00 | Q1 (<135) | 1.00 | ||
| Q2 (13.9–23.0) | 1.02 (0.92–1.13) | Q2 (135–228) | 1.08 (0.98–1.19) | ||
| Q3 (23.1–34.8) | 1.03 (0.93–1.15) | Q3 (229–356) | 1.15 (1.04–1.28) | ||
| Q4 (34.9–61.7) | 0.99 (0.88–1.11) | Q4 (357–584) | 1.10 (0.99–1.23) | ||
| Q5 (>61.7) | 1.00 (0.89–1.14) | Q5 (>584) | 1.05 (0.93–1.19) | ||
| Flavones, MI (9.3) | Anthocyanidins, MI (25) | ||||
| Q1 (<5.1) | 1.00 | Q1 (<10.3) | 1.00 | ||
| Q2 (5.1–7.8) | 1.01 (0.93–1.11) | Q2 (10.3–19.1) | 0.95 (0.87–1.04) | ||
| Q3 (7.9–11.0) | 0.94 (0.85–1.03) | Q3 (19.2–32.5) | 0.96 (0.87–1.06) | ||
| Q4 (11.1–16.5) | 1.04 (0.94–1.15) | Q4 (32.6–58.9) | 1.00 (0.91–1.10) | ||
| Q5 (>16.5) | 1.04 (0.92–1.17) | Q5 (>58.9) | 1.01 (0.91–1.13) | ||
| Nimptsch K 2016 USA Prospective cohort 2519(118,842) [22] | Flavonols, MI (-) | Flavanones, MI (-) | Age, smoking, history of colorectal cancer, history of endoscopy, regular aspirin use, BMI, PA, alcohol, total calories, vitamin D, total calcium, red meat, and processed meat intake. | ||
| Q1 (9.6) | 1.00 | Q1 (23.0) | 1.00 | ||
| Q2 | 0.92 (0.78–1.09) | Q2 | 0.99 (0.82–1.19) | ||
| Q3 (15.2) | 0.92 (0.81–1.05) | Q3 (52.0) | 1.05 (0.86–1.28) | ||
| Q4 | 1.10 (0.97–1.24) | Q4 | 0.99 (0.79–1.23) | ||
| Q5 (31.9) | 1.04 (0.91–1.18) | Q5 (56.6) | 0.96 (0.84–1.10) | ||
| Flavones, MI (-) | Flavan-3-ols, MI (-) | ||||
| Q1 (1.4) | 1.00 | Q1 (10.2) | 1.00 | ||
| Q2 | 0.99 (0.87–1.13) | Q2 | 0.96 (0.84–1.09) | ||
| Q3 (2.6) | 1.02 (0.89–1.16) | Q3 (25.0) | 0.98 (0.86–1.11) | ||
| Q4 | 1.00 (0.88–1.14) | Q4 | 1.00 (0.87–1.15) | ||
| Q5 (2.8) | 1.01 (0.89–1.15) | Q5 (141.8) | 1.07 (0.95–1.21) | ||
| Anthocyanidins, MI (-) | |||||
| Q1 (5.5) | 1.00 | ||||
| Q2 | 0.93 (0.82–1.05) | ||||
| Q3 (14.6) | 1.04 (0.92–1.18) | ||||
| Q4 | 0.97 (0.85–1.10) | ||||
| Q5 (23.6) | 0.98 (0.81–1.19) | ||||
| Simons CC Netherland 2009 Prospective cohort 2485(120,852) [23] | Flavonols, MI (26.8) men | Flavonols, MI (28.9) women | Age, FHCC, smoking, alcohol, PA, BMI and processed meat intake. | ||
| Q1 (<16.0) | 1.00 | Q1 (<18.4) | 1.00 | ||
| Q2 (16.0–22.5) | 0.95 (0.75–1.21) | Q2 (18.4–25.0) | 0.85 (0.66–1.10) | ||
| Q3 (22.5–28.3) | 0.81 (0.63–1.04) | Q3 (25.0–31.1) | 0.98 (0.76–1.25) | ||
| Q3 (28.3–36.1) | 0.89 (0.70–1.14) | Q3 (31.1–38.4) | 0.80 (0.62–1.03) | ||
| Q5 (>36.1) | 0.97 (0.76–1.23) | Q5 (>38.4) | 0.90 (0.70–1.16) | ||
| Flavan-3-ols, MI (58.6) men | Flavan-3-ols, MI (66.2) women | ||||
| Q1 (<24.2) | 1.00 | Q1 (<36.2) | 1.00 | ||
| Q2 (24.2–44.4) | 1.01 (0.79–1.28) | Q2 (36.2–51.6) | 0.90 (0.70–1.16) | ||
| Q3 (44.4–62.8) | 0.85 (0.67–1.09) | Q3 (51.6–75.4) | 0.79 (0.61–1.02) | ||
| Q3 (62.8–84.4) | 0.85 (0.67–1.08) | Q3 (75.4–95.9) | 1.02 (0.79–1.30) | ||
| Q5 (>84.4) | 0.99 (0.77–1.25) | Q5 (>95.9) | 0.79 (0.61–1.02) | ||
| Mursu J Finland 2008 Prospective cohort 55(2590) [24] | Total flavonoids, MI (131.0) | Flavanones, MI (2.9) | Age, examination years, BMI, smoking, PA, intakes of alcohol, total fat, saturated fat, fiber, vitamin C and E. | ||
| Q1 (9.1) | 1.00 | Q1 | 1.00 | ||
| Q2 (16.3) | 0.74 (0.34–1.60) | Q2 | 0.84 (0.36–1.98) | ||
| Q3 (82.7) | 0.52 (0.22–1.23) | Q3 | 1.80 (0.85–3.85) | ||
| Q5 (416.3) | 1.16 (0.58–2.34) | Q4 | 0.90 (0.37–2.20) | ||
| Flavonols, MI (9.5) | Flavan-3-ols, MI (112.3) | ||||
| Q1 | 1.00 | Q1 | 1.00 | ||
| Q2 | 0.68 (0.30–1.58) | Q2 | 1.04 (0.48–2.28) | ||
| Q3 | 0.86 (0.38–1.97) | Q3 | 0.80 (0.34–1.86) | ||
| Q4 | 1.53 (0.72–3.23) | Q4 | 1.37 (0.65–2.89) | ||
| Flavones, MI (0.3) | Anthocyanidins, MI (5.9) | ||||
| Q1 | 1.00 | Q1 | 1.00 | ||
| Q2 | 1.26 (0.59–2.68) | Q2 | 0.69 (0.30–1.60) | ||
| Q3 | 1.16 (0.54–2.50) | Q3 | 1.62 (0.80–3.31) | ||
| Q4 | 0.71 (0.30–1.65) | Q4 | 0.59 (0.24–1.41) | ||
| Knekt P Finland 2002 Prospective cohort 90(9865) [25] | Flavonol (quercetin), MI (3.3) | Flavanone (hesperetin), MI (15.1) | Sex, age, geographic area, occupation, smoking, and BMI. | ||
| Q1 (<1.7) | 1.00 | Q1 (<1.6) | 1.00 | ||
| Q2 (1.7–2.7) | 0.84 (0.48–1.49) | Q2 (1.6–10.2) | 1.49 (0.87–2.58) | ||
| Q3 (2.7–3.4) | 0.97 (0.56–1.70) | Q3 (10.2–20.9) | 1.56 (0.86–2.84) | ||
| Q5 (>3.4) | 0.62 (0.33–1.17) | Q5 (>20.9) | 0.97 (0.50–1.90) | ||
| Study | No. of Studies | RR(95% CI) | Heterogeneity Test | ||
|---|---|---|---|---|---|
| p | I2 (%) | ||||
| Flavonols | |||||
| Design | Case-control | 7 | 0.54 (0.35–0.84) | 0.00 | 93.1 |
| Prospective | 5 | 1.00 (0.92–1.08) | 0.369 | 6.6 | |
| Cancer type | Colon | 7 | 0.80 (0.68–0.94) | 0.025 | 58.4 |
| Rectum | 7 | 0.93 (0.74–1.18) | 0.009 | 64.8 | |
| Population | European | 8 | 0.75 (0.58–0.96) | 0.00 | 83.3 |
| Asian | 2 | 0.51 (0.42–0.61) | 0.00 | 98.2 | |
| USA | 2 | 1.02 (0.91–1.14) | 0.561 | 0.0 | |
| Flavones | |||||
| Design | Case-control | 5 | 0.73 (0.65–0.81) | 0.00 | 88.4 |
| Prospective | 3 | 1.02 (0.94–1.11) | 0.665 | 0.0 | |
| Cancer type | Colon | 4 | 0.88 (0.69–1.13) | 0.011 | 73.1 |
| Rectum | 4 | 0.82 (0.70–0.97) | 0.608 | 0.0 | |
| Population | European | 5 | 0.91 (0.72–1.16) | 0.002 | 76.5 |
| Asian | 2 | 0.52 (0.44–0.63) | 0.618 | 0 | |
| USA | 1 | 1.01 (0.89–1.15) | - | - | |
| Anthocyanidins | |||||
| Design | Case-control | 4 | 0.69 (0.60–0.78) | 0.196 | 36 |
| Prospective | 3 | 1.00 (0.91–1.10) | 0.488 | 0.0 | |
| Cancer type | Colon | 3 | 0.81 (0.58–1.12) | 0.022 | 73.7 |
| Rectum | 3 | 0.84 (0.59–1.21) | 0.099 | 56.7 | |
| Population | European | 4 | 0.91 (0.83–1.00) | 0.004 | 77.6 |
| Asian | 2 | 0.66 (0.45–0.98) | 0.034 | 77.7 | |
| USA | 1 | 0.98 (0.81–1.19) | - | - | |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chang, H.; Lei, L.; Zhou, Y.; Ye, F.; Zhao, G. Dietary Flavonoids and the Risk of Colorectal Cancer: An Updated Meta-Analysis of Epidemiological Studies. Nutrients 2018, 10, 950. https://doi.org/10.3390/nu10070950
Chang H, Lei L, Zhou Y, Ye F, Zhao G. Dietary Flavonoids and the Risk of Colorectal Cancer: An Updated Meta-Analysis of Epidemiological Studies. Nutrients. 2018; 10(7):950. https://doi.org/10.3390/nu10070950
Chicago/Turabian StyleChang, Hui, Lin Lei, Yun Zhou, Fayin Ye, and Guohua Zhao. 2018. "Dietary Flavonoids and the Risk of Colorectal Cancer: An Updated Meta-Analysis of Epidemiological Studies" Nutrients 10, no. 7: 950. https://doi.org/10.3390/nu10070950
APA StyleChang, H., Lei, L., Zhou, Y., Ye, F., & Zhao, G. (2018). Dietary Flavonoids and the Risk of Colorectal Cancer: An Updated Meta-Analysis of Epidemiological Studies. Nutrients, 10(7), 950. https://doi.org/10.3390/nu10070950





